Abstract
Contributors
The following document and appendices represent the third edition of the Practice Guidelines for Ocular Telehealth-Diabetic Retinopathy. These guidelines were developed by the Diabetic Retinopathy Telehealth Practice Guidelines Working Group. This working group consisted of a large number of subject matter experts in clinical applications for telehealth in ophthalmology.
The editorial committee consisted of Mark B. Horton, OD, MD, who served as working group chair and Christopher J. Brady, MD, MHS, and Jerry Cavallerano, OD, PhD, who served as cochairs. The writing committees were separated into seven different categories. They are as follows: Clinical/operational: Jerry Cavallerano, OD, PhD (Chair), Gail Barker, PhD, MBA, Christopher J. Brady, MD, MHS, Yao Liu, MD, MS, Siddarth Rathi, MD, MBA, Veeral Sheth, MD, MBA, Paolo Silva, MD, and Ingrid Zimmer-Galler, MD. Equipment: Veeral Sheth, MD (Chair), Mark B. Horton, OD, MD, Siddarth Rathi, MD, MBA, Paolo Silva, MD, and Kristen Stebbins, MSPH. Quality assurance: Mark B. Horton, OD, MD (Chair), Seema Garg, MD, PhD, Yao Liu, MD, MS, and Ingrid Zimmer-Galler, MD. Glaucoma: Yao Liu, MD, MS (Chair) and Siddarth Rathi, MD, MBA. Retinopathy of prematurity: Christopher J. Brady, MD, MHS (Chair) and Ingrid Zimmer-Galler, MD. Age-related macular degeneration: Christopher J. Brady, MD, MHS (Chair) and Ingrid Zimmer-Galler, MD. Autonomous and computer assisted detection, classification and diagnosis of diabetic retinopathy: Michael Abramoff, MD, PhD (Chair), Michael F. Chiang, MD, and Paolo Silva, MD.
Table of Contents
I. Principles of an ocular telehealth program for DRߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ498
A. Missionߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ498
B. Visionߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ498
C. Goalsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ498
D. Guiding principlesߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ498
II. Ethicsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ498
III. Clinical validationߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ498
A. Category 1ߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ499
B. Category 2ߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ499
C. Category 3ߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ500
D. Category 4ߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ500
IV. Communicationߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ501
V. Personnel qualificationsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ501
A. Medical care supervisorߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ501
B. Patient care coordinatorߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ501
C. Image acquisition personnelߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ501
D. Image review and evaluation personnelߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ501
E. Information systems personnelߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ501
I. Equipment specificationsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ501
A. Image acquisitionߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ503
B. Image displayߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ503
C. Image analysisߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ504
II. Data managementߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ504
A. Interoperabilityߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ504
B. Compressionߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ504
C. Data communication and transmissionߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ504
D. Archiving and retrievalߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ504
E. Securityߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ505
F. Reliability and redundancyߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ505
G. Documentationߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ505
I. Legal requirementsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ505
A. Facility accreditationߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ505
B. Health Insurance Portability and Accountability Actߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ505
C. Privileging and credentialingߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ505
D. Fraud and abuseߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ505
E. State medical practice acts/licensureߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ506
F. Tort liabilityߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ506
G. Consentߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ507
II. Quality controlߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ507
III. Operationsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ507
IV. Customer supportߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ507
A. Originating siteߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ507
B. Data transmissionߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ508
C. Distant siteߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ508
V. Financial factorsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ508
A. Reimbursementߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ508
B. Grantsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ508
C. Federal programsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ508
D. Other financial factorsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ508
E. Equipment costߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ509
1. Clinical Validationߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ510
2. Interoperabilityߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ511
3. Autonomous and Computer-Assisted Detection, Classification, and Diagnosis of Diabetic Retinopathyߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ513
4. Health Insurance Portability and Accountability Actߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ519
5. Privileging and Credentialingߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ519
6. Quality Controlߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ520
7. Customer Supportߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ521
8. Reimbursementߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ522
9. Telemedicine for Glaucoma: Guidelines and Recommendationsߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ524
10. Telemedicine for Retinopathy of Prematurityߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ527
11. Telemedicine for Age-Related Macular Degenerationߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙߙ533
Preamble
The American Telemedicine Association (ATA) brings together diverse groups from traditional medicine, academia, technology, telecommunications companies, e-health, allied professional and nursing associations, medical associations, government, military, regulatory, and other stakeholders to address and advance compliance with legal, ethical, and professional standards in the practice of telemedicine. The ATA has embarked on an organized effort to establish guidelines for the practice of telemedicine in various clinical applications to define patient and provider expectations, aspire to uniform quality of service for patients and providers, enhance patient experience, and enable providers to deliver appropriate care using evidence-based practices.
The guidelines are developed by panels that include experts from the field and other strategic stakeholders, and are designed to serve as a standard reference and educational tool for professionals using telehealth tools for health care service delivery. The process for developing these guidelines is based on evidence, professional consensus, and a rigorous review, including open public commentary period, with final approval by the ATA Board of Directors. Guidelines are reviewed and updated periodically.
The purpose of these guidelines is to assist providers in pursuing a sound course of action in providing safe and effective medical care using telehealth tools based on current scientific knowledge, technological requirements, and patient needs. Safe and effective practice requires technical training, professional knowledge and skill, and explicit processes as described in each document.
Adherence to these guidelines alone will not guarantee accurate diagnoses, appropriate clinical treatment, or optimal outcomes. Appropriate divergence from the guidelines may be indicated under certain conditions, such as emergency situations or locations with limited resources or other unavoidable constraints. Similarly, technological advances may alter prevailing practices or provide new and expanded opportunities.
The guidelines in this document are based on the accumulated knowledge and experience of the ATA workgroups, eye care and telemedicine professionals, and other stakeholders, and generally describe the evidenced-based best practices for ocular telehealth. However, the technical and administrative guidelines do not purport to establish binding legal standards for delivering telemedicine services.
The previous ATA Ocular Telehealth Diabetic Retinopathy Practice Guidelines were issued in 2011. This third edition reflects new evidence, new technologies, and expanded scope of the ocular telehealth domain. All guidelines issued by the ATA are properties of the ATA. Any modification or reproduction of the published guidelines must receive prior approval by the ATA.
Scope
The following document includes fundamental requirements to be followed when providing medical and other health care services using telecommunication technologies, and any other electronic communications between patients, practitioners, and other health care providers, as well as “best practice” recommendations. The guidelines apply to individual practitioners, group and specialty practices, hospitals and health care systems, and other providers of health-related services where there are telehealth interactions between patients and health care service providers.
When guidelines, position statements, or standards from any other professional organization or society exist, health professionals should also review these documents and, as appropriate, incorporate them into practice.
These guidelines pertain primarily to health care professionals and patients located in the United States. In situations wherein either or both parties are not within the United States, these guidelines may be referenced, but any local guidelines that are in place should take precedence. 1 –3
These guidelines are intended to be used as a companion to the ATA Core Operational Guidelines for Telehealth Services.
4
Recommendations in the core guidelines are not repeated herein except to emphasize or expand upon a particular point, or to provide domain-specific detail. The reader
The guidelines address three aspects of service delivery: clinical, technical, and administrative. Based upon the quantity and quality of peer-reviewed evidence, the guidelines are classified into four levels of adherence:
“
“
“
“
These indications are found in bold throughout the document.
Introduction
These guidelines present recommendations for designing, implementing, and operating an ocular telehealth diabetic retinopathy (DR) program in a broad range of clinical settings and targeted outcomes. This document also addresses current clinical, technical, and administrative issues that form the basis for evaluating DR telehealth techniques and technologies. These guidelines are intended to be consistent with federal regulations and industry best practices at the time of publication that emphasize clinical quality, data security and integrity, and interoperable health information exchange. Federal, state, and regional regulations supersede the recommendations in these guidelines. This document will be reviewed periodically and revised to reflect evolving technologies, evidence, regulations, and clinical guidelines.
This third edition of the guidelines includes four new clinical appendices that introduce additional ocular telehealth domains (Appendices A3, A9–A11). These are planned for future development into independent guidelines to be included in an ocular telehealth suite of practice guidelines.
Practice Guidelines
Clinical Guidelines
I. Principles of an ocular telehealth program for DR
Private individuals, public and private organizations, and national and international agencies may undertake telemedicine programs for DR that have been shown to be efficacious, cost-effective, and scalable means to identify diabetes-related eye disease and thereby prevent visual loss. Designing, building, implementing, and sustaining an ocular telehealth DR program require clearly defined mission, vision, goals, and guiding principles. The following statements are a guide for leadership and staff in developing and sustaining appropriate and effective programs.
A. Mission
Increase cost-effective and culturally sensitive access and adherence to accepted standards of eye care for people with diabetes mellitus (DM).
B. Vision
Ocular telehealth can be an integral component of primary care for people with DM by expanding patient-centric access to retinal examinations consistent with evidence-based recommendations for eye care in diabetes.
C. Goals
Improve access to diagnosis and evidence-based management of DR.
Reduce the incidence of vision loss due to DR.
Decrease the cost of identifying patients with DR.
Promote telehealth to enhance the efficiency and clinical effectiveness of evaluation, diagnosis and management of DR.
Promote telehealth to enhance the availability, patient centricity, quality, efficiency, and cost-effectiveness of remote evaluation and management of DR.
Facilitate integration of diabetes eye care with primary and specialty medical care.
Promote widespread adoption of telehealth services for DR. 5
D. Guiding principles
Although ocular telehealth programs offer new opportunities to improve access and quality of care for people with DR, programs
DM adversely affects the entire eye and has a diverse influence on visual function. Patients should be aware that a validated teleophthalmology examination of the retina may substitute for a traditional onsite dilated retinal evaluation for DR, but patients
II. Ethics
Regardless of the program, the care of the patient
An explicit code of ethics.
Compliance with applicable federal, state, and jurisdictional laws and regulations, and institutional policies.
Nondiscrimination clause regarding denial of service to people on the basis of disability, gender, gender preference or sexual orientation, ethnicity, national origin, or religious affiliation.
Provision of care in a culturally sensitive manner.
Provision of service not conditional upon receipt of payment by the patient.
III. Clinical validation
Multicenter national clinical trials provide evidence-based criteria for clinical guidelines in diagnosing and treating DR (
Appendix A1). Telehealth programs for DR
Early Treatment Diabetic Retinopathy Study (ETDRS) 30°, stereoscopic, seven-standard field, color, 35 mm slides (based on the ETDRS extension of the modified Airlie House classification of DR, 6 ETDRS photographs) have been the gold standard for evaluating DR in major clinical trials of DR. Although no standard criteria have been widely accepted as performance measurements of digital imagery used for DR evaluation, current clinical trials sponsored by the National Eye Institute have transitioned to digital images for DR assessment. 7,8
Telehealth programs for DR
It is recognized that severity levels of DR other than those defined by the ETDRS are used clinically for grading DR (see Table 1 for comparisons between ETDRS levels of DR and the International Clinical Diabetic Retinopathy Disease Severity Scale, and Table 2 for comparisons between ETDRS DME and the International Clinical Diabetic Retinopathy Disease Severity Scale).
14
Protocols
International Clinical Diabetic Retinopathy Scale Compared with Early Treatment Diabetic Retinopathy Study Levels of Diabetic Retinopathy
DR, diabetic retinopathy; ETDRS, Early Treatment Diabetic Retinopathy Study; NPDR, nonproliferative diabetic retinopathy; PDR, proliferative diabetic retinopathy.
International Clinical Diabetic Macular Edema Scale Compared with Early Treatment Diabetic Retinopathy Study Where Noted
CSME, clinically significant macular edema; DME, diabetic macular edema; HE, hard exudates.
This Practice Guidelines for Ocular Telehealth-Diabetic Retinopathy defines four major categories of validation for DR telehealth programs using ETDRS photographs as the reference standard. The validation study
Validation categories are not a quality continuum, but rather performance categories that describe distinct clinical outcomes of public health relevance reflecting program goals. In addition, they provide a standardized language for communicating performance for clinical, research, reimbursement, request for proposal (RFP), and regulatory compliance purposes. Information about the program's validation study design and performance
A. Category 1
Category 1 validation indicates a system can separate patients into one of two groups: (1) those who have no or very mild nonproliferative diabetic retinopathy (NPDR) (ETDRS level 20 or below) and (2) those with levels of mild NPDR or greater (greater than or equal to ETDRS level 35). Functionally, category 1 validation allows screening for presence versus absence of DR.
B. Category 2
Category 2 validation indicates a program accurately determines if sight-threatening diabetic retinopathy (STDR) or potentially STDR is present or not present as evidenced by any level of DME, severe or worse levels of NPDR (ETDRS level 53 or worse), or proliferative diabetic retinopathy (PDR) (ETDRS level 61 or worse). Functionally, category 2 allows screening for presence versus absence of STDR or potentially STDR.
C. Category 3
Category 3 validation indicates that a program accurately identifies ETDRS-defined clinical levels of NPDR (mild, moderate, or severe), PDR (early and high risk), and DME (central-involved DME or not central-involved DME). Functionally, category 3 validation provides a clinical diagnosis of DR/DME severity to match conventional clinical retinal examination through dilated pupils or ETDRS photographs, allowing remote management of the patient.
D. Category 4
Category 4 validation indicates that a program accurately identifies the presence and degree of specific lesions of DR to match the ability of ETDRS photographs to determine all specific lesions and levels of DR and DME, ranging from levels 10 to 90. Functionally, category 4 validation indicates a program can replace or coexist with ETDRS photographs as a gold standard and
The validation categories entail all components of a program (end-to-end) and do not refer to any single element such as the retinal imaging device, imaging protocol, image manager, compression protocol, image display, and image review protocol.
Determination of the validation category
Threshold sensitivity and specificity for validation categories 1 and 2
The study that establishes the program's clinical performance and validation category applies to all its implementations. Clinical fidelity with the validation study is maintained by standardized implementation and ongoing quality assurance (QA) (
Appendix A6). Accordingly, individual implementations within the original program need not be restudied. However, substantial changes in technology or clinical operations
A telehealth program's validation category impacts clinical, business, and operational features. The category influences hardware and software technology, staffing and support, clinical workflow and outcomes, participant licensure, QA, and business plan. Equipment cost, technical difficulty, operational complexity, and training requirements increase with increasing program performance as measured by validation category. 18
A telehealth program's goals and desired performance
Pupil dilation is associated with a small risk of angle-closure glaucoma. Although the risk of inducing angle-closure glaucoma with dilation using 0.5% tropicamide is minimal with no reported cases in a large meta-analysis,
24,25
programs using pupil dilation
Depending on the telehealth program operational preferences and validation category, images
It is possible that a program without stereoscopic capabilities or OCT may be validated to identify macular edema with acceptable sensitivity,
28,31
even though stereoscopic evaluation of DME is significantly more sensitive and specific than monoscopic techniques.
21
Artificial intelligence (AI) algorithms may offer another indirect measure of DME that has sufficient accuracy to warrant clinical applications in some settings
32
(Appendix A3). A program
IV. Communication
Communication is the foundation of ocular telehealth.
33,34
Communication
V. Personnel qualifications
Telehealth programs for DR depend upon a variety of functions. Distinct individuals may assume these responsibilities or a person may assume several roles depending on the size and scope of the program. Qualifications of these personnel shall be documented, including initial and recurrent training.
A. Medical care supervisor
An appropriately licensed ophthalmologist or optometrist with expertise in evaluation and management of DR
B. Patient care coordinator
The patient care coordinator ensures that each patient receives DR education and completes appropriate follow-up, especially for those meeting criteria for referral. A program
C. Image acquisition personnel
Image acquisition personnel (“imagers”) are responsible for acquiring retinal images. A licensed eye care professional may not be physically available at all times during a telehealth session, so imagers
Understanding of basic ocular telehealth technology and principles.
Qualifications for obtaining appropriate image fields of diagnostic quality.
Understanding of the clinical appearance of common retinal diseases requiring immediate or urgent evaluation.
Communication skills for acquiring patient informed consent and providing patient education.
Basic understanding of angle-closure glaucoma if pupil dilation is performed, including entry-level skills in screening for shallow anterior chamber and recognition of angle-closure signs and symptoms.
D. Image review and evaluation personnel
Image review and evaluation specialists (readers) are responsible for timely grading of images for retinal lesions and determining levels of DR. Only qualified readers
A licensed qualified optometrist or ophthalmologist with expertise in DR and familiarity with program technology
E. Information systems personnel
An information systems specialist is responsible for system privacy/confidentiality protocols, connectivity, data integrity, availability of stored images, and disaster recovery.
36,37
The specialist
Technical Guidelines
I. Equipment specifications
Telehealth systems used in the United States Image acquisition hardware (computers, cameras, and other peripherals). Image transmission, storage and retrieval, and display systems. Image analysis and clinical workflow management (scheduling follow-up examinations, clinical communication management, and decision support tools). Security and confidentiality of protected health information (PHI) and images.
Equipment specifications will vary with program needs, validation category, and available technology (Fig. 1). Equipment
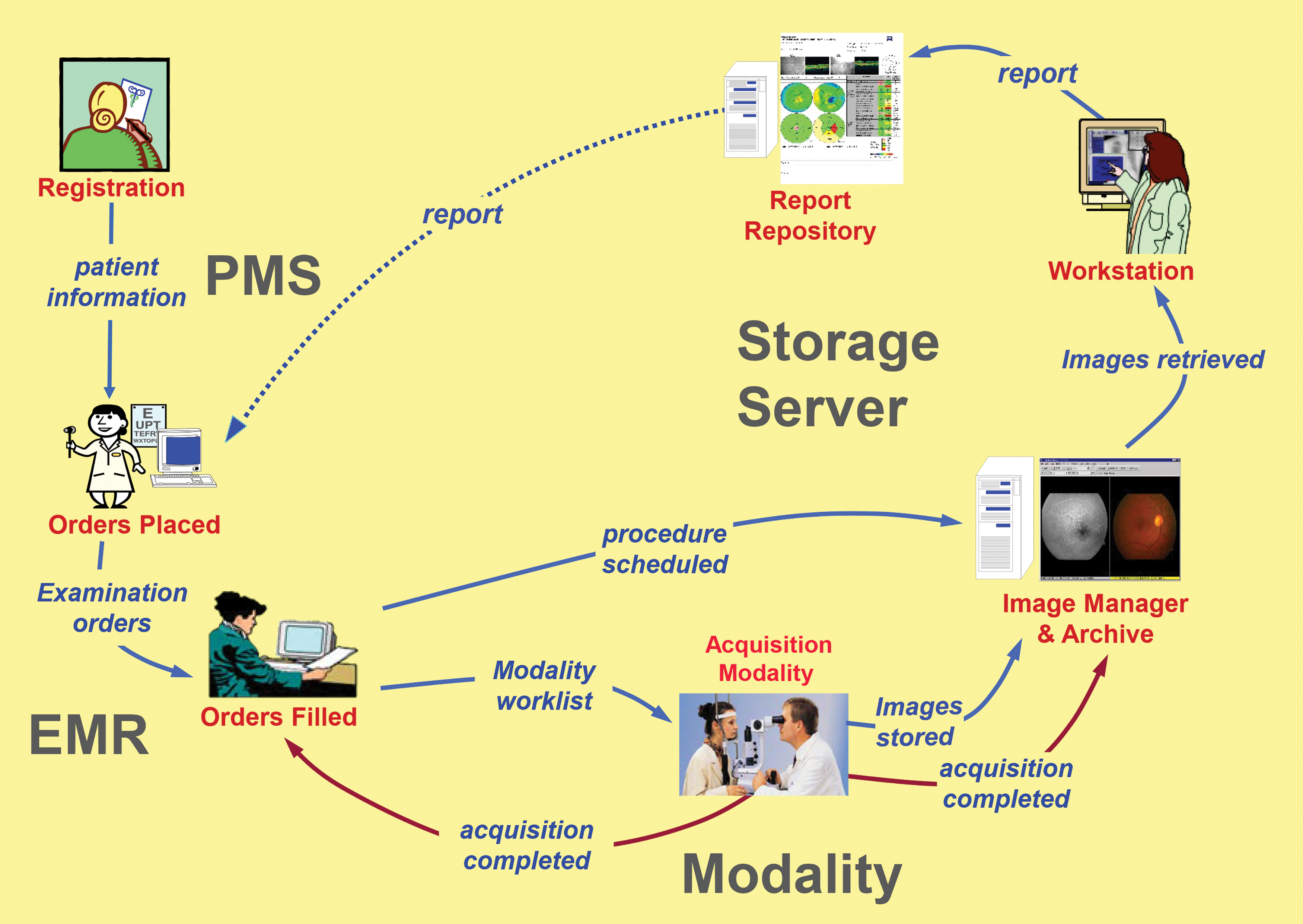
IHE-Eye Care, unified eye care workflow (Reprinted with the permission of the IHE Eye Care Domain). EMR, electronic medical record; IHE, Integrating the Healthcare Enterprise; PMS, patient management system.
All relevant technologies, including image acquisition, image management/Picture Archiving and Communication System (PACS), and interfaces to patient management systems (PMSs), and electronic medical records (EMRs)/electronic health records (EHRs),
A. Image acquisition
To provide alignment with the accepted standards for medical imaging, retinal image data sets
There are many equipment options available for image capture, but most devices currently used in telemedicine for DR (Tmed-DR) are flash-based fundus cameras designed for eye clinic settings and adapted for telemedicine use.
21
The device selected
Many factors must be considered when selecting a particular retinal imaging device and imaging protocol. Most commercially available retinal imaging devices have sufficient resolution for Tmed-DR. The minimum resolution for this purpose is 20 pixels per degree. 49 Diagnostic accuracy of the system is the pivotal feature that enables a particular validation category. The ungradable rate is a related feature since this rate can affect the system's functional specificity.
Important features influencing diagnostic accuracy include field of view (FOV) and mydriatic versus nonmydriatic imaging. 21 Although variation in methodology makes it difficult to compare existing reports, in general, larger aggregate FOV and mydriasis are associated with the highest diagnostic accuracy and lowest ungradable rate when using flash photography. (Table 3) The total FOV is the most influential feature in this consideration, with nonmydriatic ultrawide field imaging performance roughly equivalent to multifield mydriatic systems. 21
Meta-analysis of a 20-Year Review of the Telemedicine for Diabetic Retinopathy Literature
The predominate format of 45° FOV systems are shown. Mean sensitivity and specificity to match program goals (diabetic retinopathy detection, severity, or referral rate) and study ungradable rate (± standard deviation) of 45° FOV systems using 1, 2, or 3 mydriatic or nonmydriatic 45° fields.
Note: Adapted from Horton et al. 24
FOV, field of view.
The form factor of the imaging station (retinal camera and supporting equipment) is an early consideration during equipment selection. A system that can be easily transported between sites allows an increased and adaptable catchment area for the program while limiting equipment costs. Most retinal imaging devices for this purpose must be adapted from devices designed and marketed for conventional clinic applications.
Mobile systems based upon a smartphone platform have a favorable form factor and cost features, and carry the additional advantage of integrated image transmission. Although clinical potential has been demonstrated with these devices, 50 limited sensitivity and specificity for DR detection and severity level diagnosis limit their use. Moreover, a lack of standardization and a short product cycle life create significant business and interoperability challenges. 51
Portable systems using handheld imaging devices are larger and more costly than smartphones, and may suffer from some of the same limitations. 52 –54 High-quality evidence of their efficacy is lacking, although studies are ongoing to validate these devices. 55
Another alternative for portable Tmed-DR operations is the conversion of a conventional fundus camera for portable use by use of a transportable case. This method retains the performance and connectivity benefits of the conventional retinal imager but often requires the construction of a customized hardened case for device protection, resulting in a large and heavy item that may be cumbersome to move, and requires a desktop configuration.
B. Image display
Retinal images used for diagnosis
C. Image analysis
Computer algorithms to enhance digital retinal image quality or provide automated identification of retinal pathology are emerging technologies. Image analysis tools for enhancing image quality (histogram equalization, edge sharpening, image deconvolution, etc.) or identifying lesions such as microaneurysms, hemorrhages, or hard exudates can be used to aid retinopathy assessment. Computer algorithms may also be used to facilitate and standardize reader assessment of DR and DME severity using rules based upon accepted standards. Appendix A3 summarizes the use of autonomous and computer-assisted detection for classification and diagnosis of DR image processing.
Computer algorithms for DR assessment of retinal images
The nature of telemedicine allows clinical and related patient data to be reviewed remotely in a nonclinical setting where ambient conditions and privacy are less controlled. Staff involved in assessment of Tmed-DR images and related data
II. Data management
A. Interoperability
Health information technology (HIT) interoperability is the ability of systems to exchange and use electronic health information from other systems without special effort on the part of the user to advance the health status of and the effective delivery of health care for individuals and communities. 56 HIT interoperability has been recognized as a key element in moving the health care system toward improved outcomes, patient safety, and efficiencies. 57
In the United States, an integrated digital health care system has been described by federal regulations and its implementation heavily incentivized. Initially these incentives occurred through supplemental payments, but more recently this approach has transitioned to a system of financial penalties for nonconforming providers and health care facilities. This emphasis stems from evidence that harmonized communication of HIT improves operational efficiency, patient safety, and public health reporting through the availability of patient health information at the right place and the right time. The current regulatory roadmap suggests continued regulatory attention to interoperability,
58,59
so ocular telehealth programs
B. Compression
Data compression may facilitate efficient transmission, storage, and retrieval of retinal images, and
C. Data communication and transmission
A variety of technologies are available for data communication. Ocular telehealth programs
If ocular telehealth applications are integrated with existing health information systems, interoperability
D. Archiving and retrieval
Ocular telehealth systems
Each facility
E. Security
Ocular telehealth systems
F. Reliability and redundancy
Written policies and procedures
G. Documentation
Readers rendering reports on DR or other ocular abnormalities
Administrative Guidelines
I. Legal requirements
Legal and regulatory issues relating to the practice of ocular telehealth are generally the same as other telemedicine modalities and carry the risk management considerations of conventional medical practice.
33,70,71
A DR telehealth program
A. Facility accreditation
Some hospital telehealth programs fall within regulatory jurisdictions of The Joint Commission (TJC) and/or Centers for Medicare and Medicaid Services (CMS). 72 TJC and Accreditation Association for Ambulatory Health Care accredit ambulatory health care facilities. 73,74 These accrediting bodies publish standards that apply to telemedicine activities, making regulatory compliance a mandatory component for most hospital-based telehealth programs. There are specific references to telemedicine in TJC Environment of Care and Medical Staff sections, including LD.04.03.09, MS.13.01.01, and MS.13.01.03. 75
CMS requirements also occur indirectly through related activities, such as standards for contract care. There are other accreditation standards that may apply to a specific program and clinical setting, with similar, but not identical requirements. Awareness and understanding of these standards and the applicable CMS regulations can be daunting.
76
Ocular telehealth programs
B. Health Insurance Portability and Accountability Act
Ocular telehealth programs
C. Privileging and credentialing
Ocular telehealth providers
D. Fraud and abuse
Telemedicine programs are subject to the fraud and abuse statutes and regulations concerning health care-related kickbacks and other financial inducements for referrals. The antikickback statute prohibits payment or any receipt of remunerations for referrals or purchasing equipment reimbursable under federal health programs. 83 The language in this law is so broad that “Safe Harbors” were created to lessen the impact on legitimate ventures. 84
The Stark Act prohibits physicians from making a referral for designated health services to an entity with which the physician (or immediate family member) has a financial relationship. 85,86 Self-referrals occur when physicians refer patients to medical facilities in which they or their immediate family have a financial interest. For example, an ophthalmologist places a retinal imaging workstation in a primary care provider's office at deep discount or gratis and reads images at little or no charge. The Stark statute may have been violated if patients needing treatment are referred to the ophthalmologist. This practice may be avoided by charging the primary care provider full market value for equipment and services and offering the patient a choice of referral ophthalmologists for treatment. 87
Ocular telehealth programs
Another area of risk under the general category of fraud and abuse is antitrust. Although telemedicine and other e-health practices offer the opportunities of improved business efficiencies, reduced incremental costs of services, and new product offerings, in certain settings they may also be interpreted as restraining trade. To mitigate antitrust risks, the ocular telehealth program
E. State medical practice acts/licensure
In general, telehealth legal issues assume telemedicine is the practice of medicine, and telemedicine and telehealth programs are subject to the ordinary laws and regulatory oversight that govern all medical providers. These issues are addressed variably by state medical practice acts, but even in the absence of specific statutory or regulatory definitions, telehealth legal claims would be difficult to defend against otherwise. 33
All 50 states, the District of Columbia, and the U.S. territories require licensure for rendering medical care to patients located in their jurisdiction, and a physician is considered subject also to the medical practice laws and regulations where the patient is located. Many states provide for some degree of telemedicine-friendly licensure or license “portability” for telemedicine, including a small number of states with telemedicine or special purpose licensure, and a larger number with participation in the Interstate Medical Licensure Compact. 88 This compact allows qualified physicians seeking to practice in multiple states to be eligible for expedited licensure in all states participating in the compact.
The ATA Interstate Telehealth Special Interest Group (SIG) is a source of current information on cross-border practice developments.
89
Since this is an active topic for legislative attention in many states, all programs
F. Tort liability
Telemedicine may reduce overall liability risks through improved access and quality of care and improved documentation. However, experience indicates that telemedicine may increase the risk for liability for providers and facilities that use it and for those who chose to not use it. The elements of a medical malpractice claim are well established, but telemedicine can also complicate traditional tort liability. Issues include which entity or physician owes a duty to the patient, standards-of-care, jurisdiction, and choice of law. 33 Although telemedicine providers should consult an attorney familiar with telemedicine law, the fundamental aspects of tort law are fairly uniform across jurisdictions:
A physician has a duty to a patient to act within the accepted standards-of-care.
Standards-of-care were violated.
A patient suffered an injury due to the violation of standard-of-care.
Duty
A physician's duty arises from the physician–patient relationship. 92 Telemedicine alters the traditional context of this relationship but a telemedicine encounter is sufficient to establish the relationship. 33,93
Standards-of-care
The American Medical Association believes medical specialty societies should develop or participate in the development and implementation of telemedicine clinical guidelines and position statements. 94 Because telemedicine standards-of-care are not universally established and recognized, questions could arise regarding appropriateness of a telemedicine DR evaluation, whether appropriate technology was selected (e.g., Validation Category 1, 2, 3 or 4), or whether the outcome was appropriate for a particular setting or case. An example of a controversial outcome is failure to diagnose nondiabetic retinopathy pathology evident in images (e.g., venous occlusion and choroidal neovascular membrane [CNVM]), or not evident in images (e.g., choroidal melanoma anterior to the equator and peripheral retinal tear/retinal detachment).
Issues of jurisdiction, choice of laws, and apportionment of liability are additional issues that are incompletely defined by statute and case law.
95
Telehealth providers
G. Consent
Patients have the right to autonomous informed participation in health care decisions,
96
but this right cannot be exercised without enough information to allow an informed choice.
97
Informed consent is required for clinical treatments and procedures, including those delivered through telemedicine. When treatments or procedures delivered through ocular telehealth are considered low risk and within commonly accepted standards of practice, oral consent may be sufficient and a written and signed consent may not be required.
81
Ocular telehealth services for DR may satisfy these criteria. Patients
Whether the services is novel or experimental.
Differences between care delivered using ocular telehealth and face-to-face examination.
Benefits and risks of using ocular telehealth in the patient's situation.
Description of what is to be done at the patient's site and the remote site.
Informed consent requirements vary from state to state, and currently, only a few states have laws that mandate informed consent for telemedicine treatment. However, ocular telehealth providers and programs
II. Quality control
A structured process for quality control and ongoing performance improvement is fundamental in health care,
98
and no less so for Tmed-DR. A codified method for collection, analysis, and reporting of programmatically relevant data
The technical quality of images and completeness of associated clinical data
Corrective action of undesired trends and context-sensitive continuing education (CE)
III. Operations
An operations manual is a comprehensive documentation of how a program functions on a daily and ongoing basis. A DR operations manual contains operational information and description of key processes in sufficient detail to provide standardized performance at all levels of the program, and also guide new leadership and staff. It can also describe QA and staff training procedures, but is not intended to function as an employee handbook. A comprehensive manual enables normal operations during leadership absence, and provides a pathway to programmatic sustainability during staffing changes at any level. Ocular telehealth programs
IV. Customer support
Ocular telehealth programs use advanced technology in a broad range of settings, operated by diverse staff with varying training and expertise. Ocular telehealth programs
A. Originating site
Imager: imaging process, hardware/software, initial training and provisional certification, recurrent training, QA, and evidenced-based recertification.
Imaging device: image acquisition, operator-based service, device faults, preventive maintenance, site-based calibration, and diagnostics.
Provider/clinical contact: report retrieval/delivery and interpretation, patient recall, and billing.
B. Data transmission
Connectivity/network errors.
Data loss/recovery.
C. Distant site
Reader adjudication, initial training and provisional certification, QA/peer review, recurrent training, and evidence-based recertification.
Diagnostic display equipment and software.
OS and DS may be in the same facility with data transmission contained within a single local area network. Support for such systems is typically less complex than geographically distributed programs involving multiple networks and servers. Technical support can be divided into levels, or tiers, depending on difficulty or urgency. Tiered help desks are common and a convenient way to accommodate program needs efficiently. A DR telehealth program
V. Financial factors
Telehealth program sustainability depends on a well-developed and executed business plan. The actual cost of services can be a complex calculation, and reimbursement depends upon accurate diagnostic and procedural coding, and pays for performance and quality incentives. The specifics of these issues vary between regions, payers, and clinical settings, so each program should tailor billing protocols with Medicare, Medicaid, and private insurance intermediaries.
A. Reimbursement
Billing codes and reimbursement coverage are pivotal components for successful reimbursement. Billing is usually divided into technical or image capture (Current Procedural Terminology [CPT] suffix TC) and professional or interpretation components (CPT suffix 26). Before 2011, most DR telehealth programs used the 92250 (Fundus Photography with Interpretation and Report) CPT code. Infrequently, programs used CPT 92499 (Unlisted Ophthalmic Service or Procedure), which requires negotiated use with the fiscal intermediary or carrier.
In 2011, the CMS approved two new codes specific for remote retinal imaging, CPT 92227 and 92228. The reimbursement landscape is highly dynamic, and has substantial state, regional, and payer differences. Failure to attend to these changing differences appropriately can result in failed reimbursement and, in some instances, costly penalties. For these reasons, programs
B. Grants
Grants have been used to establish telemedicine programs for defined circumstances and duration. Although an important method for proof of concept, grants are usually not viable for sustained clinical operation. As telehealth programs become more common as routine tools for health care, grants have become less common business plans for DR ocular telehealth. DR telehealth programs
C. Federal programs
There are several large telemedicine programs that reside within federal agencies and are funded by recurring federal appropriations. Examples include the Indian Health Service and the Veterans Health Administration. These programs sometime supplement their federal appropriations with external reimbursements, but their predominant business plan is cost avoidance through improved outcomes stemming from increased compliance with standards-of-care.
D. Other financial factors
Nonrevenue financial benefits of a DR telehealth program may include cost savings over traditional care delivery; however, benefits may not be realized by the entity creating them. For example, patients and third-party payers may realize financial savings through cost avoidance produced by a DR telehealth program, whereas the primary care physicians funding the program realize little or no direct savings.
Under current reimbursement policies in the United States, DR telehealth may be a better business model in closed systems, such as managed care, where costs and return on investment are realized by the same entity that funds and operates the program. However, it is important to recognize that cost-avoidance benefits occur over time and may not immediately offset day-to-day operational expenses. Government pay-for-performance incentive programs may change the relationship between program funding and reimbursement in the future. Appendix A8 contains financial information on logistic efficiencies, disease prevention, and resource utilization.
E. Equipment cost
Imaging costs depend on many factors, but with the decreasing cost of computing and telecommunications, a retinal camera is frequently the largest capital investment for a DR telehealth program. Retinal imaging devices range from $3,500 to >$85,000, including fundus camera, camera back, auxiliary lenses, computer, software, and network hardware. Almost all retinal imagers used by ocular telehealth DR programs are adaptations of devices designed for conventional eye clinic use. Consequently, they have technical and operation features and price points that are not optimized for the telehealth setting. The specific imaging device selected for a particular ocular telehealth DR program depends on its target clinical goals, business plan, clinical design, and other factors tied to clinical outcome and program scalability and sustainability. An ocular telehealth DR program
Abbreviations
adverse events
artificial intelligence
age-related macular degeneration
Age-Related Eye Disease Study
American Telemedicine Association
binocular indirect ophthalmoscopy
critical access hospital
computer-based image analysis
continuing education
Centers for Medicare and Medicaid Services
convolutional neural networks
choroidal neovascularization
choroidal neovascular membrane
conditions of participation
Current Procedural Terminology
continuous quality improvement
clinically significant macular edema
Digital Imaging and Communications in Medicine
diabetes mellitus
diabetic macular edema
diabetic retinopathy
distant site
electronic health records
electronic medical record
Early Treatment Diabetic Retinopathy Study
Food and Drug Administration
field of view
geographic atrophy
hard exudates
Health Insurance Portability and Accountability Act
health information technology
Health Level 7
Integrating the Healthcare Enterprise
licensed independent provider
Medicare Administrative Contractors
neonatal intensive care unit
nonproliferative diabetic retinopathy
optical coherence tomography
originating site
Picture Archiving and Communication System
proliferative diabetic retinopathy
protected health information
patient management system
quality assurance
quality-adjusted life year
request for proposal
retinal nerve fiber layer
receiver operating characteristics
retinopathy of prematurity
relative value units
referral-warranted ROP
Special Interest Group
Systematic Nomenclature of Medicine Clinical Terms
Systematized Nomenclature of Medicine
sight-threatening diabetic retinopathy
Stanford University Network for Diagnosis of Retinopathy of Prematurity
The Joint Commission (formerly Joint Commission for the Accreditation of Healthcare Organizations)
telemedicine for DR
Telehealth Resource Centers
vascular endothelial growth factor
Glossary
DICOM: Digital Imaging and Communication in Medicine.
An international standard for distributing, storing, and viewing medical images.
HL7: Health Level 7.
An international framework for the electronic exchange of clinical, financial, and administrative information among computer systems in hospitals, clinical laboratories, pharmacies, etc.
IHE: Integrating the Healthcare Enterprise.
A global initiative by health care professionals and industry to improve computer sharing of health care information through coordinated use of established standards such as DICOM and HL7.
SNOMED CT®: Systematic Nomenclature of Medicine Clinical Terms.
A system of clinical health care terminology covering diseases, findings, procedures, microorganisms, pharmaceuticals, etc.
