Abstract
Occlusion or severe stenosis, with a reduction in the diameter of more than 70% of the extracranial arteries may lead to hypoperfusion of the brain with an increased risk of cerebral infarction. The aim of this study was to investigate whether endarterectomy of stenosed internal carotid arteries leads to alterations in cerebral metabolism in regions in which no infarcts were visible with magnetic resonance imaging (MRI). We studied 10 healthy control subjects and 20 patients with transient or nondisabling cerebral ischemia with MRI and 1H magnetic resonance spectroscopic imaging. All patients underwent carotid endarterectomy. Patients were examined 1 week before and 3–6 months after carotid endarterectomy. The N-acetyl aspartate (NAA)/choline ratio in the symptomatic hemisphere before endarterectomy (2.29 ± 0.42) was significantly (p < 0.001) lower than for control subjects (3.18 ± 0.32). In five of the patients lactate was detected preoperatively in regions that were not infarcted. The NAA/choline ratio in the symptomatic hemisphere of these five patients did not increase significantly after endarterectomy (1.99 ± 0.22 vs. 2.23 ± 0.48). The NAA/choline ratio in patients without lactate preoperatively increased significantly (p < 0.01) after endarterectomy to a normal level (from 2.39 ± 0.42 to 2.92 ± 0.52). These results indicate that the presence of cerebral lactate may predict whether the NAA/choline ratio increases after carotid endarterectomy.
The process of cerebral infarction is characterized by a reduced oxygen and glucose supply to the brain or to parts of the brain. In the acute and subacute phase of infarction this may result in an increased lactate (Lac) concentration and acidosis, caused by anaerobe glycolysis, and in a reduced N-acetyl aspartate (NAA) concentration, probably caused by neuronal damage (Bruhn et al., 1989; Gideon et al., 1992; Hugg et al., 1992; Houkin et al., 1993; Barker et al., 1994; Gideon et al., 1994). In chronic brain infarction acidosis has changed into alkalosis and all metabolite concentrations are reduced compared to those in the acute phase of infarction (Duijn et al., 1992; Felber et al., 1992; Ford et al., 1992; Graham et al., 1992; Hugg et al., 1992; Petroff et al., 1992; Houkin et al., 1993; Barker et al., 1994; Gideon et al., 1994). In patients with a severe stenosis or an occlusion of the internal carotid artery (ICA), flow adjustments in the circle of Willis, collateral blood flow by the ophthalmic artery or by leptomeningeal vessels via the external carotid artery, may compensate for the reduced blood flow through the stenosed or occluded ICA. Although collateral flow is often sufficient to keep the cerebral blood flow at a level at which brain function is not affected, in some cases it is not sufficient to accommodate the metabolic demands, causing cerebral infarcts. Regions surrounding these infarcts and regions with low flow, which are not infarcted yet, may contain cells that still receive sufficient oxygen and glucose to survive but insufficient to maintain function. Although reestablishment of the cerebral blood flow in these regions may improve the cellular function, these regions may be at risk for infarction after prolonged ischemia.
The goal of this study was to investigate whether in symptomatic patients a stenosis of >70% diameter reduction in the ICA leads to altered cerebral metabolism in regions that are likely to receive a low cerebral blood flow but that are not infarcted. Furthermore, we investigated whether in these regions the altered cerebral metabolism, e.g., increased Lac and decreased NAA, returns to a normal level after reestablishment of the blood flow through the stenosed ICA.
PATIENTS AND METHODS
Patients
Fourteen patients with unilateral severe stenosis (>70% reduction in the diameter) of the ICA (12 left side, 2 right side) and 6 patients with bilateral severe stenosis (>70% reduction) of the ICA were studied. Sixteen patients were male and four were female. The age range was 53 to 74 years (66.7 ± 12.0 years, mean ± SD). All patients were selected by the Departments of Neurology and Vascular Surgery as candidates for carotid endarterectomy because they had a transient ischemic attack (TIA) or a minor ischemic stroke in the territory of the ICA to be operated between October 1993 and September 1994. All patients were examined within 12 weeks after the onset of symptoms. All patients were investigated twice; the first magnetic resonance spectroscopic imaging (MRSI) was performed within 1 week before carotid endarterectomy and the second MRSI was performed 12–30 (mean, 22.4) weeks after carotid endarterectomy. Quantification of the carotid artery stenosis was based on arterial digital subtraction angiography (aDSA). All patients were operated under general anesthesia with preoperative EEG and transcranial Doppler (TCD) surveillance. All operations were uncomplicated and there were no ischemic events between the first and the second MRSI examination. The control group consisted of 10 subjects (8 male, 2 female). The age range was 29 to 69 years (49.7 ± 13.9 years). None of the control subjects had ever suffered a cerebral event and magnetic resonance imaging (MRI) of the brain was normal in all of them (no white or gray matter signal abnormalities on T1- and T2-weighted MRI). Study protocols were approved by the Human Research Committee.
MRI and MRSI
The MRI and MRSI studies were performed on a Philips Gyroscan S15 whole-body system operating at 1.5 T. Proton MRIs were obtained: 7 sagittal T1-weighted scout slices [slice thickness, 5 mm; slice gap, 1 mm; repetition time (TR), 450 ms; echo time (TE), 30 ms], and 14 transaxial T2-weighted slices (slice thickness, 7 mm; slice gap, 1.6 mm; TR, 2,000 ms; TE, 50 and 100 ms).
After MRI, the volume of interest (VOI) for 1H MRSI was chosen from the transaxial images. For each subject a 15-mm-thick transverse slice was selected through the centrum semiovale. The anterior–posterior and left–right dimensions of the VOI were chosen such that regions containing subcutaneous lipid were excluded and were typically 110 and 90 mm in the anterior–posterior and left–right directions, respectively. For MRSI, first the 90° pulse length was determined. To minimize eddy currents and to maximize the water echo signal, localized spectroscopy was first performed without water suppression for adjustments of the gradients (“gradient tuning”). This was followed by localized shimming of the VOI, resulting typically in a water resonance linewidth of ≤6 Hz (full width at half-height). Water suppression was performed with selective inversion of the water resonance with an adiabatic inversion pulse, followed by a double spin-echo PRESS sequence (Ordidge et al., 1985; Bottomly, 1986) for VOI localization at the zero crossing of water (Luyten et al., 1990). Gradient phase encoding was applied in two dimensions: 16 × 16 phase encoding steps were used over a field of view of 200 × 200 mm, resulting in an in-plane spatial resolution of 12.5 mm and a nominal voxel size of 2.34 ml. For each phase encoding step two averages with a TR of 2,000 ms, a TE of 272 ms, 512 time-domain datum points, and a spectral width of 1,000 Hz were used. The total procedure including patient preparation, MRI, and MRSI lasted ∼40 min.
After acquisition, MRSI data were transferred to a SUN IPX workstation for further processing. The free induction decays were zero-filled to 1,024 datum points. In the time domain Gaussian multiplication of 5 Hz was used, followed by a −4-Hz (line-broadening) exponential multiplication. After Fourier transformation in the spectral and spatial dimensions, two-dimensional MRSIs were created. Individual metabolite images were generated by spectral integration over individual resonances. Metabolite maps were interpolated to a 128 × 128 matrix and were scaled individually. The display software (sunspecl; Philips Medical Systems, Best, The Netherlands) provided simultaneous display and spectral registration of the MRI and metabolic images. 1H MR spectra were selected by mouse control of a cursor on T2-weighted MRIs from regions without gray/white matter hyperintensities on MRI and away from borders of the 1H PRESS VOI to avoid lipid contamination and chemical shift artifacts. Total choline (Cho) resonances, total creatine (Cr) resonances, total N-acetyl resonances (containing mainly NAA, referenced at 2.01 ppm), and Lac resonances were identified by their chemical shifts (Frahm et al., 1989). Individual peaks were quantified by peak height measurements. Since we were unable to calculate absolute metabolic concentrations, concentrations are expressed as the ratio between peak intensities (NAA/Cho and NAA/Cr ratios). To distinguish Lac resonances from lipid resonances, Lac was defined as a resonance at 1.33 ppm with a signal-to-noise ratio >2 having a clear identifiable 7-Hz J-coupling. Lac was quantified as present or absent. In each individual three spectra were chosen in the centrum semiovale in the symptomatic hemisphere and three spectra in homologous regions in the contralateral hemisphere. In each hemisphere, one spectrum was selected in the frontal region, one in the mesial region, and one in the parietal region. All selected regions contained primarily white matter. All spectra were selected ≥2.5 cm removed from gray/white matter hyperintensities and ≥2.5 cm from each other to avoid signal cross contamination. If infarcts were present in MRI slices outside the relatively thick MRSI slice, care was taken to maintain the 2.5-cm distance between lesions and selected volumes.
Statistical analysis
For statistical analysis repeated-measures analysis of variances (ANOVA) was used to compare metabolite ratios by groups (hemispheres) and location within the groups. The groups (n = 5) were defined as normal controls and the symptomatic and asymptomatic hemispheres of patients before and after carotid endarterectomy. Locations in the normal control group (n = 6) were defined as frontal, mesial, and parietal regions in both hemispheres in the centrum semiovale. In the patient group, locations (n = 3) were defined as frontal, mesial, and parietal regions in one (symptomatic or asymptomatic) hemisphere. Paired analysis of differences in metabolite ratios was performed using the Bonferroni t method (Dunn's multiple-comparison procedure). All data are expressed as mean ± SD; p < 0.05 was considered statistically significant.
RESULTS
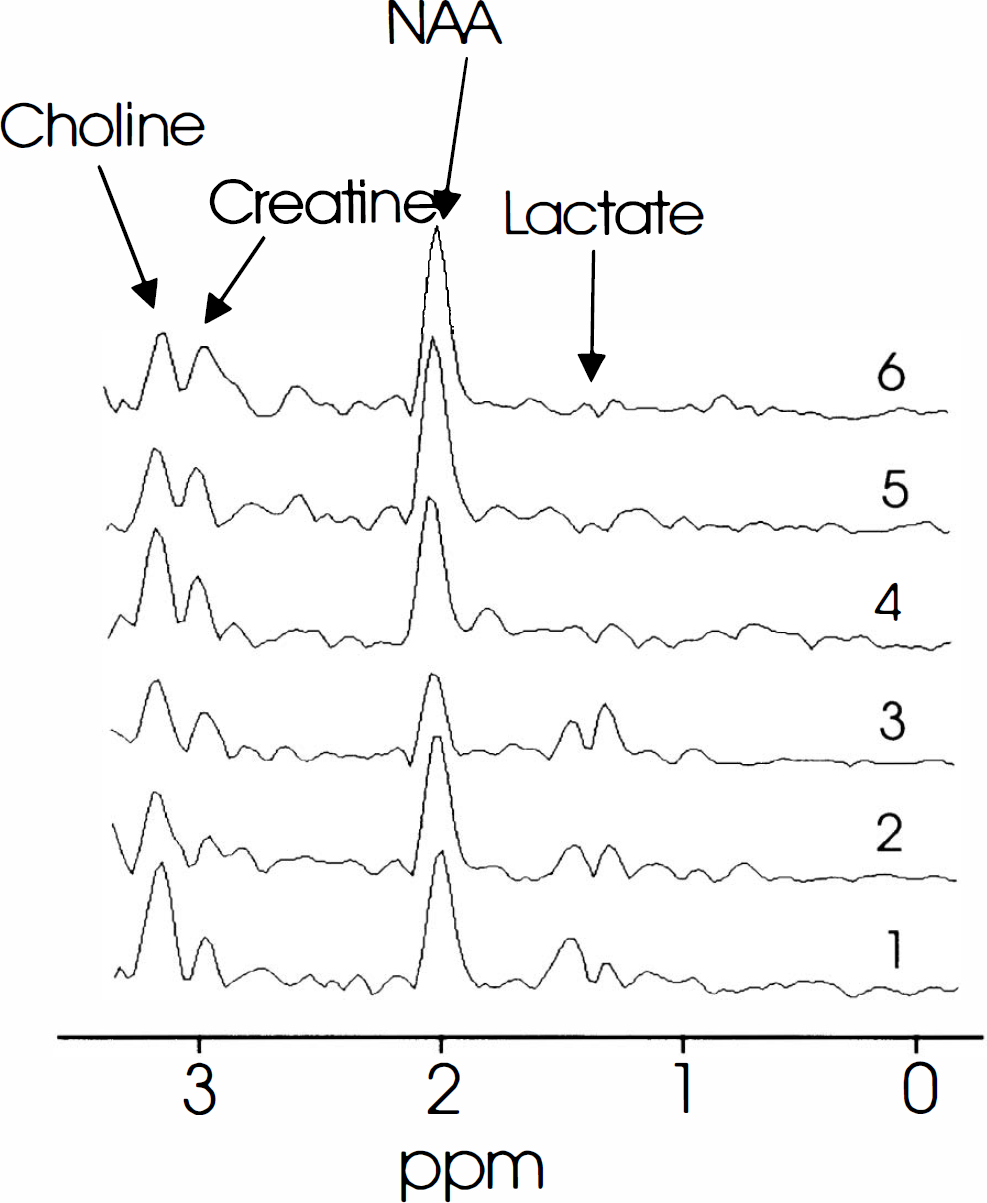
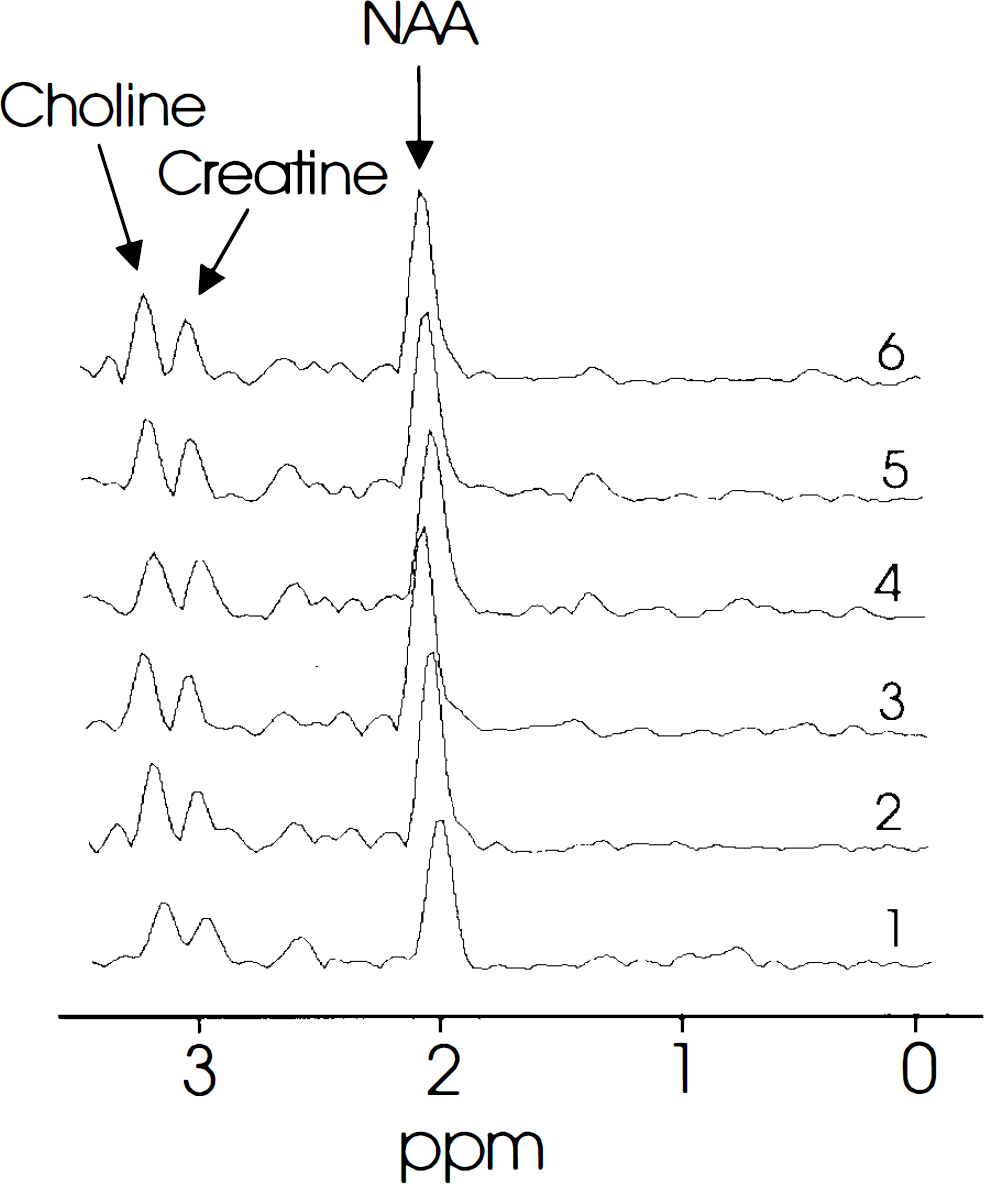
Figure 1 shows a MRSI datum set of a patient with a severe stenosis (>70% reduction in the diameter) of the left ICA. Figure 2 shows six 1H MR spectra from voxels that were selected frcm the same patient shown in Fig. 1. These figures show that Lac is present in all regions in the left hemisphere. Figure 3 shows a MRSI datum set of the same patient 3 months after carotid endarterectomy. Figure 4 shows six 1H MR spectra from voxels that were selected from the same patient 3 months after carotid endarterectomy. Figures 3 and 4 show that after carotid endarterectomy cerebral Lac is no longer present in this patient. The high signal intensities outside the VOI in the Lac image are caused by strong lipid resonances. Before and after endarterectomy no cerebral infarcts were seen on MRI in this patient.
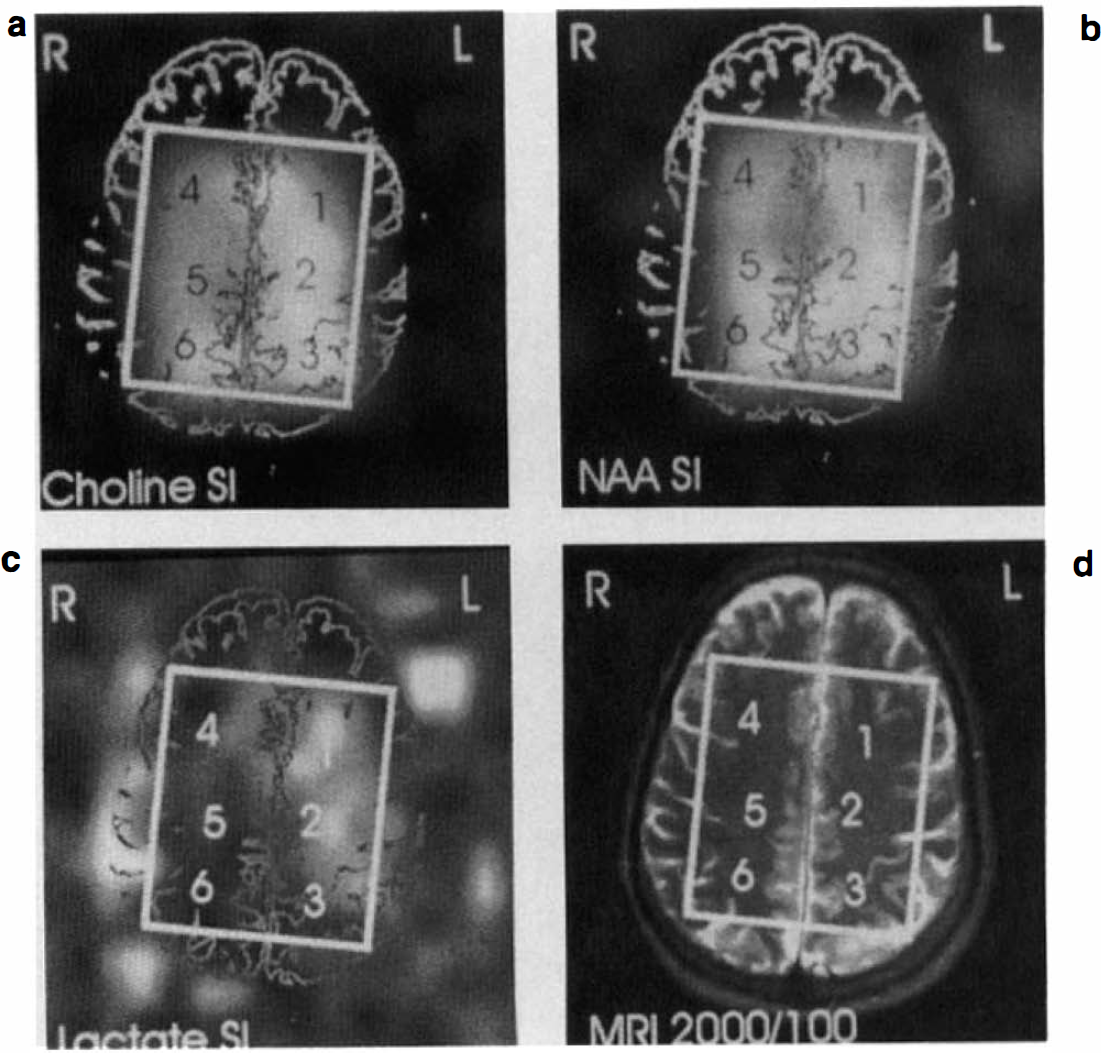
MRSI study of a 74-year-old man with severe stenosis (>70% reduction) of the left ICA.
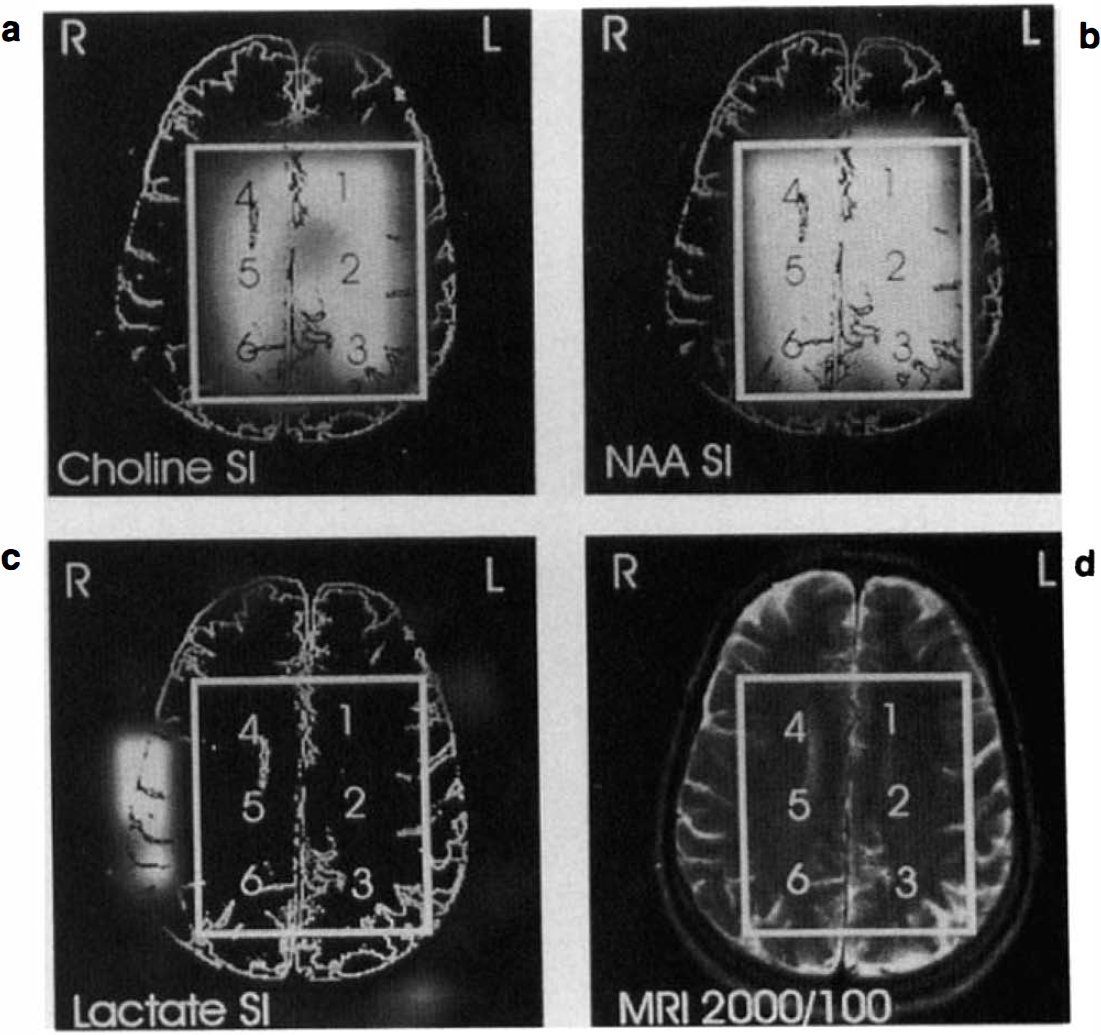
MRSI study of the same patients as shown in Fig. 1 but after carotid endarterectomy.
We did not find differences in the NAA/Cho or NAA/Cr ratio or in the absolute peak heights of Cho, Cr, or NAA among the frontal, mesial, or parietal regions within one hemisphere in the control subjects or in the patients groups. For this reason, metabolite ratios are expressed as the mean of the frontal, mesial, and parietal metabolite ratios. In the control group no left-to-right differences were found between the two hemispheres; therefore, the metabolite ratios of the control subjects are expressed as the mean of all six regions selected. Table 1 summarizes all metabolite ratios. The NAA/Cho ratio in the symptomatic hemisphere of patients with carotid lesions increases significantly after endarterectomy. No significant differences were found between the NAA/Cho ratio of the control subjects and the asymptomatic hemisphere of the patient group before and after carotid endarterectomy. No significant differences were found between groups for the NAA/Cr ratio. There was no correlation found between spectral alterations and age in the subject groups. There was also no correlation found between spectral alterations and severity of carotid lesions. In 9 of the 20 patients cerebral infarctions were seen on MRI. There was no correlation between the presence of cerebral infarcts and decreased metabolic ratios or the presence of Lac.
Metabolite ratios for all patients (n = 20) before and after carotid endarterectomy and for control subjects (n = 10) (mean ± SD)
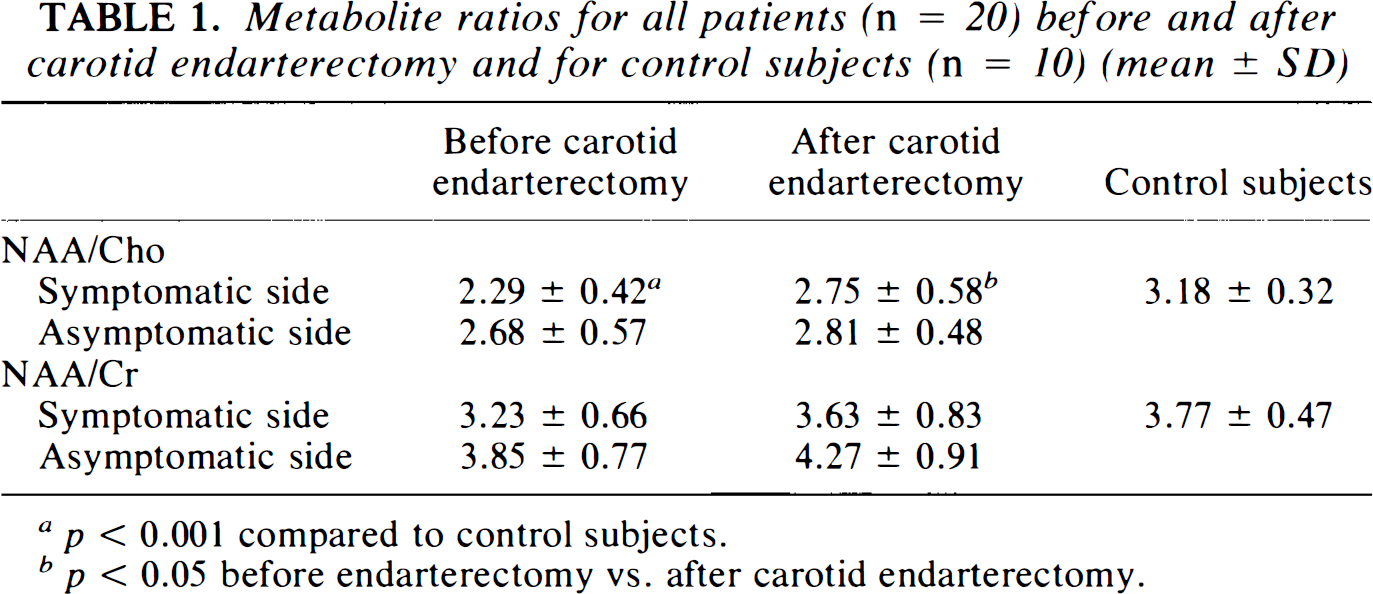
p < 0.001 compared to control subjects.
p < 0.05 before endarterectomy vs. after carotid endarterectomy.
Lac
Table 2 shows the NAA/Cho ratio of patients divided into groups with and without Lac preoperatively in the symptomatic hemisphere. The NAA/Cho ratio was significantly increased (p < 0.01) after carotid surgery in the group without Lac. In the group with Lac, the NAA/Cho ratio was not significantly increased after carotid endarterectomy. There was no significant difference in the NAA/Cho ratio between Lac and non-Lac groups pre- and postoperatively. Power analysis revealed that preoperatively, using a type I error of 0.05 and a probability of 0.90 of detecting a true difference, 23 patients in each group are needed to detect a significant difference in the NAA/Cho ratio. This implies that no significant difference was found because of the low number of patients investigated. Although there was no correlation between infarction and Lac levels, the number of patients with cerebral infarcts was significantly higher in the patient group with Lac compared to the group without Lac (p = 0.03, Fisher's exact test).
NAA/Cho ratios in the symptomatic hemisphere before and after carotid endarterectomy in patients with and patients without Lac before carotid endarterectomy (mean ± SD)

p < 0.001 compared to control subjects.
p < 0.01 before endarterectomy vs. after carotid endarterectomy.
In 14 of the 15 patients without Lac, the NAA/Cho ratio increased after endarterectomy, versus 2 of the 5 patients with Lac preoperatively (p = 0.015, Fisher's exact test). There was no relationship between the relative intensity of Lac (Lac/NAA ratio) and the behavior of the NAA/Cho ratio after endarterectomy. There was also no correlation between the presence of infarcts and the behavior of the NAA/Cho ratio after endarterectomy.
DISCUSSION
This study shows that patients with severe stenosis of the ICA who suffered TIA or minor permanent ischemic attack have decreased NAA/Cho ratios, in noninfarcted brain tissue in the symptomatic hemisphere. This ratio increased significantly after endarterectomy. However, in the subgroup of patients that showed cerebral Lac before surgery the NAA/Cho ratio did not increase.
A decrease in the NAA/Cho ratio and the presence of Lac have been reported before in patients with cerebral infarction (Bruhn et al., 1989; Duijn et al., 1992; Felber et al., 1992; Ford et al., 1992; Gideon et al., 1992; Graham et al., 1992; Hugg et al., 1992; Petroff et al., 1992; Houkin et al., 1993; Gideon et al., 1994). Brain infarction results in a marked decrease in cellular density: dead neurons and dead glial cells are removed by macrophages, while edema, astrocytes, and new glial cells partially fill the infarcted regions, resulting in a considerable decrease in NAA but also in a decrease in Cho (Bruhn et al., 1989; Duijn et al., 1992; Felber et al., 1992; Ford et al., 1992; Gideon et al., 1992; Graham et al., 1992; Hugg et al., 1992; Petroff et al., 1992; Houkin et al., 1993; Gideon et al., 1994). Although the NAA/Cho ratio is commonly used as a marker for the metabolic function of the brain, it is not clear from our data whether it is an increase in NAA or a decrease in Cho that causes the improvement of the NAA/Cho ratio after carotid endarterectomy. In the brain the NAA concentration is higher in gray than in white matter (Michaelis et al., 1993) and is located mainly in axons (Tallan, 1957; Birken and Oldendorf, 1989). NAA has been shown to be present only in cultured neurons, and not in other cell cultures except for oligodendrocyte precursor cells (Urenjak et al., 1993). Since the concentration of NAA correlates with the number of intact neurons (Koller et al., 1984), it is unlikely that the NAA concentration is increased after endarterectomy, because it is believed that neuronal loss is irreversible (Gideon et al., 1992). The most prominent contributions in cerebral “choline” originates from phosphorylated cholines (Miller, 1991). Therefore, changes in phospholipid metabolism may be the cause of the observed change in the NAA/Cho ratio after endarterectomy. In particular, increased phospholipid breakdown and/or reduced phospholipid biosynthesis are probably the main cause of increased cerebral Cho before endarterectomy.
Previous studies of human stroke using 1H magnetic resonance spectroscopy have shown that Lac is present in the acute stage of infarction and may decrease slowly for the following 6 months or longer (Felber et al., 1992; Graham et al., 1992; Houkin et al., 1993; Barker et al., 1994; Gideon et al., 1994). The metabolic role of Lac in the process of infarction and subsequent recovery is not yet completely understood. However, recently the clinical predictive value of cerebral Lac in patients after acute cerebral infarction has been shown (Graham et al., 1995). The results are in agreement with animal studies showing that the presence of cerebral Lac is related to an increased amount of cerebral damage after endarterectomy (Chopp et al., 1987; Combs et al., 1990; Chang et al., 1992; Wagner et al., 1992). Although it is not clear whether acidosis or the neurotoxic effect of Lac (Myers and Yamaguchi, 1977) causes cerebral damage, a relation between the Lac concentration and acidosis in the acute phase of infarction seems obvious (Rehncrona et al., 1981; Combs et al., 1990; Chang et al., 1992). Studies in which Lac-free acid solutions were injected extracellularly (Kraig et al., 1987) or in which the pH was lowered by an increased CO2 tension (Xu et al., 1991) have identified a pH threshold of 5.8–6.2 for necrosis in normoxic brain without the observation of increased Lac. However, cell death occurs at a higher pH in neuronal/glial cultures subjected to lactic acidosis than in those exposed to hydrochloric acid (Goldman et al., 1989).
In the acute phase of infarction Lac accumulation and acidosis in the infarcted area depend on the level of serum glucose, suggesting that in the acute phase of infarction Lac accumulation is caused mainly by anaerobe glycolysis (Rehncrona et al., 1981; Gardiner et al., 1982; Chopp et al., 1987; Siesjö, 1988; Combs et al., 1990; Wagner et al., 1992). The relation between the presence of increased carbohydrates in the brain and the increased risk of stroke in the diabetic patient has been known for years (Plum, 1983) and was confirmed in a recent study showing that diabetes influences stroke in age, in speed of recovery, and in mortality (Jørgensen et al., 1994). In our patient population there was one diabetic patient. This patient showed the highest Lac resonance of all patients before endarterectomy. In spite of the high Lac resonance preoperatively, after endarterectomy the NAA/Cho ratio was increased and the Lac resonance was decreased considerably.
In the chronic phase of infarction it has been shown that in patients with severe stenosis or occlusion of the ICA, infarcted tissue is still perfused at a low level (Myers, 1979; Symon et al., 1979; Hossman and Schuier, 1980). This low regional perfusion is probably still sufficient to accommodate for the metabolic demands, since positron emission tomography (PET) studies have shown luxury perfusion in chronic infarcts (Syrota et al., 1985; Hakim, 1987; Senda et al., 1989). Although Lac is continuously produced from blood glucose in chronic infarctions (Rothman et al., 1991), anaerobe glycolysis is not the only source of Lac production in the chronic stage of infarction. It has recently been shown that in the subacute and chronic stage of infarction cerebral regions showing low NAA and high Lac concentrations are also associated with the occurrence of large numbers of macrophages that infiltrate 3 days after the onset of infarction (Petroff et al., 1992). The latter study suggested that brain macrophages could be a major source of elevated brain Lac signals that persist for months after stroke.
We selected our regions outside infarcted regions, therefore the observed metabolic changes seem to be more compatible with changes associated with the initial phase of infarction rather than with the chronic phase of infarction. The observed difference in the NAA/Cho ratio after carotid endarterectomy in patients with and without Lac (NAA/Cho ratio unchanged and increased, respectively) supports the suggestion that at the early stage of infarction cerebral lactate could be indicative of the reversibility of the occurring ischemic damage (Hossman and Kleihues, 1973; Rehncrona et al., 1981; Siesjö, 1988; Chang et al., 1992; Graham et al., 1995). Although in our study the number of patients with cerebral infarcts was relatively high in the patient group with Lac, there was no correlation between the initial NAA/Cho ratio and infarction or Lac levels. Although Lac and decreased NAA/Cho ratios are not directly related to each other, they may indicate what our patients have in common, i.e., cerebral damage, probably caused by hypoperfusion. Although the incidence of cerebral infarcts was significantly higher in the patient group with Lac, infarcts and Lac are not necessarily directly related to each other, since we selected our regions 2=2.5 cm away from signal abnormalities on MRI. It could be that in these patients collateral flow is insufficient to maintain cellular function, resulting in infarcts but also in the presence of Lac outside these infarcts. This hypothesis could be tested if the cerebral blood flow were also measured pre- and postendarterectomy.
In conclusion, the presence of Lac may predict whether the NAA/Cho ratio increases after carotid endarterectomy, and this increase may be associated with benefit from the procedure. However, further studies are required to correlate an increase in NAA/Cho with improved outcome.
Footnotes
Acknowledgment:
This study was supported by Dutch Heart Foundation Grant D94.012.
