Abstract
Background:
A prospective randomized study of prophylactic mesh placement at the time of cystectomy and ileal conduit found a significant reduction the cumulative incidence of parastomal hernias by ∼50%.>1 We present a novel simple sutureless prophylactic sublay permanent mesh placement at the time of ileal conduit creation.
Materials and Methods:
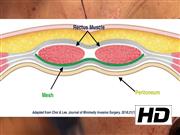
Robotic cystectomy is performed in the standard manner with intracorporeal diversion (n = 34). Open cases (n = 4) were performed through a lower midline incision. The stoma site is marked by the stoma nurse specialists during an outpatient visit before the procedure. The stomal skin and subcutaneous tissue is excised to the anterior rectus sheath. The anterior rectus sheath is incised in a cruciate manner revealing the rectus muscle. The rectus muscle is gently split bluntly between retractors revealing the posterior sheath. The space between the posterior sheath and the rectus is then bluntly dissected circumferentially with a finger. This is the space where the mesh will be placed. The mesh is prepared on the backtable. A 4″ × 6″ Bard Ventralight™ ST mesh is used. This is a permanent medium weight monofilament propylene mesh with an absorbable hydrogel barrier on the posterior side based on Sepra® Technology (ST). A circular mesh with 6 cm in diameter is formed and a cruciate cut is made centrally to fit the diameter of the ileal conduit. The conduit is brought out of the stoma site and the mesh is thread over the conduit with the shiny ST side down; the ST side is in contact with the conduit and prevents adhesions. The mesh is then gently tucked into the dissected space. The rest of the conduit is matured in standard manner. We present two cases in this video of mesh placement at the time of ileal conduit.
Results:
Initially, after observing our colorectal colleagues place mesh in this manner, we used subjective criteria when deciding to perform mesh placement. These factors primarily included body habitus, smoking history, and history of abdominal hernias. Using these criteria we performed mesh placement in 23 patients between March 2019 and July 2020. After having subjectively favorable outcomes without any mesh-related complications, mesh placement became standard for all ileal conduit patients after August 2020. To date, 38 cases have been performed; 34 through robotic intracorporeal diversion. Median follow-up is 4 months (IQR 2–8). There have been no mesh-related complications such as mesh erosion, mesh infection, mesh pain, or need for mesh explant. The follow-up is too short to determine efficacy; however, no clinically apparent parastomal hernias have developed. Twenty-one patients had available postoperative imaging for review by our team with specific attention to subclinical parastomal hernias. Of those, the median radiographic follow-up was 5 months (IQR 3–10); one patient from the robotic group (5%) had radiographic evidence of a parastomal hernia.
Conclusion:
We present a novel simple sutureless technique for prophylactic mesh placement at the time of ileal conduit creation. With short follow-up, there are no evidence of mesh-related complications. Longer follow-up is needed to determine efficacy.
No competing financial interests exist.
Runtime of video: 6 mins


Get full access to this article
View all access options for this article.
