Abstract
Introduction:
Cancer diagnosis in thyroid nodules is typically based on ultrasound examination and fine-needle aspiration (FNA) of suspicious nodules with subsequent cytological evaluation. Whereas ~80% of thyroid nodules are definitively diagnosed as benign or malignant on cytology, 20%–30% of them have indeterminate cytology. Most of these patients undergo diagnostic surgery, which yields a benign diagnosis in ~70% of cases. These surgical procedures, with their costs and complications, could be avoided if a reliable diagnosis of benign nodule could be established without surgery.
Materials and Methods:
PubMed review of English-language literature between 2010 and April 2023 and selected prior publications related to thyroid nodules and molecular diagnostics.
Results:
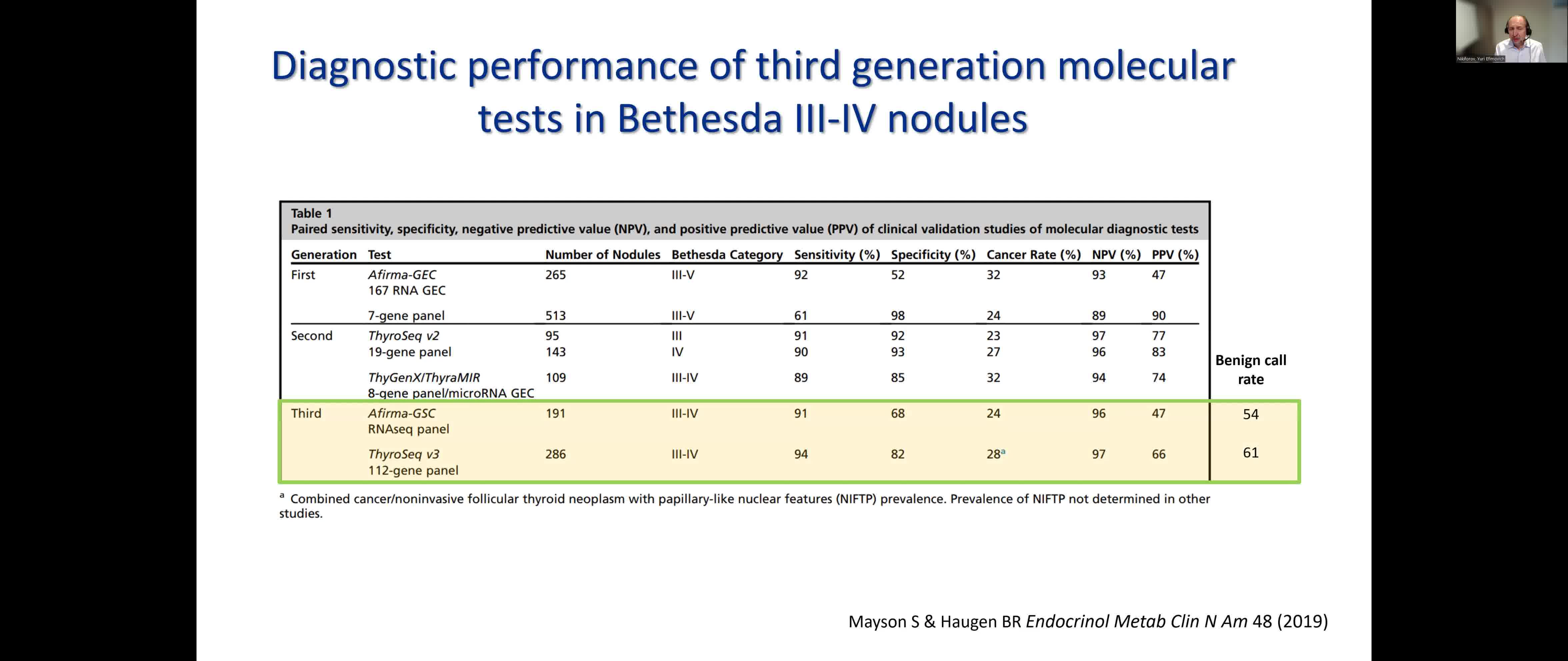
Over the past decade, molecular testing has gradually become part of a routine diagnostic work-up for thyroid nodules with indeterminate cytology in the United States and few other world regions. Third-generation molecular tests for thyroid FNA samples, which include Afirma GSC and ThyroSeq v3 GC, are based on next-generation sequencing and provide high sensitivity and relatively high specificity, and with the benign call rate of ~60% allow to avoid diagnostic surgery in most of patients with indeterminate cytology nodules. Furthermore, information on specific genetic alterations provided by molecular tests offers preoperative cancer risk stratification for test-positive nodules, which can offer additional information informing the optimal extent of surgery, that is, thyroid lobectomy vs total thyroidectomy.
Conclusions:
Molecular testing for indeterminate cytology nodules is necessary to avoid unneeded diagnostic surgeries in most of the patients with indeterminate thyroid nodules. An additional benefit of molecular testing is in informing the extent of surgical excision in patients with test-positive nodules.
IP ownership and receive royalties related to ThyroSeq from the University of Pittsburgh, Consultancy agreement with Sonic Healthcare USA
.
Author(s) have received and archived patient consent for video recording/publication in advance of video recording of procedure.
Runtime of video: 11 mins 14 secs


Get full access to this article
View all access options for this article.
