Abstract
This study evaluated the antimicrobial activity of Estericide® QX (super oxidized solution) in 524 bacterial clinical isolates causing nosocomial infections. The minimum inhibitory concentration (MIC) was determined by the serial broth microdilution method. The bacterial viability of the isolates and control strains was tested. The bactericidal effect of the disinfectant was determined according to the European Standards (EN) Test Methods-1040 guidelines. Assay of stability in Estericide QX after 1 year of storage was performed. The microdilution assays showed that the isolates were inhibited at concentrations of 10–40 parts per million (ppm). For gram-positive bacteria, the MIC values 20 and 40 ppm were more predominant (95%), whereas for gram-negative bacteria, the MIC values 10 and 20 ppm had the highest percentage (91.7%). The difference between the two groups was statistically significant (p<0.001). The results of the assay of bactericidal activity showed that all tested bacteria (99.999%) were killed within 30 sec of contact time. The stability test showed that Estericide QX maintained its disinfectant action over time. In conclusion, the results of the present study showed that the super oxidized solution of Estericide QX provides a high antibacterial activity on both gram-positive and gram-negative bacteria. Based on these results and under the conditions of the present study, we believe that Estericide QX can be used efficiently against multiresistant nosocomial bacteria, providing an opportunity for new disinfection alternatives.
Introduction
C
At present, the use of disinfectants in the hospital environment is paramount, which is why new formulations of antiseptics have been developed. Several disinfectants are available in the market such as quaternary ammonium compounds, glutaraldehyde, ethylic alcohol, super oxidized solutions, povidone iodine, and chlorhexidine.4,5,38 Super oxidized solutions are broad-spectrum disinfectants with a promising antimicrobial activity on microorganisms and therefore widely used in clinical practice.2,28,33 Super oxidized solutions are disinfectants capable of destroying bacteria, fungi, viruses, and spores through different mechanisms of action. 33 These compounds are presented in various commercial presentations (Sterilox®, Sterisol®, Medilox®, Dermacyn®, Microcyn®, Varul®, Esterilife® and Estericide® QX), each with different compositions.
Estericide QX is a commercially available solution which, according to the manufacturer is nontoxic and noncorrosive to biologic tissues and surgical instruments, and contains active chlorine equivalent to 0.004% (40 parts per million [ppm]), has a neutral pH (6.4–7.5) and oxidation reduction potential (ORP) of 650–900 mV. Previous studies have demonstrated the successful application of Estericide QX as a disinfectant and antiseptic.3,7,14,20,25,26,31 However, this is the first experimental study where the effectivity of Estericide QX against multiresistant bacteria causing nosocomial infections is demonstrated.
The aim of the present study was to evaluate the antimicrobial activity of Estericide QX in clinical isolates causing nosocomial infections. Moreover, we evaluated the stability of this biocide after 1 year of storage.
Materials and Methods
Bacterial isolates
A total of 524 isolates were evaluated, including different bacterial species: Staphylococcus aureus methicillin resistant (n=242), Escherichia coli beta-lactam resistant (n=81), Staphylococcus epidermidis methicillin resistant (n=82), Pseudomonas aeruginosa beta-lactam resistant (n=64), and Enterococcus faecium (n=55). Strains were collected from several clinical sources: blood, wound infections, catheter-related infections, abscesses, peritoneal fluid, and cerebrospinal fluid. All isolates included in this study were characterized in the previous studies and were resistant to multiple antibiotics (more than three antibiotics belonging to different classes).6,8,30,34,35
Control strains
S. aureus ATCC 6538, E. coli ATCC 11229, P. aeruginosa 15442, Enterococcus faecalis ATCC 29212, and E. faecalis ATCC 51299 were used as controls in all assays.
Test solution analysis and stability testing
According to the manufacturer, Estericide QX has a shelf life of 18 months and stored at room temperature (25°C±2°C) in containers of high-density polyethylene (HDPE). As part of Estericide QX quality control, long-term stability tests were periodically performed in different product batches under the following storage conditions: 25°C±2°C/60%±5% RH (relative humidity), and all measured parameters were kept within expected ranges; pH (7.3–7.5); ORP (886–895); HClO/ClO (5.23–5.45); ClO2 (0.34–0.50); NaCl (2.30–2.40); and O3 (0.03–0.05). In addition, studies were carried out using different materials for manufacturing containers (HDPE, polyvinylchloride, ethyl vinyl acetate, and polypropylene), and HDPE gave the best results. These studies were performed by an external laboratory.
Determination of minimal inhibitory concentrations and bacterial viability
The minimum inhibitory concentration (MIC) of Estericide QX was determined by the broth microdilution method using the Mueller–Hinton broth (Becton Dickinson). Each bacterial culture was adjusted to a turbidity equivalent to 0.5 McFarland standards (108 UFC/ml) in saline solution. Serial dilutions of Estericide QX were performed using 96-well microtiter plates. The dilutions were prepared in sterile water. Each well was filled with 50 μl of defined disinfectant dilution and 50 μl of test microorganism suspension. In the first column, concentrated disinfectant (40 ppm) was added; in the next columns, the following dilutions were performed: 1:2–1:16 equivalents to 20–2.5 ppm, respectively. All assays included 2 sterility controls (disinfectant with water and Mueller–Hinton broth), growth control (bacteria and Mueller–Hinton broth), and ATCC control strains. After 24 h of incubation at 37°C, the plates were read; the first test well not showing visible growth or turbidly after incubation was considered as the MIC value. All assays were performed in duplicate. To measure the bacterial viability, 10 μl of suspension from each well not showing visible growth were plated in Trypticase soy agar plates (Becton Dickinson) and incubated for 24 h at 37°C.
Bactericidal activity
The bactericidal effect of the disinfectant was measured using a quantitative test according to the European Standards (EN) Test Methods-1040 guidelines with some modifications. 1
Suspension test (traditional plate count method)
The bacterial test suspensions were adjusted to a concentration of 108 UFC/ml for each strain. One milliliter of bacterial test suspension was added to 99 ml of disinfectant solution (Estericide QX), mixed thoroughly, and left at room temperature for a specified contact time of 30 sec. Following contact, an aliquot (1 ml) of the reaction mixture (bacterial cells and disinfectants) was transferred to 9 ml of neutralizer solutions and serially diluted in phosphate-buffered saline (PBS) (pH 7.2). After dilution, 1 ml of the mixture was spread immediately onto standard methods agar plates and incubated for 24 h at 37°C. The number of colonies surviving on each plate was counted and cell survival rates were calculated. The control test did not contain the disinfectant, but only the serially diluted suspension in PBS (pH 7.2), which was plated and counted. Each assay was performed in duplicate and the average number of colonies was considered for analysis. Data were reported as the reduction in colony counts based on 30 sec of exposure time.
Shelf life testing
The solution used for this assay was maintained at room temperature (25°C±2°C) in its original container made of HDEP, thereby preventing overheating of the product and exposure to sunlight, conditions favoring stability of the solution.
According to the results of the MIC values obtained by the microdilution test, a sample of the initial study population was selected to determine the MIC and bactericidal activity using a solution of Estericide QX opened 1 year before. All the assays were performed in duplicate.
Statistical methods
The sample size to stability assays was calculated based on the frequency of MIC value in each species with a confidence interval of 95% and a finite sample; the sample selection was performed randomly.
U Mann–Whitney, a nonparametric test, was used to compare bactericidal activities between bacterial species. A value of p<0.05 was considered statistically significant.
Results
Microdilution assays with Estericide QX showed that the isolates were inhibited at concentrations of 10–40 ppm. The results showed that 52.1% (n=273) of the 524 strains tested in this study had a MIC of 20 ppm, 36.6% (n=192) were inhibited at a concentration of 40 ppm, and 11.3% (n=59) had a MIC of 10 ppm. For gram-positive bacteria (S. aureus, S. epidermidis, and E. faecium), the MIC values of 20 and 40 ppm were more predominant (95%; 363/379) and for gram-negative bacteria (E. coli and P. aeruginosa), the MIC values of 10 and 20 ppm had the highest percentage (91.7%; 133/145); the difference between the two groups was statistically significant (p<0.001). The highest value of MIC (40 ppm) was more abundant in S. aureus and E. faecium, 57.9% and 65.5% of the isolates, respectively (p<0.001). The MIC value of 10 ppm was observed in lower percentages (19–32%) (Fig. 1). All control strains showed a MIC value of 20 ppm with the exception of E. faecalis ATCC 29212 and E. faecalis ATCC 51299 that showed a MIC value of 40 ppm under the same assay conditions.
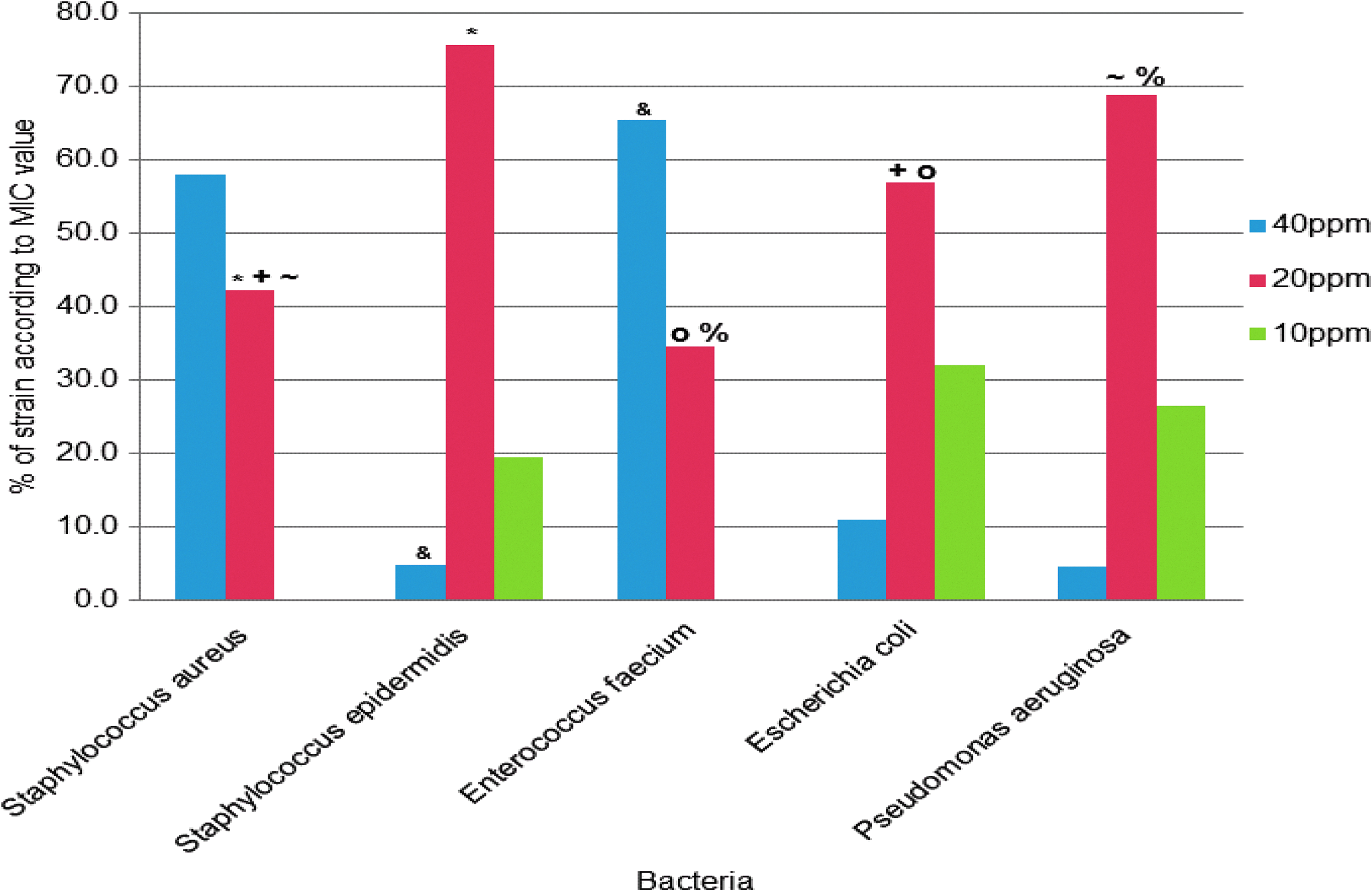
Percentage of minimal inhibitory concentration (MIC) of Estericide® QX and comparison of median MIC values with statistically significant differences (p<0.001) between the bacterial species tested. *Staphylococcus epidermidis versus Staphylococcus aureus (20 ppm); &Enterococcus faecium versus S. epidermidis (40 ppm); +Escherichia coli versus S. aureus (20 ppm); oE. coli versus E. faecium (20 ppm); ∼Pseudomonas aeruginosa versus S. aureus (20 ppm); and %P. aeruginosa versus. E. faecium (20 ppm).
According to the median MIC values obtained, we found significant differences between gram-positive and gram-negative bacteria. The only exception was S. epidermidis that showed median values similar to the ones observed for E. coli and P. aeruginosa. Statistically significant differences of Estericide QX median MIC values found between bacterial species are shown with different symbols (Fig. 1).
In none of the viability tests bacterial growth was observed, confirming the bactericidal activity of Estericide QX in the well where the MIC was obtained.
The bactericidal activity assays showed that all tested bacteria (control strains and clinical isolates) were killed (99.999%) within 30 sec of contact time.
The stability test of Estericide QX was carried out in 157 strains: S. aureus, n=39; S. epidermidis, n=30; E. faecium, n=25; E. coli, n=35; and P. aeruginosa, n=28; selected according to the MIC value obtained in the microdilution assay (40, 20, or 10 ppm). The results showed that the disinfectant was able to inhibit bacterial growth after 1 year of storage. The stability results showed that 49.7% (n=78) of the 157 strains tested for the second time had a MIC of 20 ppm, 29.9% (n=47) showed a MIC of 40 ppm, and 20.4% (n=32) had a MIC of 10 ppm. For gram-positive bacteria, the MIC values of 20 and 40 ppm were more predominant (89.3%; 84/94), and for gram-negative bacteria, the MIC values of 10 and 20 ppm had the highest percentage (84.1%; 53/63); these results were identical to those obtained in the first trial.
The MIC value (40 ppm) was higher in S. aureus and E. faecium, 51.2% and 56%, respectively, equal to what was observed before 12 months between these two species. The MIC value 20 ppm was detected in percentages that ranged from 44% to 57% between different species of bacteria analyzed, S. epidermidis and P. aeruginosa showed the highest percentages, 57% and 53%, respectively. The MIC value 10 ppm was observed only in E. coli, P. aeruginosa and S. epidermidis. The MIC values for control strains were unchanged. The bactericidal activity assays showed that all tested strains were killed (99.999%) within 30 sec of contact time.
Discussion
Disinfection is one of the most important factors for preventing nosocomial infections. The selection of appropriate disinfectants that have proven the broad-spectrum activity against different microorganisms is very important. This super oxidized solution, which has been widely used in different environments, has many advantages such as being safe for patients and the environment, not being toxic, not harming to the human tissue, and being of low cost. 27
In this study, we tested the super oxidized solution Estericide QX obtained from a laboratory in Mexico, to evaluate its antimicrobial activity among several nosocomial pathogens. All tested strains showed three different susceptibility MIC values of 10, 20, and 40 ppm. None of the bacteria showed a MIC exceeding 40 ppm. The effectivity of super oxidized solutions has been described in other works. Landa-Solis et al. treated pure cultures of S. aureus, E. coli, P. aeruginosa, Salmonella typhi, and Candida albicans with Microcyn and found it was active on all bacteria and C. albicans tested. 18 Vorobjev et al. reported that super oxidized solution was effective on spores, gram-positive and gram-negative bacteria causing nosocomial infections. 36 Gunaydin et al. found good antimicrobial activity of Medilox super oxidized solution in ATCC strains and clinical isolates. 12 Tanaka et al. have compared super oxidized water with 2% Dialox-c and 3.8% formalin and reported that super oxidized water was more effective than the other disinfectants. 32 Nishimura et al. have reported that hand disinfection using super oxidized water was 7.5% more effective than povidone iodine. 22
The results showed that among gram-positive bacteria, E. faecium and S. aureus showed the highest value of MIC (40 ppm) in more than 50% of the population. A similar behavior was described for E. faecium by Kampf et al. when testing chlorhexidine; they found E. faecium to be less susceptible than E. faecalis. 16 Park et al. reported that S. aureus was more resistant to super oxidized solution than Enterobacter aerogenes; the population of E. aerogenes decreased to an undetectable level, whereas the surviving population of S. aureus was 3.9 log10 CFU/ml. 24 These results are important considering that S. aureus and E. faecium have developed wide resistance to antibiotics, including vancomycin, and their behavior against disinfectants should be monitored.
In the present study, all isolates were susceptible to Estericide QX and a reduction of 99.999% in viability was observed in all tested species within 30 sec of contact time. The microbicidal activity of super oxidized solution has been demonstrated in previous studies. Banerjee and Anupurba found that among the disinfectants tested in their research, the super oxidized solution showed maximum reduction (99.999%) in bacterial count at different exposure times. 2 Guentzel et al. reported a reduction of the bacteria load of 100% within 10 min of contact time using neutral electrolyzed oxidizing water in pure cultures of E. coli, Salmonella typhimurium, S. aureus, Listeria monocytogenes, and E. faecalis. 11 Landa-Solis found that a super oxidized solution (Microcyn™) with neutral pH was effective in all pathogens tested: E. coli, S. aureus, P. aeruginosa, S. typhi, and C. albicans were killed after 30 sec with a reduction of 99.999%. 18
The stability tests showed that Estericide QX maintained its disinfectant action at least after 1 year; as shown in the results, the control strains and clinical isolates showed MIC values equal to those found in the first trial. Several factors may contribute to the stability observed in the disinfectant tested, for example, the material of the container where the product is stored (HDEP), which prevents exposure to sunlight and temperature storage conditions (25°C±2°C). These storage conditions could help the disinfectant to be active. The stability of super oxidized solutions has been a controversial topic. There are marked differences between the pH of super oxidized solutions (acidic, neutral, or alkaline) and these differences determine the germicide activity, corrosion potential, and shelf life of the disinfectant. Additional studies are now being conducted to compare the activity of Estericide QX and other commercial disinfectants.
Some authors have reported that these solutions have a short half-live due to storage conditions, sun exposure, range of pH, and ORP.17,19,29 However, other studies have shown the stability of neutral super oxidized solutions, attributing its stability to pH and ORP.13,21 To maintain the HOCl solution in a stable form, maximize its antimicrobial activities, and minimize undesirable side products, a neutral pH is required. 37 At pH lower than 3, an appreciable amount of Cl2 gas forms, which may cause the rapid loss of all active chlorine in an open container. The ORP increases with decreasing pH due to the Nernst Law, which found that the effect of lighting on chlorine loss was not clearly observed from OPR profiles.23,37
In conclusion, the results of the present study showed that super oxidized solution of Estericide QX provides a high antibacterial activity on both gram-positive and gram-negative bacteria. Based on these results and under the conditions of the present study, we believe that Estericide QX can be used efficiently against multiresistant nosocomial bacteria providing an opportunity for new disinfection alternatives. Future studies are needed to test the microbiocidal activity of Estericide QX against other pathogenic bacteria of epidemiological importance. Further work needs to be carried out to investigate any role of organic surface contaminants on the efficacy of Estericide QX.
Footnotes
Acknowledgments
We thank Ms. Patricia Cornejo Juárez (Instituto Nacional de Cancerología, México, DF), Guadalupe Miranda Novales, Fortino Solórzano Santos (Hospital de Pediatría del Centro Médico Nacional Siglo XXI-IMSS, México, DF), and Rayo Morfin Otero (Hospital Civil de Guadalajara “Fray Antonio Alcalde,” Guadalajara, México) for giving authorization for the use of the strains. Laboratory work was performed at the National Institute of Public Health, Cuernavaca, Morelos, Mexico. We thank Lilia Chihu Amparán, PhD, Marta Aires de Sousa, PhD, and Veronika Wirtz, PhD, for the critical review of the manuscript.
Disclosure Statement
This study was supported by a grant from Esteripharma, Mexico, SA de CV.
