Abstract
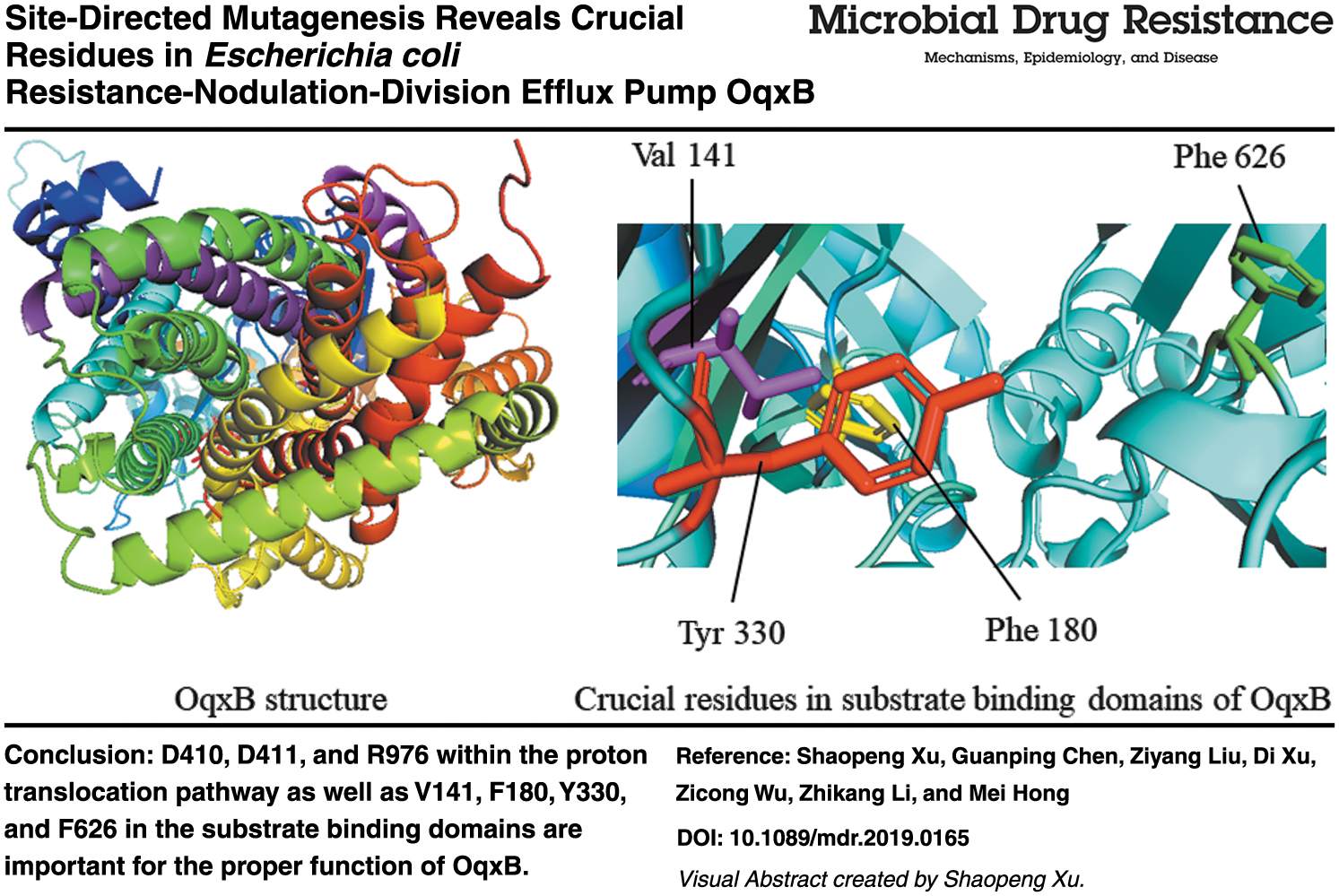
Resistance-nodulation-division (RND) efflux pumps are important determinants of multidrug resistance in Gram-negative bacteria. As one of the typical members of the RND superfamily, Escherichia coli OqxAB multidrug efflux pump confers resistance to antimicrobial agents, such as olaquindox and fluoroquinolone. In the present study, site-directed mutagenesis and antimicrobial susceptibility measurement assay were applied to identify the crucial residues within OqxB, the transporter component of the OqxAB efflux pump system. It was found that alanine substitution of proton translocation pathway residues D410, D411, and R976 resulted in a complete loss of the transport function. Further studies revealed that the charge property of these residues is important for proper function of OqxB. Alanine replacement of residues involved in substrate-binding domains, including V141, F180, Y330, and F626, exhibited different responses toward different antimicrobial agents. Conservative replacement of Y330, F626, and F180 with amino acids having similar aromatic ring structure resulted in full or partial recovery of the efflux function. Molecular docking analysis demonstrated that olaquindox may form hydrogen bonds with F626, Y330, and V141, whereas only Y330 and F180 may interact with ciprofloxacin, implicating the different roles played by these residues when transporting different kinds of substrates.
Graphical Abstract
Color images are available online.
Get full access to this article
View all access options for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
