Abstract
In the cat, cerebral hemispherectomy sustained neonatally results in a remarkable degree of recovery and/or sparing of function as compared with the effects of a similar lesion but sustained in adulthood. We have proposed that this effect is due to a combination of reduced neuronal loss within partially denervated structures and a lesion-induced reorganization of corticofugal projections arising from the remaining intact hemisphere in the neonatally lesioned animal. The current study was designed to assess the physiological consequences of these anatomical changes utilizing [14C]2-deoxy-D-glucose autoradiography. A total of 17 adult cats were studied. Seven animals served as intact controls, five received a left cerebral hemineodecortication as neonates (NH; mean age 11.4 days), and five sustained the same lesion in adulthood (AH). Histological analysis indicated that the lesion was very similar between the two age groups and essentially represented a unilateral hemineodecortication. Local CMRglc (LCMRglc; μmol 100 g-1 min-1) values were calculated for 50 structures bilaterally and indicated that in the remaining intact contralateral (right) cerebral cortex (including all areas measured), AH cats exhibited a significantly (p < 0.05) lower level of LCMRglc (ranging from 20 to 72 μmol 100 g-1 min-1) than NH (ranging from 49 to 81 μmol 100g-1 min-1). In comparison, the rates of NH cats within the cerebral cortex were very similar to those seen in intact animals (ranging from 48 to 119 μmol 100 g-1 min-1). Ipsilateral to the lesion in AH cats, the structures spared by the resection, including the basal ganglia and thalamus, exhibited LCMRglc rates of between 23 and 69 μmol 100 g-1 min-1, which were significantly lower (p < 0.05) than in NH cats (range 47–72 μmol 100 g-1 min-1). Considering all structures, both age-at-lesion groups exhibited a lower level of metabolism compared with similar measurements for intact control animals (LCMRglc range 45–75 μmol 100 g-1 min-1). However, this depression of glucose metabolism was more pronounced in the AH cats (p < 0.05). These results indicate that following neonatal hemineodecortication, LCMRglc is maintained at a higher level in many regions of the brain than in animals that sustain the same resection in adulthood. This higher level of glucose metabolism in NH animals suggests that the lesion-induced anatomical reorganization of structures not directly injured by the lesion plays a functional role that is probably responsible for the greater degree of recovery and/or sparing of function in these early lesioned cats.
Keywords
In previous work we have documented the degree and extent of neurological and behavioral sparing and/or recovery of function following cerebral hemispherectomy when it is sustained neonatally as compared with when this lesion is performed in adult cats (Burgess and Villablanca, 1986; Burgess et al., 1986; Villablanca et al., 1986b; Hovda and Villablanca, 1989b, 1990). In terms of neurological and behavioral measurements, animals sustaining a neonatal hemispherectomy performed much better as adults on sensorimotor tasks as a consequence of marked improvement of neurological functions, including reflexes and visual field perception, than animals that sustained hemispherectomy in adulthood. This age-at-lesion-induced differential response to cerebral hemispherectomy is most likely due to a complex interaction of the effects that this lesion has on neuroanatomical degeneration (Villablanca et al., 1986a; Hovda and Villablanca, 1989a; Villablanca and Hovda, 1989; Shook and Villablanca, 1991) and reorganization of axonal terminal fields (Olmstead et al., 1983; Villablanca et al., 1984; Gómez-Pinilla et al., 1986; Villablanca and Gómez-Pinilla, 1987; Fisher et al., 1988; Villablanca et al., 1988; Sutton and Villablanca, 1990; Hovda et al., 1992c,) both of which exhibit different degrees of expression depending on the age at which the resection is conducted.
Assessed as adults, neonatal hemispherectomized animals exhibit far less retrograde neuron loss and shrinkage (Villablanca et al., 1986a; Hovda and Villablanca, 1989a; Shook and Villablanca, 1991) as well as transsynaptic degeneration (Villablanca and Hovda, 1989) in every structure that we have examined than adult hemispherectomized cats. These differential effects were most striking within the thalamus ipsilateral to the hemispherectomy (Villablanca et al., 1986a; Hovda and Villablanca, 1989a; Shook and Villablanca, 1991); however, other structures, including the mammillary nucleus (Villablanca and Hovda, 1989) and the superior colliculus (Villablanca and Hovda, 1989), also exhibited differential degeneration depending on the age at which animals were lesioned. Furthermore, complementing this change in neuronal loss, there was a marked degree of gliosis coupled with structural atrophy, both of which were far more evident in adult than neonatal hemispherectomized animals.
Although being a very striking finding, these subcortical changes due to degeneration following hemispherectomy do not provide the entire picture of differential anatomical changes seen between neonatal and adult hemispherectomized animals. Specifically, the brain of the neonatal hemispherectomized animals exhibited a marked degree of reorganization in terms of the descending pathways arising from both the primary sensorimotor (Villablanca et al., 1984; Gómez-Pinilla et al., 1986; Villablanca and Gómez-Pinilla, 1987; Fisher et al., 1988; Villablanca et al., 1988; Sutton and Villablanca, 1990) and the visual (Houda et al., 1992c) cortices of the remaining intact hemisphere. This reorganization consisted of reinnervation by novel terminals (presumably sprouting of axons from the sensorimotor cortex that normally contribute to unilateral projections only) of the thalamus (Villablanca and Gómez-Pinilla, 1987) and red nucleus (Villablanca et al., 1988) ipsilateral to the lesion as well as of the dorsal column nuclei and spinal cord (Gómez-Pinilla et al., 1986) contralateral to the hemispherectomy. In addition, the ipsilateral superior colliculus (Hovda et al., 1992c) received reinnervation from the remaining primary visual cortex (areas 17 and 18). This reinnervation of subcortical structures following cerebral hemispherectomy was far more pronounced in neonatally lesioned than adult lesioned cats and was correlated with the degree and extent of neural sparing and with the amount of behavioral recovery.
Although the correlation between these anatomical and behavioral changes was quite strong (Burgess and Villablanca, 1986; Burgess et al., 1986; Villablanca et al., 1986; Hovda et al., 1992c), direct physiological assessment of the affected structures has yet to be conducted. To begin to assess the functional characteristics of these age-at-lesion structural changes, we utilized [14C]2-deoxy-D-glucose (2DG) autoradiography (Sokoloff et al., 1977) to determine the degree and the anatomical extent of cerebral glucose metabolic alterations following neonatal (NH) as compared with adult cerebral hemineodecortication (AH) in the cat. As glucose metabolism is a direct reflection of neuronal firing (Shinohara et al., 1979; Mata et al., 1980; Sokoloff, 1981), the calculated values of metabolic rates from the present work provide a functional anatomical map of activity relating the degree of structural change to neurological recovery.
MATERIALS AND METHODS
Subjects
Seventeen adult cats underwent studies of cerebral glucose utilization. Seven animals served as intact adult controls (1–4 years of age), five received a left cerebral hemineodecortication as a neonate (NH; mean age 11.4 days), and five had the same surgery in adulthood (AH; 1–4 years of age). All animals were allowed to survive at least 9 months following the surgical procedure before being studied for cerebral metabolism.
Surgery and maintenance
Kitten surgery was performed under combined chlor-promazine (5 mg/kg i.p.) and pentobarbital (10–15 mg/kg i.p.) anesthesia, while only the standard dose of pentobarbital (35 mg/kg i.p.) was used for adults. Hypothermia (30–35°C rectal temperature) was used in all cases. The surgical procedure has been described in detail (Villablanca et al., 1986b). In brief, by blunt dissection with a suction pipette, the caudal aspect of the left cerebral hemisphere was first retracted from the midline. The hemisphere was then penetrated at the lower level of the gyrus cinguli, just above the corpus callosum. Thereafter, the callosum was penetrated and the lateral ventricle was opened. Continuing the dissection rostrally, the head of the caudate nucleus was exposed. The dissection was continued lateral to the head of the caudate nucleus, rostrally, and through the internal capsule and lateral to the thalamus, caudally. Thereafter, the pedicle of the middle cerebral artery was ligated and sectioned, the frontal pole was dissected from the olfactory bulb, and the entire hemisphere was removed in a block. The cranial defect was covered with the temporal muscle, which was reattached to the midline (in cats), or with the skull bone flap, which was sutured in place (in kittens). The hemisphere removed was always the left. After surgery the animals were gradually warmed, and the kittens were maintained for the night in a thermostatically controlled incubator.
All animals were closely monitored for the first few days of postsurgical recovery, and all cats received periodic neurological testing using selected tests from the battery described previously (Burgess, and Villablanca, 1986; Burgess et al., 1986; Villablanca et al., 1986). There were no complications, and neurological evaluations (to be reported separately) indicated no striking deviations from our previous reports (Burgess and Villablanca, 1986; Burgess et al., 1986 Villablanca et al., 1986b) examining hemispherectomized cats.
Metabolic rate determinations
Measurements of local CMRglc (LCMRglc; μmol 100 g−1 min−1) were obtained using the 2DG method of Sokoloff et al. (1977) utilizing a lump constant of 0.411 as previously described for cats (Chugani et al., 1991). Animals were anesthetized with halothane gas (1%), the femoral triangle was surgically exposed, and the femoral artery and vein were cannulated with polyethylene tubing (PE-50). Local anesthesia (Xylocaine HCl) was topically applied to the wound to minimize discomfort, and the skin was sutured closed. The animals were then restrained with a loose-fitting padded plaster cast from the lower thorax to the abdomen and by securing the hind limbs to an aluminum splint. At least 3 h was allowed for recovery from anesthesia. During this time, the body temperature was thermostatically controlled (38°C) using a rectal probe and heating pad.
The 2DG (specific activity 50–55 mCi/mmol; New England Nuclear Corp.), in the amount of 100 μCi/kg in 0.5–1.0 ml of normal saline, was administered intravenously over a 30-s period. Timed arterial blood samples were drawn during the subsequent 45 min. The samples were immediately centrifuged in a Beckman Microfuge B, and the plasma was separated and stored on ice until further analysis. One arterial blood sample was used to analyze pH and blood gases (Po2, Pco2) to ensure a normal physiological state. The animals were monitored during the entire 45 min and their general behavior noted. If animals became drowsy (closing their eyes), they were awakened by a gentle tap to the nose. To ensure a standardized and stable environment, the animals were otherwise not disturbed and noise in the laboratory was kept to a minimum.
At the end of the 45 min, the cat was killed with an intravenous overdose of pentobarbital, and the brain quickly removed and frozen in pulverized dry ice. The frozen brain was stored at −70°C and subsequently coated with chilled embedding medium (Lipshaw) just prior to sectioning into 20-μm coronal slices at −22°C in a cryostat (American Optical Cryo-Cut II). The sections were then autoradiographed together with calibrated [14C]methylmethacrylate standards (Amersham). Adjacent sections were taken and stained with thionine to assist in identification of anatomical regions on the autoradiographs.
The arterial plasma samples were assayed for concentrations of 2DG by liquid scintillation counting and of glucose by means of a glucose analyzer (Beckman). Optical densities of 50 selected brain structures were determined from the autoradiograms using a computer-supported image analysis system (JAVA; Jandel Scientific). For each structure, at least 10 readings were taken across different sections and averaged into one value. The optical densities, the 14C-standard curve, the plasma glucose values, and the 2DG input function were used to calculate LCMRglc (μmol 100 g−1 min−1) according to the operational equation of Sokoloff et al. (1977).
Statistical analysis
A repeated measures analysis of variance was conducted with the individual structure serving as the repeated variable. Comparisons were made to determine the significance of the main effect (lesion group) with simple main effect comparisons made between intact, NH, and AH using appropriate contrasts (Hays, 1973; Myers, 1979).
RESULTS
Histological analysis
The results from the thionine-stained sections for both NH and AH cats indicated that the extent of the resection was similar in the two age-at-lesion groups. Analysis of the animals used in the current study demonstrated that the extent of the lesion was similar to that in our previous studies (Villablanca et al., 1986b) except for sparing of the neostriatum (see Fig. 1). Because we are now interested in assessing possible caudate nucleus anatomical changes (Loopuijt et al., 1992) including atrophy and reinnervation as well as metabolic changes in this structure, our previously used lesion model was modified for the present experiment to spare the neostriatum ipsilateral to the resection. In brief, serial reconstructions of the thionine-stained sections indicated that in all cats the lesion consisted of a removal of almost the entire left neocortex [including all of the primary visual cortex (areas 17 and 18)]. Since the caudate nucleus was spared, portions of the neocortex lateral and ventral to the head of this nucleus were not removed. These included the lower one-third to one-half of the anterior and posterior ectosylvian sulci, the anterior and posterior sylvian gyri and the ectosylvian sulci, the anterior and posterior sylvian gyri, and the lower one-third to one-half of the medial sylvian gyrus. The caudate nucleus as well as the dorsal thalamus (including the lateral geniculate bodies and optic tracts) were intact in all brains. Other than that described, the ablation was similar to our hemispherectomy as described in detail elsewhere (Villablanca et al., 1986b). Briefly, other telencephalic areas that were spared on the left side were olfactory bulb and tract, ventral aspect of the gyrus proreus, gyrus presylvius, areas of the ventral brain surface medial to the olfactory tract and sulcus rhinicus anterior–posterior, amygdala nuclei, nucleus accumbens, claustrum, and putamen. Dor-sally, there were cortical remnants only in the midline, rostral to the genus of the corpus callosum, including variable portions of the gyri rectus and cinguli. The septum and midline fornix were not directly lesioned, but most fimbria fibers were interrupted and/or removed together with the dorsal hippocampus. The lower brainstem and the contralateral hemisphere were not damaged in any of the cats.
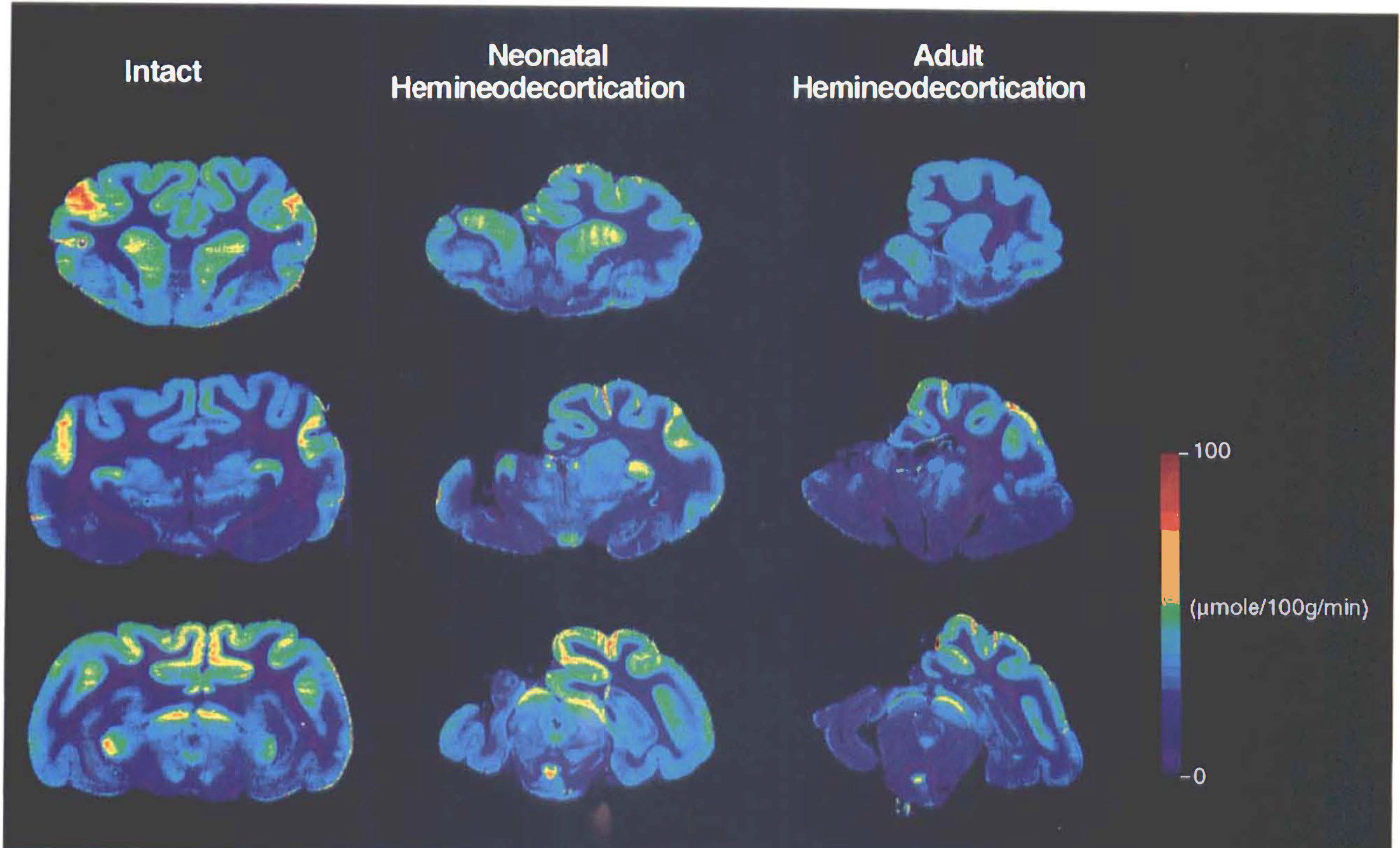
Selected color-coded, coronal autoradiographs taken at the level of the caudate
Measurements of cerebral glucose metabolism
Across all structures measured, intact animals exhibited a mean LCMRglc of 69 μmol 100 g−1 min−1. In both age-at-lesion groups, this mean rate was generally (47 of 50 structures) depressed bilaterally. In NH animals, structures ipsilateral (left) to the lesion exhibited a 17% decrease of metabolism (mean LCMRglc 57 μmol 100 g−1 min−1) with contralateral structures showing a 26% decrease (mean LCMRglc 51 μmol 100 g−1 min−1) compared with intact controls. The metabolic depression was more dramatic in AH cats with measurements in the ipsilateral hemisphere showing a 34% decrease (mean LCMRglc 45 μmol 100 g−1 min−1) and with contralateral structures exhibiting a 31% (mean LCMRglc 47 μmol 100 g−1 min−1) decrease compared with intact animals.
These comparisons revealed that throughout all areas measured, LCMRglc was reduced bilaterally in all lesioned animals irrespective of when they were operated on. This was particularly the case for regions that had undergone extensive degeneration. Furthermore, this metabolic depression was more severe in animals that had sustained cerebral hemineodecortication in adulthood. The neonatally lesioned cats exhibited a remarkable degree of sparing of glucose metabolism, with some structures being only slightly (nonsignificant) depressed (Fig. 1; Table 1).
Mean local CMRglc (μmol 100 g−1 min−1) with corresponding SD and p values for each brain region studied in intact and hemineodecorticate animals
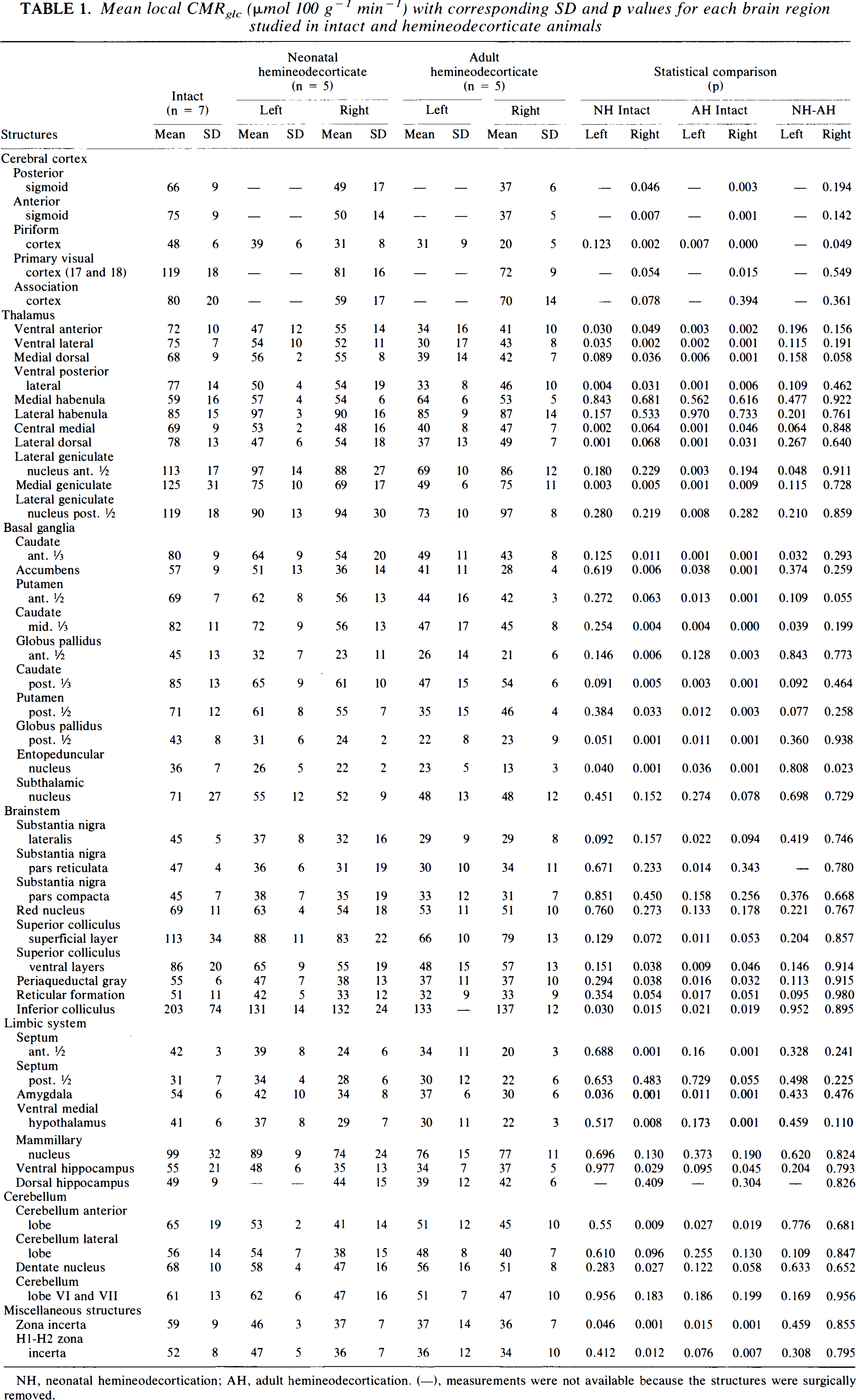
NH, neonatal hemineodecortication; AH, adult hemineodecortication. (−), measurements were not available because the structures were surgically removed.
The LCMRglc values as well as statistical comparisons for all 50 structures studied are shown in Table 1.
Cerebral cortex
The remaining intact (right) cerebral cortex of the AH animals exhibited a decrease in LCMRglc of 12%—49% across all areas measured compared with intact controls. This depression was greater than that seen in the NH animals for which the decrease ranged from 18 to 34% (except for the association cortex, which exhibited more depression in NH than AH cats). All regions of the remaining cerebral cortex were significantly (p < 0.015) depressed in the AH animals except for the right association cortex and the left piriform cortex, whereas in the NH cats, only the visual and right piriform regions were significantly (p < 0.05) depressed compared with intact controls (see Table 1 and Fig. 2).
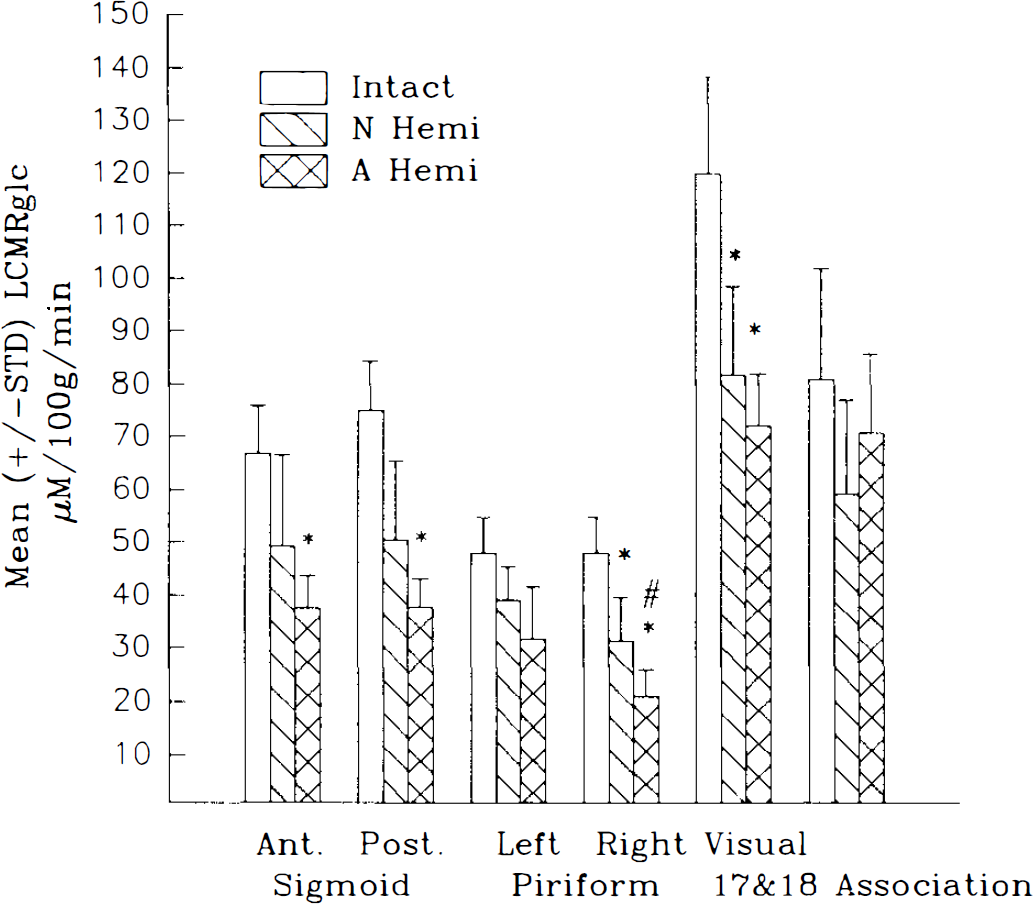
Mean ± SD local CMRglc (LCMRglc; μmol 100 g−1 min−1) for regions in the cerebral cortex. Note that the greatest degree of metabolic depression was seen in the adult lesioned animals. N Hemi, neonatal hemineodecortication; A Hemi, adult hemineodecortication. *p < 0.05, **p < 0.01 compared with intact; #p < 0.05 compared with N Hemi.
Thalamus
The thalamic nuclei ipsilateral (left) to the lesion exhibited a pronounced difference between NH and AH cats. The NH animals exhibited a modest although significant metabolic depression across all nuclei measured within the ipsilateral thalamus (p < 0.03, except for the lateral geniculate nucleus) ranging from 13 to 39% compared with intact controls. In contrast, AH animals showed a marked depression of LCMRglc ranging from 3 8 to 60% among all the nuclei measured (p < 0.008). The nucleus most affected by the lesion for both age-at-lesion groups was the medial geniculate. The nucleus least affected was the lateral geniculate nucleus. However, regardless of the degree of the lesion effect, it was a consistent finding for each thalamic nucleus that the metabolic depression was more severe in AH than NH animals, reaching statistical significance for the lateral geniculate nucleus (p < 0.048) (see Table 1 and Fig. 3).
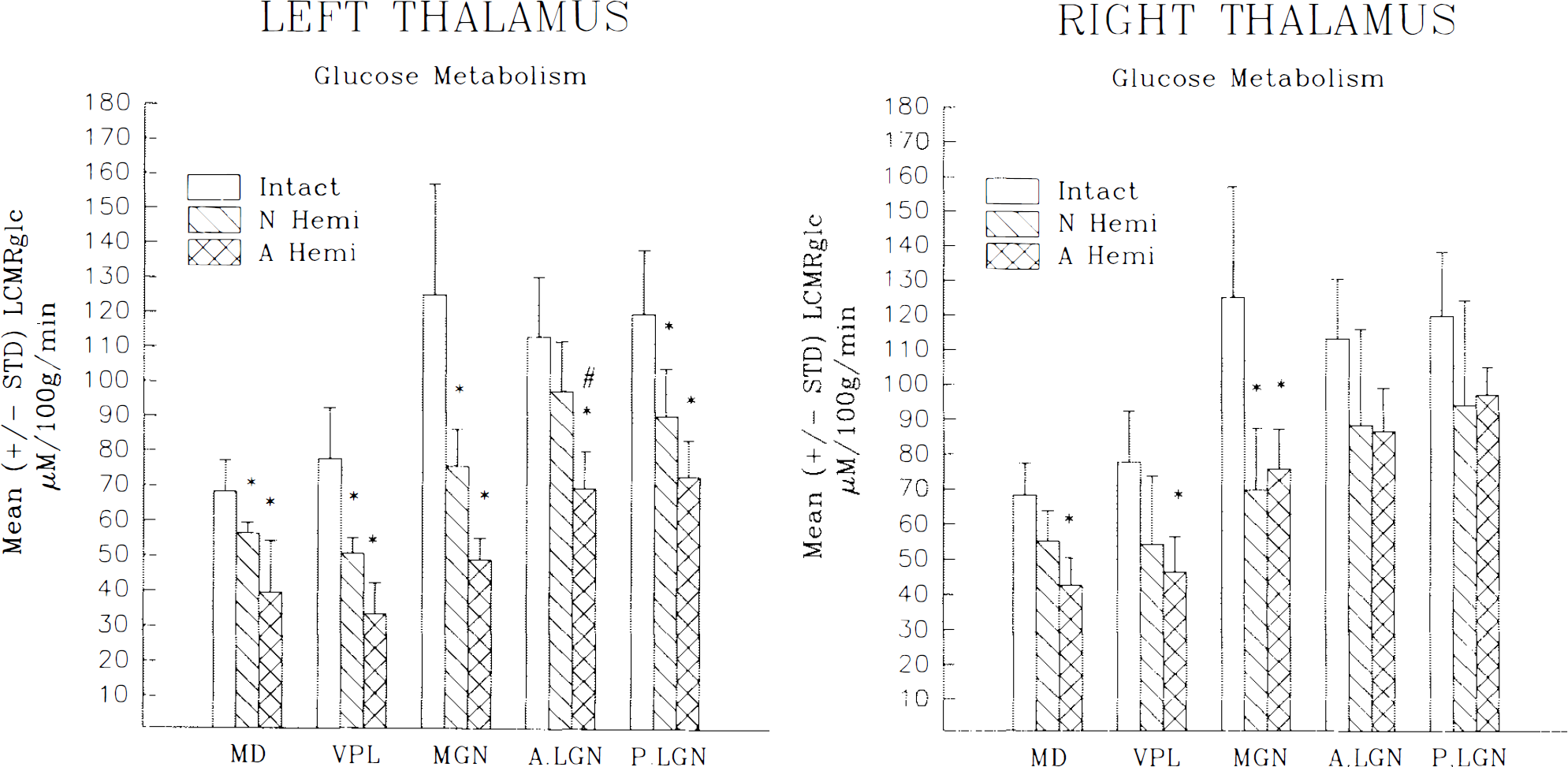
Mean ± SD local CMRglc (LCMRglc; μmol 100 g−1 min−1) for nuclei within the ipsilateral
The thalamus contralateral (right) to the hemineodecortication also exhibited some degree of metabolic depression. However, unlike the ipsilateral thalamus, the degree and extent of the depression were not as affected by the age at lesion. Neonatally lesioned animals exhibited a reduction ranging from 21 to 49% for all nuclei measured compared with controls. This severity of depression was similar to that seen in adult lesioned cats in which the LCMRglc decrease ranged from 18 to 45% (see Table 1 and Fig. 3 for statistical significance). As for the ipsilateral thalamus, the medial dorsal and the lateral geniculate nuclei were the most and least affected, respectively, for both age-at-lesion groups.
Basal ganglia
All structures measured within the basal ganglia (BG) exhibited some degree of metabolic depression with many measurements reaching statistical significance when compared with intact controls (see Fig. 4). This depression was evident bilaterally. Considering all BG structures of both sides, the depression ranged from 26 to 52% in AH animals and from 9 to 47% in NH cats. Although comparisons between the age-at-lesion groups did not result in statistical significance, there was a tendency for the depression to be more pronounced in the AH animals. For these AH cats, the anterior portion of the caudate, putamen, and globus pallidus all exhibited a greater degree of metabolic depression on the side contralateral (right) to the injury, whereas ipsilaterally LCMRglc measurements taken in the posterior portions of these structures revealed a greater depression compared with intact controls. The subthalamic nucleus and substantia nigra appeared to be equally depressed irrespective of side. In NH animals, all BG structures exhibited a marked asymmetry in terms of LCMRglc. Without exception, all structures showed a lower level of LCMRglc on the side contralateral (right) to the side of the lesion. In these animals this contralateral depression ranged from 18 to 47% compared with measurements taken within the left (ipsilateral) hemisphere, which ranged from 9 to 27%. In contrast, such asymmetry was not observed in AH cats (ipsilateral depression, 26–50%; contralateral, 26–52%), although for both sides the decrease tended to be greater for AH cats.
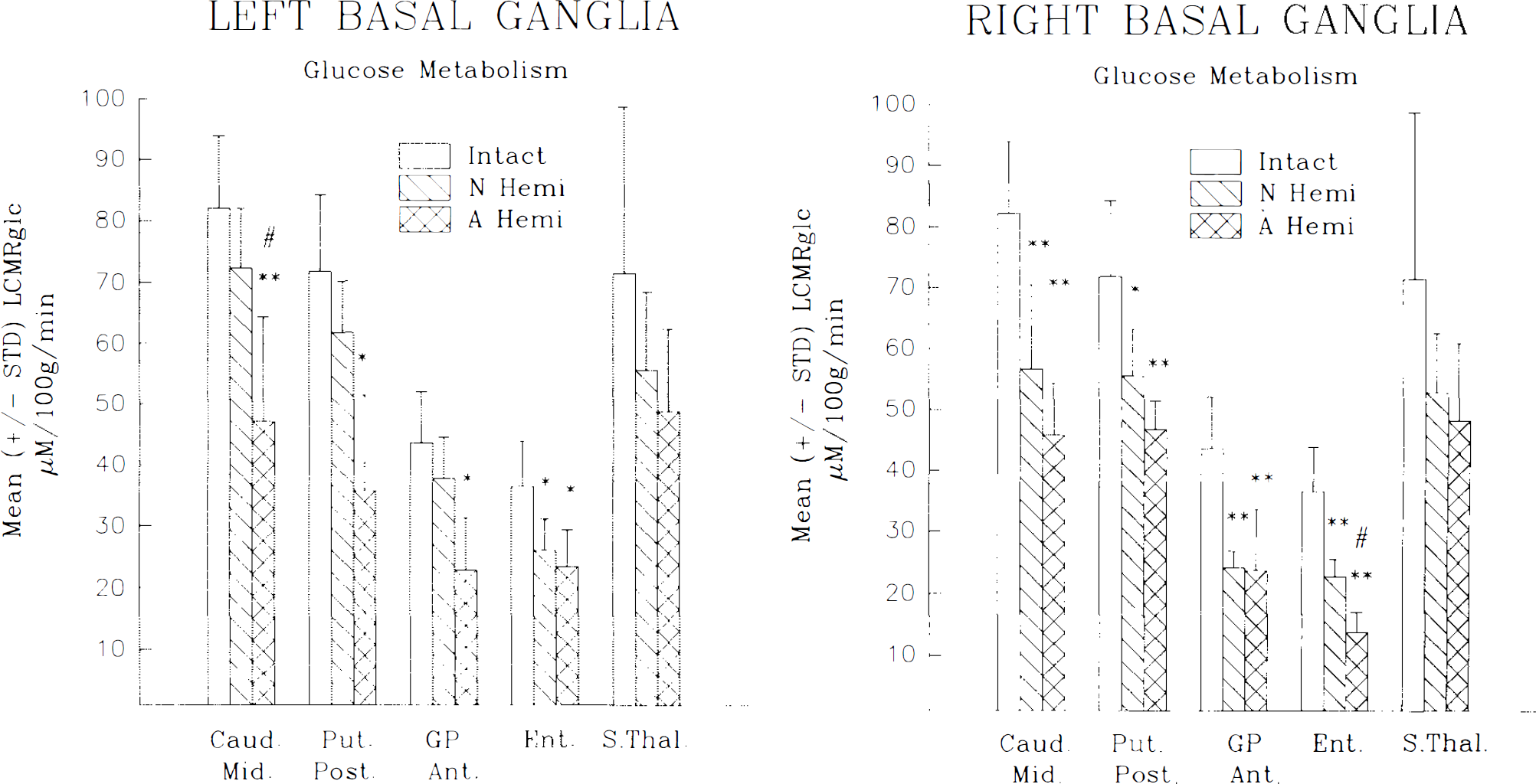
Mean ± SD local CMRglc (LCMRglc; μmol 100 g1 min1) for nuclei within the ipsilateral
Brainstem
Structures within the brainstem of both age-at-lesion groups tended to be uniformly depressed, with LCMRglc measurements indicating a decrease of between 22 and 43%. Structures tended to be more depressed if they were located on the side ipsilateral (left) to the hemineodecortication, particularly in AH animals. This was most striking for the superficial layer of the superior colliculus in AH cats, which on the left exhibited a 41% depression, but on the right showed only a 29% decrease in LCMRglc. An exception to this finding in AH animals was seen in measurements for the red nucleus. In this structure the left side exhibited rates that were similarly depressed (22%) as in the side contralateral to the lesion (26%).
In NH animals all structures exhibited some degree of metabolic depression; however, this depression was not as remarkable as seen in adult lesioned animals and was more pronounced on the side contralateral (right) to the lesion. This contralateral depression in the NH cats was particularly evident for the ventral layer of the superior colliculus and the periaqueductal gray, exhibiting a 35 and 31% depression, respectively (p < 0.05). In addition, the reticular formation was also markedly depressed (34%) contralateral; however, this did not quite reach statistical significance (p < 0.54). The inferior colliculus was equally depressed in both the left (35%) and the right (34%) side as was the superficial layer of the superior colliculus, which exhibited a 22 and 26% depression in both the left and the right sides, respectively. Finally, for the red nucleus, the ipsilateral depression was minimal (8%), whereas contralaterally it was higher (21%).
Limbic system
For both age-at-lesion groups, structures within the limbic system exhibited a depression of LCMRglc of between 9 and 45%. However, the NH animals did exhibit some sparing from this reduction primarily in the ventral hippocampus and ventral medial hypothalamus. As seen in other subcortical structures, this partially sustained metabolic activity was seen exclusively on the side ipsilateral (left) to the lesion. On this side the ventral hippocampus of NH cats exhibited only a 11% and the ventral medial hypothalamus only a 9% depression of LCMRglc.
Cerebellum
For the cortical regions measured, all the metabolic depression was seen in the right cerebellar hemisphere. This decrease was very similar for the two age-at-lesion groups ranging from 22 to 29% in AH animals and from 23 to 35% in NH cats. This pattern was also similar to that seen in the deep cerebellar nuclei (e.g., dentate). Within the left hemisphere, there was very little evidence for any metabolic depression with the exception of the anterior lobe in the AH cats, which did exhibit a 21% depression (p < 0.05).
Miscellaneous structures
For the medial and lateral habenulas, there was little change in LCMRglc for either AH or NH animals. The zona incerta (including H1 and H2) exhibited a bilateral depression of up to 34%, which was highly significant compared with intact controls but was unremarkable in terms of a difference between age-at-lesion groups.
DISCUSSION
Although the depression of cerebral glucose metabolism has been previously reported following traumatic and/or ischemic injury to the cerebral cortex (Pappius, 1981, 1982; Pappius and Wolfe, 1983; Feeney et al., 1985; Colle et al., 1986; Hovda et al., 1987, 1992a; Yoshino et al., 1991, 1992), the current work represents the only metabolic study conducted following an anatomical resection while at the same time addressing developmental issues. The fact that we have extensive knowledge regarding the degree and extent of anatomical and behavioral changes following cerebral hemispherectomy contributes to making this work unique in nature. Consequently, the results of the current study provide an opportunity to assess functional–structural relationships and indicate that the metabolic differences within the two age-at-lesion groups most likely reflect the marked changes in anatomical reorganization that we have previously reported (Gómez-Pinilla et al., 1986; Villablanca and Gómez-Pinilla, 1987; Fisher et al., 1988; Villablanca et al., 1988; Hovda et al., 1992c).
Remaining cerebral cortex and its projections
As was demonstrated in the current study, the remaining contralateral sensorimotor (anterior and posterior sigmoid gyri) and primary visual (areas 17 and 18) cortices exhibited a distinct age-at-lesion effect, with the greatest severity of metabolic depression occurring within the cerebral cortex of the AH animals. Our previous work demonstrated that following hemispherectomy early in life, the remaining undamaged sensorimotor cortex and primary visual cortex alter their descending projections to innervate subcortical structures bilaterally. In contrast, the adult lesioned animals basically retained the normal unilateral projection pattern. The metabolic results described herein are our first assessment of a physiological measure that provides a direct evaluation of functional activity in projection nuclei of these novel crossed pathways.
Sensorimotor cortex. In the intact animal, neurons located within the anterior and posterior sigmoid gyrus project unilaterally to the ventral basal complex of the thalamus (Gómez-Pinilla and Villablanca, 1989), red nucleus (Villablanca et al., 1988), and corresponding gracilis and cuneatus nuclei (Villablanca et al., 1988) prior to entering the spinal cord. Following adult hemispherectomy, this pathway remains primarily unilateral with only the red nucleus exhibiting some significant bilateral innervation (Villablanca et al., 1988). In contrast, neonatal hemispherectomized animals exhibit extensive sprouting, resulting in a descending pathway that innervates all these structures bilaterally (Gómez-Pinilla et al., 1986; Kolb and Tomie, 1988; Villablanca et al., 1988). This reinnervation is robust and, in terms of topography (Villablanca et al., 1988) and synaptic ultrastructure (Fisher et al., 1988), resembles the innervation normally exhibited by intact animals. Structures receiving this reinnervation also show substantial sparing from the degenerative process that is so typical following removal of the cerebral cortex, which implies that it may play an important role in restricting the extent of retrograde neuronal degeneration and the corresponding atrophy and gliosis.
In terms of physiology, our previous behavioral results strongly suggest that this newly formed bilateral projection provides the anatomical conduit by which the animal can maintain neurological functioning. This enhanced recovery of function in the neonatal hemispherectomized animals includes the maintenance of more normal reflex and postural responses in the forelimb contralateral to the lesion, which contribute to a greater efficiency for directed movements. The current metabolic study supports this contention since the nuclei along this newly formed pathway in NH cats exhibited more metabolic activity than seen in AH cats.
Primary visual cortex. In previous work (Hovda and Villablanca, 1990; Hovda et al., 1992c), we described that neonatal hemispherectomized animals, unlike adult lesioned cats, do not exhibit a complete hemianopsia. A similar finding has also been reported in patients who had undergone cerebral hemispherectomy as children (Ptito et al., 1987; Perenin and Jeannerod, 1978). We (Hovda and Villablanca, 1990; Hovda et al., 1992c) as well as others (Sprague, 1966; Sprague and Meikle, 1965; Sherman, 1974, 1977; Wallace et al., 1989), have proposed that the relationship between the primary visual cortex and the superior colliculus, as well as how they interact following cortical resection, may provide some insight into the mechanism(s) behind the sparing of the visual field in animals and in children with visual cortex lesions.
The classic experiment by Sprague (1966) and followed up by Wallace et al. (1989) showed that visual orienting, which is lost in the contralateral visual field after a unilateral ablation of the occipitotemporal cortex, can be immediately and completely restored by subsequently lesioning the contralateral superior colliculus or by sectioning the collicular commissure. According to the original hypothesis put forth by Sprague (1966), these behavioral results supported the concept that each superior colliculus receives selective, unilateral facilitative input from the ipsilateral visual cortex and inhibitory input from the contralateral superior colliculus. Consequently, unilateral removal of the visual cortex results in an inhibition within the ipsilateral superior colliculus, given the loss of facilitative input. The subsequent removal of the inhibitory input from the contralateral superior colliculus (e.g., via sectioning of the collicular commissure) would alleviate this unbalanced inhibition, thereby restoring visual perception to the affected visual field.
The present work complements our previous efforts (Hovda and Villablanca, 1990; Hovda et al., 1992c) to determine if the novel cortical–tectal relationship described is matched by metabolic changes in NH and AH animals in which the optic tracts and the lateral geniculate nuclei remain intact (see Results). The results indicate that AH cats exhibit a more marked metabolic depression, particularly within the superficial layer of the superior colliculus ipsilateral to the lesion, than neonatal hemipherectomized animals, which exhibit the sparing of visual field perception. The reasons for this sustained metabolic function within the superior colliculus of the NH cats appear to be a combination of lack of neuronal loss (Villablanca and Hovda, 1989) and the formation of a bilateral cortical–tectal pathway (Hovda et al., 1992c). Consequently, this anatomical-metabolic relationship provides a better understanding of the mechanism behind the different sparing/recovery of visual functioning following cortical lesions sustained early in life.
Subcortical structures
Other subcortical structures that receive this new innervation also demonstrate less neuronal degeneration presumably (at least partially) because of this sustaining input. Specific examples of reduced degeneration within the thalamus following neonatal cerebral hemispherectomy include the ventral posterior lateral nucleus (Villablanca et al., 1986a), the medial geniculate nucleus (Hovda and Villablanca, 1989a), and the lateral geniculate nucleus (Shook and Villablanca, 1991). All of these nuclei exhibit neuronal loss and shrinkage as well as gliosis following cerebral hemispherectomy. However, these anatomical changes are quite reduced in animals sustaining the lesion early in life (with gliosis being minimal or nonexistent). Taken together with the current metabolic finding, this suggests that these anatomical age-at-lesion differences within the thalamic nuclei play an important role for their postlesion metabolic vitality.
In addition to the thalamus, the current finding within the BG and cerebellum complements previous studies and adds to our understanding of how these structures react to a dysfunctional or damaged cerebral cortex. Specifically, these results regarding the lateral lobe of the cerebellum support the concept of a crossed cerebellar metabolic diaschisis following unilateral damage to the cerebral cortex, a process that has been reported in many other species including humans (Kushner et al., 1984; Pantano et al., 1986; Broich et al., 1987; Shimoyama et al., 1988).
Regarding the BG, we have previously shown (Loopuijt et al., 1992) that in AH, but not NH, cats, there is a loss of neurons as well as glia proliferation within the caudate nucleus ipsilateral to the hemineodecortication. Moreover, there is a study (Goldman, 1980) indicating that following unilateral removal of the dorsolateral prefrontal cortex early in life in monkeys, the corticostriatal pathway, arising from the remaining prefrontal association cortex, provides robust reinnervation to the caudate nucleus ipsilateral to the lesion. Both of these reports correlate well with the relative sparing of glucose utilization in the current study seen in the caudate nucleus of the NH versus AH cats (mean LCMRglc depression in the ipsilateral caudate: NH 18%; AH 41%). Pertinent to these findings is the report (Chugani et al., 1989) that at 6 months after hemineodecortication in an infant, cerebral glucose utilization (as measured using positron emission tomography) was reduced in the ipsilateral caudate nucleus. However, 2.5 years after surgery, glucose utilization had increased to reach normal levels.
Overall, it is important to emphasize that, in general, in any of the subcortical structures reviewed herein, there is an interplay between the amount of atrophy-degeneration (tending to depress the metabolic rates) and reinnervation (tending to sustain the rates). For example, (a) the metabolic decrease was maximal in sites with the most atrophy-degeneration (Villablanca et al., 1986a; Hovda and Villablanca, 1989a; Villablanca and Hovda, 1989; Shook and Villablanca, 1991) and the least reinnervation (Villablanca et al., 1982; Olmstead et al., 1983; Villablanca and Gómez-Pinilla, 1987; Sutton and Villablanca, 1990), as in the ventrobasal thalamus of adult hemispherectomized cats; (b) the depression was intermediate in degree in sites with mild atrophy-degeneration and robust reinnervation, like in the superficial layer of the superior colliculus of NH animals (Hovda et al., 1992c;) and (c) the metabolic decrease was minimal or nonexistent in areas with no degeneration and with robust reinnervation, like in the ipsilateral red nucleus of neonatal hemispherectomized cats (Villablanca et al., 1988).
Changes in contralateral hemisphere
Cerebral glucose metabolic depression within the nonsurgically damaged contralateral hemisphere was not surprising, given our previous work indicating that this hemisphere also exhibits atrophy-degeneration following a similar lesion (Villablanca et al., 1986a; Hovda and Villablanca, 1989a; Shook and Villablanca, 1991). What was surprising, however, was that with the exception of the neocortex, these metabolic rate decreases were more marked in the NH than in the AH cats. In our atrophy-degeneration studies of the contralateral hemisphere, we found subcortical changes similar to those seen ipsilaterally, i.e., loss and shrinkage of neurons together with atrophy of subcortical nuclei, although less pronounced (Villablanca et al., 1986a; Hovda and Villablanca, 1989a; Shook and Villablanca, 1991). However, an important age-at-lesion difference was that these changes were generally less marked in neonatal hemispherectomized cats (only occasionally reaching statistical significance) than in the AH animals (reaching significance most of the time). Therefore, we must conclude that in the contralateral hemisphere, the degree of metabolic depression did not match, as seen ipsilaterally, the degree of atrophy–degeneration such that the metabolic depression was higher in NH cats. Consequently, hitherto unknown additional factors might contribute to the tendency to a greater subcortical metabolic depression in the contralateral hemisphere of NH animals. One suggestion is that the massive transhemispheric reduction in synaptic activities produced by the removal of the left neocortex might have had a greater proportional impact following the lesion in the developing versus the adult brain.
Regarding the neocortex, we have shown that, at least for the visual cortex, the neurons that after hemispherectomy project bilaterally tend to be larger in size (Hovda et al., 1992c). This may be related to the relatively less pronounced decrease in metabolic rates in neocortical versus subcortical areas and also to the fact that, for the neocortex, the contralateral depression tended to be less marked for the NH than the AH cats (since the NH animals show a larger number of bilaterally projecting neurons).
Other published studies
In reviewing the literature, we found very few studies attempting to correlate cerebral glucose metabolic shifts with quantitative anatomical changes (including reinnervations) following neocortical resections and none addressing the issue of development.
In a nonquantitative study, Cooper et al. (1984) reported that 9 months following an extensive posterior cortical lesion in rats (n = 2), there was a bilateral decrease of 2DG uptake particularly within the thalamus. Unfortunately, the only anatomical changes reported in this article consisted of a single photograph to “illustrate the considerable shrinkage which took place in the thalamus of the damaged hemisphere.” In another study (Hosokawa et al., 1985), 2DG experiments performed 7 days after a unilateral ablation of the frontal cortex in rats revealed a depression of glucose metabolism within the ipsilateral thalamic, red, and pontine nuclei. The only other structure reported to exhibit a decrease in metabolism was the contralateral cerebellar cortex. In contrast, the ipsilateral striatum did not exhibit any changes in metabolism, and the globus pallidus exhibited a lesion-induced increase in glucose utilization. The only anatomical description was that within the thalamic nuclei, “the number of larger cells was reduced and several spots of gliosis … were observed on the lesioned side.” There was no apparent attempt to assess the anatomical changes in any other structures.
Studies in nonhuman primates (Gilman et al., 1987) focused on glucose metabolic changes primarily within the BG after unilateral cortical ablation of Brodmann areas 4 and 6. These authors reported a moderate to substantial reduction in metabolic rates in all nuclei at 1 week postlesion, with only marginal recovery of metabolism when the animals were studied 2 months after surgery. Contralaterally, there was a small to moderate reduction of glucose utilization that after 6 weeks was almost recovered in the neostriatum but showed only a marginal recovery in the globus pallidus. Anatomically, these authors reported only that there was “a more prominent astrocytic reaction at eight weeks” and that “shrinkage became apparent only after eight weeks.”
SUMMARY
Following cerebral hemineodecortication, the remaining intact areas of the brain are chronically metabolically depressed. This depression is more pronounced in regions that have undergone extensive degeneration and is less evident in areas that have been reinnervated due to the reorganization of pathways arising in the remaining intact cerebral cortex. Animals that have sustained this lesion early in life exhibit less metabolic depression than adult lesioned cats, and this age-at-lesion difference provides important information regarding structure–functional relationships. In previous work we demonstrated that during the age at which we performed the lesion in neonatal animals, the brain is going through a stage of accelerated maturation of both glucose (Chugani et al., 1991) and oxidative (Hovda et al., 1992b) metabolism. This condition, together with the morphological maturational events occurring during this period of the cat's life (Villablanca et al., 1993), appear to provide an optimal developmental environment for neuroplasticity and recovery of function after brain injury. Finally, given the direct relationship of this study to clinical positron emission tomography investigations, the current results may have diagnostic and prognostic implications with regard to patients who sustain damage to the central nervous system early in life.
Footnotes
Acknowledgment:
This work was supported by USPHS grants: HD05958, NS5780, HD04612, NS27544, NS30308, MH37916, NS15654, and DOE DE-AC03-SF 7600012.
