Abstract
The selectin family of glycoproteins facilitates the early phase of polymorphonuclear leukocyte adhesion to the endothelial cell and, thus, may promote ischemic cell damage. To evaluate E-selectin in the pathogenesis of focal cerebral ischemia and reperfusion injury, we cloned rat E-selectin cDNA and measured the temporal profiles E-selectin mRNA (Northern blot) and protein (immunohistochemistry) during (1 h of ischemia) and after (up to 1 week) transient (2 h) middle cerebral artery (MCA) occlusion in the male Wistar rat. We also tested the effect on these rats of administration of CY-1503, an analog of sialyl Lewisx (SLex), on ischemia cell damage. mRNA for E-selectin was first detected in the ischemic hemisphere at 2 h of reperfusion and persisted to 46 h of reperfusion. E-selectin (protein) was localized to microvessels within the ischemic lesion at 0 h of reperfusion and persisted to 70 h of reperfusion. Treatment of the ischemic animals with CY-1503 (50 mg/kg) (n = 8) significantly reduced infarct volume by 42% (p < 0.05) and significantly reduced myeloperoxidase immunoreactive cells in the ischemic lesion by 60% (p < 0.05). These findings provide the first direct evidence for the involvement of E-selectin in transient MCA occlusion in rats and suggest that the E-selectin may facilitate neutrophil adhesion and subsequent cerebral ischemic cell damage.
Leukocytes contribute to cerebral ischemic cell damage (Hallenbeck et al., 1986; del Zoppo et al., 1991; Garcia et al., 1994; Zhang RL et al., 1994a). Accumulation of leukocytes in an ischemic lesion is a dynamic process that is sequentially regulated by selectin, integrin and immunoglobulin families of related adhesion molecules (Patarroyo et al., 1990; Butcher, 1991; Bevilacqua, 1993). The initial leukocyte endothelial low-affinity interaction promoting leukocyte rolling is mediated by the selectin family, including L-selectin, P-selectin, and E-selectin. Subsequently, a high-affinity interaction between activated leukocytes and the endothelium that promotes leukocyte endothelial adhesion occurs. This high-affinity interaction is primarily mediated by β2 integrins (CD1 1a/CD 18, CD1 1b/CD18) and their ligand, the endothelial intercellular adhesion molecule-1, ICAM-1 (Patarroyo et al., 1990; Butcher, 1991; Bevilacqua, 1993). P-selectin and ICAM-1 are upregulated in the primate (Okada et al, 1994) and rat (Wang et al., 1994; Zhang RL et al., 1995b) after middle cerebral artery (MCA) occlusion.
Depletion of neutrophils and inhibition of neutrophil endothelial adhesion reduce infarct volume in animal models of focal cerebral ischemia (Chen et al., 1992a; Chopp et al., 1994; Matsuo et al., 1994; Zhang RL et al, 1994b; Zhang ZG et al., 1995). Administration of monoclonal antibodies (mAb) to CD 18, CD 11b and ICAM-1 after MCA occlusion in the primate improves perfusion of cerebral microvessels (Mori et al., 1992) and significantly reduces cerebral infarct volume concomitantly with a reduction of myeloperoxidase activity after MCA occlusion in the rat (Bevilacqua, 1993; Okada et al, 1994; Zhang RL et al., 1995b). Induction of E-selectin mRNA has recently been reported in permanent MCA occlusion in the rat (Wang et al., 1995). However, little is known concerning the functional significance of E-selectin in transient focal cerebral ischemia. E-selectin as a single chain glycoprotein expressed on endothelial cells (Bevilacqua et al., 1989). E-selectin binds to a myeloid-specific cell surface carbohydrate, sialyl Lewisx (SLex) (Phillips et al, 1990; Lasky, 1992). Administration of SLex analogs attenuate neutrophil accumulation and myocardial (Lefer et al., 1994) and lung (Seekamp et al., 1994) injury after ischemia and reperfusion. In the present study, we cloned the rat E-selectin cDNA and demonstrated that transient (2 h) MCA occlusion in the rat induces E-selectin mRNA and E-selectin expression within ischemic tissue and that administration of a novel SLex analog reduces ischemic cell damage.
METHODS
Animal model
Male Wistar rats weighing 270–310 g (n = 123) were used in this study. Transient MCA occlusion was induced as previously described (Zea Longa et al., 1989; Chen et al., 1992b). Briefly, rats were anesthetized with halothane (1–3.5% in a mixture of 70% N2O and 30% O2, vol/vol) using a face mask. The right common carotid artery (CCA), external carotid artery (ECA), and internal carotid artery (ICA) were isolated via a ventral midline incision. A 4–0 nylon monofilament, with its tip rounded by heating near a flame, was introduced into the ECA lumen through a puncture of the ECA and advanced into the ICA to block the origin of the MCA. Restoration of MCA blood flow in animals subjected to 2 h of MCA occlusion was achieved by withdrawing the suture to the stump of the ECA. Rectal temperature was maintained at 37°C throughout the surgical procedure using a feedback regulated water heating system.
Immunohistochemistry for E-selectin expression
The rats were randomly assigned to the following groups: experimental (n = 42), in which rats were subjected to 2 h of MCA occlusion and killed at time points spanning the period from 0 to 166 h after onset of reperfusion; control (n = 4), in which animals were not subjected to any surgical procedure; and sham (n = 4), in which animals were subjected to the same surgical procedures as those in the experimental group; however, the intravascular monofilament that occludes the MCA was withdrawn within 1 min, and the same group of animals were killed at 24 h.
Rats were anesthetized with intraperitoneal administration of ketamine (44 mg/kg) and xylazine (13 mg/kg) and transcardially perfused with heparinized 0.9% sodium chloride. The brains were rapidly removed and frozen in 2-methylbutane (Fisher Scientific, Itasca, IL, U.S.A.) cooled on dry ice. Coronal brain sections (8 μm thick) were cut on a cryostat and thaw-mounted onto gelatin-coated slides. Monoclonal mouse anti-human E-selectin antibody (clone ENA 1) (Caltag Laboratories, San Francisco, CA, U.S.A.) was used for immunohistochemistry. Immunohistochemical staining for the E-selectin antibody was performed according to the manufacturer's instructions. Tissue was fixed for 10 min in pure acetone, and incubated for 10 min in chloroform, and then incubated with an E-selectin antibody at 1:20 dilution in 0.5% bovine serum albumin for 1 h at room temperature. Biotinylated rat-absorbed anti-mouse IgG (Vector, Burlingame, CA, U.S.A.) was used as a secondary antibody. Antibody binding was detected using an Elite ABC Kit (Vector) and with 3.3 diaminobenzidine-H2O2 as the chromogen. Slides were counterstained with Mayer's hematoxylin to identify cell nuclei. Nonimmune mouse IgG1 (Becton Dickinson, Mountain View, CA, U.S.A.), used at the same concentration as the primary antibodies, served as the negative control. A coronal section located at interaural 8.2 mm, bregma 0.8 mm was selected from each animal (Chen et al., 1992b) for evaluation of E-selectin expression, and an adjacent section was stained with hematoxylin and eosin for histopathological evaluation.
The numbers of E-selectin immunoreactive microvessels were counted throughout the ipsilateral hemisphere using a light microscope at 400× magnification.
Double immunostaining with antibodies to E-selectin and von Willebrand factor (Factor VIII)—a polyclonal anti-human von Willebrand antibody developed in rabbit (code A 082; Dako, Carpineria, CA, U.S.A.)—was performed. Sections were incubated with the polyclonal anti-human von Willebrand antibody (1:100) for 1 h at room temperature after E-selectin antibody binding was detected with 3,3 diaminobenzidine-H2O2 as the chromogen. Fluorescein isothiocyanate (FITC) conjugated anti-rabbit immunoglobulin antibody was used as secondary antibody. Sections were observed with fluorescent microscopy for detection of FITC.
cDNA
The cDNA encoding rat E-selectin was isolated by homology polymerase chain reaction (PCR) cDNA library screening, using a homology-based strategy similar to that described previously (Manning et al., 1995). Briefly, gene-specific oligonucleotides were designed based upon an alignment of reported cDNA sequences for human (Bevilacqua et al., 1989), mouse (Becker-Andre et al., 1992), and rabbit (Larigan et al., 1992) E-selectin. Using oligo-dT-primed first strand cDNA synthesized from total RNA isolated from the lungs of endotoxin-treated rats as template and the oligonucleotides 5′-TTACTACTGGATTGGAATCAG-3′ and 5′-TGTTTCTGAT-TGTTTTGAACTTA-3′, a specific product of 1.6 Kb in length was generated by PCR (Sambrook et al., 1989). Molecular cloning and sequence analysis confirmed that this cDNA fragment encompassed sequences encoding the (EGF)-like and complementary regulatory-like repeats, and the transmembrane and cytoplasmic domains of rat E-selectin. An additional PCR fragment of 0.75 Kb, encoding the Signal sequence, and lectin and EGF domains, was generated using the oligonucleotides 5′-TGAAAACAACTGCTGGAGTCATG-3′ and 5′-TCAACCACATGGCAGGCTGGA-3′. This fragment was subsequently used to screen an endotoxin-treated rat lung cDNA library prepared in the ΛZAPII vector (Stratagene, La Jolla, CA, U.S.A.). From the cDNA clones isolated, the complete cDNA sequence for rat E-selectin was determined by dideoxynucleotide sequencing (Sambrook et al., 1989). The rat E-selectin cDNA sequence has been deposited with the Gen-EMBL database under the accession no. L25527. For Northern blot analysis, the 1.6 Kb PCR product was nick-translated and radiolabeled with 32P-dCTP (Sambrook et al., 1989).
Generation of stably-transfected Chinese hamster ovary (CHO) cells expressing rat E-selectin
The full-length rat E-selectin cDNA (RESEC) was sub-cloned into the eukaryotic expression vector pcDNA1 neo (Invitrogen, San Diego, CA, U.S.A.). To confirm the ability of this construct to direct rat E-selectin cell surface expression, COS-7 cells were transiently transfected with 10 μg of RESEC construct using the DEAE-dextran method (Sambrook et al., 1989). After 72 h, cells were harvested by scraping and cell surface expression of E-selectin evaluated by flow cytometry using a rabbit anti-human E-selectin polyclonal antibody (a kind gift of Dr. J. G. Geng, Upjohn Labs) and FITC-labeled sheep anti-rabbit IgG (Sigma Co., St. Louis, MO, U.S.A.). Analysis of antibody binding was performed using a FACScan flow cytometer (Bectin-Dickinson). COS-7 cells transfected with vector alone or with the full-length human E-selectin cDNA were included as controls. For the generation of a cell line stably expressing rat E-selectin, CHO cells were transfected with 10 μg of the RESEC construct by the calcium phosphate method (Sambrook et al., 1989). Following transfection, cells were grown in DMEM high glucose medium in the presence of 800 μg/ml G418. Single G418 resistant colonies were isolated using sterile glass pipettes, trypsinized, and serially cloned. E-selectin surface expression was examined by flow cytometry as described above.
HL-60 cell adhesion assay
Stably-transfected CHO cell lines expressing either rat or human E-selectin, or nontransfected CHO cells were plated into 35-mm Petri dishes and grown to confluence. HL-60 cells were washed twice in ice-cold (RPMI)-1640 medium, resuspended at 3.33 × 106 cells/ml, and added to the assay plates (0.6 ml), which had been washed twice with RPMI-1640. Plates were rocked from side to side for 20 min at 4°C and washed five times with RPMI-1640, after which the number of adherent HL-60 cells were counted on 10–12 independent confluent fields. The requirement for Ca2+ was evaluated by including 2.5 mM (EGTA) in the assay media.
Northern blot analysis of E-selectin expression
Rats were randomly assigned to the following groups: experimental (n = 36), in which rats were subjected to MCA occlusion and reperfusion, and were killed at time points spanning the period from 1 h of occlusion to 166 h after onset of MCA occlusion and reperfusion; control (n = 3), in which the animals were not subjected to any surgical procedure; and sham (n = 3), in which animals were subjected to the same surgical procedures as in the experimental group; however, the intravascular monofilament that occludes the MCA was withdrawn within 1 min. Total RNA was extracted from the ipsilateral and contralateral hemispheres by the acid guanidine thiocyanate-phenol-chloroform method (Chomczynski and Sacchi, 1987). The level of E-selectin mRNA was determined by Northern blot analysis using a nick-translated 32P-labeled cDNA rat E-selectin. Total RNA (20 μg) was electrophoresed in 1% agarose gels containing formaldehyde, and then transferred by capillary action to Nytran membranes and left overnight. Blots were prehybridized for 5 h at 42°C in 50% formamide, 1% SDS, 5× SSC, 1× Denhardt's solution, 0.25 mg/ml denatured herring ptoses DNA, and 50 mM sodium phosphate buffer, pH 6.5. Hybridization was carried out at 42°C for 12–18 h with 107 cpm of denatured probe. Using a solution 0.1% SDS/–2× SSC, filters were washed for 30 min at room temperature followed by a wash at 65°C in 0.1% SDS/–0.2× SSC. Blots were then exposed to XAR-5 x-ray film with Du Pont (Du Pont, Wilmington, DE, U.S.A.) Cronex Lightening Plus intensifying screens at −70°C. Expression of α-tubulin was used as an internal control to quantify total mRNA.
Treatment with SLex
Rats were subjected to 2 h of MCA occlusion and 46 h of reperfusion. The right femoral artery and vein were cannulated for measuring blood gases (pH, Po2, Pco2) before ischemia and for drug administration, respectively. CY-1503, an oligosaccharide analog of SLex, which attenuates neutrophil accumulation and myocardial necrosis (Lefer et al., 1994), was employed to investigate the potential benefits of this compound on focal cerebral ischemia. Sialyl-N-acetyllactosamine (SLN), a nonfucosylated form of SLex, which has no blocking activity for selectin-dependent adhesion pathways for neutrophils, was used as a control. CY-1503 and SLN were synthesized (Mulligan et al., 1992) by Cytel Corporation (San Diego, CA, U.S.A.). Rats were randomly assigned to the following three experimental groups: (a) SLex analog CY-1503 (25 mg/kg): group A: rats (n = 6) received CY-1503 immediately after withdrawal of the suture (0 h of reperfusion) via i.v. administration and at 1, 2, and 3 h of reperfusion via i.p. administration; group B: rats (n = 7) received CY-1503 at the same time points and via the same route as animals in group A; however, four additional doses were given at 4, 5, 6, and 22 h of reperfusion; (b) SLex analog CY-1503 (50 mg/kg): rats (n = 9) received CY-1503 immediately after withdrawal of the suture (i.v.) and at 1, 2, 3, 4, 5, 6, and 22 h of reperfusion (i.p.); (c) control: rats received either a SLN (50 mg/kg) (n = 4) or an equal volume of 0.9% normal saline (n = 5) immediately after withdrawal of the suture (i.v.) and at 1, 2, 3, 4, 5, 6, and 22 h of reperfusion (i.p.).
Rats were anesthetized via i.m. administration of ketamine (44 mg/kg) and xylazine (13 mg/kg) at 46 h after reperfusion. After transcardial perfusion with heparinized saline, brains were removed. Using a rat brain matrix, each brain was cut into 2-mm thick coronal blocks, for a total of seven blocks per animal. Brain sections were incubated in isotonic phosphate-buffered saline, pH 7.4, containing 2% triphenyltetrazolium chloride (TTC) (Sigma Chemical) at room temperature for 30 min and then stored in 10% neutral buffered formalin. Brain sections were photographed after 3–5 h fixation, and brain volume was measured using a Global Lab Image analysis program (Data Translation, Malboro, MA, U.S.A.). For each coronal section, the area of infarct and the ipsilateral and contralateral hemispheric areas (mm2) were calculated by tracing the right and left hemispheres and the ischemic lesion on the computer screen. Infarct volume (mm3) was determined by integrating the appropriate area with the section interval thickness (Paxinos and Watson, 1986). The indirect method for calculating infarct volume, in which the intact area of the ipsilateral hemisphere is subtracted from the area of the contralateral hemisphere, was used (Jiang et al., 1994). Infarct volume is presented as the volume percentage of infarct to the contralateral hemisphere.
Brain myeloperoxidase
Inflammatory cells, primarily neutrophils, within the brain were quantified immunohistochemically using an antibody against myeloperoxidase (MPO) (Zhang RL et al., 1995a) in rats treated with CY-1503, SLN, and saline. A coronal section located within the center of the ischemic lesion at the level of the anterior commissure (coordinates: interaural 8.2 mm, bregma 0.8 mm; Zhang et al., 1995a) was selected from each animal. Tissue was processed and embedded, and a 2 μm-thick paraffin section was cut from the block and stained with a polyclonal rabbit anti-human MPO antibody for evaluation of MPO expression on inflammatory cells.
From each animal, immunoreactive MPO cells within the ipsilateral hemisphere were counted in a 2 μm-thick section in 25–35 nonoverlapping fields (200×) under the light microscope. Correlation between the area of the ischemic lesion and numbers of immunoreactive MPO cells, as well as numbers of immunoreactive MPO cells per unit area of the ischemic lesion, were calculated from data obtained for the same coronal tissue section.
Statistics
Analysis of variance (ANOVA) was performed to compare means across the groups and to determine differences between groups, Bonferroni corrections for multiple testing were performed. All data are presented as means ± SD. All measurements were performed blindly.
RESULTS
Isolation and characterization of the E-selectin gene
The rat E-selectin gene was cloned by a combination of PCR and cDNA library screening. The deduced rat E-selectin sequence had a primary structure similar to that of E-selectin from other species, including a hydrophobic signal sequence, a C-type lectin domain, five complement regulatory (CR) repeats, a single transmembrane domain, and a short cytoplasmic domain (Fig. 1).
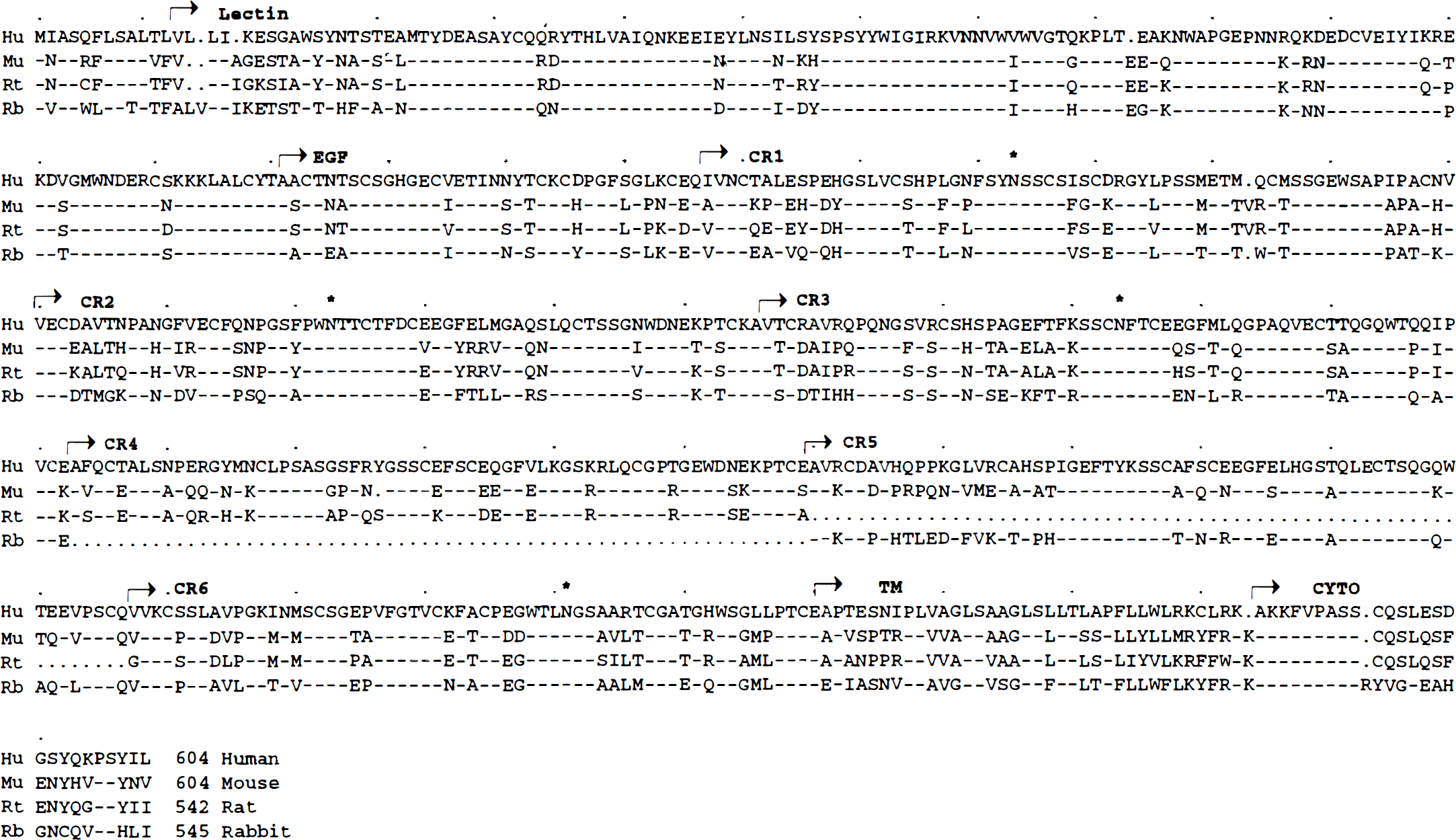
Alignment of E-selectin amino acid sequences of human, mouse, rat and rabbit E-selectins. Human E-selectin (top) is designated by the single letter amino acid code. Dashes (–) represent identical amino acid and dots (·) represent gaps introduced to maximize the alignment; amino acid differences are indicated. The boundaries of the lectin, EGF, complement regulatory repeat (CR), transmembrane (TM), and cytoplasmic (CYTO) domains are marked by arrows. Conserved sites for N-linked glycosylation are also marked (*).
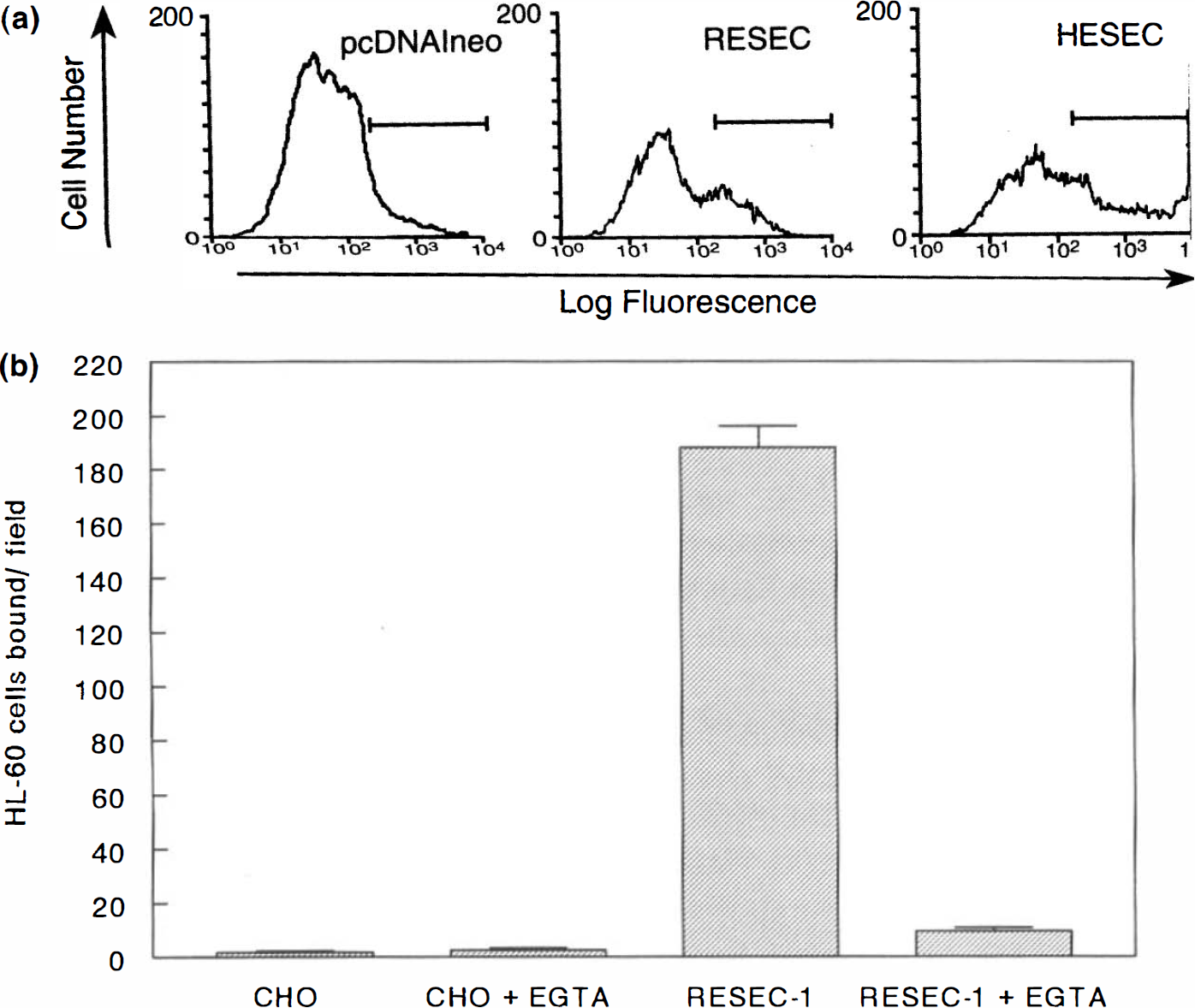
To confirm that rat E-selectin is functionally homologous to human E-selectin, we measured its ability to bind HL-60 cells, a human myelogenous leukemia cell line that displays the E-selectin ligand (Zhou et al., 1991). A eukaryotic expression construct containing full-length rat E-selectin cDNA was generated, and the ability of the construct to direct the cell surface expression of rat E-selectin was confirmed by flow cytometry with an E-selectin polyclonal antibody (Fig. 2A). COS-7 cells transfected with vector alone displayed only background binding of the antibody [3% positive cells with mean channel fluorescence (MCF) of 409], whereas COS-7 cells transfected with the same vector containing the complete rat E-selectin cDNA demonstrated enhanced binding of the E-selectin antibody (30% positive, MCF 840). COS-7 cells transfected with human E-selectin were strongly positive for antibody binding (40%, MCF 3,428). A CHO cell line stably transfected with the RESEC construct bound significant numbers of HL-60 cells (Fig. 2B), whereas nontransfected CHO cells did not. Addition of 2.5 mM EFTA to the incubation abolished binding activity of the rat E-selectin, consistent with the fact the E-selectin is a member of the family of Ca2+-dependent lectins (Drickamer, 1988).
Induction of E-selectin mRNA in ischemic brain
Figure 3 shows the Northern hybridization of E-selectin mRNA expression in the ipsilateral hemisphere in rats at various reperfusion times after onset of MCA occlusion. E-selectin mRNA was not detected in the contralateral hemisphere (data not shown) in control or sham-operated animals. E-selectin mRNA was first detected in the ipsilateral hemisphere at 2 h of reperfusion (Fig. 3, lane 4) and was markedly elevated at 10 h of reperfusion (Fig. 3, lane 6); thereafter, expression of E-selectin mRNA decreased at 22 and 46 h of reperfusion (Fig. 3, lanes 7,8). E-selectin mRNA was no longer detectable after 46 h of reperfusion.
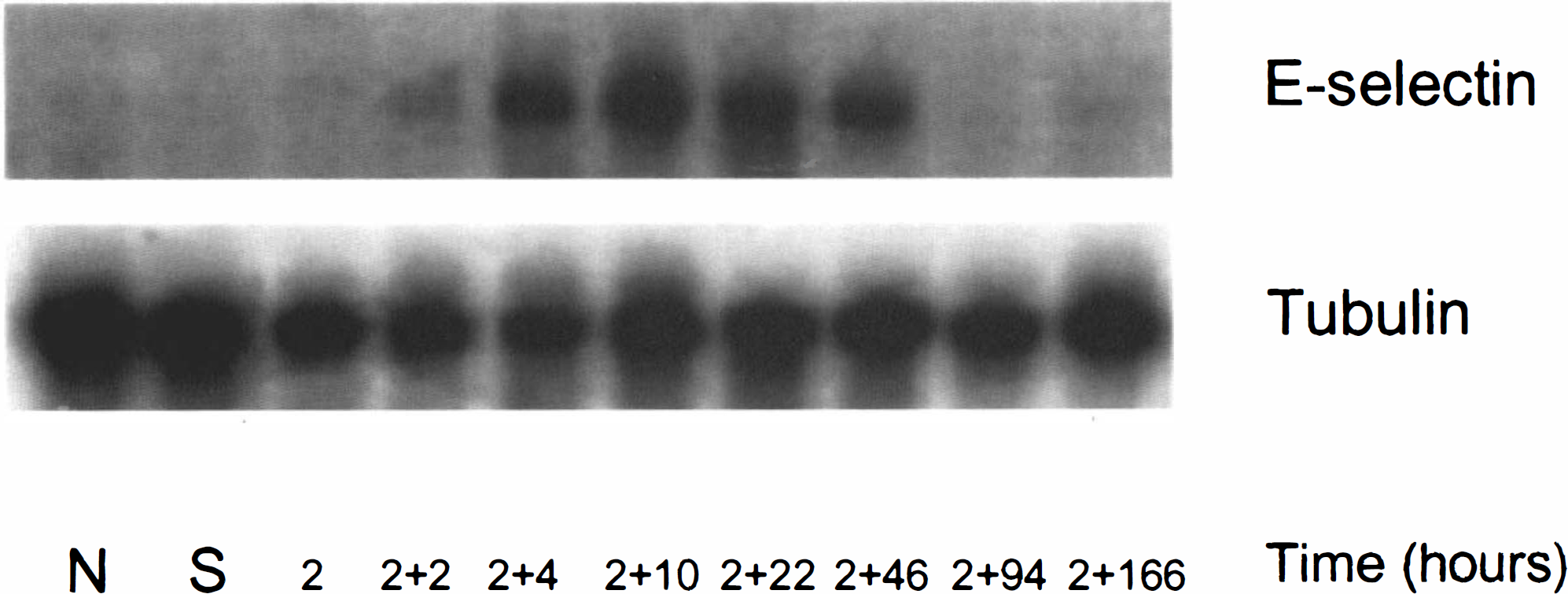
Northern blots for E-selectin mRNA in control, sham, or ischemic brain at various time points of reperfusion. E-selectin mRNA was first detected at 2 h of MCA occlusion and 2 h of reperfusion. Maximal expression was found at 10 h of reperfusion. E-selectin was not detected after 46 h of reperfusion. α-Tubulin was used as an internal control. (N, control; S, sham).
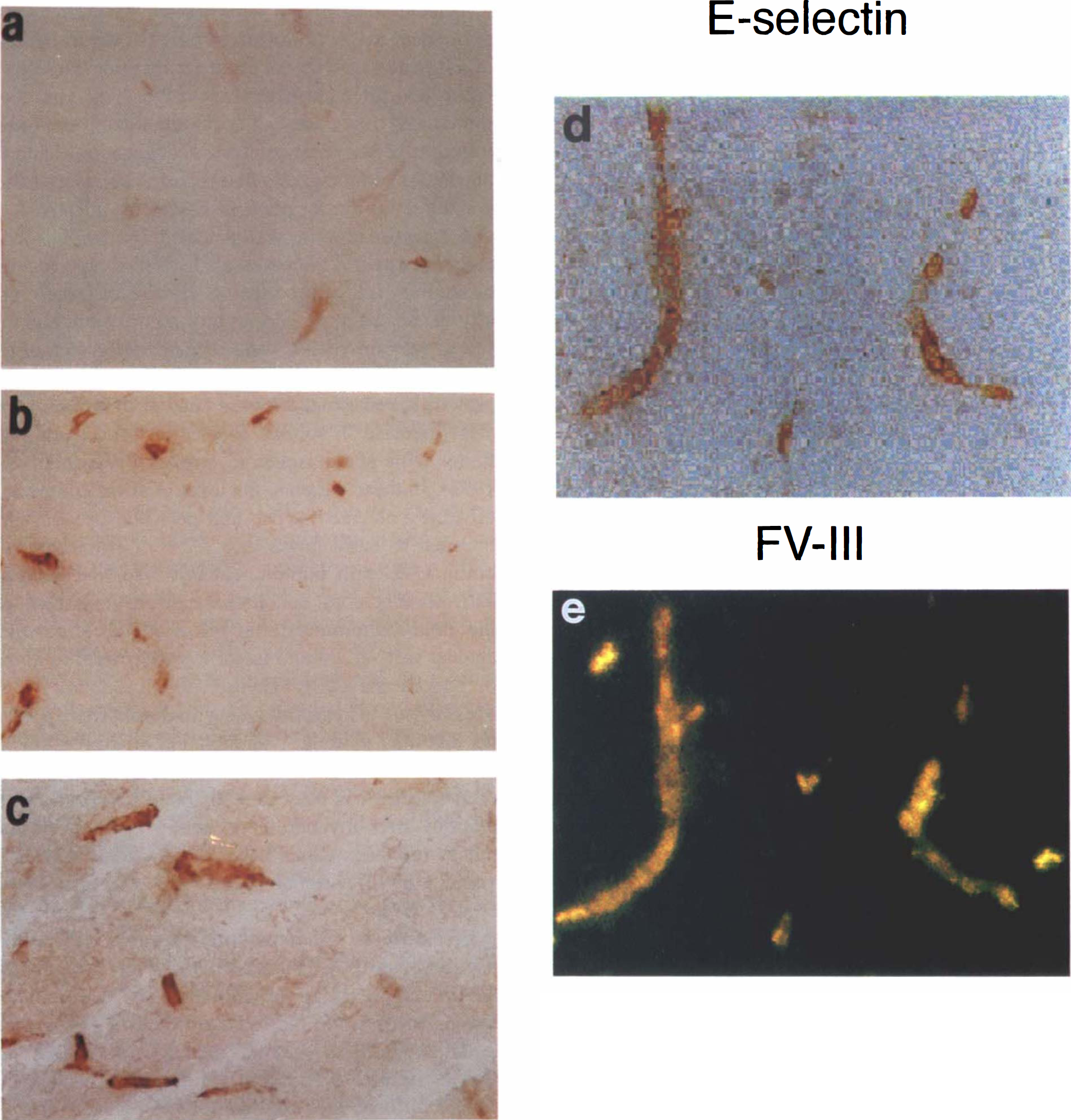
Expression of E-selectin protein in ischemic rat brain. Cryostat coronal sections of brain were immunostained using mAb ENA1 and polyclonal anti-von Willebrand antibody as described in Methods. The immunoreaction of E-selectin was restricted on microvessels within lesion of the ipsilateral hemisphere from
Immunostaining of E-selectin in ischemic brain
Expression of E-selectin was not observed in brain tissue of normal control or sham-operated rats. E-selectin immunoreactivity was first observed on microvessels within the ischemic lesion at 0 h of reperfusion (Fig. 4A). The intensity and number of E-selectin immunoreactive vessels increased at 2 h, peaked at 4 and 10 h of reperfusion in ischemic areas (Fig. 4B,C), and decreased after 72 h of reperfusion; few E-selectin vessels were detected at 166 h of reperfusion. Upregulation of E-selectin on endothelial cells was confirmed by double immunostaining (Fig. 4D,E). E-selectin immunoreactivity was colocalized with factor VIII immunoreactive microvessels. All E-selectin immunoreactive vessels localized in the ischemic core lesion. Figure 5 summarizes E-selectin expression as a function of reperfusion time.
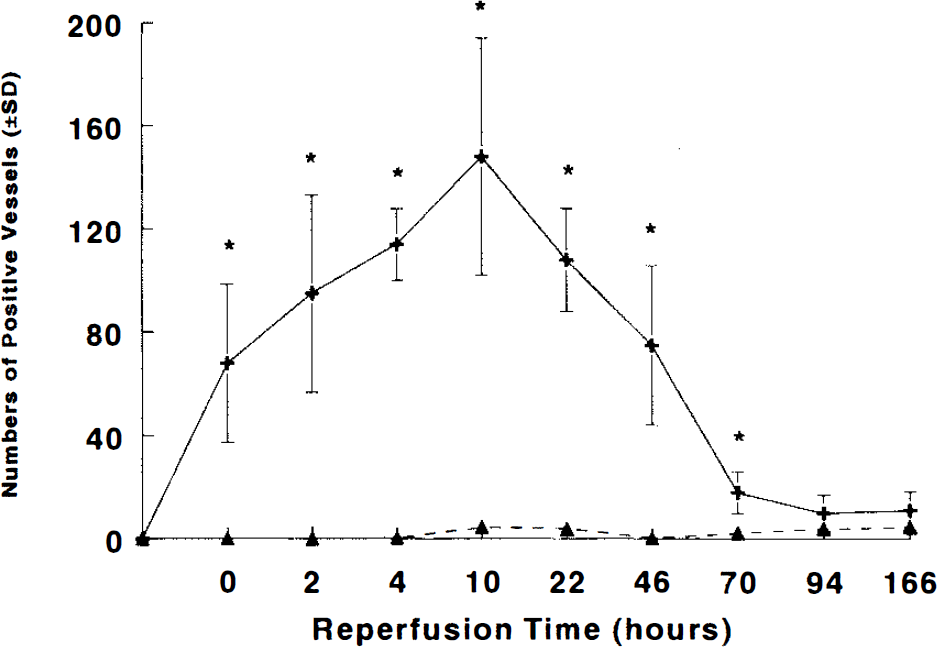
The temporal profile of numbers of microvessels expressing E-selectin within the ipsilateral (+) and contralateral (▴) hemisphere in rat subjected to 2 h of MCA occlusion (n = 3–8 per time point). Statistically significant increases were detected after 2 h of MCA occlusion. *,p < 0.05.
Effect of CY-1503 on ischemic volume
Blood gases were within normal physiological range in SLex-treated and control groups (Table 1). Figure 6 presents the percentage of infarct volume in the four treatment groups. The infarct volume in CY-1503 50 mg/kg treated group was significantly (p < 0.05) less than that in the control group. No difference in the infarct volume was detected between the CY-1503 25 mg/kg (X 4 times or X 8 times) group and the control group. Figure 7 shows the percentage of area of infarction in each of the seven coronal sections from CY-1503 50 mg/kg and the control groups. The area of infarction was significantly (p < 0.05) reduced in sections 4, 5, and 6 in the CY-1503 50 mg/kg group.
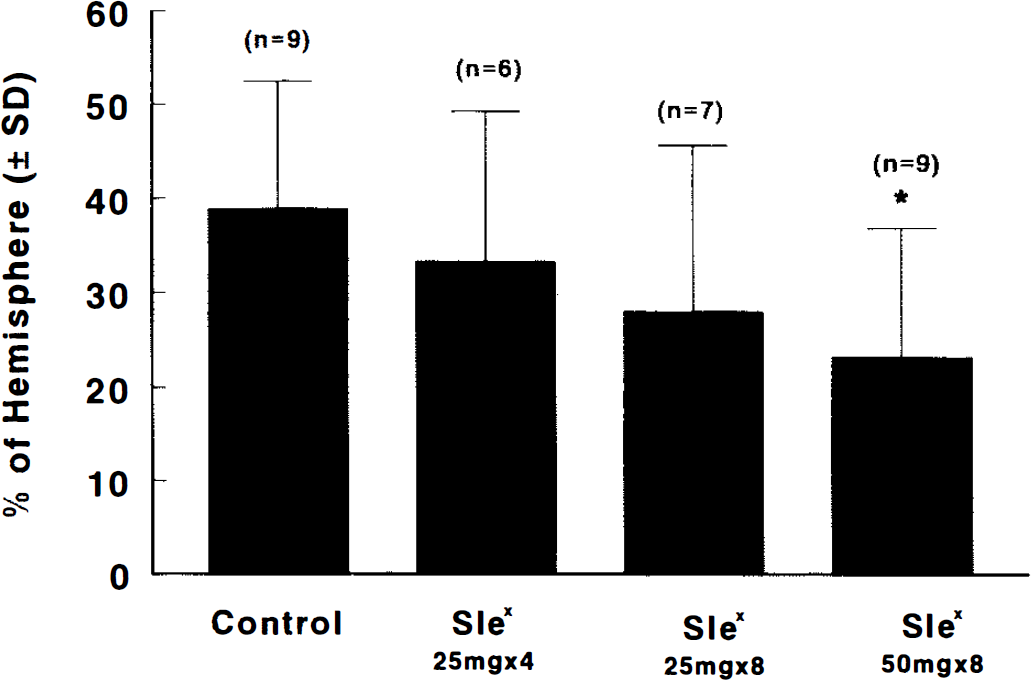
Bar graph shows the effects of SLex and SLN on infarct volume. Rats were subjected 2 h of MCA occlusion and 46 h of reperfusion. Significant reduction of infarct volume was found in a group at a dose of 50 mg/kg of SLex. Values are mean ± SD. *,p < 0.05 as compared with the control group.
Physiological parameters in the four groups
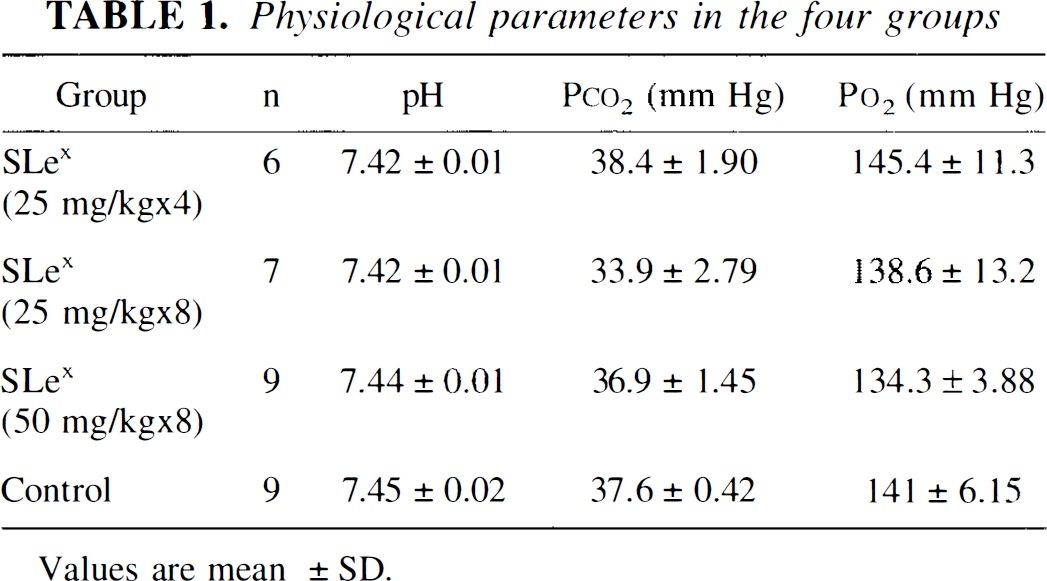
Values are mean ±SD.
Effect of CY-1503 on neutrophil accumulation
Treatment of rats with 50 mg/kg of CY-1503 significantly (p < 0.05) reduced the number of MPO immunoreactive cells accumulated in the ischemic lesion, whereas administration of four times or eight times of CY-1503 25 mg/kg did not significantly reduce the number of MPO immunoreactive cells (Fig. 8). The area of the ischemic lesion was highly correlated with the number of the immunoreactive MPO cells (r = 0.79; p < 0.05), as illustrated in Fig. 9.
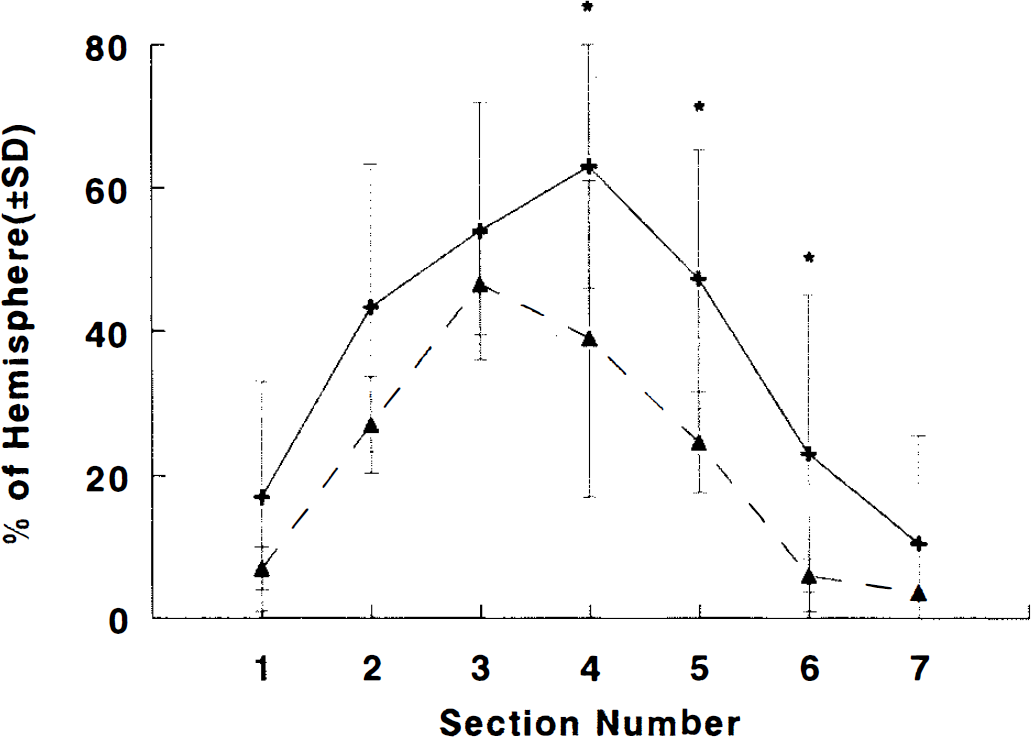
Graph shows percentage of infarct area for each coronal section (2 mm) from anterior to posterior in SLex (50 mg/kg, A, n = 9) and in control (+,n = 9) groups. Infarct areas were significantly (*,p < 0.05) reduced in coronal sections 4, 5, and 6 in SLex treated animals relative to control animals.
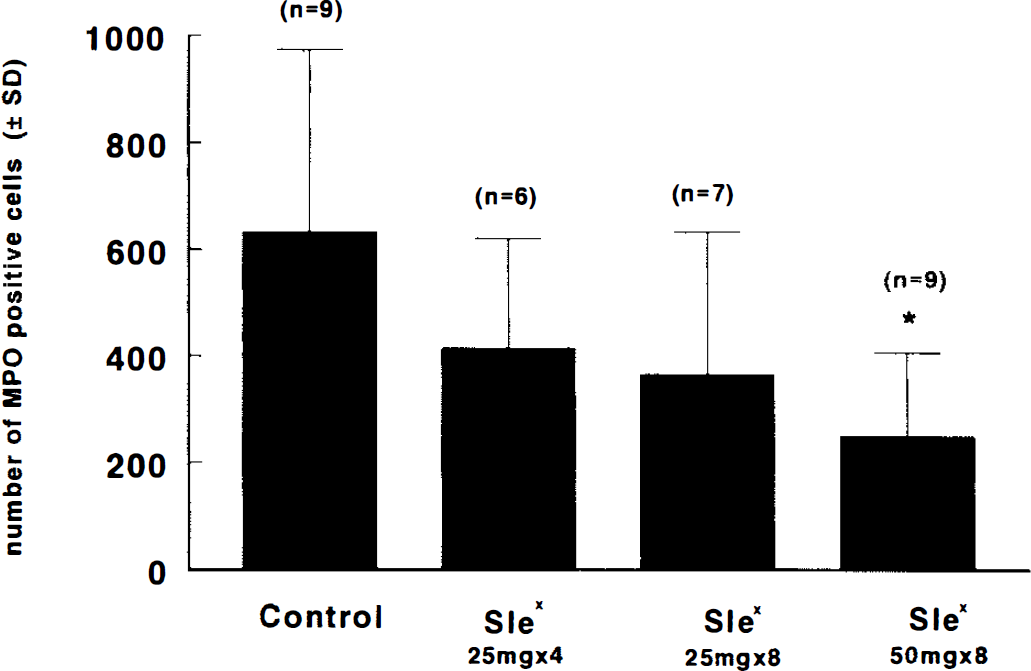
Bar graph shows the effect of SLex, SLN, and saline on MPO immunoreactive cells in the ipsilateral hemisphere. Numbers (mean ± SD) are obtained at 48 h after reperfusion. *,p < 0.05 as compared with the control group. The control group includes SLN- (n = 4) and saline- (n = 5) treated animals.
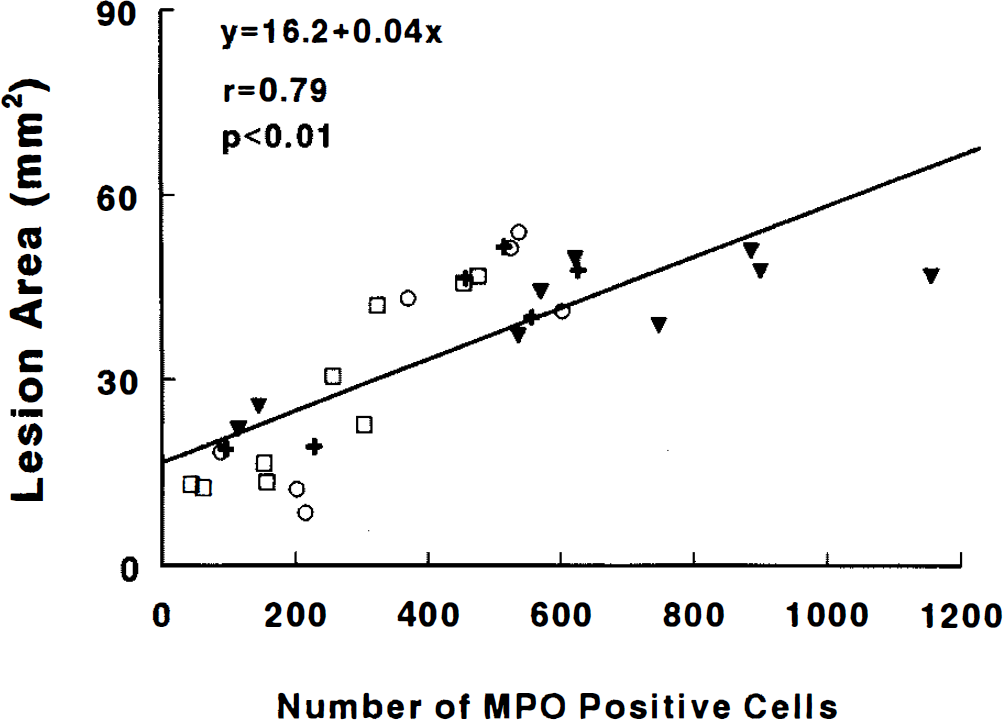
Shows correlation of lesion area with the MPO immunoreactive cells within the lesion. (+, SLex 25 mg/kg ×4; O, SLEx 25 mg/kg ×8; □, SLex 25 mg/kg ×8, ▾ = control).
DISCUSSION
The present study provides the first direct evidence for the involvement of E-selectin in transient MCA occlusion in rats. We cloned rat E-selectin cDNA and showed that E-selectin mRNA and protein were induced in the ischemic region during reperfusion and that administration of a novel carbohydrate analog of SLex, CY-1503, at a dose of 50 mg/kg, significantly reduced infarct volume concomitantly the number of MPO immunoreactive cells. In contrast, treatment with the nonfucosylated oligosaccharide, SLN, neither reduced infarct volume nor decreased MPO immunoreactive cells in the ischemic area.
Recently, a rat E-selectin gene sequence was derived by PCR alone (Fries et al., 1993). Comparison of the authentic cDNA sequence obtained in this study with that sequence revealed four differences in the deduced amino acid sequence. These differences may be the result either of DNA polymorphism or sequence artifacts introduced during the PCR reaction.
In the lectin domain, those residues in human E-selectin that have been demonstrated to be critical for SLex carbohydrate recognition were found to be conserved in rat E-selectin (Erbe et al., 1992). Rat E-selectin epidermal growth factor-like domain displays high identity to that of other species, with conservation of the arrangement of cysteins involved in disulfide bonding. Of note, the deduced rat E-selectin sequence encodes five CR repeats, whereas both human and mouse E-selectin contain six. Rat E-selectin does not contain the equivalent of CR 5 in the human. Rat and rabbit E-selectins are similar in that they both contain only five CRs; however, the rabbit lacks the fourth CR domain (Larigan et al., 1992). Our results also demonstrate that the polypeptide encoded by rat E-selectin cDNA is functionally similar to human E-selectin and that CR 5 does not appear to be critical for function, at least as measured by this in vitro assay. Similar species differences in the number of CR domains has been reported in P-selectin, a closely related member of the selectin gene family (Auchampach et al., 1994).
E-selectin mRNA was detected in the ischemic hemisphere as early as 2 h of reperfusion and was markedly elevated at 10 h of reperfusion. Elevated E-selectin mRNA persisted for up to 22 h, then decreased at 46 h of reperfusion. E-selectin mRNA was no longer detected after 46 h of reperfusion. The temporal profile for induction of E-selectin mRNA in transient MCA occlusion is generally consistent with data obtained from permanent MCA occlusion (Wang et al., 1995). Wang et al. (1995) showed that the level of E-selectin mRNA increased at 6 h, peaked at 12 h, and persisted for 48 h after onset of permanent MCA occlusion in rats. However, expression of E-selectin mRNA occurred earlier after transient ischemia than after permanent ischemia. This parallels the more rapid time course of accumulation of neutrophils in transient versus permanent MCA occlusion (Zhang RL et al., 1994a) and suggests that reperfusion injury is a trigger for rapid transcription of E-selectin mRNA.
E-selectin protein was detected immunohistochemically on cerebral microvessels by 0 h of reperfusion and peaked at 10 h of reperfusion. E-selectin protein gradually decreased at 46 and 70 h of reperfusion, and only a few E-selectin vessels were detected after 96 h of reperfusion. The temporal profile of upregulation of E-selectin protein is consistent with a recent observation of a transient increase in soluble E-selectin (sE-selectin) in patients with acute ischemic stroke (Fassbender et al., 1995). In these patients, the level of sE-selectin was significantly elevated at 8 h and persisted up to 24 h and returned to basal levels after 72 h of stroke. In the rat brain, E-selectin immunoreactivity was exclusively localized on cerebral endothelial cells, as demonstrated by the double immunostaining technique. The antibody against von Willebrand factor is widely used as a marker of endothelial cells (Theilen and Kuschinsky, 1992). This finding is in accordance with those of previous studies that only vascular endothelial cells express this molecule (Bevilacqua et al., 1989). Furthermore, induction of E-selectin has been demonstrated on coronary endothelium after myocardial ischemia and reperfusion in rhesus monkeys (Shen and Verrier, 1994) and on lung vasculature after ischemia-reperfusion in rats (Mulligan et al., 1991).
E-selectin protein expression appeared prior to the induction of mRNA. It is possible that the upregulation of the protein preceding the mRNA is attributed to the relative insensitivity of the Northern blot method compared to other, e.g., PCR, methods. Wang et al. failed to detect E-selectin mRNA signal in focal cerebral ischemic rats using Northern blot methodology. However, they were able to detect E-selectin mRNA by using a more sensitive reverse transcription (RT) and PCR (RT-PCR) method (Wang et al, 1995).
E-selectin is not constitutively expressed on human umbilical vein endothelial cells (HUVECs) (Bevilacqua et al., 1987). In vitro studies have revealed induction of E-selectin in HUVECs within 1–4 h (Bevilacqua et al., 1989) by interleukin (IL)-1β and tumor necrosis factor a (TNF-a). In vivo, airway administration of TNF-α into rat lung results in intense vascular expression of E-selectin (Mulligan et al., 1993). In addition, administration of an inhibitor of TNF-α, cloricromene, significantly decreases serum levels of TNF-α and sE-selectin and increases survival time in rats subjected to splanchnic artery occlusion shock (Altavilla et al., 1995). Expressions of IL-1β mRNA and TNF-α have been documented in ischemic brain by Northern blots and by immunohistochemistry method, respectively, after MCA occlusion in rats (Liu et al, 1993, 1994). These studies suggest that expression of the E-selectin described here could be induced by IL-1β as well as TNF-α.
Administration of mAbs directed against neutrophils and adhesion molecules have provided strong evidence for the role of neutrophils in contributing to ischemic cell damage in animal models of transient focal cerebral ischemia. Administration of anti-CD 11 b or anti-CD 18 reduced infarct volume in rats (Zhang ZG et al., 1995) and improved cerebral microcirculation in nonhuman primates (Mori et al., 1992) after MCA occlusion. Treatment with anti-ICAM-1 reduced infarct volume by 43% and decreased MPO activity by 41% in rats subjected to transient focal cerebral ischemia (Zhang RL et al., 1994b). The marked induction of E-selectin parallels neutrophil accumulation in rat brain after transient MCA occlusion (Zhang RL et al., 1994a). Leukocyte rolling is commonly regarded as the initial step in a cascade of events leading to transendothelial leukocyte migration into the extravascular space (Lasky 1992). Both P- and E-selectin promote leukocyte rolling under conditions of shear flow (Lawrence and Springer, 1991; Abbassi et al., 1993). E-selectin mediates leukocyte rolling by interacting with its neutrophil counter ligand containing the tetrasaccharide SLex (von Andrian et al, 1993). Neutrophil expression of SLex is well correlated with E-selectin-dependent adhesion (Munro et al., 1991; Fukuda et al., 1984). In the present study, treatment with a unique carbohydrate analog of SLex, CY-1503, at a dose of 50 mg/kg resulted in a significant reduction of infarct volume and MPO immunoreactive cells in ischemic lesion. Taken together, these data suggest a hypothesis that E-selectin may be vital for neutrophil adhesion and subsequent cerebral ischemic cell damage. A crucial role for E-selectin in neutrophil-mediated myocardial ischemia has also been reported. Administration of mAb against E-selectin significantly reduced myocardial injury and decreased MPO activity in a rat model of myocardial ischemia-reperfusion injury (Altavilla et al., 1994).
The protective effect of CY-1503 is dose dependent, as treatment with CY-1503 at a dose of 25 mg/kg did not significantly reduce infarct volume. The effective dose is higher than that previously reported in other animal models. This was not unexpected given the experimental design, which involved prolonged reperfusion and pharmacokinetics of CY-1503 in rat. Clearance of CY-1503 is through renal excretion, with >95% of the injected dose in rats recovered in the urine. The compound has a high volume of distribution (887 mg/kg) and an elimination half-life of 46 min in rat (Wing and Stoudemire, 1994). In our model, E-selectin expression peaked between 10 and 22 h following reperfusion. Therefore, a high dose was required to maintain an effective serum concentration in rats for the first 24 h of the reperfusion period. It is of note that no toxic effects of this compound have been observed in rats given up to 1,500 mg/kg/day of CY-1503 for 7 days (unpublished observation). Treatment with the nonfucosylated analog SLN did not result in any salvage of cerebral tissue or reduction in neutrophil accumulation. The protective intervention of SLex analog in the present study is consistent with results of previous studies in canine (Lefer et al., 1994) and cat (Buerke et al., 1994) models of myocardial ischemia and reperfusion, a murine model of lung ischemia and reperfusion (Seekamp et al., 1994), and a rat model of traumatic shock (Skurk et al, 1994).
Our data strongly suggest that an interaction between E-selectin and an analog of SLex exerts beneficial effects in rats subjected to transient MCA occlusion. However, we are uncertain as to which of the selectins mediates cerebral ischemia-reperfusion injury. Immunohistochemical staining revealed an upregulation of P-selectin on cerebral postcapillary microvessels of an ischemic region in a primate model of transient MCA occlusion (Okada et al., 1994). Studies from P- or E-selectin-deficient mice have indicated that P- and E-selectin are partially redundant and can replace each other as mediators of neutrophil rolling at inflammatory sites in vivo (Ley and Tedder, 1995). In addition, L-selectin is constitutively present on neutrophil membranes and application of SLex could inhibit L-selectin interaction with P- or E-selectin expressed on the endothelial surface (Picker et al., 1991). Thus, treatment with CY-1503 might effectively block the actions of all of the selectins simultaneously and could inhibit selectin-mediated neutrophil rolling along the ischemic reperfused cerebral vascular endothelium. In summary, transient MCA occlusion induced E-selectin mRNA and protein expression within ischemic tissue, and administration of an oligosaccharide analog of SLex, CY-1503, reduced infarct volume as well as MPO immunoreactive cells in rats subjected to transient MCA occlusion. These studies indicate that expression of E-selectin has an important role in facilitating neutrophil rolling and that CY-1503 offers a novel therapeutic approach to reduce selectin-mediated ischemic reperfusion brain injury.
Footnotes
Abbreviations used
Acknowledgment:
This work was supported NINDS grant RO1 NS33627 and RO1 NS34184. The authors wish to thank Denice Janus for manuscript preparation and Cecylia Powers, Xiu Li Zhang, Jonathan Prostak, and Qing Wang for technical assistance.
