Abstract
Chronically implanted ultrasonic Doppler flowmeters were used to obtain simultaneously recorded flow velocity signals from internal carotid and vertebral arteries, and the sagittal sinus, in rabbits. All three signals increased to 144 ± 7–215 ± 35% of baseline during hypercapnia (arterial Pco2 55 mmHg) in both anesthetized and conscious animals. During the period of change in inspired CO2, the relationship between simultaneously recorded mean internal carotid and mean sagittal sinus signals was linear, with the correlation ranging from 0.83 to 0.96. Since forebrain arterial inflow must approximate forebrain venous outflow, the high correlation between internal carotid and sagittal sinus signals indicates that these measures provide reliable and valid indices of cerebral blood flow (CBF). Vertebral and internal carotid angiography confirmed the location of Doppler probe. Chronically implanted ultrasonic Doppler flowmeters can, thus, provide continuous noninvasive measurements of cerebral arterial flow in both anesthetized and conscious rabbits.
Procedures providing continuous beat-to-beat indices of regional blood flow in experimental animals have not commonly been applied to the arteries supplying the brain. Conventional electromagnetic and Doppler ultrasonic flow probes have been too bulky to fit around the internal carotid artery, thus their use in the assessment of cerebral blood flow (CBF) has been via measurement of common carotid flow after ligation of external carotid (Meyer et al., 1967; Goadsby et al., 1982; Scremin et al., 1982). Interpretation of such studies may be problematic for animal species in which the anatomical arrangement of the carotid vessels permits significant interchange between internal and external carotid circulations. This occurs in the cat and the sheep, in which a rete form of communication exists; and in the dog, in which part of the cerebral circulation derives from communications between the external carotid and intracranial circulations (Hayward and Baker, 1969; Heisted et al., 1980; Abboud, 1981). Currently used procedures to measure CBF in experimental animals are applicable mainly to anesthetized preparations. This is paradoxical, since the brain is the bodily organ most affected by anesthesia. Basal CBF is substantially reduced during the anesthetized state, and there is no opportunity to observe rapid changes in flow that might occur as a consequence of an animal's normal interaction with the environment.
In rabbits, the cerebrovascular architecture is similar to that in primates, including humans, with no significant communication between external and internal carotid circulations normally occurring (Chungcharoen et al., 1952; Scremin et al., 1982). The construction of Doppler ultrasound probes has been improved (Haywood et al., 1981; Koerfer et al., 1993), suggesting their applicability to assessment of flow in the internal carotid and vertebral arteries of the rabbit. Although relatively global measures of flow are provided, many physiological questions may be addressed using such measures, as evidenced by recent studies of the role of nitric oxide in the control of the cerebral circulation (Iadecola et al., 1994).
We now describe the use of chronically implanted ultrasonic Doppler flowmeters for continuous measurement of internal carotid and vertebral arterial blood flow in both anesthetized and conscious rabbits. We assessed the reliability and validity of our procedures by comparing internal carotid and vertebral signals with that of the sagittal sinus simultaneously recorded, when CBF changed in response to alterations in the concentration of CO2 in the inspired gas (Reivich, 1964). We confirmed that our probes were positioned on the appropriate vessel by carrying out digital subtraction angiography on internal carotid and vertebral arteries.
METHODS
Implantation of flow probes
Male New Zealand white rabbits (2.5–3.5 kg) were intubated after being anesthetized with sodium thiopentone (40 mg/kg i.V.); anesthesia was maintained with 1–2% halothane in O2. Antibiotic (Penstrep, Troy Laboratories, Australia) was administered (0.1 ml/kg i.m.). The left common carotid bifurcation was exposed, then viewed with an operating microscope (Seiler Instrument, St. Louis, MO, U.S.A.). The internal carotid artery was gently isolated for a distance of ∼7 mm distal to the carotid sinus region. Lignocaine (2%) (Delta West Pty Ltd., Bentley, WA, Australia) was applied to minimize spasm. The anatomical arrangement of the occipital artery was noted (Scremin et al., 1982 and Lee et al., 1994). A directional pulsed Doppler flow probe (Iowa Doppler Products, IA, U.S.A.), lumen 0.8 mm, ultrasonic frequency 20 MHz was placed around the internal carotid artery, distal to the carotid sinus and proximal to the foramen lacerum. The probe was held in place by suturing the connecting wires near the probe to surrounding muscle. Probe wires were passed dorsally through the cervical muscles and positioned subcutaneously behind the head, with the wire tips protruding ∼2 cm. A cervical left paramedian skin incision was then made over the medial one-third of the clavicle. The vertebral artery on the lateral and posterior wall of the thoracic outlet was exposed by gentle dissection in the region bounded by the trachea, esophagus, and common carotid artery. Then, ∼7 mm of the artery was isolated from surrounding tissue and a second Doppler probe, lumen 0.8–1.0 mm, was positioned around this artery. Probe wires were passed dorsally and positioned subcutaneously behind the head, a little caudal to the wires from the internal carotid probe. Antibiotic powder was applied to each wound before it was sutured. Animals recovered from anesthesia and were returned to the animal house.
One week later, rabbits were reanesthetized and intubated as above. A burr-hole was drilled over lambda to expose the dorsal surface of the sagittal sinus. The cuff of a third Doppler probe was trimmed, leaving the silastic base with the piezoelectric crystal and connecting wires intact. This modified probe was inserted through the burr-hole so that it rested on the dorsal surface of the sagittal sinus. The position of the probe was adusted until a stable signal was apparent. The probe was then fixed in position via dental cement anchors to three stainless steel screws inserted into the skull. The first blood flow experiment with modulation of inspired CO2 was then performed with the animal still under anesthesia (see details below). After this, the rabbit recovered and was taken back to the animal house. The next day, the animal was brought to the laboratory and placed in a small wooden cage. A CO2 concentration experiment was then performed on the conscious rabbit (see details below). In two animals the experiment was repeated twice after an interval of 1 week. In another animal, experiments were repeated after 12 and 16 weeks.
Changing the CO2 concentration in the inspired gas
In the anesthetized animal study, 10% CO2 in oxygen (B.O.C. gases, Adelaide, Australia) was gradually introduced into the tracheal tube, with continuation of 1–2% halothane. In the conscious animal study, the rabbit's box was enclosed in a plastic bag, and either air or 10% CO2 in oxygen (4–6 L/min) was supplied via a length of silastic tubing (Dow Corning, Midland, MI, U.S.A.) entering at one end of the box. Gas outflow was via plastic tubing at the other (rabbit's head) end of the box, where the percentage of CO2, was monitored (Datex Normocap CO2 monitor, Finland). During a ∼3-min period, the concentration of CO2 was gradually increased 10%. This concentration was then maintained for 0.5–1 min. The 10% CO2 in oxygen was then replaced with 100% oxygen or air so that the concentration of CO2 returned to normal baseline levels over a ∼3-min interval.
Measurement of arterial blood pressure, analysis of blood gas, and recording of Doppler flow signals and CO2 levels
Arterial pressure was measured via a catheter inserted temporarily into the central ear artery under general or local (2% lignocaine) anaesthesia. The catheter was advanced towards the external carotid artery. Pressure was measured by connecting the catheter to a Statham P23 ID transducer and to the MacLab system (see below). The wires from internal carotid and vertebral probes were attached to leads from a Triton Technologies (San Diego, CA, U.S.A.) flowmeter (model 200 mainframe, model 202, PLSD Doppler, 20MHz). Flow signal, on directional setting, was calibrated using the Triton internal standard signals. Range was adjusted for maximal signal amplitude. The blood pressure signal was amplified by a Mac Lab Bridgeamp (ADInstruments, Sydney, Australia). Doppler signal, blood pressure, and percentage CO2 signals were digitized (40 Hz) with MacLab. Digitized signals were stored and displayed on a Macintosh Quadra 700 computer. Arterial blood samples (100 μl) were taken from the ear artery line before inspired CO2 was increased and again at maximum CO2 level. Arterial Po2, Pco2, and pH were measured on an ABL520 blood gas analyzer (Radiometer, Denmark). At the end of the day's experiment, the ear artery catheter was removed.
Data analyses
Analysis was performed either with Chart software (ADInstruments), IGOR Pro (WaveMetrics, Lake Oswega, OR, U.S.A.), or Statview II (Abacus Concepts, Berkeley, CA, U.S.A.). A 1-min sample of simultaneously recorded pulsatile internal carotid and vertebral artery signals was taken from individual rabbits while the animals were at rest in their box with no experimental perturbations. Vertebral pulse signals occurred ∼25 ms (one datum point) before the corresponding point of the carotid pulse signal, reflecting the relative proximity of the vertebral probe to the heart. Signals were adjusted to correct for this phase difference, and the Pearson correlation between the phasic signals from internal carotid and vertebral arteries for the 1-min period was calculated for each rabbit.
Simultaneously recorded internal carotid and sagittal sinus signals were averaged over 1-s intervals, and the relationship between the two averaged signals was analysed using linear regression, with data recorded during both increasing and decreasing CO2 levels. For each vessel, a pre-CO2 Doppler signal baseline for each procedure was then defined as 100%. The percentage value during the maximum Doppler signal level observed in response to increased inspired CO2 was obtained and compared with the baseline using analysis of variance and Fisher's least significant difference test for post-hoc comparison of means. Signals recorded during the period of increasing CO2 concentration were averaged into a 100-point data sample for each rabbit. In conscious and anesthetized groups, averaged mean values from individual animals for internal carotid, vertebral, and sagittal sinus vessels were expressed as Doppler signal versus corresponding percentage CO2. IGOR Pro software was then used to fit a power function (y = a + bxc) to the relationship.
Digital subtraction angiography
Digital subtraction angiography was performed on four rabbits after CO2 experiments were completed. Animals were anesthetized with urethane (1.5 g/kg body wt). The right femoral artery was exposed and a Cordis catheter sheath (Cordis Corporation Miami, FL, U.S.A.) was inserted. The left common and external carotid arteries were then exposed. A catheter was inserted anterogradely into the left common carotid artery. The external carotid artery was ligated. The rabbit was then taken from the laboratory to the Radiology Department of Flinders Medical Centre for angiography. Using fluorescence screen observation, a 3-French catheter was introduced through the femoral sheath to the left subclavian artery. Then, 1–2 ml of iopromide 300 mg/ml (UHiavist, Schering, Germany) was injected by hand, and appropriate images obtained. Iopromide, 1–2 ml, was then injected into the left common carotid artery, and further images obtained. Angiograms were scanned (RFS 2035 Plus Films Scanner, Kodak) and processed on a Macintosh computer using AdobePhotoshop (Adobe System Inc., CA U.S.A.) and Canvas (Deneba Systems, Inc. Miami, FL, U.S.A.).
Number of rabbits
Successful Doppler flow signals were obtained from at least one implanted probe in 13 rabbits. Simultaneously recoded signals were obtained from internal carotid and sagittal sinus probes in eight rabbits (four animals studied while under anesthesia and four rabbits studied while conscious). Vertebral flow signal was also simultaneously obtained in all eight rabbits. Occasionally, the sagittal sinus signal did not reliably increase with hypercapnia, even though typical increases were observed in simultaneously recorded internal carotid and/or vertebral signals. This presumably reflected change of position of the probe with respect to the sinus (see Discussion). In these cases, we excluded sagittal sinus data from subsequent arterial-sagittal sinus flow correlation analysis. However, internal carotid and vertebral signals were used in other analyses. Five animals were used for angiography studies. The number of rabbits in each individual experimental condition is specified in the appropriate Results section.
RESULTS
Change in Doppler signals with increasing inspired CO2
Simultaneously recorded Doppler signals from all three vessels in response to changes in inspired CO2 are shown in Fig. 1. In anesthetized animals, when the concentration of CO2 in inspired air was increased to 9.3 ± 0.9% (mean ± SD, n = 8), the sagittal sinus signal increased to 215 ± 71% of baseline (n = 4). At the same time, the internal carotid signal increased to 187 ± 61% of baseline (n = 8) and the vertebral signal increased to 144 ± 19% of baseline (n = 7). In conscious animals, when the concentration of CO2 in inspired air increased to 9.3 ± 0.41% (n = 11), the sagittal sinus signal increased to 185 ± 46% of baseline (n = 4). At the same time, the internal carotid signal increased to 181 ± 40% of baseline (n = 11) and the vertebral signal increased to 161 ± 21% of baseline (n = 10). All these increased values were significantly greater than baseline control values, p < 0.01, but they were not significantly different from each other (analysis of variance and Fisher post-hoc comparisons, p > 0.05) in either the anesthetized or the conscious state.
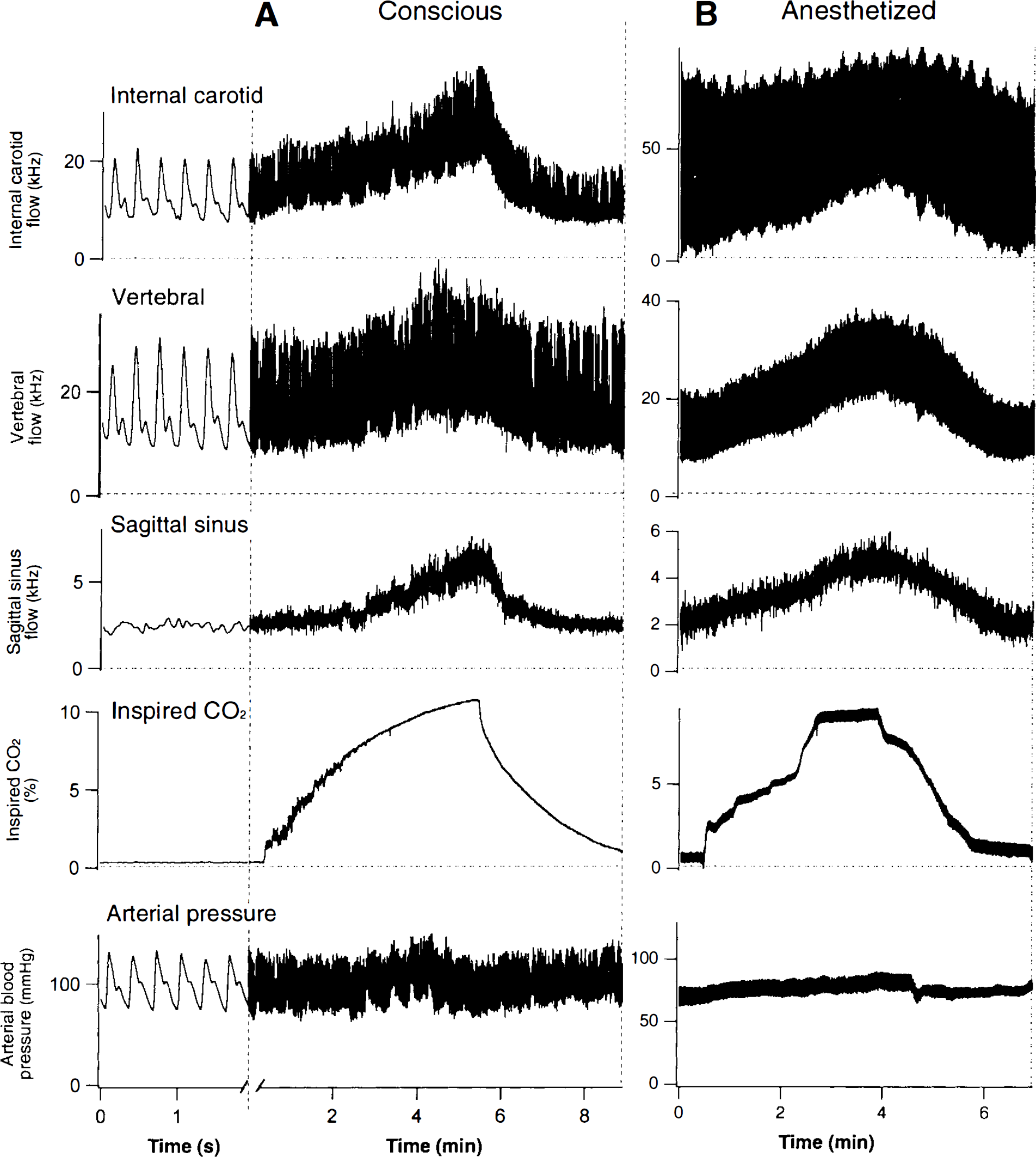
MacLab records of ultrasonic Doppler pulsatile cerebral blood flow signals, inspired CO2 concentration and arterial blood pressure in conscious
When animals were breathing normal air (conscious animals) or a mixture of oxygen and halothane (anesthetized animals), arterial pH was 7.47 ± 0.03 and Pco2 was 39 ± 5 mmHg (n = 6). When inspired CO2 was increased to 9 ± 1%, arterial pH was 7.34 ± 0.05 and Pco2 was 55 ± 4 mmHg (n = 6). Arterial Po2 during inhalation of 10% CO2 in oxygen was always >400 mmHg.
Correlation between internal carotid and sagittal sinus signals during changes in percentage inspired CO2
Data from internal carotid and sagittal sinus signals recorded during the period of increasing levels of CO2 and during the washout period, were analysed for eight rabbits (four anesthetized, four conscious). Correlations between the two signals (averaged over 1 s) in anesthetized animals were 0.96, 0.96, 0.96, and 0.87, with significant linear regressions as shown in Fig. 2A. Correlations between the two signals (averaged over 1 s) in conscious animals were 0.87, 0.82, 0.91, and 0.95, with significant linear regressions as shown in Fig.
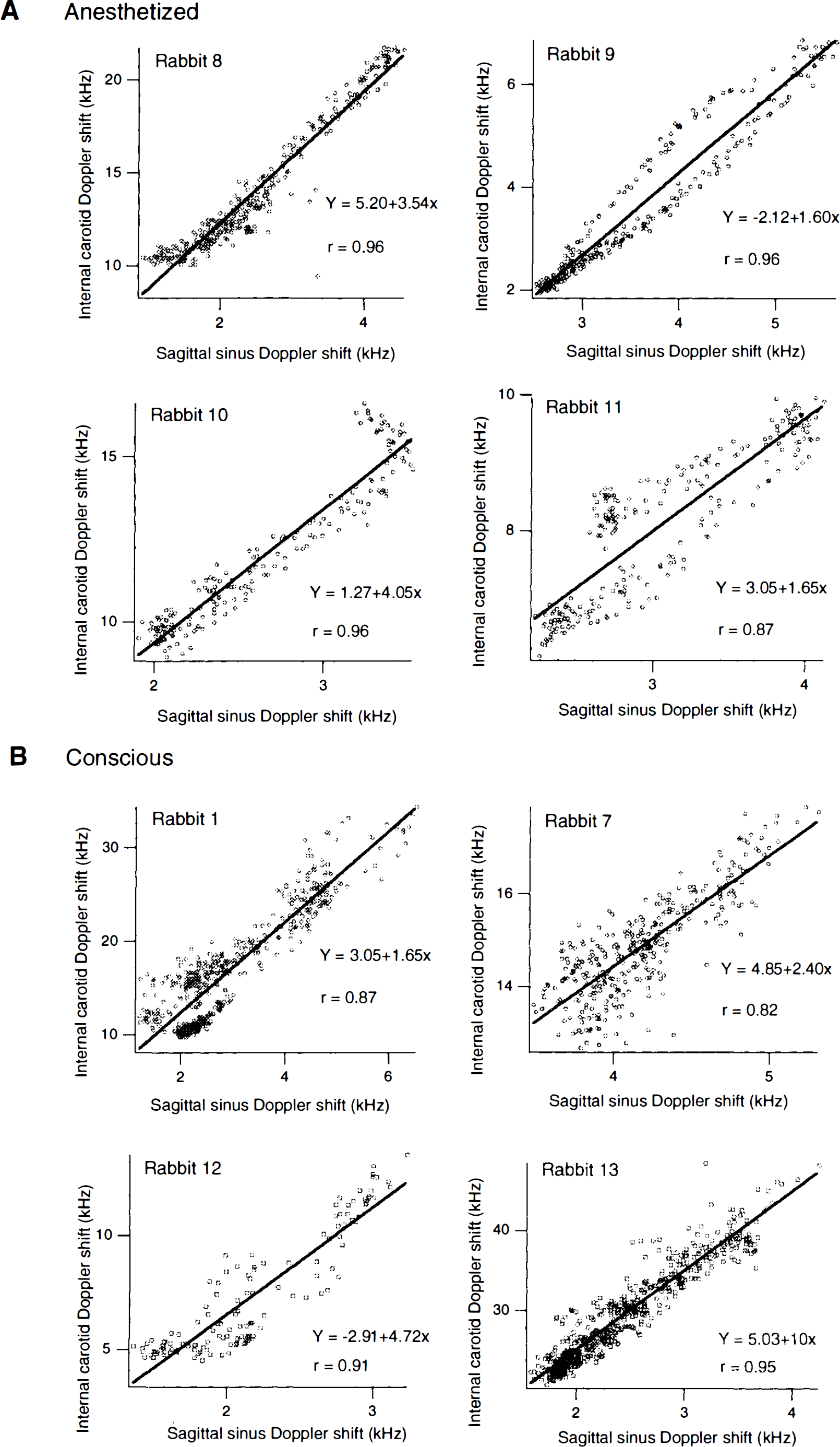
Relationship between mean internal carotid and sagittal sinus flow signals in individual rabbits obtained during experimental increase of inspired CO2 concentration in either anesthetized
Relationship between signals from each vessel and increasing CO2 levels in anaesthetized and conscious animals
Relationships for different vessels between mean Doppler signals and percent inspired CO2 are shown in Fig. 3, in which data have been fitted to a power function (y = a + bxc). The initial slope of the curve appears greater in anesthetized than in conscious animals. When inspired CO2 reaches ∼5% in conscious animals, cerebral flow increases more rapidly so that eventual peak values are similar in conscious and anesthetized animals.
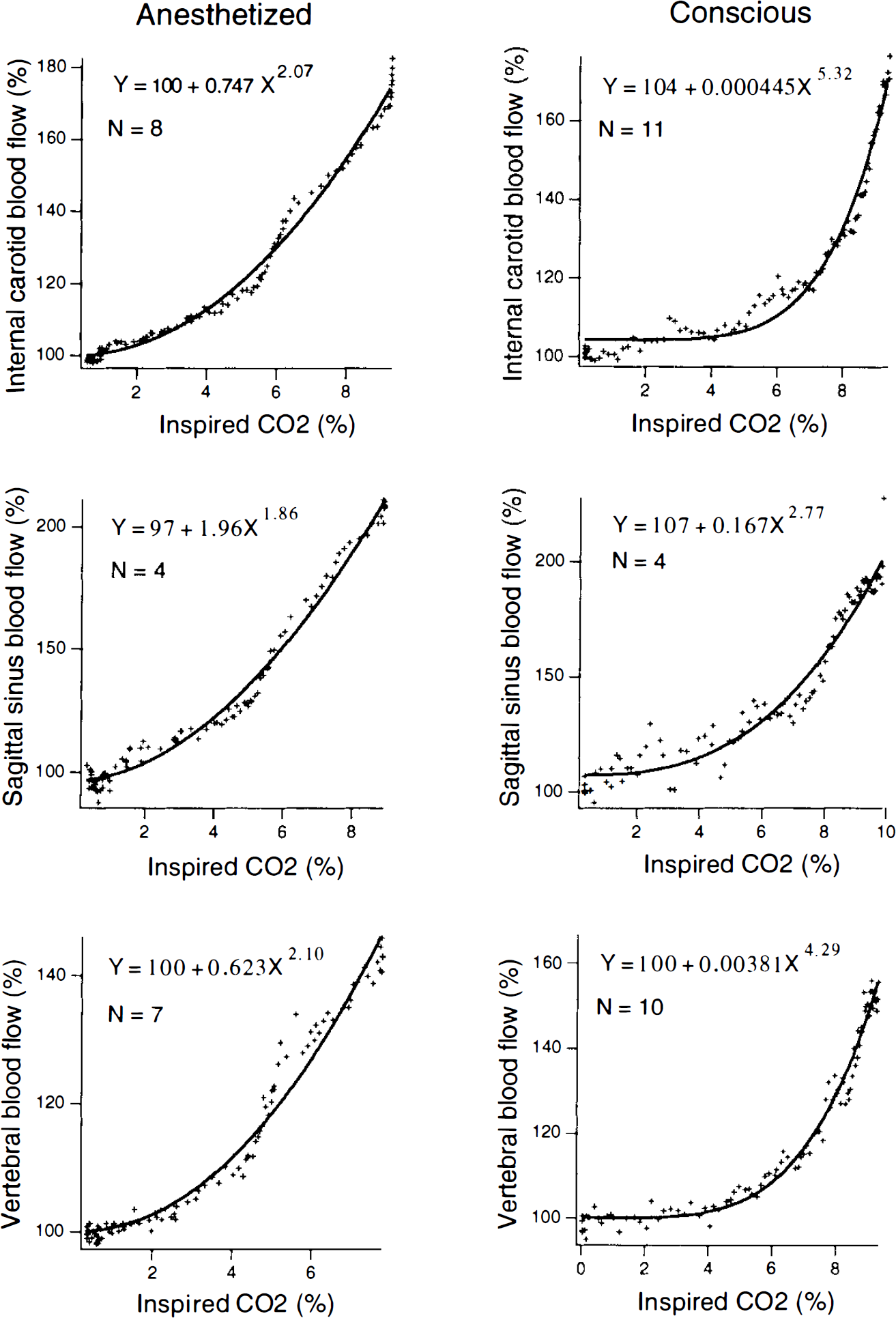
Relationship between mean internal carotid, sagittal sinus, or vertebral flow signal and concentration of CO2 in inspired gas. Data points (+) are averages from the indicated number (n) of rabbits. Curves were fitted with IGOR software, using a power function.
Comparison of blood flow and internal carotid signals
Clear, pulsatile flow signals were obtained from the vertebral as well as the internal carotid artery. When simultaneously recorder vertebral and internal carotid pulsatile signals were compared in conscious rabbits in the resting state, the correlation between data points on 1-min traces (corrected for phase difference) ranged between 0.87 and 0.98 (n = 8 rabbits). This high correlation between resting pulsatile records attests to the reliability and low noise level of the signal recordings. When percent inspired CO2 was increased, the signal from the vertebral artery increased in a manner generally similar to that observed in the internal carotid artery in both anesthetized and conscious animals (Figs. 1 and 3). In anesthetized animals, the correlation between simultaneously recorded internal carotid and vertebral signals (1 s average) during the period of increasing and decreasing CO2 ranged between 0.80 and 0.97 (n = 7 rabbits).
Long-term repeatability of internal carotid flow measurement
In two rabbits, the change in internal carotid signal in response to alterations in inspired CO2 was reexamined in the conscious state after intervals of 1–3 weeks. The example shown in Fig. 4 demonstrates that the signal changes in response to increasing CO2 after a 1-week interval was quite similar to the initial response, with similar equations describing the power relationship. A third rabbit was examined on three occasions (1, 12, and 16 weeks), and similar relationships were again obtained (Fig. 4).
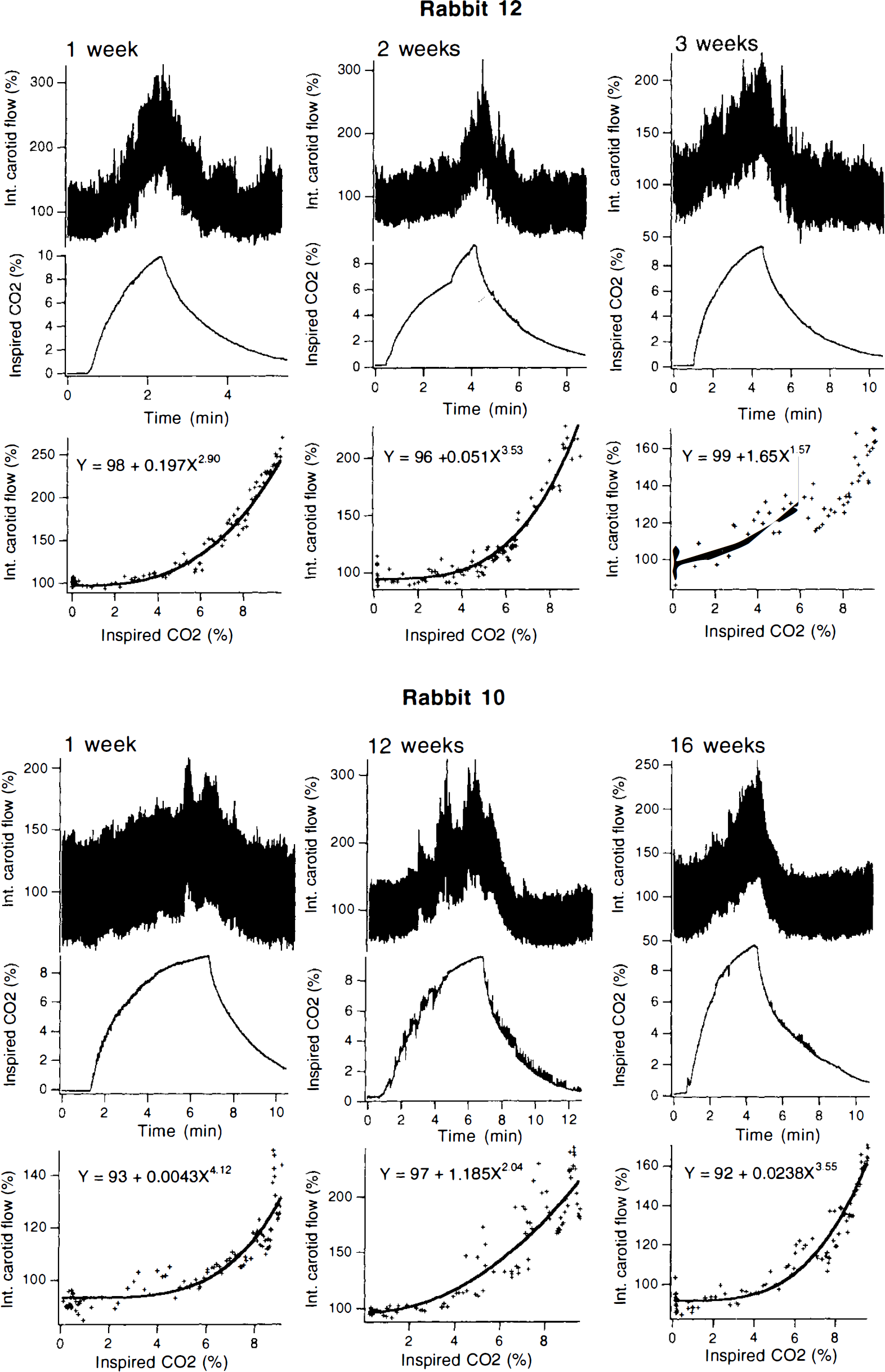
Examples of internal carotid Doppler signals in response to increases in inspired CO2. Experiment repeated at intervals from weeks to months after implantation of probe.
Angiography
Vertebral and common carotid angiography, including early venous phase images, demonstrated the recording probes implanted around the appropriate arteries and in relation to the sagittal sinus (Fig. 5). The radiological contrast was injected under relatively high pressure, displaying a high proportion of the cerebral arterial tree. Obviously, this does not mean that one artery normally supplies the whole brain. Vertebral angiograms defined occasional small branches originating from the extracranial portion of the artery. The common carotid angiogram was performed after external carotid ligation. No communication between internal carotid and extracranial vessels was demonstrated. The ophthalmic artery was seen to originate from the internal carotid artery, as demonstrated by Scremin et al., (1982).
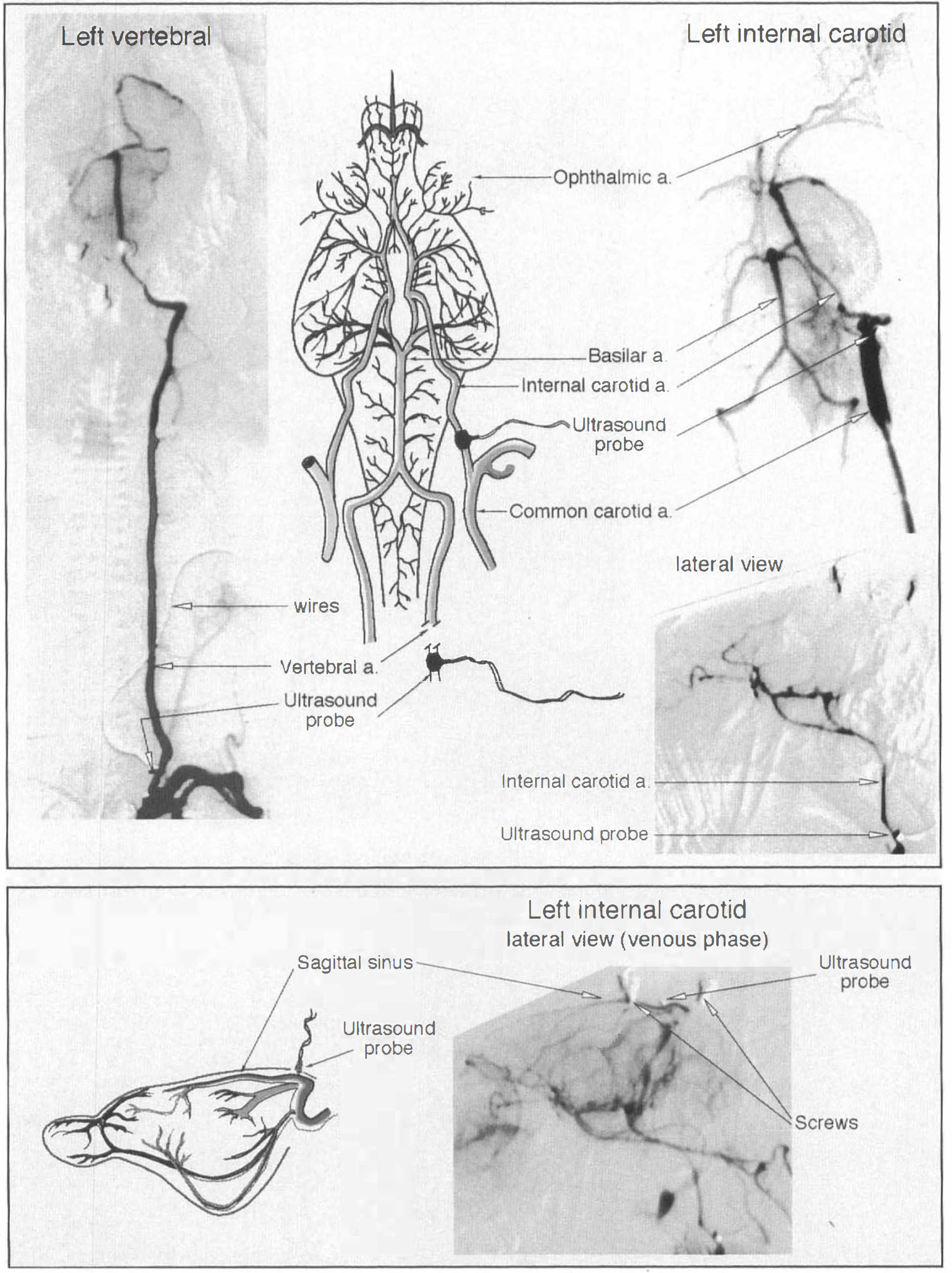
Cerebral angiograms demonstrating the Doppler probes in situ around vertebral and internal carotid arteries (top panel), and in relation to the sagittal sinus (bottom panel). Vascular diagrams were copied and modified from Scremin et al., (1982) with permission.
DISCUSSION
We believe that our study is the first to provide relatively noninvasive continuous indices of beat-to-beat internal carotid and vertebral arterial flow in conscious experimental animals (Heistad and Kontos, 1983; Goldman, 1993). Our study is also the first to record forebrain flow via a probe implanted on the internal carotid artery in anesthetized animals. Previous studies have measured common carotid flow with the external carotid ligated (Meyer et al., 1967; Goadsby et al., 1982; Scremin et al., 1982). One of these studies also used electromagnetic flowmeters to assess vertebral arterial flow in anesthetized monkeys (Meyer et al. 1967).
Ultrasonic Doppler probes measure blood cell velocity, with the relationship between velocity and flow depending upon the angle between the probe crystal and long axis of the vessel, and upon the diameter of the vessel in the region surrounded by the probe. Any variability in these parameters during an experiment alters the Doppler signal independently of any real change in flow. The commercial probes used in the present study are constructed so that the angle of the crystal is constant at 45°. Probes of appropriate size were chosen to fit snugly around the vessel, with chronic implantation allowing time for fibrotic tissue to hold the probe in place and minimize diameter change in the portion of the vessel surrounded by the probe. If changes in crystal angle or vessel diameter did occur, they should have had independent effects on carotid and the sagittal sinus flow signals, thereby reducing the correlation between forebrain inflow and forebrain outflow. However, we found in both anesthetized and conscious animals that the correlations between simultaneously recorded internal carotid and sagittal sinus Doppler signals were always >0.80, and usually >0.90, both at rest and during the period when flow was experimentally varied by increasing the concentration of inspired CO2. Our correlation data strongly suggest that the arterial Doppler signals are valid indices of CBF in the rabbit. Although our methodology does not specify absolute flow, proportional changes in the Doppler signal reflect proportional changes in absolute flow.
Correct positioning of arterial probes was affirmed by the presence of clear and obvious pulsatile arterial signals at the time of probe implantation; such affirmation was not available for the sagittal sinus probe. A strong sagittal sinus signal was not always obtained, and, on some occasions, the sagittal sinus signal did not increase when inspired CO2 was increased. The probe was cemented to the skull, and any change in the position of the probe relative to the sinus, as might occur if the brain moves in relation to the skull during hypercapnia, could have interfered with the signal from the sinus probe. In contrast, the signals from internal carotid and vertebral probes increased reliably with increases in inspired CO2. Thus, although we used sagittal sinus flow measurement to validate arterial flow measurements, in practice, probes implanted around the major cerebral arteries provide more reliable flow measurements than do probes positioned over the sagittal sinus.
Our results in anesthetized rabbits suggest a relationship between internal carotid flow and concentration of inspired CO2, similar to that observed in studies in rabbits using an electromagnetic probe to measure common carotid flow with ligated external carotid (Scremin et al. 1982). In separate rabbits, Scremin and colleagues measured sagittal sinus blood flow (volumetric and H2-clearance techniques), demonstrating that cerebral venous outflow also increased with increases in the concentration of CO2 in inspired air. Our study, with simultaneous recordings of both flows, also demonstrates that arterial inflow and venous outflow vary together. Our sagittal sinus flow changes are also similar to those obtained by Upton et al. (1994) in unanesthetized sheep.
Our angiography confirmed the findings of Scremin et al. (1982) concerning the anatomical arrangements of cerebral blood vessels in the rabbit. There is, normally, little or no communication between internal and external carotid circulations. The internal carotid artery supplies the forebrain. Sometimes, as noted by Scremin and colleagues and by Lee et al., (1994), the occipital artery may arise from the internal carotid. Occasionally, we found that it was either difficult to identify the different vessels or difficult to place the internal carotid probe distal to the origin of the aberrent occipital vessel. In these cases, the rabbit was not used for CBF measurements. The internal ophthalmic artery, a branch of the internal carotid, supplies the retina and optic nerve. There is a small communication between internal and external ophthalmic arteries. This communication is more likely to open when the external carotid artery is ligated. In the case of the vertebral artery, flow to small muscular branches in the neck would account for a small percentage of the recorded flow.
Implanted ultrasonic Doppler flowmeters are commonly used in general cardiovascular physiology to provide continuous assessment of various regional blood flows in both anesthetized and unanesthetized animals (Vatner et al., 1970; White et al., 1974; Haywood et al., 1981; Koerfer et al., 1993). Their applicability to cerebral arterial flows has been affirmed by simultaneous comparison of Doppler ultrasonic and electromagnetic flowmetry to measure common carotid flow in anesthetized newborn pigs with ligated external carotid arteries, with correlations >0.9 between the electromagnetic signal and Doppler signals recorded from probes positioned against major intracerebral arteries (Haaland et al., 1994). The materials from which the implanted Doppler probes are constructed are stable in vivo and the measurement techniques are relatively noninvasive. Therefore, with chronically implanted probes, it is reasonable to expect that blood velocity measurements could be made in the same animal on repeated occasions, as has already been documented for other arterial beds (Vatner et al., 1970). We have confirmed this for the cerebral arteries, documenting generally similar internal carotid responses to similar increases in inspired percentage of CO2 when the study was repeated at intervals of 1 week to 3 months.
Transcranial and transcutaneous ultrasonic Doppler techniques for measuring CBF are routinely used in clinical settings to assess flow in extra- and intracranial vessels in humans (Schöning et al., 1994; Bladin and Burns 1994). However, these procedures are not particularly suited to studies in the smaller vessels of laboratory animals or to mobile unanesthetized animals. Changes in probe crystal orientation and vessel diameter during an experiment may confound results when external probes are used (Hansen et al., 1983; Sonesson and Herin, 1988). Chronically implanted probes, as used in our study, are preferable for studies in experimental animals. Laser Doppler flowmetry can also provide continuous measurement of CBF, and the technique can be used in conscious animals (Sato et al., 1994). However, a single probe records from only a very small sample of brain tissue. In the future, valuable information may derive from combined use of an ultrasonic Doppler probe on an internal carotid or vertebral artery, and a Laser Doppler probe in a particular cerebral region.
Our study thus demonstrates that chronically implanted Doppler probes can provide continuous measurement of internal carotid and vertebral arterial flow in rabbits, thereby providing beat-to-beat indices of CBF. The method is quite suitable for studies in conscious animals, with repeated measures available in the same rabbit under different experimental conditions. Such indices of CBF should be especially useful for measuring rapid serial changes in flow in conscious animals. Our preliminary studies (Blessing and Yu, 1995) indicate that forebrain blood flow increases within seconds, when the rabbit detects a novel or potentially dangerous environmental event.
Footnotes
Acknowledgment:
The work was supported by the National Health and Medical Research Council, the National Heart Foundation of Australia, and the National SIDS Council of Australia. Dr. B-S Zhu holds an Australian Postdoctoral Fellowship from the National Health and Medical Research Council of Australia. We thank Dr. M. Sage, Department of Diagnostic Imaging at Flinders Medical Centre, for providing angiographic facilities and Dr. Alan Wilson for helping us obtain angiograms.
