Abstract
We investigated the role of nitric oxide (NO) in leukocyte-endothelium interaction, blood-brain barrier (BBB) function and oxygen free-radical production in the rat pial microcirculation. In a closed cranial window preparation (dura removed) over the parietal cortex of pentobarbital-anesthetized Wistar rats, NO synthase (NOS) was inhibited by systemic and/or topical application of Nω-nitro-
Keywords
Nitric oxide (NO) or a related compound has been shown to play an important role in the blood flow regulation of various tissues, including the regulation of resting and stimulated cerebral blood flow (CBF) during global (i.e., with CO2) and functional activation of the cerebral cortex (for review, see Iadecola et al., 1994a).
In addition to its vascular effects via modulation of smooth muscle function, NO may play a role in the microcirculation by affecting leukocyte-endothelium interaction. Impairment of endothelial NO production in mesenteric venules in vivo leads to a drastic increase in leukocyte adherence to the vascular endothelium (Kubes et al., 1991) possibly due to a superoxide anion radical mechanism (Gaboury et al., 1993). In addition, increases of neutrophil adhesion during NOS inhibition have also been observed in the heart (Ma et al., 1993), liver (Harbrecht et al., 1992), and skeletal muscle (Persson et al., 1990). So far, the role of NO in leukocyte-endothelium interaction has only been studied in detail in the circulation of noncerebral tissues. The brain microvascular endothelium, which forms the blood-brain barrier (BBB) differs functionally, antigenetically, and morphologically from extracerebral vascular endothelium. In addition, in the brain, NO is constitutively not only produced by the endothelium, but also by the surrounding parenchyma (neurons, astrocytes, etc.). Data from extracerebral tissues concerning the role of NO in leukocyte–endothelium interaction may, therefore, not be applicable to the brain.
The aim of this study was to investigate whether NO also plays a role in leukocyte-endothelium interaction during physiological as well as leukotriene B4 (LTB4)-activated conditions in the cerebral microcirculation. Leukocyte-endothelium interaction in pial vessels of the cerebral cortex of anesthetized rats were studied before and during systemic and/or topical application of the irreversible nitric oxide synthase (NOS) inhibitor Nω-nitro-
MATERIALS AND METHODS
General preparation
Male Wistar rats (280–320 g), were anesthetized with pentobarbital (Trapanal, 100 mg/kg body wt, Byk Gulden, Konstanz, Germany), tracheotomized, and mechanically ventilated. The left femoral artery and vein were cannulated for physiological monitoring, intravenous saline infusion (infusion rate 1 ml/h), and application of drugs. Body temperature was monitored and maintained at 38 ± 0.5°C using a heating pad. Systemic arterial blood pressure (SAP) and endexpiratory Pco2 (Heyer EGM I, Bad Ems, Germany) were monitored continuously and blood gases measured periodically (AVL LIST, Graz, Austria). Animals were placed in a stereotactic frame and a closed cranial window was implanted over the right parietal cortex (Dirnagl et al., 1991). The dura mater was removed and the exposed brain continuously superfused (superfusion rate, 1 ml/h) with artificial cerebrospinal fluid (aCSF) (composition in mM):K+, 3; Na+, 150; Ca2+, 1.25; Mg2+, 0.6; Cl−, 132; HCO3−, 24.5; glucose 3.7; urea 6 (Levasseur et al., 1975), which was equilibrated with a gas mixture containing (in %) O2, 6.6; CO2, 5.9; and N2, 87.5 leading to the following gas tensions and pH: Po2, 119 ± 8 mm Hg; Pco2, 34.6 ± 3.9 mm Hg; and pH 7.36 ± 0.03.
CLSM setup
For monitoring leukocytes within the pial circulation, we used a Bio-Rad MRC 600 CLSM system (Bio-Rad Microscience, Watford, U.K.) (for details, see Dirnagl et al., 1992). In brief, the CLSM was attached to a Nikon Optiphot microscope with a Zeiss water immersion objective corrected for a cover slip with a numerical aperture of 0.75, a working distance of 1.6 mm, and ×40 magnification. We used a Krypton laser (Ion Laser Technology, Salt Lake City, UT, U.S.A.) with principle lines at 488, 568, and 647 nm as light source. Confocal microscopy was performed through the cranial window. To visualize leukocytes, rhodamine 6G (Sigma Chemicals) was injected intravenously (200 μg/ml in 0.9% saline, 1 ml as bolus injection, followed by continuous infusion at an infusion rate of 1 ml/h). After intravenous injection of rhodamine 6G, circulating leukocytes (polymorphonuclear leukocytes, lymphocytes and monocytes) and platelets were labeled, whereas endothelial (ECs) and red blood cells remained unstained (Dirnagl et al., 1994; Villringer et al., 1991). Rhodamine 6G staining, according to this protocol, does not affect leukocyte function (Lorenzl et al., 1993; Dirnagl et al., 1994; Villringer et al., 1991). Appropriate filter sets for rhodamine 6G imaging were used. An average of seven different sites of exposed pial microvasculature per animal was recorded (area, 317 × 211 μm) at a frame rate of 16/s for 60 s and digital/analog converted for recording on a video recorder (Panasonic NV-V 8000 EG, Osaka, Japan). Images were analyzed off-line from the video recording by redigitalization. The CLSM was equipped with a computer-controlled x-y-z stage, permitting movement of the object with an accuracy of 0.1 μm in every dimension. It was, therefore, possible to return exactly to each of the seven recording sites during the course of the experiment.
In vivo CL recording setup
Reactive oxygen species were detected by lucigenin-enhanced CL in vivo. For a detailed description of this technique, see Dirnagl et al. (1995). In brief, the animal in the stereotactic frame was housed in a dark box and covered with aluminum foil to exclude photons from sources other than CL and to prevent spontaneous ultraweak CL. The unshielded cranial window was positioned under a reflector focused to the photon sensitive area of a cooled (–20°C) photomultiplier (Hamamatsu R943–02) with a dark count of 3 cps at −20°C. The animal within the dark box was housed in a second, larger dark box, equipped with metal shielding (Faraday cage) to exclude photons and other electromagnetic waves unrelated to brain CL from outside. To record oxygen free-radicals optically, brain topical superfusion of the CL enhancer lucigenin (10−3 M, 1 ml/h) was used. Lucigenin reacts with oxygen free-radicals producing photons in the blue/green wavelength range. Lucigenin-induced CL is particularly sensitive to the intracellular production of the superoxide anion radical (Faulkner and Fridovich, 1993), regardless of the cell type within the brain. Counts of the photomultiplier were amplified and counted by a Hewlett Packard Universal counter (HP 5316 B9) connected to a PC for data recording and storage.
NOS activity determination
Immediately after experiments, tissue specimens were removed from the cortex in situ to depth of 1 mm under the cranial window and frozen in liquid N2 for later analysis. The NOS assay was based on assessing the conversion of
NOS activity in sham-operated animals (n = 2) was assayed and used as control. Percent inhibition was calculated by comparing enzyme activity in tissue exposed to the NOS inhibitor
Experimental paradigm (Fig. 1)
In the first two studies (I and II), leukocyte-endothelium interaction was investigated before and 30 and 60 min after
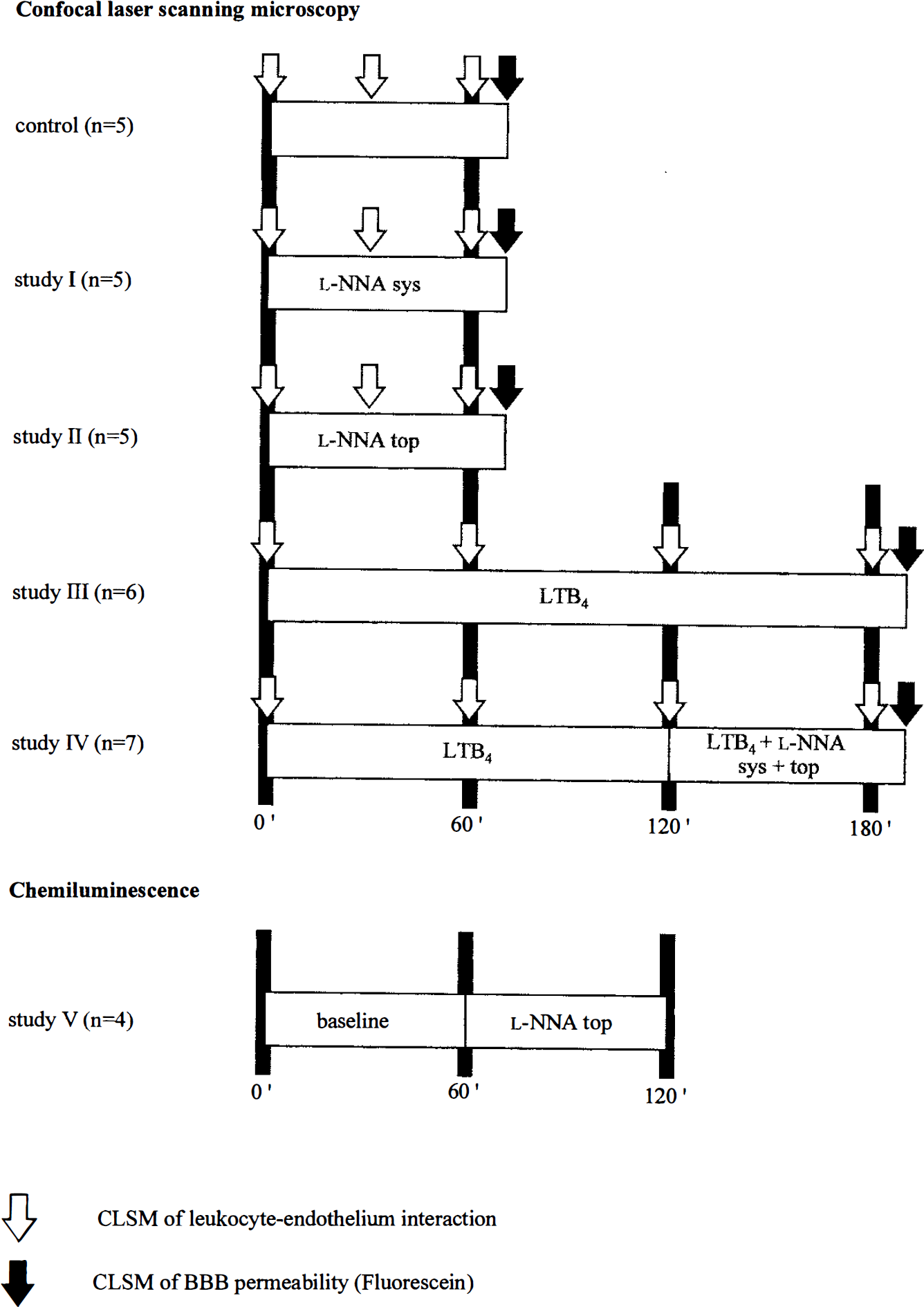
Experimental paradigm.
In studies I and II, a laser-Doppler flow (LDF) probe monitored rCBF through the closed cranial window in the area of confocal imaging. In addition to the ex vivo NOS activity determination assay, a decrease of resting rCBF and appearance of vasomotion marked the effectivity of the NOS blockade in the brain cortex underlying the cranial window in all animals (Dirnagl et al. 1993a,b).
In studies III and IV, the leukotactic compound LTB4 (7.5 × 10−9 M) was superfused over the cranial window for 3 h. Experiments of 3 h of topical superfusion of LTB4 without
To study BBB integrity, at the end of each experiment in studies I-IV, sodium-fluorescein (MW 376, Sigma Chemicals) was injected intravenously (2 mg/100 g body wt) (Leakage of this low molecular weight dye is a sensitive marker for BBB disruption). Extravasation of sodium-fluorescein was investigated in vivo using CLSM (Dirnagl et al., 1994). At 10 min after the bolus injection, leakage of sodium-fluorescein into the extravascular space was graded qualitatively as no extravascular fluorescence (grade 0), slight appearance of fluorescence outside the pial vessels along the vessel wall (grade 1), slight level of fluorescence not only along pial vessels but also throughout the brain surface (grade 2), and high fluorescence on brain surface (grade 3).
Because the role of NO in leukocyte-endothelium interaction has been implicated as being closely related to the oxygen free-radical superoxide, in study V, lucigenin-enhanced CL was recorded at baseline and during topical application of
Data analysis
Numbers of rollers or stickers per 100 μm vessel length before and after 30 and 60 min of
RESULTS
Physiological variables
During experiments, all physiological parameters were within normal ranges, except for SAP, which increased significantly after systemic
Physiological variables: systemic arterial pressures (SAP) and arterial gas tensions
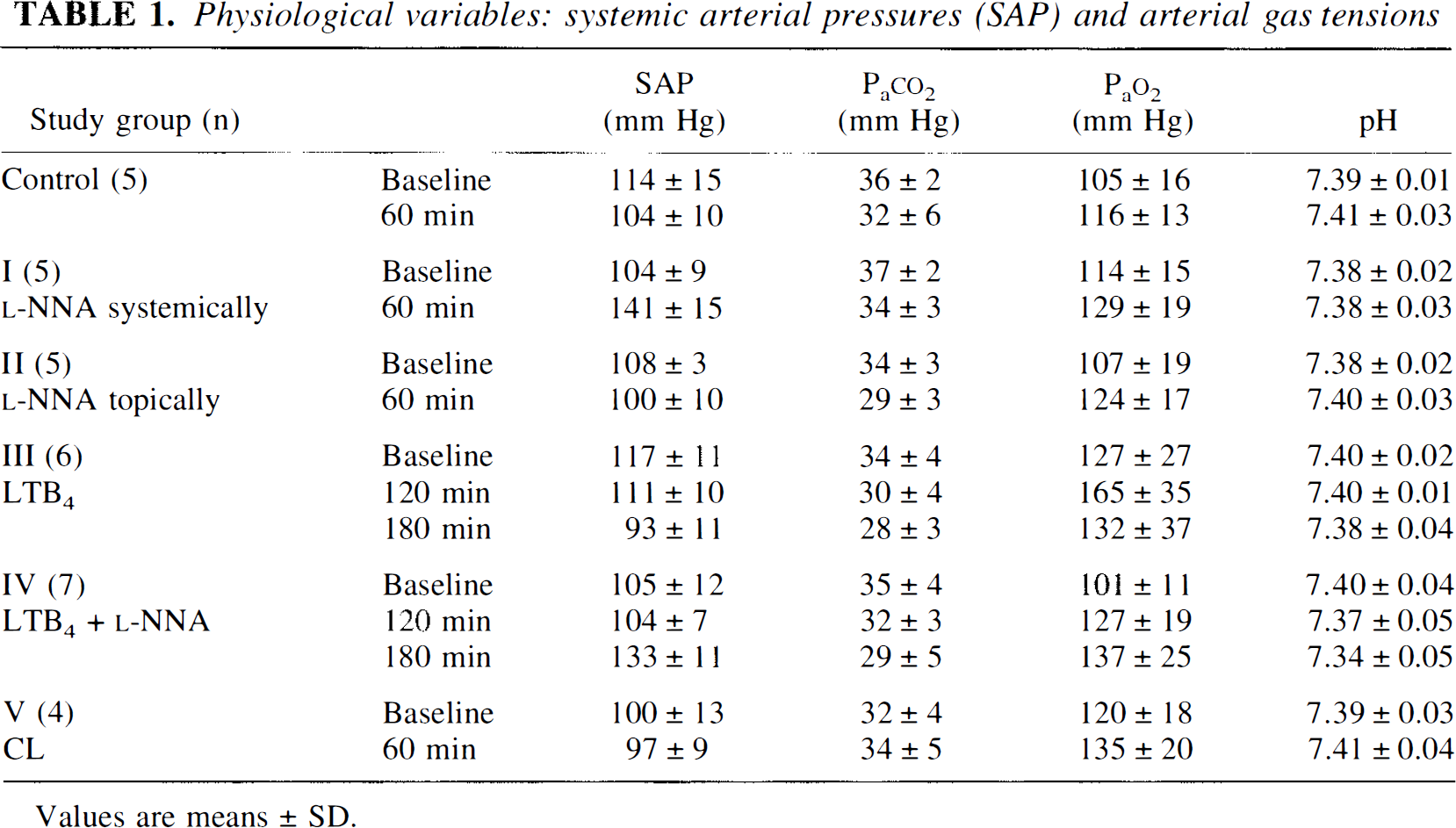
Values are means ± SD.
NOS activity determination
NOS activity in sham-operated animals was assayed and set at 100% as control. Whereas systemic application of the NOS inhibitor
The NOS activity assay as described here does not distinguish between constitutive and inducible NOS. In the ischemic brain, expression of inducible NOS mRNA starts as early as 12 h after onset of ischemia (Iadecola et al., 1995), suggesting that the LTB4-activation period in studies III and IV is too short to cause expression of inducible NOS during the course of the experiment. Therefore, the main NOS activity measured in our experiments is expected to originate from the constitutive NOS in neurons, astrocytes, and endothelial cells in the brain.
CLSM
The mean length of the pial vascular network investigated per site per animal was 277 ± 52 μm in study I (systemical
There was only a very slight and nonsignificant increase in the numbers of rolling or sticking leukocytes during the 60-min observation times in studies I and II (Table 2) and during the combination of systemic and topical
Leukocyte-endothelium interactions in pial veins at seven cortical sites per animal
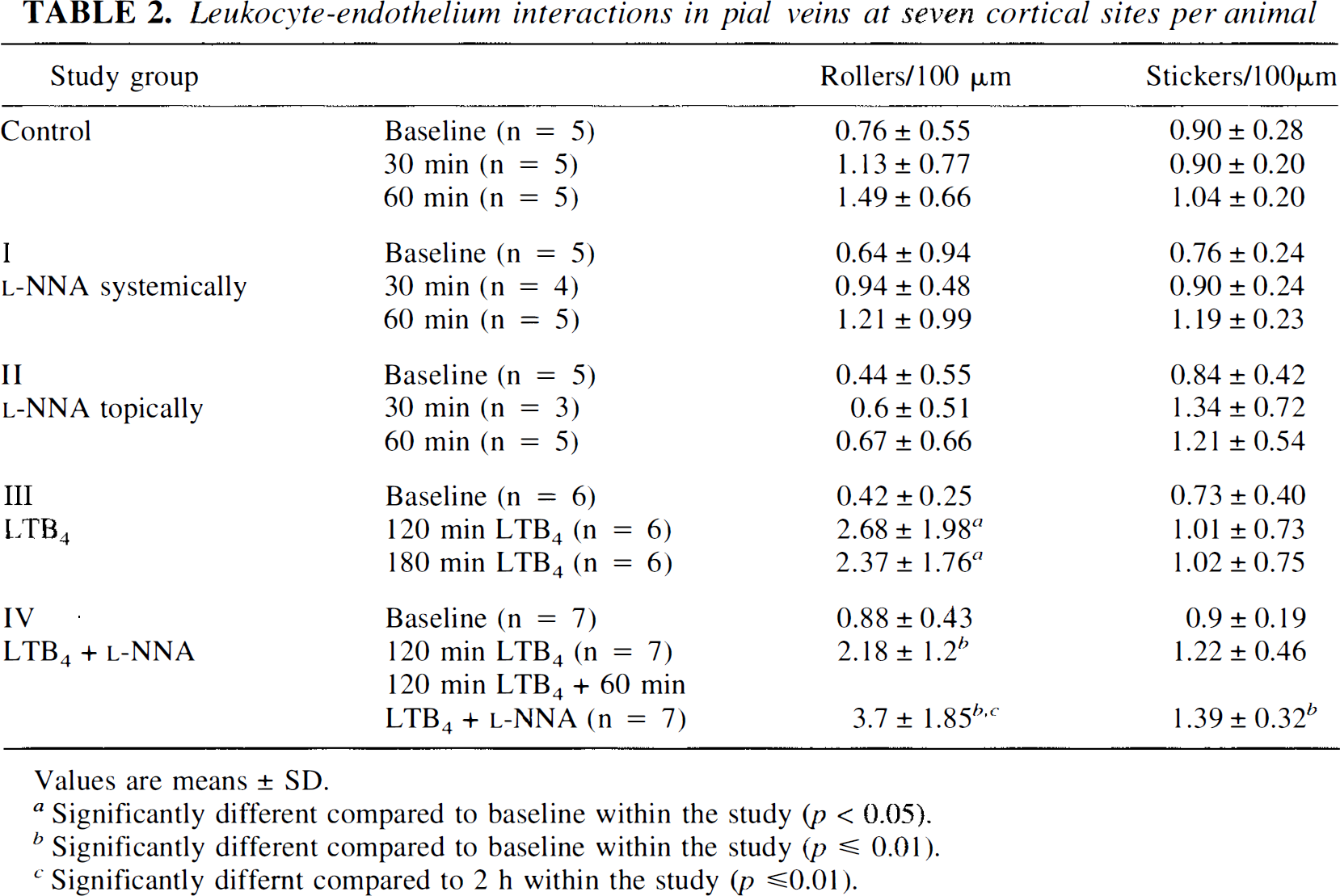
Values are means ± SD.
Significantly different compared to baseline within the study (p < 0.05).
Significantly different compared to baseline within the study (p ≤ 0.01).
Significantly differnt compared to 2 h within the study (p ≤0.01).
In studies III and IV, leukocyte rolling was significantly increased during continuous superfusion of a low concentration of the leukoattractant compound LTB4. In contrast to the physiological state in studies I and II, NOS inhibition due to systemic and topical application of the NOS inhibitor following 2 h of LTB4 activation (study IV) led to a significant increase in the numbers of rolling leukocytes in the pial venous system (Table 2, Figs. 2 and 3) compared to LTB4 administration alone (3 h in study III). Due to high variations in the absolute numbers of increased rolling leukocytes during LTB4 superfusion, there was only a slight, but not significant, difference in the absolute values seen after the third hour of the observation period between studies III and IV. Nevertheless, a significant difference (p ≤ 0.01) exists when comparing the % increase of rolling leukocytes in the third hour (second hour = 100%) between studies III and IV. This discrepancy in the significance between absolute and percentage values can be explained by high variations in the degree of activation of leukocyte-endothelium interaction due to LTB4 application from animal to animal. Regardless of the absolute degree of activation of leukocyte-endothelium interaction in each individual animal, the level reached after the second hour of the observation period did not increase further during the third hour of superfusion of LTB4 alone throughout all experiments in study III.
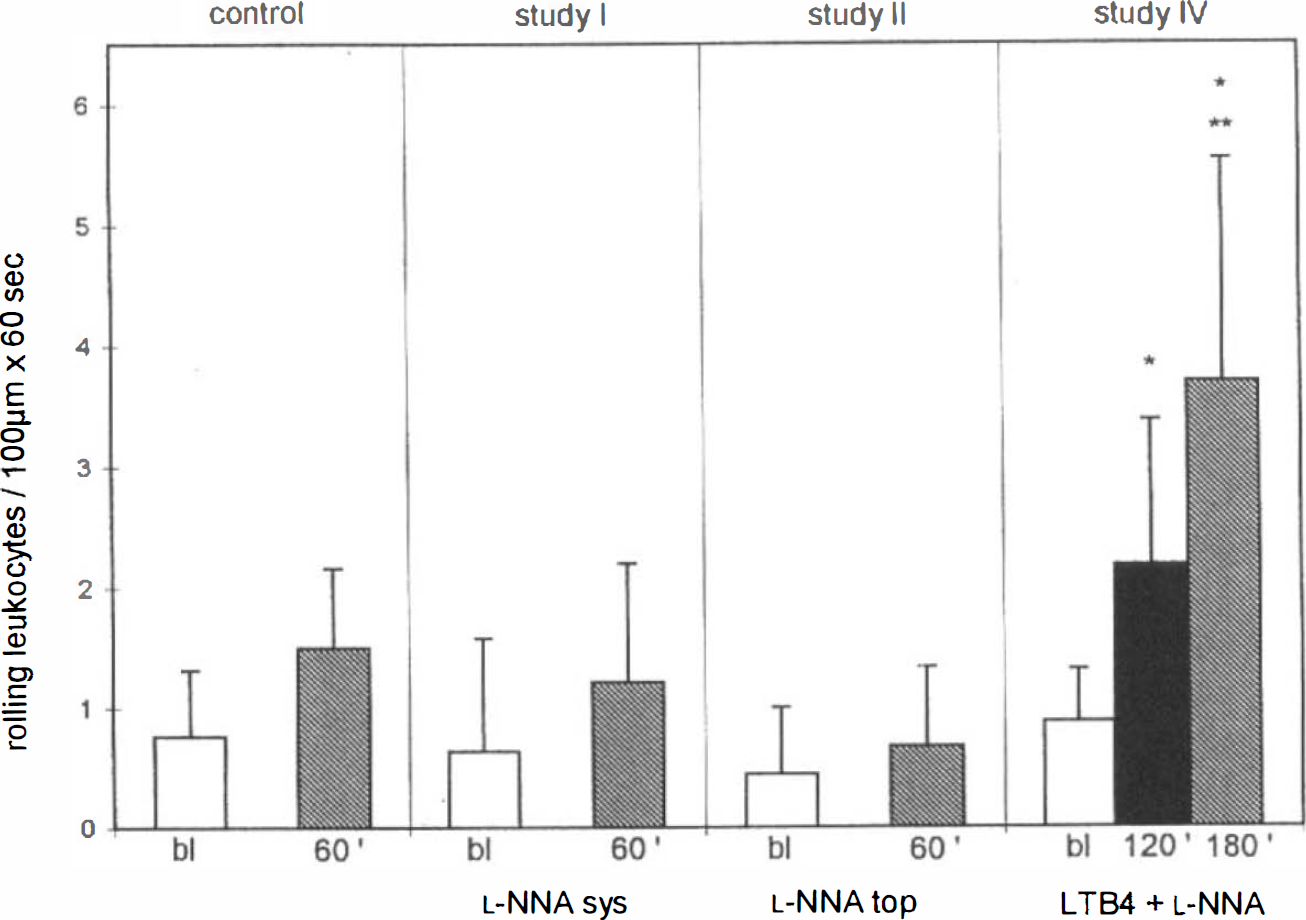
Effect of
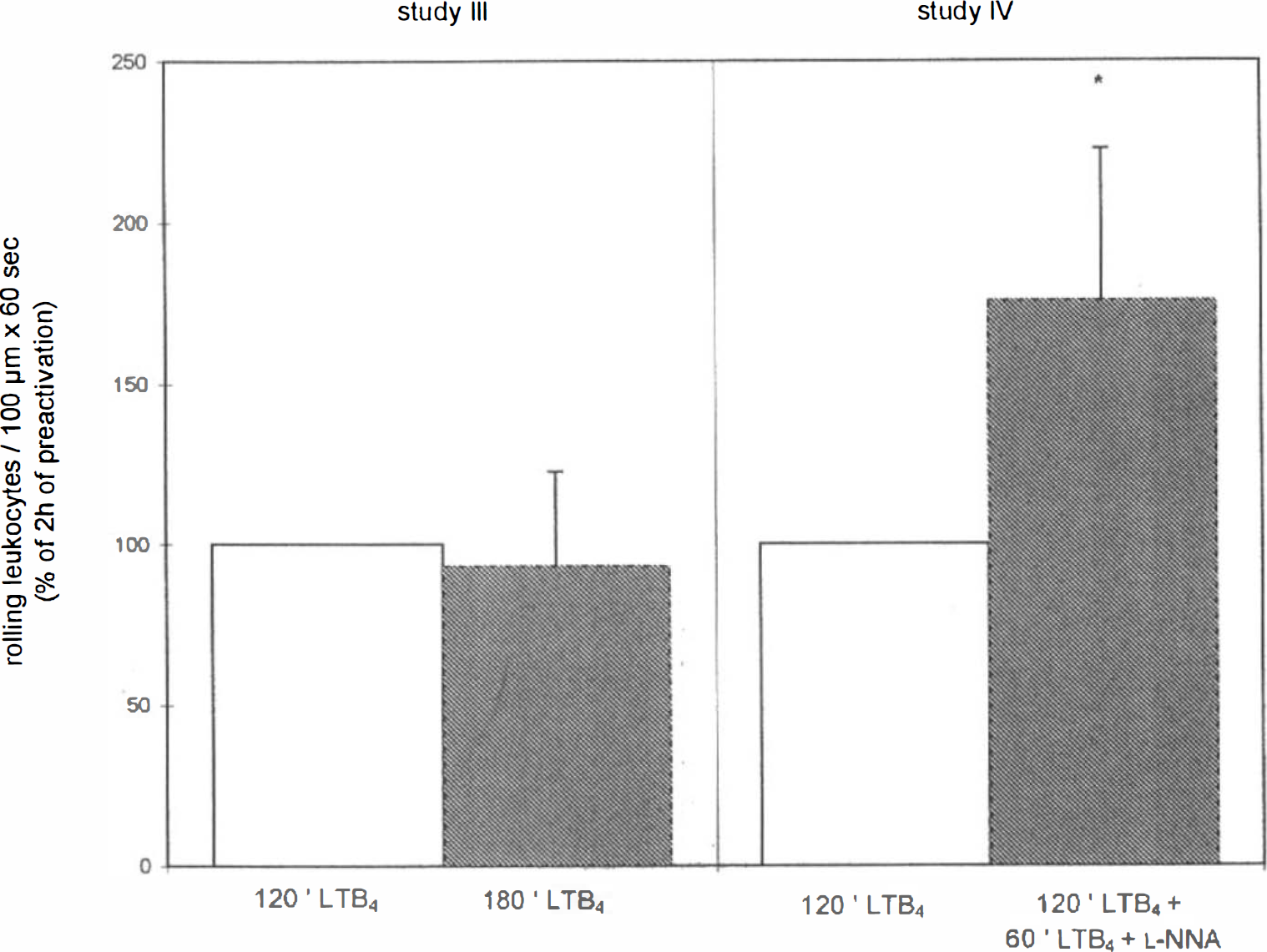
Effect of NOS inhibition on numbers of leukocytes per 100 μm of pial veins rolling along the vessel wall (60 s) under LTB4-activated conditions. Whereas after 120 min of LTB4 superfusion (7.5 × 109– M) a plateau was reached and another 60 min of LTB4 superfusion alone did not further increase the numbers of rolling leukocytes (study III, n = 6), LTB4, +
In previous experiments in our laboratory (Dirnagl et al., 1994), it was shown that, under physiological conditions, there is no change in leukocyte-endothelium interaction during a 240-min observation period (a prolonged control group). This information is provided in addition to the 60-min control group for comparison with studies III and IV.
A typical example of the imaging of leukocytes within a pial vein with CLSM before and after 2 h of LTB4 application is shown in Fig. 4.
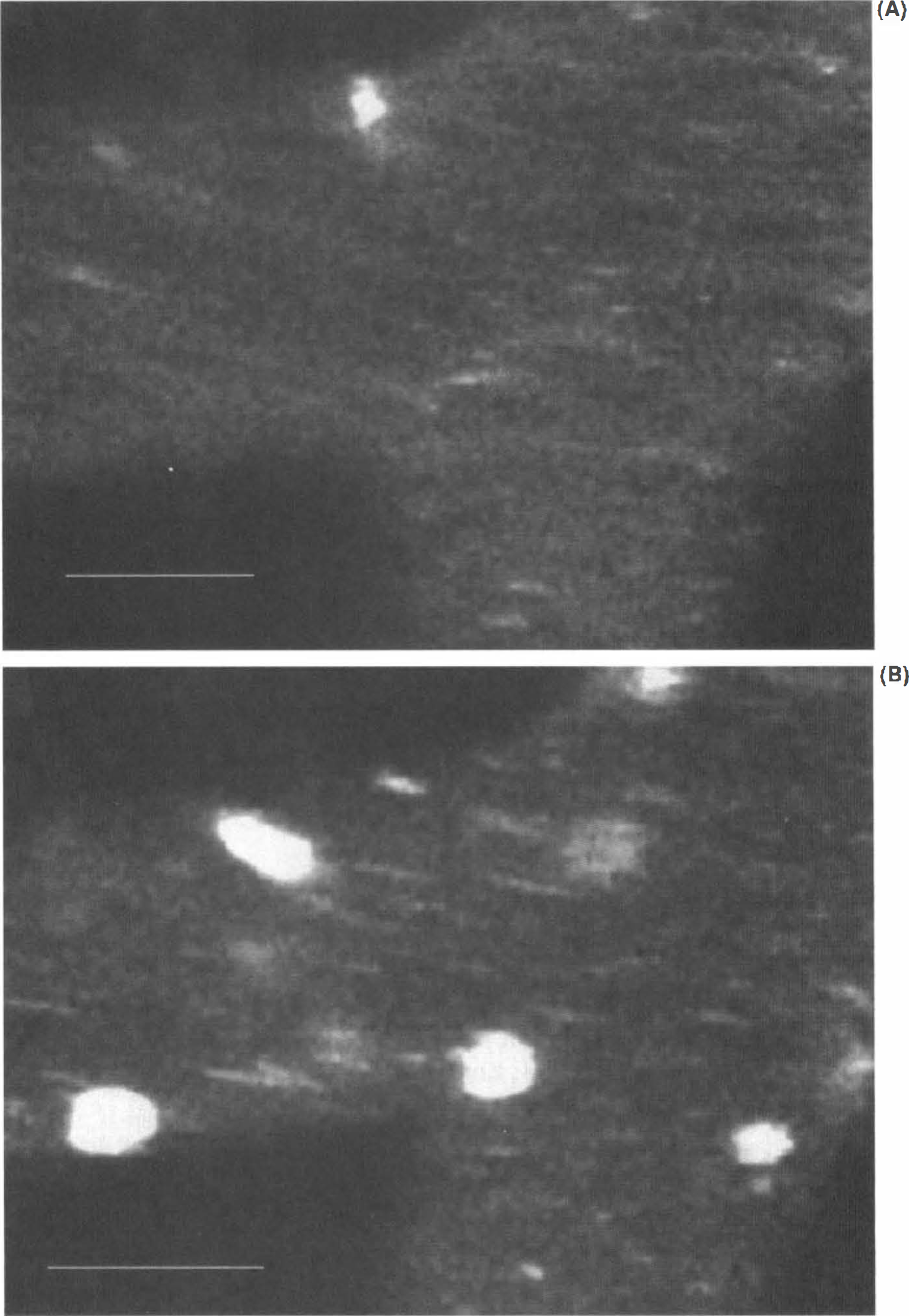
Imaging of rhodamine 6G-labeled leukocytes within a pial vein before and after 2 h of LTB4 superfusion using CLSM.
LTB4 application alone (study III) did not lead to a significant change in the numbers of sticking leukocytes within the pial circulation of the cerebral cortex. LTB4 superfusion followed by
In pial arteries, no significant rolling or sticking of leukocytes was observed at any time point in any experimental group.
NOS inhibition alone did not affect BBB integrity during systemic and/or topical
LTB4 application alone led to only slight leakage of sodium-fluorescein into the extravascular space in all animals of study III (grade 1). This slightly enhanced BBB permeability was significantly increased due to NOS inhibition following LTB4 superfusion (study IV) (grade 2 in all animals).
Lucigenin-enhanced CL
Topical application of the NOS inhibitor
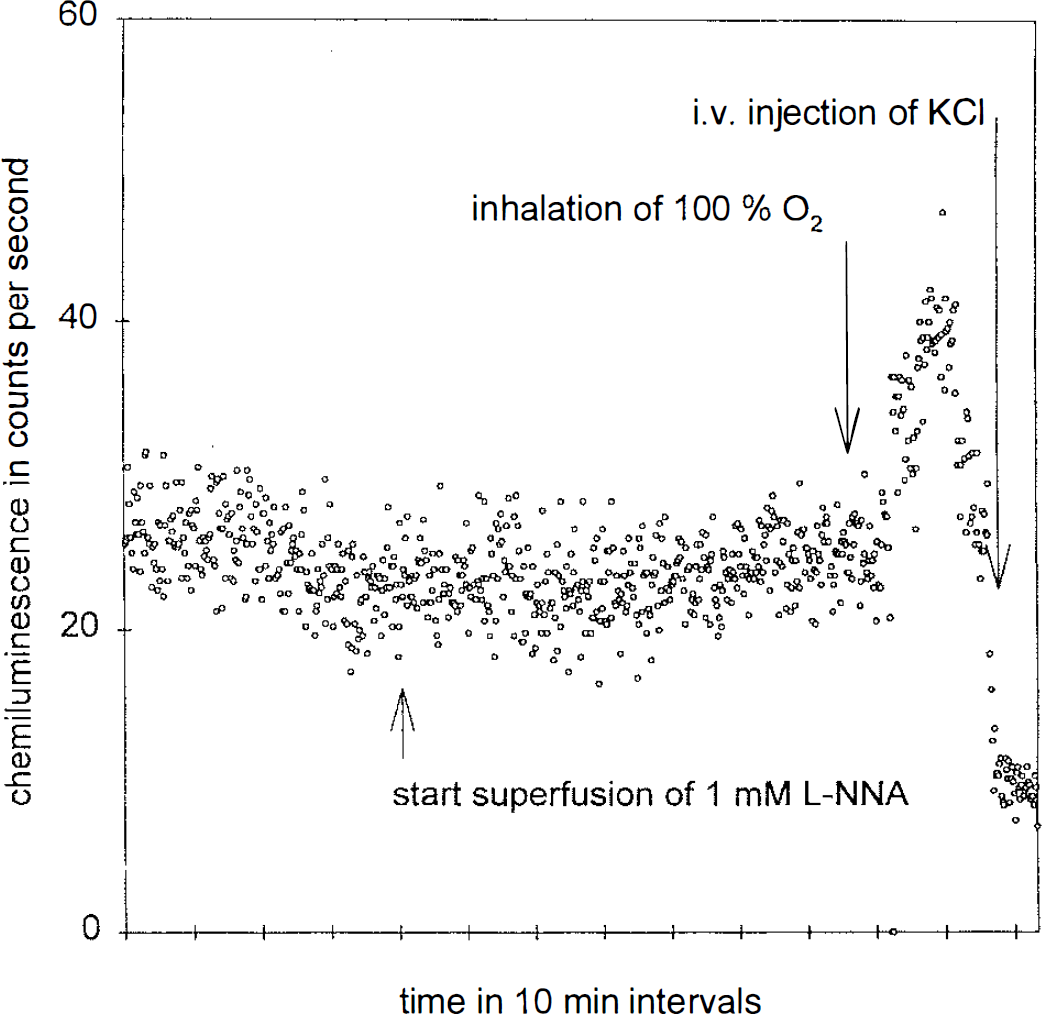
Effect of topical application of
DISCUSSION
In the present study, we found that neither systemic nor topical application of the NOS inhibitor
Effect of NOS inhibition on leukocyte-endothelium interaction and vascular permeability
In several noncerebral tissues (e.g., heart, skeletal muscle, liver), an important role for NO in the regulation of leukocyte-endothelium interaction has been demonstrated (Persson et al., 1990; Ma et al., 1993; Harbrecht et al., 1992). Most of the studies investigating the increase in leukocyte-endothelium interaction during NOS inhibition were performed in intestinal and mesenteric tissue. In the gastrointestinal tract, systemic as well as topical NOS inhibition increased leukocyte-endothelium interaction in postcapillary venules, leading to adhesion and emigration of leukocytes (Kubes et al., 1993; Kubes et al., 1991).
The role of NO in the regulation of vascular permeability is discussed controversely. In some studies, inhibition of NO production induced albumin and horseradish peroxidase leakage in venules of mesenteric and intestinal tissue (Kanwar et al., 1994; Kurose et al., 1993; Kubes and Granger, 1992) as well as cerebral arterioles (Prado et al., 1992). In contrast, other investigators showed that NOS inhibition did not affect basal levels of vascular permeability but reduced augmented permeability in various tissues, including brain (Haberl et al., 1994; Hughes et al., 1990; Mayhan, 1993).
Mechanisms of NO effects on leukocyte-endothelium interaction
The mechanisms by which NO modulates microvascular endothelium function are not sufficiently resolved. Because superoxide anion (O2−) can be inactivated by NO, it has been suggested that NO suppression may enhance O2− levels in the tissue, thus leading to functional changes in microvascular endothelium, such as leukocyte-endothelium interaction and vascular permeability (Gaboury et al., 1993). In addition, mast cell degranulation, which releases proinflammatory agents that cause leukocyte adhesion to endothelium, can be induced by superoxide. These findings suggest that basal levels of NO in vivo may continuously inactivate the proadhesive molecule superoxide.
Besides this scavenging effect of NO on superoxide, other mechanisms of enhanced leukocyte-endothelium interaction are discussed. Recently, it was shown that NO suppression-induced enhancement of endothelial adhesiveness may be mediated by a direct intracellular increase in oxidative stress in arteriolar and venular endothelial cells, and in mast cells in the mesenteric microcirculation, that is followed by venular leukocyte adhesion (Suematsu et al., 1994).
Another mechanism of NO-associated antiinflammatory properties was shown recently (Peng et al., 1995). Those authors found that NO inhibits the activation of the transcription factor NF-κB (which itself, when activated, leads to the expression of proinflammatory mediators) through induction and stabilization of the NF-κB inhibitor IκBα, even under basal conditions in a cell culture system.
NO and leukocyte-endothelium interaction in the brain: comparison to other tissues
In our cranial window preparation studies on the brain cortex, very low baseline levels of rolling and sticking leukocytes were detected and no increase in leukocyte-endothelium interaction occurred due to NOS inhibition. In addition, we found no increase in superoxide production of the cerebral cortex using lucigenin-enhanced CL during topical
Whereas, in the mesentery in the intra-abdominal situs under physiological conditions, neither rolling nor sticking of leukocytes was present, leukocyte-endothelium interaction increased rapidly after exteriorization in experimental studies and remained high over a long period (Fiebig et al., 1991). Therefore, Fiebig et al. suggested that inflammation is present in experimental mesentery preparations, and this results in relatively high levels of rolling or sticking leukocytes at the beginning of the experiments. In addition, Ma et al. (1993) have shown, that NOS inhibition in myocardial ischemia promotes neutrophil adherence only after reperfusion, in which free radical production is enhanced.
To mimic these experimental conditions in other tissues, in which leukocyte-endothelium interaction was increase before NOS inhibition (i.e., by the experimental preparation), we produced a slight activation of leukocyte-endothelium interaction by topical LTB4 application. This induces a weak respiratory burst in neutrophils (Gaboury et al., 1993). In addition, leukocytes have been shown to produce superoxide anion radicals due to LTB4 application in vitro (Maghni et al., 1991; Murohara et al., 1995). In a previous video microscopy study, it was shown that cerebral superfusion of LTB4 induces an increase in leukocyte-endothelium interaction in the same range as in our study, without an increase in BBB permeability (Schürer et al., 1994).
Under these LTB4-activated conditions, we found a slight but significant increase of leukocyte-endothelium interaction and BBB permeability after NOS inhibition, findings that are in accordance with those of other studies in noncerebral tissues.
Besides these methodological issues, other tissue-specific mechanisms may also be involved in the differences in the role of NO in leukocyte-endothelium interaction and vascular permeability in the brain compared to noncerebral tissues: (a) smaller lymphocyte binding capacities of cerebral endothelial cells compared to endothelial cells of other tissues (Male et al., 1990); (b) tight junctions of high electrical resistance between cerebral endothelial cells, presenting an effective barrier even to ions. Peripheral endothelial cells are either fenestrated or have tight junctions of low resistance between the cells (Rowland et al., 1991); (c) low mast cell content of the cerebral cortex; mast cells in the brain are found particularly in the leptomeninges, thalamus, and hypothalamus (Theoharides, 1990). In the brain, stationary interstitial immunocytes are mainly represented by microglial cells. During activation of interstitial immunocytes, proinflammatory agents such as superoxide anion are released. In the normal brain, microglial cells are not activated, and lack phagocytic and secretory activities (Giulian, 1992).
In conclusion, our data support a modulatory role for NO in leukocyte-endothelium interaction and BBB integrity in the pial microcirculation of the brain when this interaction is increased. This may be important for pathophysiological states of the brain, e.g., cerebral ischemia, during which induction of leukocyte-endothelium interaction occurs (Garcia et al., 1994) and NO levels in the brain tissue increase (Malinski et al., 1993). However, our data imply that basal NO production is not involved in modulating leukocyte-endothelium interaction. This latter finding is in contrast to results of a number of studies of the role of NO in leukocyte-endothelium interaction in noncerebral tissues. It remains open whether this discrepancy is due to methodological factors, particularly the degree of activation of leukocytes at the beginning of NOS inhibition, or to differences in the endothelium and/or surrounding tissues, resulting in different levels of superoxide in the resting state.
Footnotes
Abbreviations used
Acknowledgment:
This study was supported by the Deutsche Forschungsgemeinschaft (Di 454/4-2, Di 454/8-1. DFG-SFB 507) and the Wilhelm Sander Stiftung.
