Abstract
The effects of the centrally acting anticholinesterase metrifonate (MFT) and its metabolite dichlorvos (2,2-dichlorovinyl dimethyl phosphate; DDVP) on local cerebral glucose utilization (LCGU) have been studied in 3- and 27-month-old rats, using the autoradiographic [14C]deoxyglucose technique. In 3-month-old rats, MFT (80 mg/kg i.p.) increased LCGU significantly in 17 of the 54 regions studied, including insular, cingulate, and temporal cortices, ventral hippocampus, thalamus, lateral habenula, substantia nigra, and superior colliculus. In these regions, the average MFT-induced increase in LCGU was 23% above control. The average hemispheric LCGU increased by 10% (p < 0.01). DDVP (5 mg/kg) increased LCGU in 19 regions (average increase 26%). The average hemispheric LCGU increased by 9% (p < 0.01). Regional distributions of MFT- and DDVP-induced increases in LCGU were similar and overlapped the distribution of the acetylcholinesterase activity. In 27-month-old rats, MFT was active in 18 regions (average increase 25%). The whole-brain mean LCGU increased by 10% (p < 0.01). MFT compensated for the age-related hypometabolism in some brain areas including insular, temporal, and retrosplenial cortices, substantia nigra, and superior colliculus. The effects of MFT on LCGU were preserved in old rats, at variance with other anticholinesterases (tacrine, physostigmine), which are less active in the aged rat brain.
One of the earliest and most characteristic changes in Alzheimer's disease (AD) is a decreased level of cholinergic markers in cortical areas and subcortical cholinergic nuclei (Whitehouse et al., 1982). This cholinergic deficit correlates with the degree of memory impairment of AD patients. A possible therapeutic approach in AD may be to compensate for the loss of central cholinergic neurons by potentiating the activity of the remaining intact neurons. This can be achieved by inhibitors of acetylcholinesterase (AChE), the hydrolytic enzyme of acetylcholine (ACh), making more ACh available to postsynaptic neurons. A number of studies report that Cholinesterase inhibitors such as physostigmine (PHY) and the recently approved agent tacrine (tetrahydroaminoacridine; THA) are moderately effective in some mildly demented AD patients (for review see Lamy, 1994). However, the therapeutic effects of PHY and THA are limited in terms of both number of responders and quality of improvement.
There is a need for other agents that ideally would be effective in a larger number of patients, would have a long duration of action, and would be devoid of adverse effects. The organophosphorus compound metrifonate (MFT) is a member of the so-called second generation of cholinergic AD therapeutics. Dichlorvos (2,2-dichlorovinyl dimethyl phosphate; DDVP), hydrolyzed nonenzymatically from MFT, is the active AChE-inhibiting principle. Based on results obtained in the rat, Hallack and Giacobini (1989) suggested that MFT might be more likely to produce therapeutic effects than other anticholinesterase compounds, because of its long duration of action, low rate of side effects, and capability to reach high levels of AChE inhibition (Nordgren et al., 1978; Hallack and Giacobini, 1987). An open trial study gave evidence that MFT improves cognitive performance in AD (Becker et al., 1990). Further studies are required to confirm the efficacy of MFT and to determine its mechanisms of action. One way of investigation is to assess the effect of MFT on cerebral metabolism. The therapeutic effect of MFT might involve an effect on local cerebral glucose utilization (LCGU). Indeed, LCGU decreases in a number of brain areas in AD patients (Benson et al., 1983; de Leon et al., 1983; Foster et al., 1984). There is evidence that THA increases LCGU in AD patients in brain areas such as parietal and temporal cortex, which show the most consistent alterations in AD (Nordberg et al., 1992; Nordberg, 1993). The aims of the present study were to analyze the effects of MFT and DDVP on LCGU in young rats and to compare these results with those obtained in aged rats where reductions in brain metabolism occur (Smith et al., 1980; Wree et al., 1991; Bassant et al., 1994) to determine whether MFT-induced changes in LCGU observed in young rats are also found in a situation of age-related hypometabolism.
MATERIALS AND METHODS
Animals
Male Sprague-Dawley rats were obtained from IFFA Credo (France). Forty-two rats aged 3 months (body wt 280 ± 21 g) were used. The rats were housed at 22°C under a 12-h light/dark cycle and had free access to water and food. Aged rats (27 months, 730 ± 120 g) were obtained at 24 months of age and housed in our animal facility for 3 months (three animals per cage). Approximately 40% survived to an age of 27 months. None of the sixteen aged rats used in the experiment had apparent tumor mass. Two had pituitary adenoma.
Chemicals
MFT is not by itself an AChE inhibitor. Nonenzymatic metabolism (dehydrochlorination) of the agent is required to form the active component dichlorvos, which is an irreversible Cholinesterase inhibitor. MFT and DDVP were obtained from Bayer AG-Tropon (Germany). PHY, hexamethonium bromide, and atropine methylbromide were obtained from Sigma (France). [14C]2-Deoxy-D-glucose ([14C]DG; specific activity 50–55 mCi/mmol) in ethanol was purchased from Amersham (France). The solution was evaporated under nitrogen, and [14C]DG was resuspended in isotonic saline.
Measurement of LCGU
LCGU was measured by the quantitative [14C]DG technique of Sokoloff et al. (1977). Following 16 h of food deprivation, rats were lightly anesthetized (1% halothane in oxygen) and catheters were inserted in a femoral vein and artery. The operative sites were infiltrated with 1% xylocaine and closed with wound clips. Rats were placed in a loose-fitting plaster body cast in which the hindlimbs were restrained, permitting free movements of the head and forelimbs. Body temperature was monitored via a rectal thermoprobe connected to a thermostatic device (Harvard, UK) that activated an external warming element when body temperature fell below 35.5°C. Mean arterial blood pressure and heart rate were assessed before and at fixed times after drug treatment by connecting the arterial catheter to a pressure transducer (Barovar; Alvar Electronic, France), which was attached to a paper chart recorder (Gould, U.S.A.). Baseline and subsequent measurements of plasma glucose concentration were made throughout the experiment (Glucose Analyzer II; Beckman, U.S.A.). Hematocrit was determined prior to the injection of [14C]DG. Animals were excluded from the study if baseline values were outside of the following limits: mean arterial pressure 90–130 mm Hg, temperature 35–38°C, plasma glucose concentration 100–200 mg/dl.
After a 4-h recovery period, [14C]DG 125 μCi/kg was administered by rapid intravenous bolus injection. Arterial blood samples (100 μl) were taken at 6, 18, 30 s and 1, 3, 5, 7.5, 15, 25, 35, and 45 min after [14C]DG injection. Samples were centrifuged immediately and plasma radioactivity (liquid scintillation counter 1209; LKB Wallac, U.S.A.) and glucose concentration were measured. Animals were killed 45 min after [14C]DG by an overdose of pentobarbital. Brains were removed rapidly and frozen in isopentane chilled with dry ice at −45°C, then stored at −70°C. Coronal brain sections (20 μm thick) were cut on a cryostat (Reichert-Jung, Cambridge Instrument, Germany) at −20°C. Sections were mounted on coverslips and dried immediately at 55°C on a hot plate. Autoradiographs were then prepared by exposing X-AR5 x-ray film (Kodak) to the mounted sections and [14C]methyl-methacrylate standards (Amersham) for 7 days.
Autoradiograms were analyzed by quantitative densitometry with a computerized image-processing device (Biocom, France). To identify brain structures and cortical layers, sections adjacent to those used for autoradiography were stained with cresyl violet. Stained sections and corresponding sections in the autoradiogram were superimposed and the delineation of the structures was made according to the atlas of Paxinos and Watson (1986). The neocortex was divided into superficial layers (layers I, II, and upper part of layer III), middle layers (lower part of layers III and IV), and deep layers (layers V and VI). Five separate optical density measurements for each structure were made in consecutive brain sections. For each rat and each brain region, LCGU value was calculated as the arithmetic mean of five independent measurements. As right-left differences were sometimes found, only measurements in the left hemisphere were considered. Tissue 14C concentrations were determined from the optical densities and a calibration curve obtained by a densitometric analysis of the autoradiograms of the calibrated standards. LCGU was then calculated from the local tissue 14C concentrations, the time course of the plasma [14C]DG and glucose concentration, and appropriate kinetic constants according to the operational equation published by Sokoloff et al. (1977).
Time course of drug effects
To inject the [14C]DG at the peak of the drug effects, a pilot experiment had been performed to determine the time course of the behavioral effects of MFT and DDVP. Young rats received an intraperitoneal injection of MFT (80 mg/kg; n = 5) or DDVP (5 mg/kg; n = 5). Control rats (n = 5) received an equal volume of saline. Central body temperature was assessed at fixed times (10 min) during 2 h. Behavioral signs (tremor, gnawing, teeth chattering) were quantified at fixed times by a single experimenter who rated each animal on a scale of 0 (no effect) to + + + (maximum effect). On the basis of these observations (data not shown) MFT and DDVP were respectively injected 30 and 5 min before [14C]DG. The same experiment was repeated in 27-month-old rats with MFT (80 mg/kg). As compared with 3-month-old rats, the time course and the amplitude of the behavioral effects were the same in aged rats. Therefore, the same schedule of drug administration was used in the aged rats.
Drug treatments
To block the peripheral effects of anticholinesterases, animals were pretreated with hexamethonium bromide (5 mg/kg i.p.) and atropine methylbromide (4 mg/kg s.c), respectively, 30 and 20 min before drug administration. Animals then received an intraperitoneal injection of isotonic saline (control group) or MFT (freshly prepared, pH 6.0) or DDVP. Under acidic conditions, the rate of decomposition of MFT into DDVP is very slow and can be considered as negligible. In 3-month-old rats, LCGU was assessed in six groups of seven rats: control group, MFT 50 or 80 mg/kg 30 min before [14C]DG, DDVP 5 mg/kg 5 min before [14C]DG; to assess early or late effects, MFT (80 mg/kg) was also administered 10 min or 2 h before [14C]DG. In 27-month-old rats, LCGU was assessed in two groups of eight rats: control and MFT 80 mg/kg 30 min before [14C]DG.
Statistical analysis
Physiological parameters were compared between age-matched control and treated rats by one-way analysis of variance (ANOVA), followed by the Scheffé method. Within each experimental group, physiological parameters at different times after [14C]DG were compared using paired-sample t test.
For each brain region, LCGU values were analyzed as follows: Three-month-old control rats and the five groups of MFT- or DDVP-treated rats were compared by one-way ANOVA followed by Dunnett's test for multiple comparisons. The 27-month-old control and treated rats were evaluated by ANOVA followed by the Scheffé method. Then LCGU values were subjected to two-way ANOVA (region, drugs). Values from 3- and 27-month-old treated rats were compared by one-way and two-way ANOVA (age, treatment). Differences were considered significant if p < 0.05.
RESULTS
Physiological parameters during [14C]DG experiment
As compared with age-matched control rats (Table 1), body temperature significantly decreased in MFT-treated rats. Plasma glucose concentration increased in 27-month-old MFT-treated rats. As compared with the baseline conditions (before [14C]DG), body temperature decreased and plasma glucose concentration increased in MFT-treated rats of both ages. However, physiological parameters in treated rats were within the required limits of Sokoloff's method.
Physiological variables in experimental groups
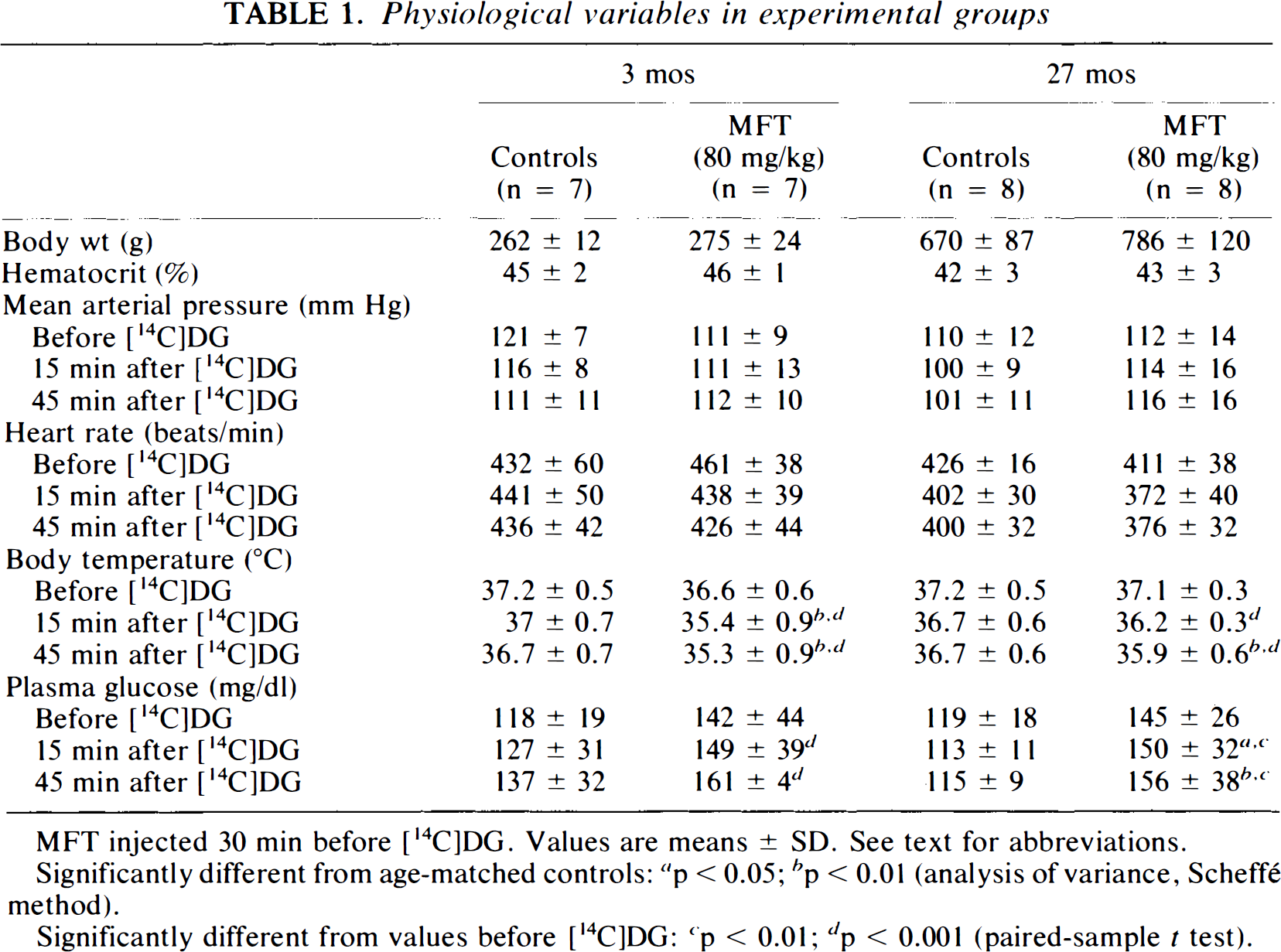
MFT injected 30 min before [14C]DG. Values are means ± SD. See text for abbreviations.
Significantly different from age-matched controls:
p < 0.05
p < 0.01 (analysis of variance, Scheffé method).
Significantly different from values before [14C]DG:
p < 0.01
p < 0.001 (paired-sample t test).
LCGU in 3-month-old rat
Effects of MFT 30 min. before [14C]DG
MFT (50 mg/kg) significantly increased LCGU in the insular and temporal cortex and in the thalamus, lateral habenula, and substantia nigra pars compacta (7 of the 54 regions studied) (Table 2). A higher dose of MFT (80 mg/kg) produced a larger metabolic activation. LCGU significantly increased in 17 of the 54 regions studied (Tables 2–5). In these regions, LCGU increased by 23% as compared with controls. In addition to the regions where changes were observed after the 50-mg/kg dose, increases in LCGU were found in the superior colliculus (50%), the cingulate (19%) and retrosplenial cortex (16%), the preoptic area (21%), the ventral hippocampus CA3 (18%), the horizontal limb of the diagonal band of Broca (13%), the basolateral amygdala (15%), the interpeduncular nucleus (16%), and the interpositus nucleus (20%) (Figs. 1 and 2).
LCGU in 3-mo-old rats following MFT and DDVP treatment
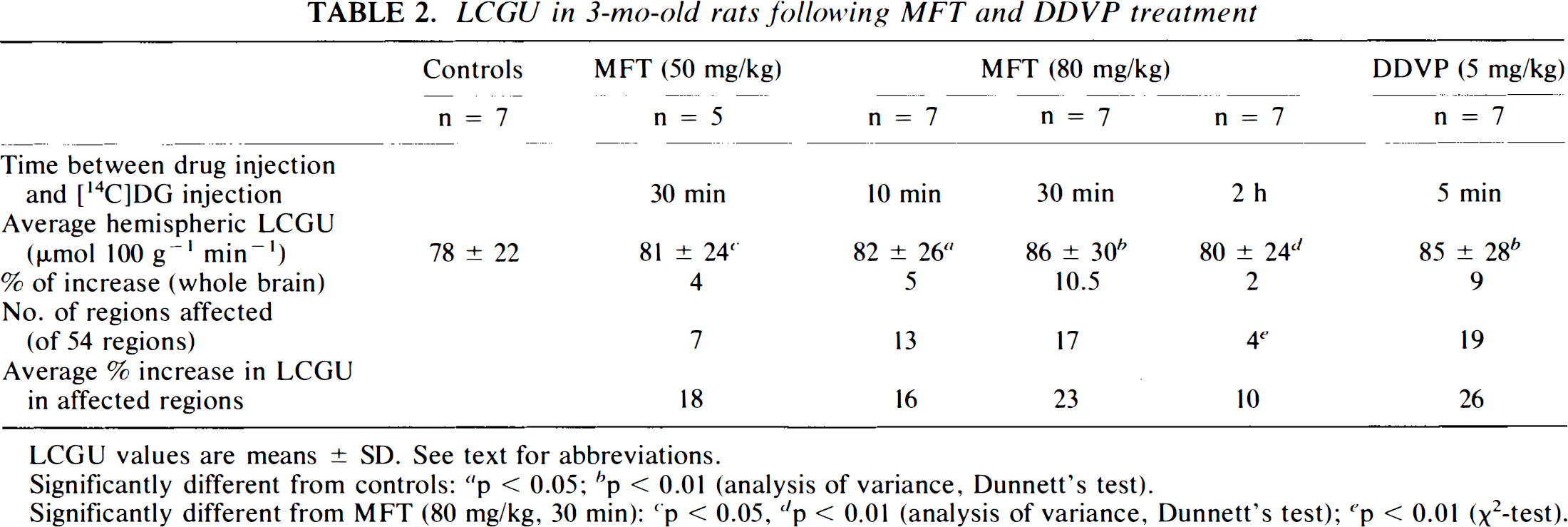
LCGU values are means ± SD. See text for abbreviations.
Significantly different from controls:
p < 0.05
p < 0.01 (analysis of variance, Dunnett's test).
Significantly different from MFT (80 mg/kg, 30 min):
p < 0.05
p < 0.01 (analysis of variance, Dunnett's test)
p < 0.01 (χ2-test).
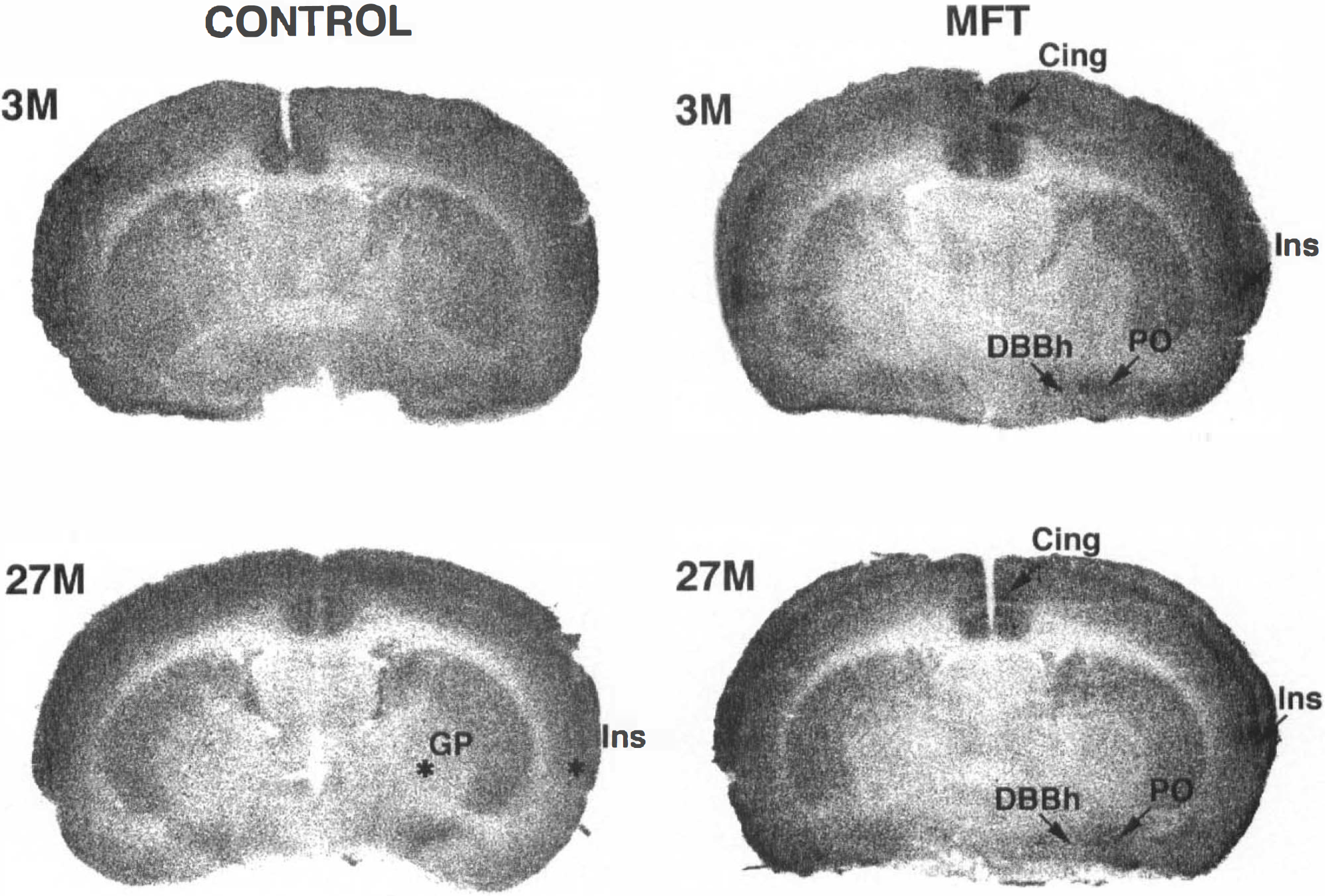
Autoradiograms from control and metrifonate (MFT)-treated rats (80 mg/kg, 30 min).
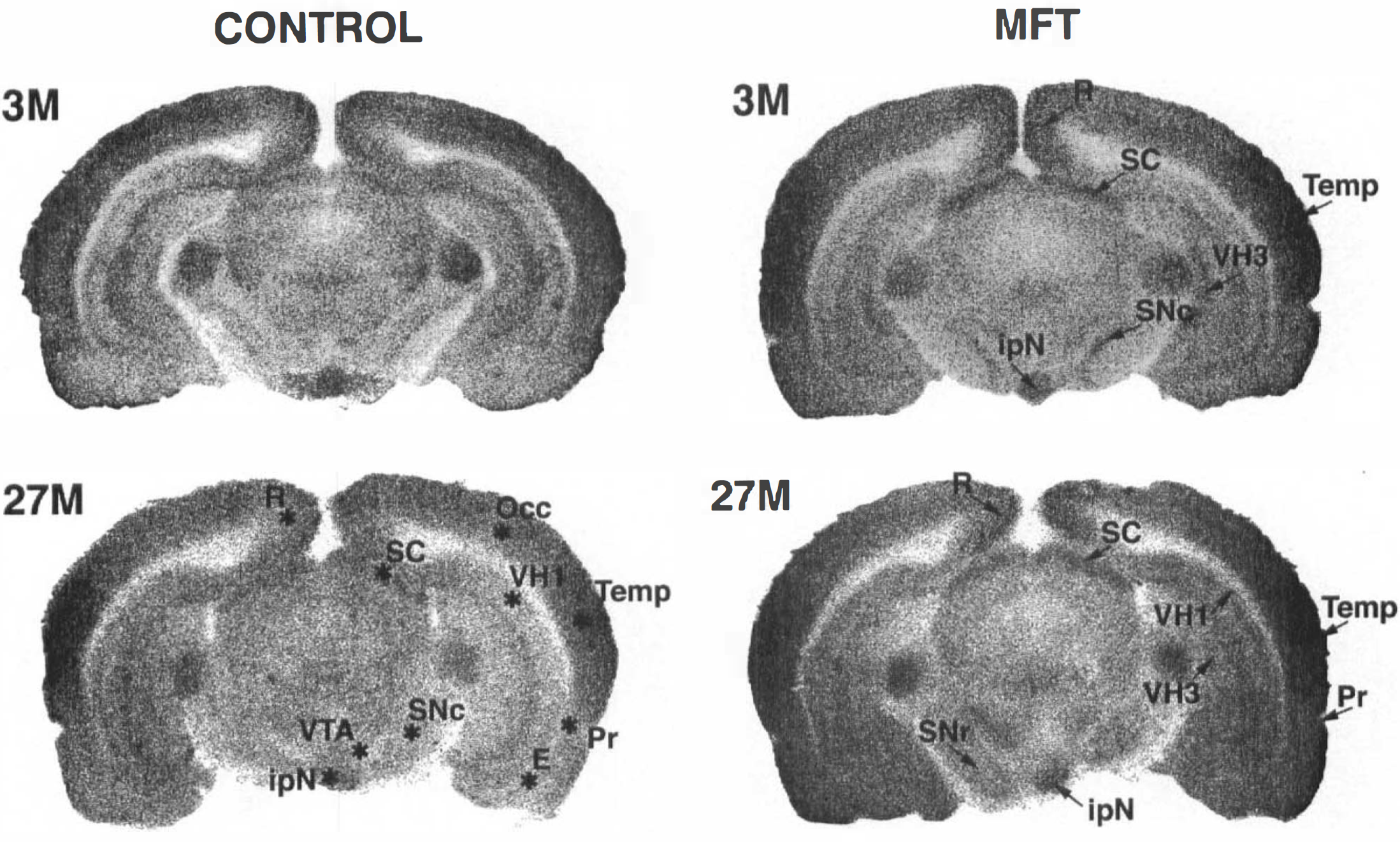
Autoradiograms from control and metrifonate (MFT)-treated rats (80 mg/kg, 30 min). Brain sections more caudal than in Fig. 1.
The average hemispheric LCGU (unweighted arithmetic mean of LCGU in all the regions studied) increased by 4% (n.s.) after 50 mg/kg and by 10.5% (p < 0.01) after 80 mg/kg.
MFT 10 min and 2 h before [14C]DG
Looking for possible early or late effects of MFT on LCGU, we also administered the drug (80 mg/kg) 10 min or 2 h before [14C]DG. In the first condition, the average hemispheric LCGU increased by 5% (p < 0.05). The regions where LCGU was altered after the shortest delay (13 of 54) were among the 17 regions affected by MFT 30 min before [14C]DG. However, the average increase in LCGU in the affected regions was smaller with the 10-min schedule (16%) as compared with the 30-min schedule (23%) (n.s., ANOVA) (Table 2). In case of the long delay (2 h) between drug administration and [14C]DG, MFT retained activity in the insular, temporal, and retrosplenial cortex and in the preoptic area (average increase of 10%). The average hemispheric LCGU was not different from controls (Table 2).
Effect of DDVP
It has been established in vitro that MFT is not an inhibitor of AChE. Inhibition of the enzyme is due to DDVP, the compound formed nonenzymatically from MFT (Reiner et al., 1975). However, some authors consider that MFT itself has anticholinesterase properties (Bueding et al., 1972). The effects of DDVP on LCGU were measured and compared with those of MFT to determine whether the effects of MFT on LCGU were similar to or different from those of its metabolite. DDVP (5 mg/kg) was administered 5 min before [14C]DG because of its short duration of action. Increases in LCGU were observed in 19 regions (average increase of 26%) (Table 2). The average hemispheric LCGU increased by 9% (p < 0.01). Large metabolic activations were observed in the superior colliculus (69%), the ventral thalamus (40%), the interpeduncular nucleus (32%), and the basolateral amygdala (31%) (Fig. 3). With the exception of four regions that responded only to DDVP (piriform cortex, caudate putamen, vertical limb of the diagonal band of Broca, CA3 field of the dorsal hippocampus), DDVP- and MFT-induced increases in LCGU had a similar topography.
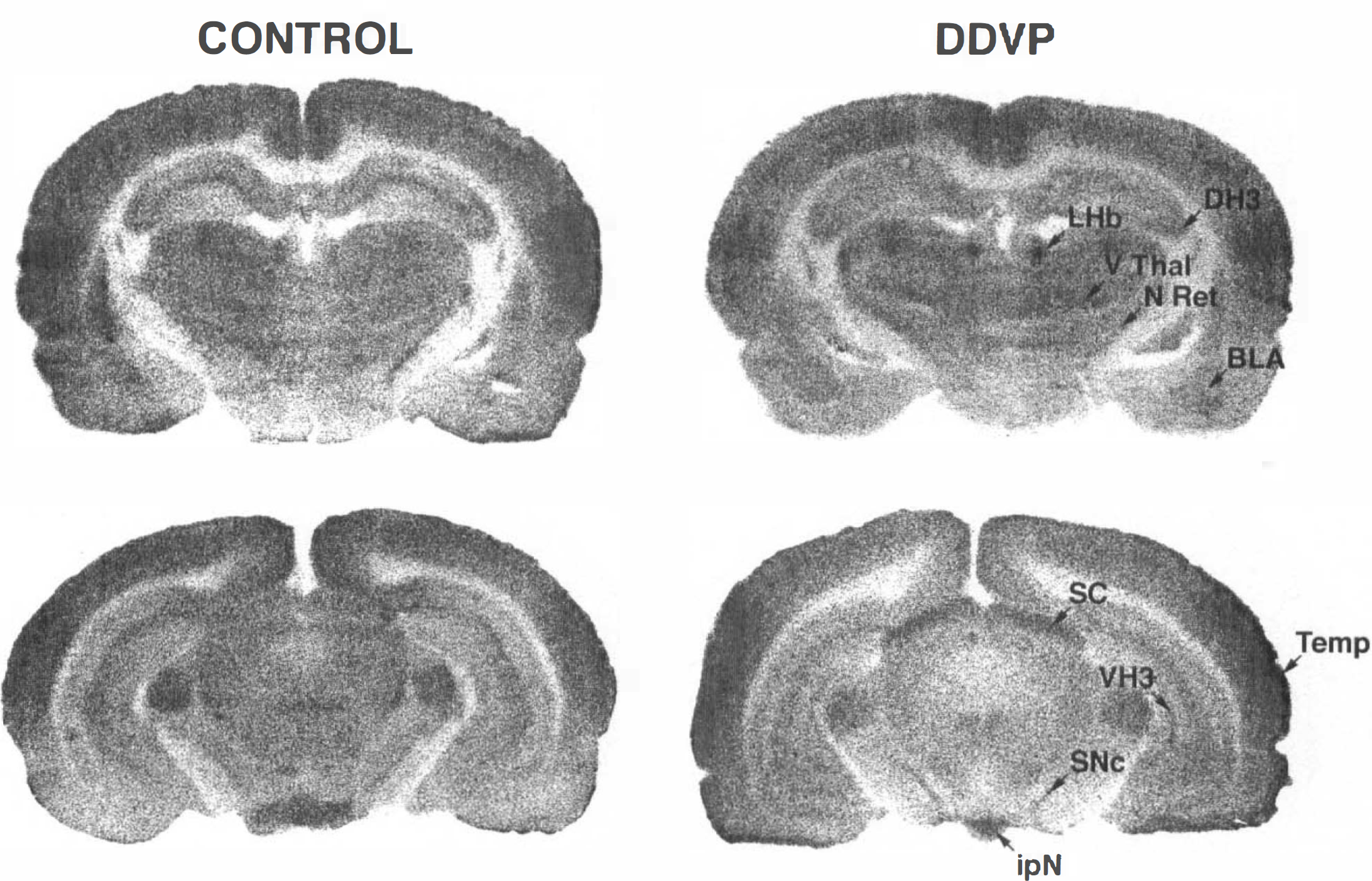
Autoradiograms from control and dichlorvos(DDVP)-treated young rats (5 mg/kg).
Comparison with other AChE inhibitors
The effects of PHY and THA have been previously studied under identical experimental conditions (Bassant et al., 1993). Results can be summarized as follows: After THA (10 mg/kg), average hemispheric LCGU increased by 15%; 23 of 54 (42%) brain regions were affected with an average increase of 28%. After PHY (0.5 mg/kg), average hemispheric LCGU increased by 13%; 23 brain regions were affected with an average increase of 29%. The effects of MFT (80 mg/kg) (Table 2), although somewhat smaller, were not significantly different (ANOVA, χ2-test).
LCGU in 27-month-old rats
LCGU in 27-month-old rats under basal conditions
As compared with 3-month-old rats, the average hemispheric LCGU was significantly reduced by 10% (p < 0.01) in 27-month-old rats (78 ± 22 and 70 ± 20 μmol 100 g−1 min−1 respectively). LCGU reduction in aged rats was not evenly distributed in the brain but restricted to specific regions (20 of the 54 regions studied) (Tables 3–5). In these regions, LCGU decreased by 10–24% in aged rats as compared with young controls (average decrease of 17%). Significant decreases were found in cortical areas (prefrontal, insular, retrosplenial, occipital, entorhinal, perirhinal, and temporal cortices). LCGU was unchanged in the limbic regions with the exception of the medial septum and the ventral hippocampal CA1 field. LCGU was also reduced in the nigrostriatal system (substantia nigra, globus pallidus, and caudate putamen), the ventral tegmental area, the superior colliculus. These results are in agreement with those previously published (Bassant et al., 1994).
LCGU (μmol 100 g−1 min−1) in cerebral cortical areas in control and MFT-treated rats aged 3 and 27 mos
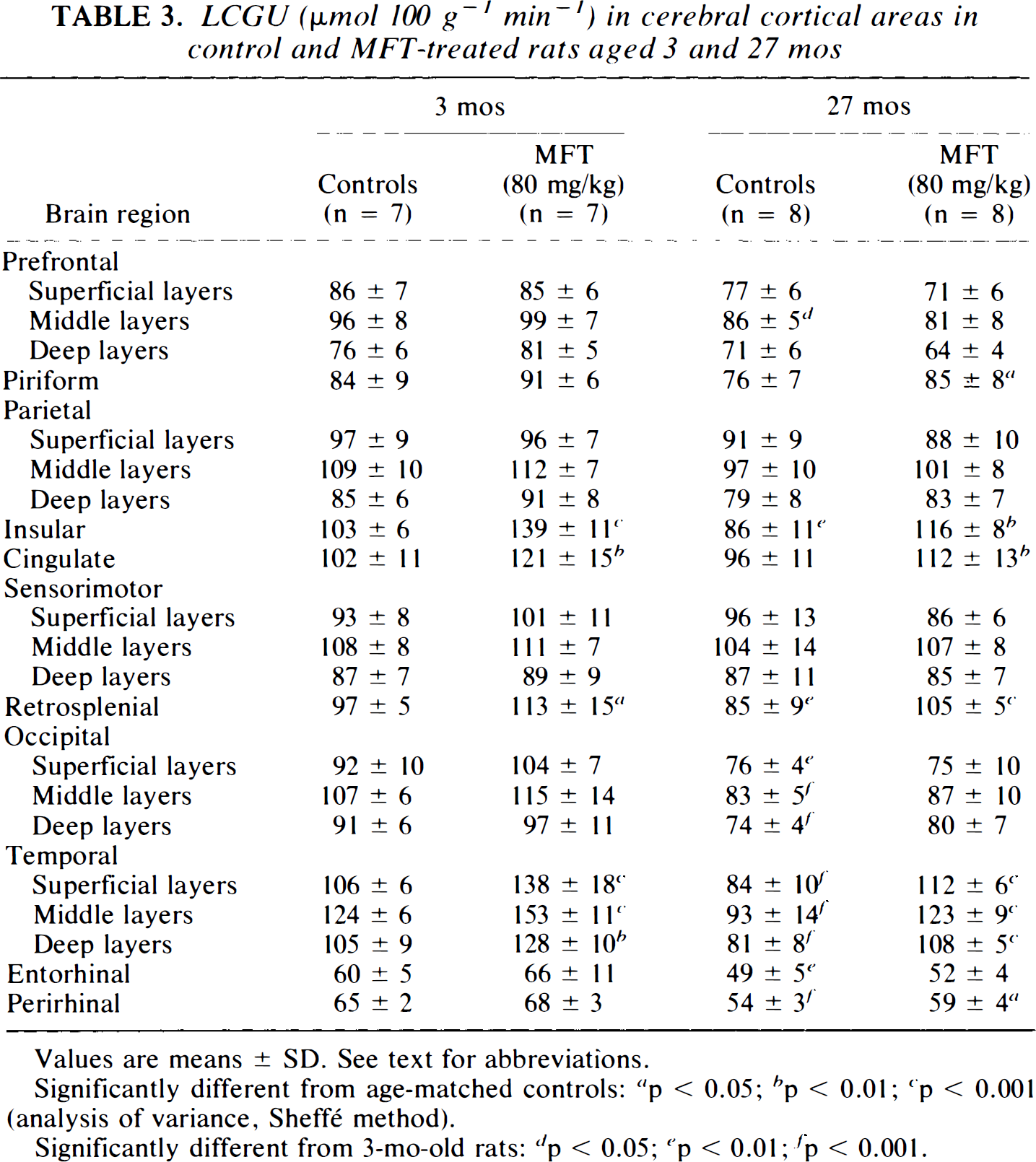
Values are means ± SD. See text for abbreviations.
Significantly different from age-matched controls:
p < 0.05
p < 0.01
p < 0.001 (analysis of variance, Sheffé method).
Significantly different from 3-mo-old rats:
p < 0.05
p < 0.01
p < 0.001.
LCGU (μmol 100 g−1 min−1) in limbic areas and white matter of control and MFT-treated rats aged 3 and 27 mos
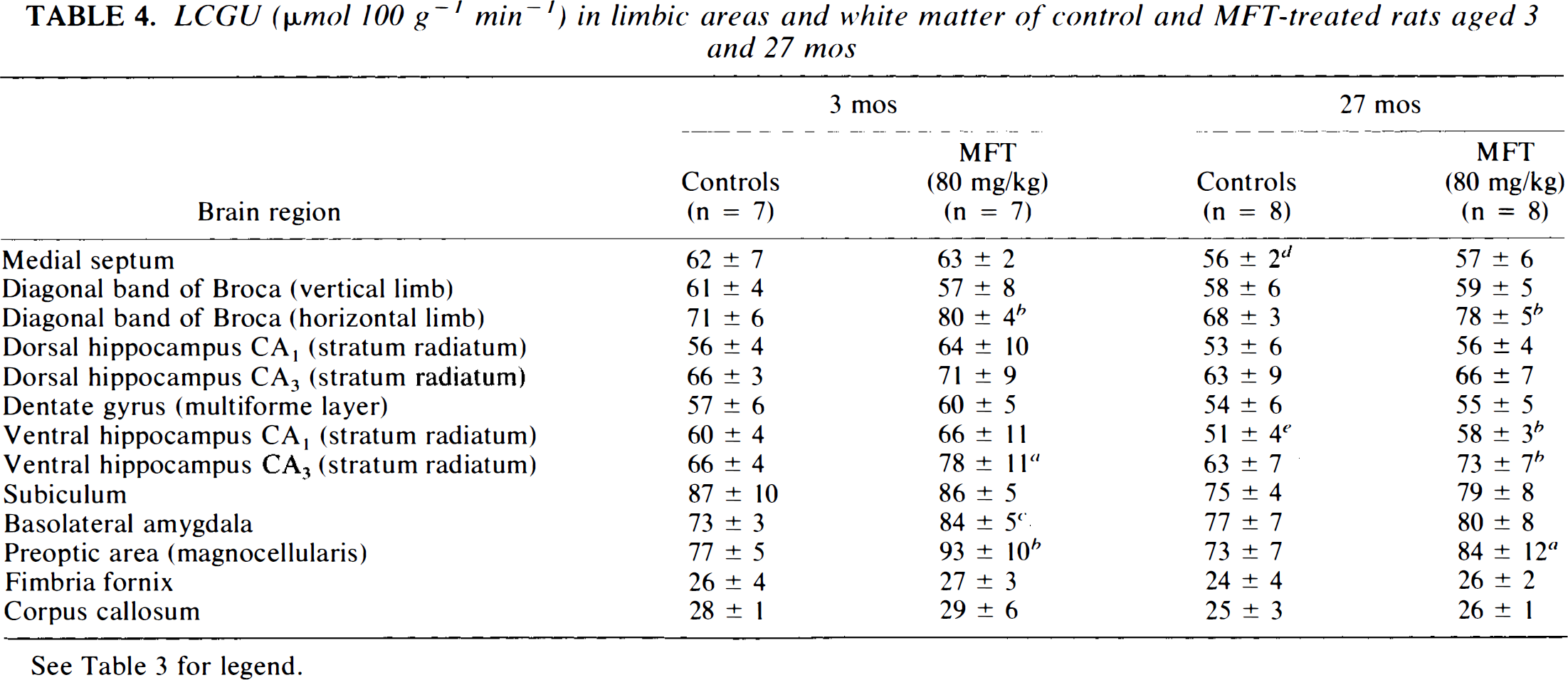
See Table 3 for legend.
LCGU (μmol 100 g−1 min−1) in basal ganglia, diencephalon, brainstem, and cerebellum of control and MFT-treated rats aged 3 and 27 mos
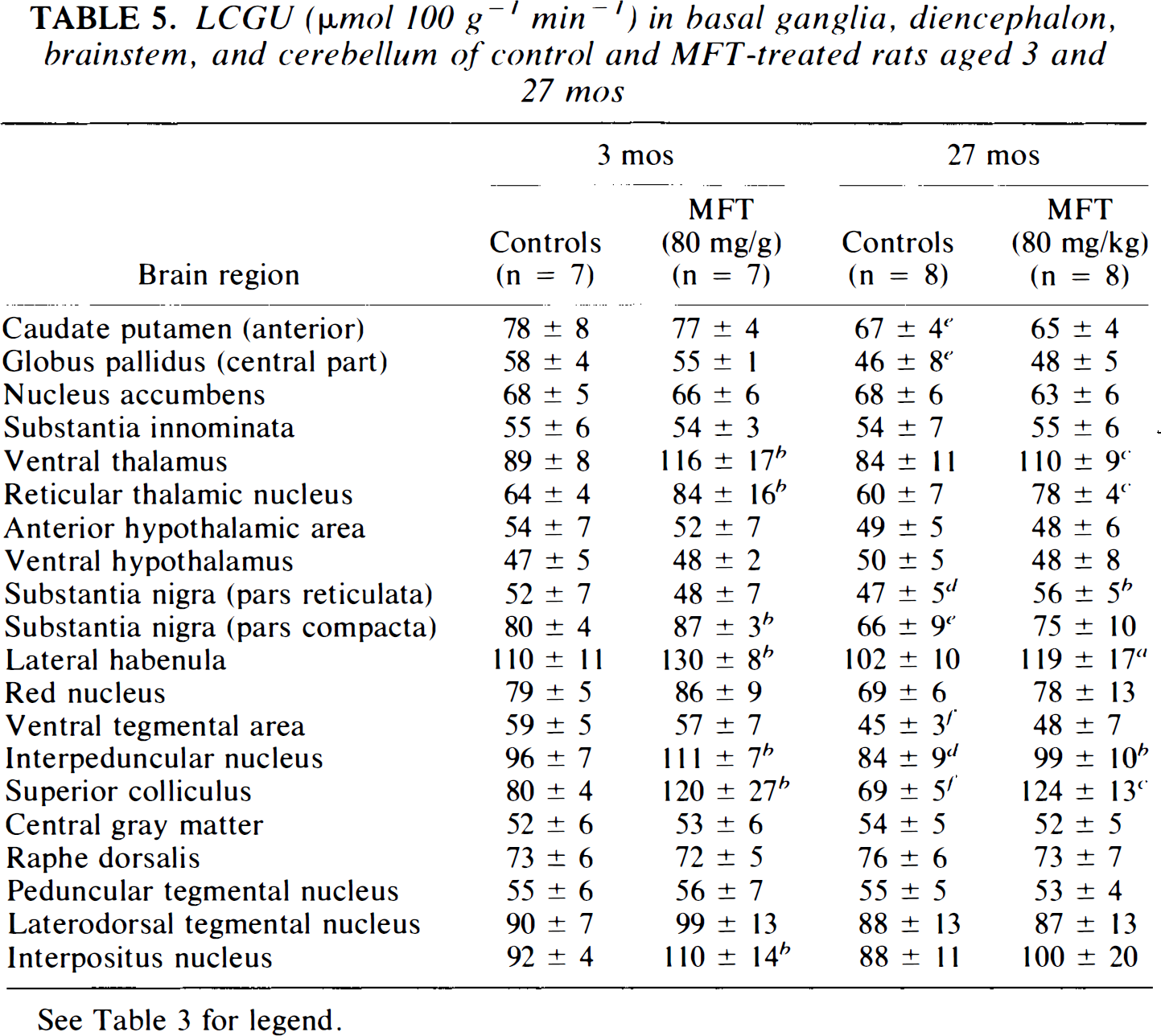
See Table 3 for legend.
Effects of MFT
MFT (80 mg/kg, administered 30 min before [14C]DG) increased average hemispheric LCGU by 10% (p < 0.01). Significant metabolic activation was observed in 18 regions (Tables 3–5). In these regions, average MFT-induced increase in LCGU was 25% with respect to the age-matched controls. In cortical areas, the largest effects were found in the insular (35%), temporal (33%), and retrosplenial (23%) cortices (Figs. 1 and 2). In the subcortical areas, pronounced effects were observed in the superior colliculus (80%), the ventral thalamus (31%), the lateral habenula (17%), and the ventral hippocampus (15%) (Fig. 2).
In the 20 regions where a significant age-related decrease in LCGU occurred (Tables 3–5), MFT was active in nine regions where it compensated for the age-related decrease in LCGU (temporal cortex) or exceeded the young control values (substantia nigra pars reticulata, insular and retrosplenial cortices, superior colliculus) (Fig. 4).
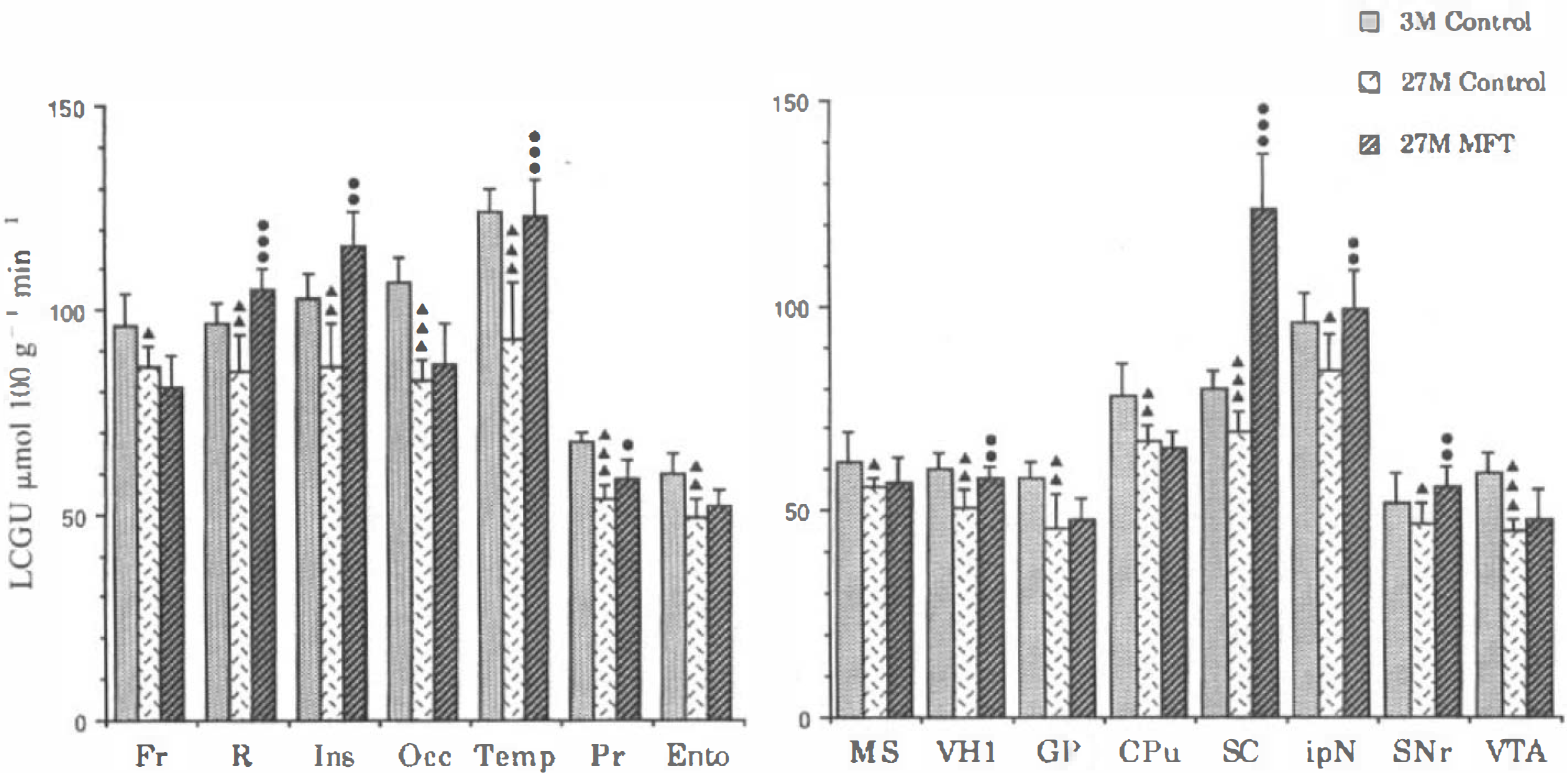
Effects of metrifonate (MFT) (80 mg/kg) in regions with age-related hypometabolism. Local cerebral glucose utilization (LCGU) values are means ± SD. Significant differences between 3- and 27-month-old control rats are indicated by triangles. Significant differences between 27-month-old control and treated rats are indicated by circles (see Table 3 for statistical analysis). MFT is still active in eight regions where it compensates for the hypometabolism (temporal cortex) or raises LCGU to the level of 3-month control values (insular and retrosplenial cortices, superior colliculus). CPu, caudate putamen; Ento, entorhinal cortex; Fr, frontal cortex; GP, globus pallidus; Ins, insular cortex; ipN, interpeduncular nucleus; MS, medial septum; Occ, occipital cortex; Pr, perirhinal cortex; R, retrosplenial cortex; SC, superior colliculus; SNr, substantia nigra; Temp, temporal cortex; VH1, ventral hippocampus (CA, field); VTA, ventral tegmental area.
Effects of MFT in 27-month vs. 3-month-old rats
Average hemispheric increase in LCGU (10.5 vs. 10%), number of affected regions (17 vs. 18), and average increase in LCGU in the affected regions (23 vs. 25%) were similar in young and aged MFT-treated rats. Two-way ANOVA did not show significant interaction between age and treatment. Effect of MFT was no longer observed in the basolateral amygdala, the substantia nigra (pars compacta), or the interpositus nucleus. On the other hand, significant increases were found in the piriform and perirhinal cortices, the ventral hippocampus (CA1 field), and the substantia nigra (pars reticulata).
Comparison with other AChE inhibitors
The effects of PHY and THA have previously been studied in the aged rat under identical experimental conditions (Bassant et al., 1995). Results can be summarized as follows: After THA, average hemispheric LCGU increased by 5% (n.s.), 13 of 54 brain regions were affected (average increase of 24%). After PHY, average hemispheric LCGU increased by 7% (p < 0.05); 10 brain regions were affected (average increase of 23%). As compared with those of 3-month-old rats, the effects of THA and PHY were significantly weaker in aged rats. MFT was somewhat more potent than the other two drugs, in terms of both magnitude of the increase in LCGU and number of regions involved (Fig. 5).
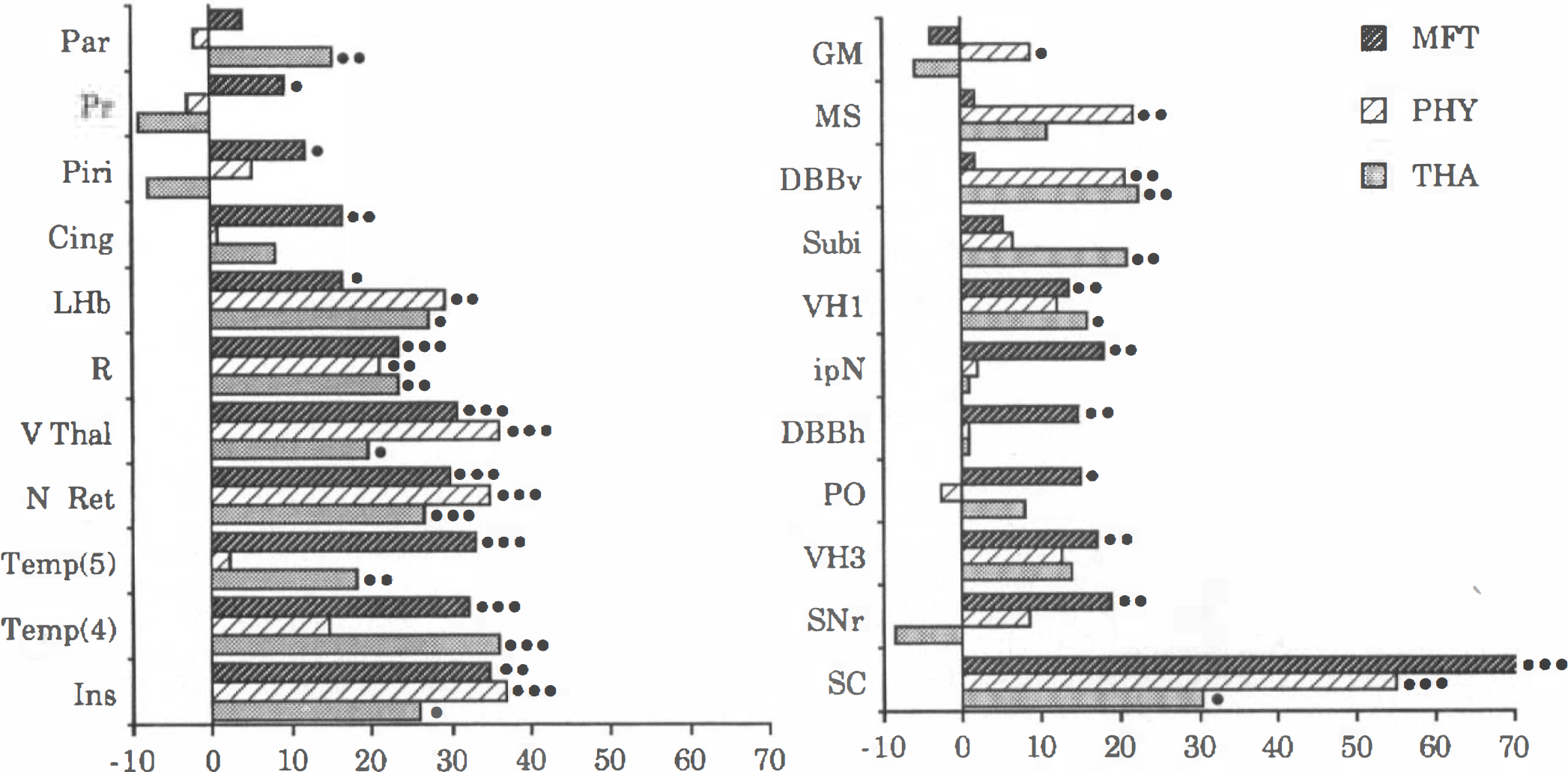
Changes in local cerebral glucose utilization (LCGU) produced by metrifonate (MFT), physostigmine (PHY), and tacrine (THA) in 27-month-old rats. Values are mean changes expressed as percents of age-matched control values. Regions are those where at least one of the drugs increased LCGU significantly (analysis of variance). See abbreviations in Figs. 1–4. GM, central gray matter; Par, parietal cortex; Subi, subiculum; DBBv, diagonal band of Broca (vertical limb); Piri, piriform cortex.
DISCUSSION
Effects of MFT on LCGU in 3-month-old rats
MFT increases LCGU in a sizable number of brain regions in the young rat, in a dose-dependent manner. DDVP, the active metabolite of MFT, which is an irreversible Cholinesterase inhibitor, has similar effects. This finding indicates that the MFT-induced metabolic activation is probably due to the anticholinesterase properties of DDVP. In addition, the effects of MFT have a regional topography similar to that observed with PHY, a reference Cholinesterase inhibitor (Bassant et al., 1993). As anticipated, a large majority of the regions where MFT increases LCGU (14 of 17) have a high AChE activity (Mash and Potter, 1986; Paxinos and Watson, 1986). The correlation, however, is not perfect because some regions with a high AChE activity, such as striatum and medial septal area, are not affected by MFT. LCGU increases in the interpositus nucleus, a structure that receives few cholinergic fibers (Goldsmith and Van Der Kooy, 1988) but is involved in the control of muscular activity. The effect observed in the interpositus might be due to the muscular hyperactivity (tremors) induced by MFT rather than to a specific effect on cholinergic transmission.
Results from in vivo and in vitro experiments indicate that glucose utilization, as measured with [14C]DG method, occurs more in the neuropil (region enriched in axon terminals, dendrites, synapses, and astrocytic processes) than in the neuronal perikarya (Magistretti et al., 1995). In case of functional activation, however, the increase of LCGU in the perikaryal layers is positively correlated with an increase of LCGU in their associated neuropil-rich layers (Wree et al., 1988). Several observations strongly suggest that astrocytes are the cells where glucose uptake occurs during activation. In astrocytes, glucose is metabolized into lactate, which is taken up by neurons and serves as substrate for oxidative phosphorylation (see ref. in Magistretti et al., 1995). MFT, by inhibiting AChE, causes ACh to accumulate at cholinergic receptor sites. As a consequence, several neuronal functions such as ion pumping, intracellular transports, re-uptake, etc., are increased. These functions have high metabolic requirements, as shown by high level of [14C]DG uptake in MFT-treated rats. In addition, the activity of astrocytes, which possess muscarinic and nicotinic receptors (Hösli and Hösli, 1988), might also be potentiated by the increased concentration of ACh and participate in the MFT-induced increase in LCGU.
MFT has been reported to improve cognitive performances in young and old rats (van der Staay et al., 1996) and in AD patients (Becker et al., 1990). To what extent an increase in LCGU is involved in the therapeutic effects of MFT remains to be established. It is interesting to note that the temporal cortex, which shows the most consistent LCGU alterations in AD (Foster et al., 1984), is among the regions that are the most “sensitive” to MFT in the rat. The metabolic activation observed in the septohippocampal system (diagonal band of Broca, ventral hippocampus) and in the cortex in the present experiment could result from an increased neuronal activity in regions involved in cognitive functions. Such an increase in LCGU might occur in AD patients receiving MFT and participate in the cognitive improvement.
The MFT-induced increase in LCGU is in the same range as that of other anticholinesterases studied under identical conditions (Bassant et al., 1993). MFT has no superiority to PHY and THA with respect to metabolic activation in the young rat. Other experimental studies, however, show that it offers several advantages such as long duration of action, maintenance of steady-state levels of ACh in the brain, absence of decrease in the synthesis rate of ACh, and reduced toxicity (Hallack and Giacobini, 1989; Giacobini, 1991).
To compare the effects of MFT with those of other anticholinesterases, the doses for each drug have been determined on the basis of equivalent behavioral effects. Under these conditions, the metabolic effects of MFT and PHY were quantitatively similar, but the dose of PHY was 160-fold smaller. This finding is consistent with the stronger anticholinesterase action of PHY (Becker and Giacobini, 1988; Hallack and Giacobini, 1989).
Effects of MFT on LCGU in 27-month-old rats
Moderate age-related reductions of brain metabolism have been observed in humans (Kuhl et al., 1984; Horwitz et al., 1986) and in rats (Smith et al., 1980; Wree et al., 1991; Bassant et al., 1994). Severe hypometabolism is a constant finding in the parietotemporal cortex of AD patients (Benson et al., 1983; de Leon et al., 1983; Foster et al., 1984). These alterations of LCGU might be due, at least in part, to deficits in neurotransmitter systems, including the cholinergic system. It was therefore important to determine if the effects of anticholinesterases on the brain metabolism observed in the young rat were still present in the aged rat, particularly in regions where an age-related hypometabolism occurs.
Our results show that MFT increases LCGU in several brain regions in the aged rat. As compared with young rats, the response of aged rats to MFT is similar, in terms of average hemispheric LCGU, number of regions affected, and amplitude of regional effect. It is interesting to note that MFT is still significantly active in regions with age-related hypometabolism. For example, the hypometabolism is completely compensated by MFT in the temporal cortex, the substantia nigra, and the interpositus nucleus. In the insular and cingulate cortex, the ventral hippocampus, the thalamus, and the superior colliculus, the balance between the effect of MFT and the age-related hypometabolism results in large increases in LCGU, which exceed the young control values.
Comparison with other anticholinesterases
In a previous study, THA and PHY were found significantly less active in aged than young rats, in terms of both number of regions affected and amplitude of metabolic activation (Bassant et al., 1995). What might be the reasons for this difference? AChE activity is not consistently affected by aging (for review, see Decker, 1987). It seems unlikely that the differences observed could be explained by a decrease in muscarinic receptor density or affinity or both because no consistent age-related changes have been reported in various strains including Sprague-Dawley rats (Strong et al., 1980; Michaleck et al., 1989; Schwartz et al., 1990). Defective coupling of postsynaptic mechanisms might alter muscarinic function in the aged brain. However, Surichamorn et al. (1989) have found that phosphatidylinositol hydrolysis, in response to activation of muscarinic receptors, does not change with aging in various brain areas. Decreases in the number or size of basal forebrain cholinergic neurons or both have been reported in the aged rat (Biegon et al., 1986; Fisher et al., 1989; de Bilbao et al., 1991). It is conceivable that a loss of cholinergic neurons, even moderate, and the resulting decrease of cholinergic input to the hippocampus and neocortex could alter the effect of THA and PHY by reducing the availability of ACh at the synapse. There is also evidence that the rate of ACh synthesis decreases with advancing age (see ref. in Mc Entee and Crook, 1992). On the other hand, studies on ACh release have shown that the depolarization-induced and the basal release of ACh are significantly decreased in synaptosomal preparations from aged rat brains (Araujo et al., 1990) and that basal ACh release is decreased in cortex and hippocampus in freely moving aged rats (Wu et al., 1988). It is therefore possible that the efficacy of AChE inhibitors may decline with age as a result of reduced ACh release. If this is true, then the effects of MFT should have also been reduced.
Are there arguments that could explain the better preservation of the effects of MFT in aged rats as compared with THA and PHY? The comparison of the effects of PHY, THA, and MFT on ACh metabolism in young rats has shown that the greatest peak of AChE inhibition was obtained 30 min after MFT administration as compared with moderate peaks at 15 and 60 min for PHY and THA; the duration of inhibition of AChE was four times longer after administration of equipotent doses of MFT than after PHY; and after a second dose, the increase in levels of ACh was more significantly prolonged as compared with the first treatment with MFT, similar with PHY, and lower with THA (Hallack and Giacobini, 1987, 1989). It is conceivable that such properties, producing a longer-lasting AChE inhibition, might partly explain the larger effect of MFT on LCGU in the aged brain, where the release of ACh is reduced.
Conclusions
In conclusion, MFT, like other AChE inhibitors, increases LCGU by activating cholinergic transmission. This effect probably plays a role in the cognitive improvement observed after treatment of AD patients with MFT. In addition, our results show that MFT is more effective than PHY and THA in the aged rat. This original finding, consistent with some properties of the compound, and the fact that MFT has more limited side effects suggest that this drug could be particularly suitable for the treatment of the cholinergic deficit in AD.
Footnotes
Acknowledgment:
This work was supported by a grant from Bayer Pharma, France. The authors thank R. Ram-bur for photographic assistance.
