Abstract
Due to their bacterial lytic action, bacteriophage endolysins have recently gained great attention as a potential alternative to antibiotics in the combat of Gram-positive pathogenic bacteria, particularly those displaying multidrug resistance. However, large-scale production and purification of endolysins is frequently impaired due to their low solubility. In addition, a large number of endolysins appear to exhibit reduced lytic efficacy when compared with their action during phage infection. Here, we took advantage of the high solubility of two recently characterized enterococcal endolysins to construct chimeras targeting Staphylococcus aureus. The putative cell wall binding domain of these endolysins was substituted by that of a staphylococcal endolysin that showed poor solubility. Under appropriate conditions the resulting chimeras presented the high solubility of the parental enterococcal endolysins. In addition, they proved to be broadly active against a collection of the most relevant methicillin-resistant S. aureus epidemic clones and against other Gram-positive pathogens. Thus, fusion of endolysin domains of heterologous origin seems to be a suitable approach to design new potent endolysins with changed and/or extended lytic spectrum that are amenable to large-scale production.
Introduction
Among the different approaches to obtain new antibacterials, one that has recently caught great attention is the exploitation of bacteriophage endolysins.27,29,48,59 Endolysins are bacterial cell wall-hydrolyzing enzymes that promote host cell lysis in the end of the lytic cycle of double-stranded DNA phages, thus allowing efficient release of the viral progeny to the extracellular medium. 71 Despite the fact that endolysins have been evolutionarily designed to operate from the inside of infected cells, when exogenously applied as purified recombinant proteins they have been shown to promote lysis of different Gram-positive pathogenic bacteria, including S. aureus.47,55,58,80
Most endolysins have a modular organization with a conserved N-terminal catalytic domain (CD) and a more diverse C-terminal cell wall binding domain (CWBD).28,32 However, several S. aureus phages produce endolysins with two CDs in their N-terminus, such as those from phages K, φ11 and φMR11. These lytic enzymes present a CHAP (cysteine, histidine-dependent amidohydrolases/peptidases) domain followed by an Amidase-2 domain (N-acetylmuramoyl-L-alanine amidase), where the CHAP seems to be the most effective in inducing lysis.54,58,63 Another feature commonly reported for S. aureus phage endolysins is their poor solubility when overexpressed in Escherichia coli, frequently leading to low yields of soluble protein.9,31,51,58
We have experienced this insolubility problem when attempting to purify a broad-host-range anti-staphylococcal endolysin available in our lab, Lys87. 11 Here, we describe a new strategy to overcome this problem that consisted of the construction of new chimerical endolysins composed of the Lys87 CWBD fused to the highly soluble CDs of two Enterococcus faecalis phage endolysins, Lys168 and Lys170. 62 By employing this strategy, complemented with optimized expression conditions, we obtained two highly soluble lytic enzymes that are broadly active against a large cohort of S. aureus strains, including MRSA, and against other staphylococcal, streptococcal, and enterococcal species. To our knowledge, this corresponds to the first report of chimeras comprising domains of S. aureus and E. faecalis phage endolysins.
Materials and Methods
Bacteria, culture media, and growth conditions
The E. coli cloning strain XL1-Blue MRF′ and its derivatives were grown at 37°C with aeration in Luria-Bertani (LB) medium. 68 The E. coli expression strain CG61 72 and its derivatives were grown in LB in the same conditions, except that incubation temperature was 28°C before induction of protein production and 16°C afterward. When appropriate, LB medium was supplemented with kanamycin (40 μg/ml) and/or ampicillin (100 μg/ml) for plasmid selection. Lytic action of chimerical endolysins was assayed in 200 bacterial clinical isolates (Table 1, Supplementary Tables S1 and S4; Supplementary Data are available online at www.liebertonline.com/mdr). Table 1 corresponds to a panel of 30 MRSA and 13 Methicillin-sensitive S. aureus (MSSA) typed strains. Table S1 lists 100 S. aureus isolates from Technophage's collection, 42 MRSA, 29 MSSA, and 29 with unknown methicillin susceptibility, which were obtained from different Portuguese community and hospital settings between 2005 and 2008. Recombinant lytic enzymes were also tested in clinical isolates of other species, namely against Staphylococcus epidermidis (n=10), Staphylococcus haemolyticus (n=9), Staphylococcus saprophyticus (n=7), Enterococcus faecalis (n=12), Enterococcus faecium (n=10), and Streptococcus pyogenes (n=9) (Technophage collection, Table S4). The growth media for these bacteria were purchased from Biokar Diagnostics, Beauvais, France. Staphylococcus and Enterococcus species were cultured in Brain-Heart Infusion and Streptococcus pyogenes in Todd Hewitt broth supplemented with 2% of yeast extract (THY). Bacteria were grown at 37°C, with aeration, except for Enterococcus that was grown without agitation. When necessary, 1.5% or 0.7% agar was added to these culture media to obtain solid or soft-agar plates, respectively. E. faecalis and S. aureus phages were propagated and purified by standard methods,15,42 either in soft-agar media or liquid broth supplemented with CaCl2 and MgCl2 (5 mM each).
Clonal complexes were determined using the E-burst software http://saureus.mist.net/, last accessed on January 25, 2012.
NA, not applied; NT, non-typeable; MLST (ST), multilocus sequence typing (sequence type); SCCmec, Staphylococcal Chromosomal Cassette mec; MRSA, methicillin-resistant S. aureus; MSSA, methicillin-sensitive S. aureus; CC, clonal complex.
Identification and bioinformatics analysis of phage endolysins
Genomic DNA from E. faecalis and S. aureus phages was extracted from CsCl-purified lysates 77 and their complete nucleotide sequence determined (service purchased to Macrogen). DNA homology searches were carried out with BLASTN, 81 using the NCBI non-redundant nucleotide sequences database. Recognition of phage genome putative genes was performed by integrating the results obtained with GeneMark.hmm and MetaGeneAnnotator web software.10,56 Endolysins Lys168 and Lys170 from E. faecalis phages F168/08 and F170/08, respectively, were characterized previously. 62 Identification of S. aureus phage F87s/06 endolysin gene was based on BLASTP homology searches 4 against the NCBI non-redundant protein sequences database, using the deduced gene product, and on prediction of protein functional domains using NCBI's CDD 52 and Pfam (http://pfam.janelia.org/search). Assignment of putative linkers connecting protein functional domains was performed with SVM 25 and the SVM-joint output. Multiple protein sequence alignments were performed with ClustalW2. 45
Construction and cloning of lys168-87 and lys170-87 chimerical genes
The coding sequences of the N-terminal regions of the enterococcal phage endolysins Lys168 and Lys170, which included CHAP and Amidase-2 CDs, respectively 62 (Fig. 1A and Supplementary Fig. S1A), were polymerase chain reaction (PCR) amplified from the corresponding phage DNA using a high-fidelity Pfu DNA Polymerase (Fermentas Molecular Biology Tools, Thermo Scientific). The sequence encoding the C-terminal region of the staphylococcal phage endolysin Lys87 (Fig. 1A and Supplementary Fig. S1A), and that harbored the putative CWBD, was similarly amplified by PCR in a separate reaction. The forward primers used for CD amplification carried an NcoI restriction site in their 5′ end, whereas the reverse primer employed in CWBD amplification contained an XmaI site. The reverse and forward primers used in CD and CWBD amplification, respectively, carried a 26-bp complementary segment in their 5′ end. This allowed fusing the coding sequence of each enterococcal endolysin CD to that of the CWBD of Lys87 by the technique of overlap-extension by polymerase chain reaction (OE-PCR), 37 using the CD forward and CWBD reverse primers and a mixture of the initial PCR products as template. The resulting PCR products, corresponding to the chimerical genes lys168-87 and lys170-87 (Fig. 1B and Supplementary Fig. S1B), were purified using the High Pure PCR Product Purification Kit (Roche Applied Science), double-digested with NcoI and XmaI, and ligated to the equally digested pIVEX2.3d expression vector (Roche Applied Science). This vector is designed to drive the expression of cloned genes under the control of the phage T7 φ10 promoter and to allow the production of the corresponding proteins C-terminally fused to a hexahistidine tag. The E. coli strain XL1-Blue MRF′ was transformed with these ligations as described previously. 14 Transformants were selected in the presence of 100 μg/ml ampicillin and screened for the presence of the desired recombinant plasmids by PCR, using insert and vector complementary primers. Plasmid DNA from positive clones was extracted (Pure Link Quick Plasmid Miniprep Kit; Invitrogen) and the correct DNA structure confirmed by endonuclease restriction and DNA sequencing (Macrogen). The constructs pSF168-87 and pSF170-87 are pIVEX2.3d derivatives carrying lys168-87 and lys170-87, respectively.
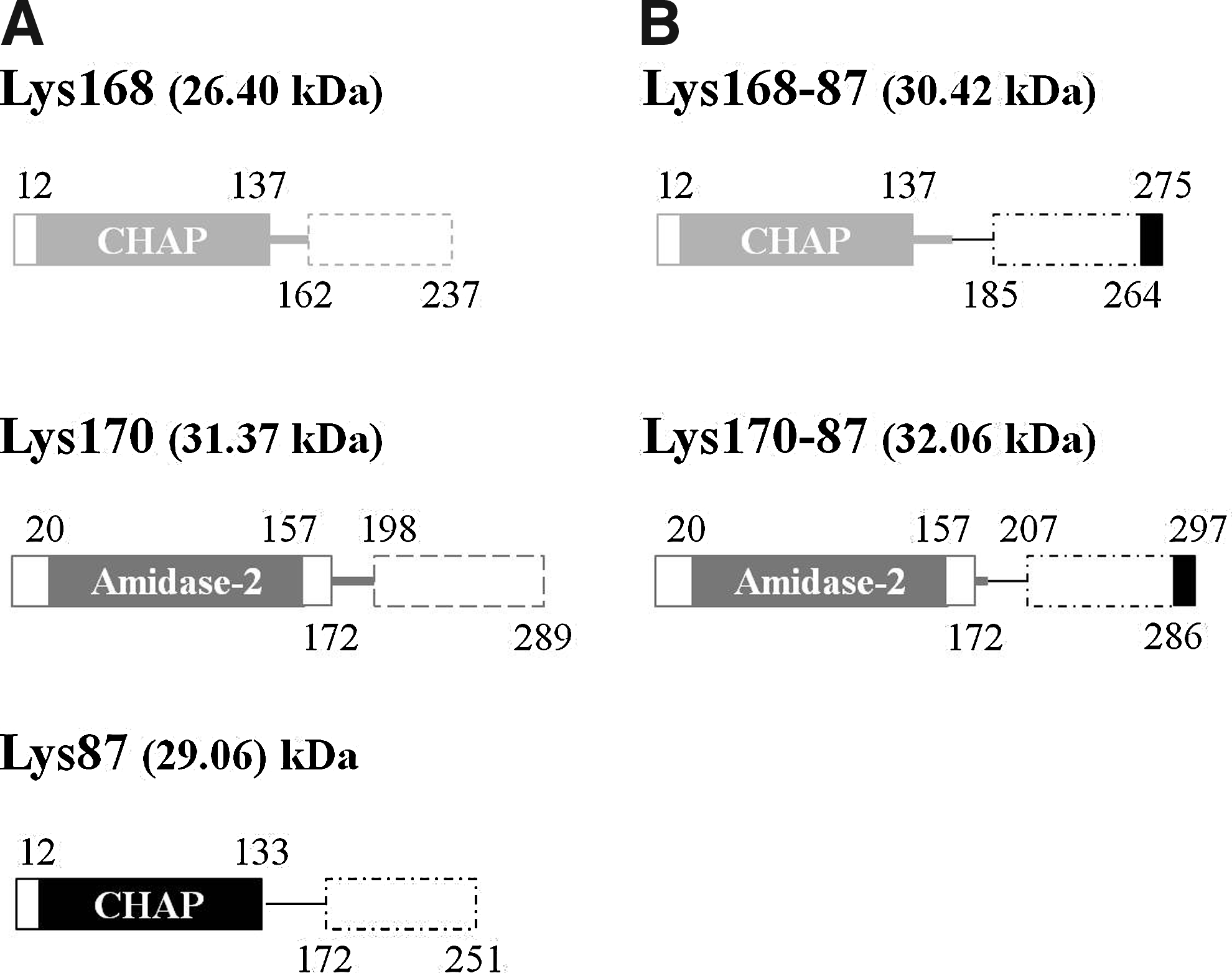
Schematic representation of parental
Production and purification of the chimerical endolysins Lys168-87 and Lys170-87
E. coli strain CG61, which overproduces phage T7 RNA polymerase upon temperate upshift 72 was transformed with plasmids pSF168-87 and pSF170-87 and transformants selected at 28°C in presence of 100 μg/ml ampicillin and 40 μg/ml kanamycin. Production of soluble and active Lys168-87 and Lys170-87 by CG61 transformants was first confirmed by growing them over a dense lawn of autoclavated staphylococcal cells, incorporated in soft-agar LB medium, and checked for the presence of lysis halos around E. coli colonies (Supplementary Fig. S2).
Selected clones of each chimera were grown at 28°C until an optical density at 600 nm (OD600) of 0.8–1.0, after which protein production was induced by changing cultures to a shaking water bath set to 42°C. After 30 min, cultures were transferred to an incubator at 16°C and agitated for an additional period of 14 hr. Cells from induced cultures were pelleted by centrifugation (8,000 g, 30 min, 4°C) and resuspended in 1/50 volume of lysis buffer (20 mM Hepes, 500 mM NaCl, 20 mM imidazole, 1% glycerol, and 1 mM DTT [pH 6.5]) supplemented with 1× Complete Mini EDTA-free Protease Inhibitor Cocktail (Roche Applied Science). Cells were kept on ice and disrupted by sonication (Vibra Cell MS2T, Sonic Materials) by performing about 10 bursts of 1 min (amplitude 50, pulse 3, 30–40 W) intercalated with pauses of 1 min. Insoluble material was sedimented by centrifugation (20,000 g, 30 min, 4°C). The supernatant corresponding to the total soluble extract was filtered through 0.22 μm and the chimerical enzymes purified by affinity chromatography using HisTrap™ HP columns (GE Healthcare) coupled to an AKTA-Prime system (GE Healthcare). The column and elution buffers had the same composition of the lysis buffer, except that the imidazole concentration in the elution buffer was 500 mM. Eluted fractions were analyzed by sodium dodecyl sulfate polyacrylanude gel electrophoresis and Coomassie blue staining. 44 Chimeras pure fractions were pooled, concentrated, and changed to an imidazole-free, phosphate-based endolysin buffer (50 mM phosphate-Na, 500 mM NaCl, 25% glycerol, and 1 mM DTT [pH 6.5]) using HiTrap™ Desalting columns (GE Healthcare). Protein concentrations were determined by the Bradford method (Bio-Rad Laboratories) using bovine serum albumin as standard. The enzymes were divided into small aliquots and kept at −20°C.
Evaluation of the lytic action of chimerical endolysins against bacterial pathogens
The ability of chimeras Lys168-87 and Lys170-87 to induce lysis of clinical strains from different bacterial species was evaluated by two different assays. When tested against a large number of bacterial isolates, these were individually cultured as indicated above until an OD600 of 0.8–1.0. Cells were recovered by centrifugation and resuspended in 1/100 volume of fresh medium. A 100 μl sample of these cellular suspensions was diluted in 10 ml of incorporation buffer (25 mM phosphate-Na and 250 mM NaCl [pH6.5]) supplemented with 0.7% agar and poured in a Petri dish. Four quantities of the purified chimeras (10, 5, 1, and 0.2 μg, in 10 μl final volume) were spotted on each bacterial lawn and after overnight incubation at 37°C checked for the presence of lysis halos. These were evaluated and scored (− to +++) according to their relative diameter and transparency.
Bacterial cell lysis was also studied in liquid medium. Selected strains were grown until an OD600 of 0.3–0.4, centrifuged, and cells recovered in 1/2 volumes of a phosphate buffer (25 mM phosphate-Na and 250 mM NaCl [pH 6.5]). One-milliliter cell suspensions were challenged with 10 μg/ml of each chimera separately or with a mixture of both enzymes, each at 5 μg/ml. The OD600 variation was followed over time. At the end of each assay the surviving colony forming units (CFU)/ml were determined. Negative controls were equally prepared except that endolysin buffer was added instead of endolysin.
Results
Production of chimerical endolysins targeting S. aureus
We have recently characterized three phage endolysins, two from E. faecalis phages F170/08 and F168/08 and another from S. aureus phage F87s/06.62,11 In silico analysis of the endolysin from phage F170/08, that is, Lys170 identified a CD of the Amidase-2 family in its amino-terminal region (Fig. 1A and Supplementary Fig. S1A). This family includes zinc amidases that have N-acetylmuramoyl-L-alanine amidase activity. 13 The same analysis performed with endolysins Lys168 and Lys87 from phages F168/08 and F87s/06, respectively, revealed a CD of the CHAP family,7,46,64 also located in the enzymes' N-terminal region (Fig. 1A and Supplementary Fig. S1A). Enzymes from this family have been shown to cleave different amide bonds in the peptidoglycan mesh, frequently displaying amidase or endopeptidase activity.
Lys170 and Lys168 could be easily produced in E. coli in its soluble form, whereas Lys87 showed a high propensity to precipitate as inclusion bodies in all tested conditions.11,62 The enterococcal endolysins exhibited a narrow lytic spectrum with their activity being basically restricted to E. faecalis cells. 62 This feature might result from CWBD binding to a specific epitope present on the cell wall of E. faecalis cells and/or dependence of the CD on CWBD binding for activity. 49 Lys170 showed better lytic performance when both enterococcal endolysins were tested against a large number of E. faecalis clinical strains. 62
Having this in mind we have envisaged that replacing the CWBD of Lys170 and Lys168 by that of Lys87 would result in chimerical endolysins (Fig 1B and Supplementary Fig. S1B) maintaining the high solubility properties of the parental enterococcal enzymes and the ability to recognize and act on S. aureus cells conferred by the parental Lys87 CWBD.
The coding sequences of the functional domains composing each chimerical endolysin were PCR-amplified, fused by OE-PCR, and cloned in the E. coli expression vector pIVEX2.3d, which allowed production of the chimeras C-terminally fused to a hexahistidine tag (Fig. 1B, Supplementary Fig. S1B, and Materials and Methods). The ability of the chimerical enzymes Lys168-87 and Lys170-87 to lyse S. aureus cells was initially confirmed by growing the resulting protein-expressing E. coli clones on a dense lawn of autoclavated staphylococcal cells and checking for the presence of lysis halos around the E. coli colonies (Supplementary Fig. S2).
By employing standard protein expression conditions (37°C, 3–4 hr production after culture induction) with selected clones we could obtain much higher soluble amounts of the chimerical endolysins than those we had previously obtained with Lys87. However, the quantity of soluble Lys168-87 and Lys170-87 did not match the levels we achieved with the parental enterococcal endolysins, as we anticipated (not shown). This could be overcome though by simply incubating the induced cultures at 16°C during 14 hr. In these conditions we achieved similar or higher soluble quantities of the chimeras when compared with the native enterococcal enzymes. The chimeras were subsequently purified from total soluble extracts by affinity chromatography using nickel columns. Pure fractions were pooled and subjected to a desalting step for protein exchange to an imidazole-free, sodium phosphate-based buffer (Fig. 2). Average yields were 20 μg and 30 μg of pure Lys168-87 and Lys170-87, respectively, per ml of induced culture.
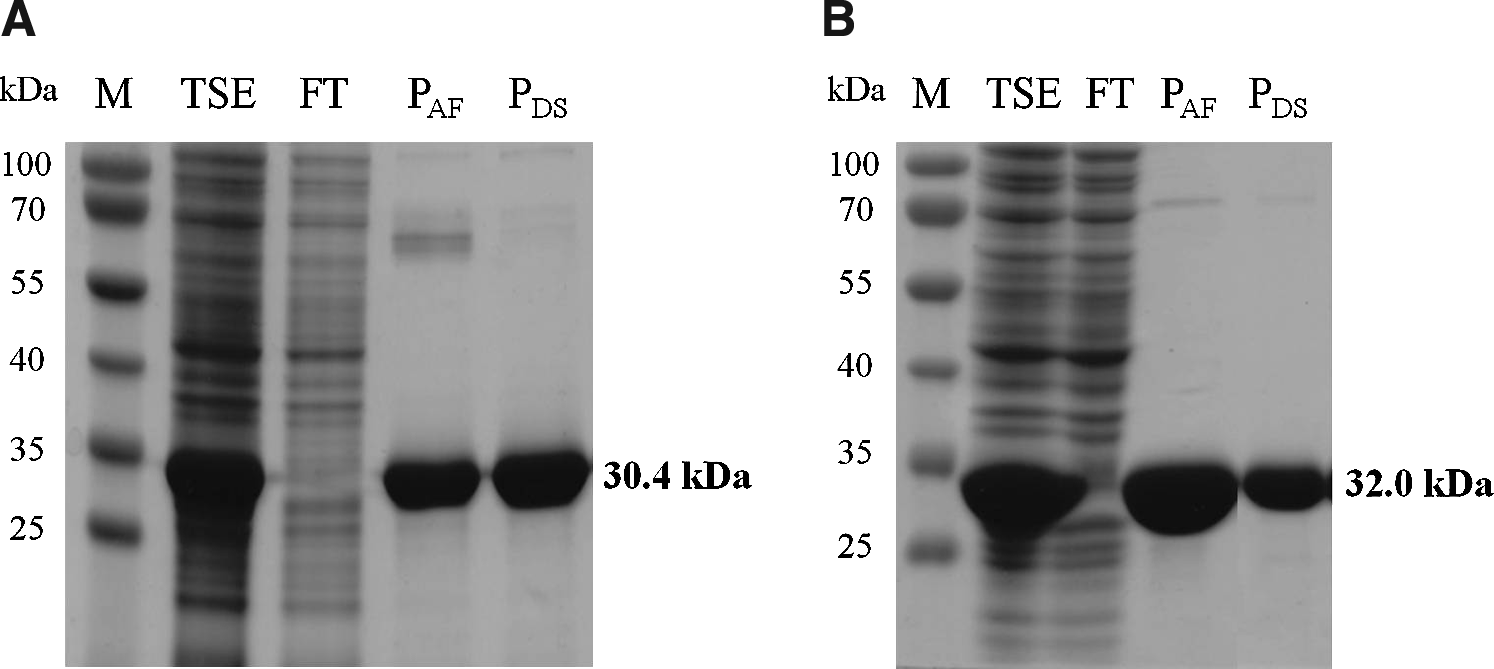
Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) analysis of Lys168-87
Lytic action of the chimerical endolysins against clinical S. aureus
The lytic action of Lys168-87 and Lys170-87 against S. aureus was first assessed using a panel of 100 clinical isolates from Technophage's collection (Supplementary Table S1), which were isolated from different infection contexts and obtained both from Portuguese community and hospital settings. This panel included 42 isolates identified as MRSA. The lytic capacity of each chimera was evaluated by spotting four different amounts of pure enzyme (10, 5, 1, and 0.2 μg) on a dense lawn of viable cells from each tested isolate, which was produced by incorporating cells from exponentially growing cultures in a soft-agar, phosphate-buffered medium (see Materials and Methods). The chimera-induced lysis was qualitatively evaluated by scoring the relative diameter and turbidity of the lysis halos produced after overnight incubation at 37°C (Supplementary Table S2).
For the highest tested protein amount (10 μg) we observed that both chimeras were able to produce lysis halos in more than 90% of the isolates (Supplementary Fig. S3). For each protein quantity though, Lys168-87 generally produced larger and more transparent lysis halos than Lys170-87 (Supplementary Table S2). The narrowing of the lytic spectrum of this chimera as the spotted protein amount decreased was more pronounced than that observed with Lys168-87 (Supplementary Fig. S3). Interestingly, these results were basically opposite to those we obtained with the parental Lys168 and Lys170, where the latter exhibited better lytic performance against E. faecalis. 62
Although both chimerical endolysins revealed to be very effective in lysing this panel of S. aureus clinical isolates (n=100), these were not typed and therefore, the diversity present in this group was unknown. To have a more precise view on the lytic potential of the chimeras against S. aureus, the enzymes were similarly tested in a panel composed of diverse and typed MRSA and MSSA strains (Table 1). This strain collection was previously characterized in terms of their genetic background and included representatives of the most relevant MRSA pandemic clones from different parts of the world and representatives of the dominant of MSSA clones.
Remarkably, the chimerical endolysins lysed about 70% to 100% of this panel of typed S. aureus clinical strains, depending on the amount of tested enzyme (Fig. 3). Lys168-87 again showed a better lytic performance when compared with Lys170-87 since 0.2 μg of the former were sufficient to produce a lysis halo in 83% of the tested MRSA strains, whereas Lys170-87 lysed only 70%. A detailed description of the lytic activity of the endolysins in each particular S. aureus strain is shown in Supplementary Table S3.
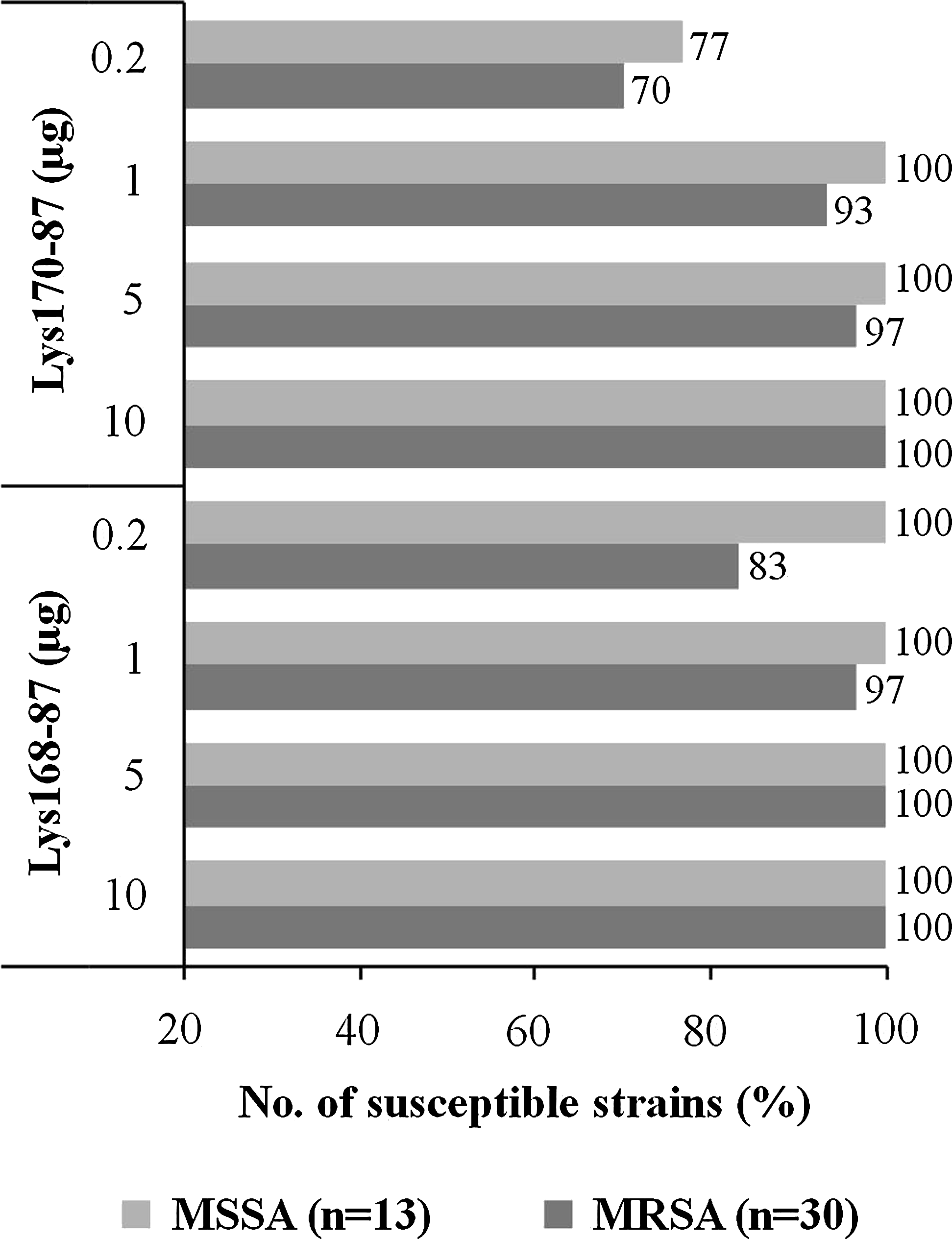
Susceptibility of a panel of diverse and typed Staphylococcus aureus clinical strains to the lytic action of Lys168-87 and Lys170-87. Typed MRSA and MSSA strains (Table 1) were challenged by the spot assay (see text) with the indicated amounts of each chimera. The percentage of strains that developed a lysis halo after overnight incubation at 37°C is plotted as a function of each enzyme amount. A detailed description of the susceptibility of each strain to both chimeras is presented in Supplementary Table S3. MRSA, Methicillin-resistant S. aureus; MSSA, Methicillin-sensitive S. aureus.
Lys168-87 and Lys170-87 have a synergistic lytic effect in liquid media
The chimerical endolysins were also able to induce lysis of viable S. aureus cells in dense liquid suspensions, as shown in the example of Fig. 4, for the MRSA epidemic clone USA200. When applied separately, each endolysin caused a drop of cell suspension turbidity to about 40% of the initial value, after 1 hr incubation, which translated into a corresponding decrease of the initial CFU/ml. However, if simultaneously added to cultures, each at half of the concentration when used separately, the chimerical enzymes decreased the cell suspension turbidity to almost 5% of the initial value and eliminated ∼99% of the initial CFU/ml (Fig. 4). These results indicate a synergistic effect of both endolysins in the effective lysis and killing of S. aureus.
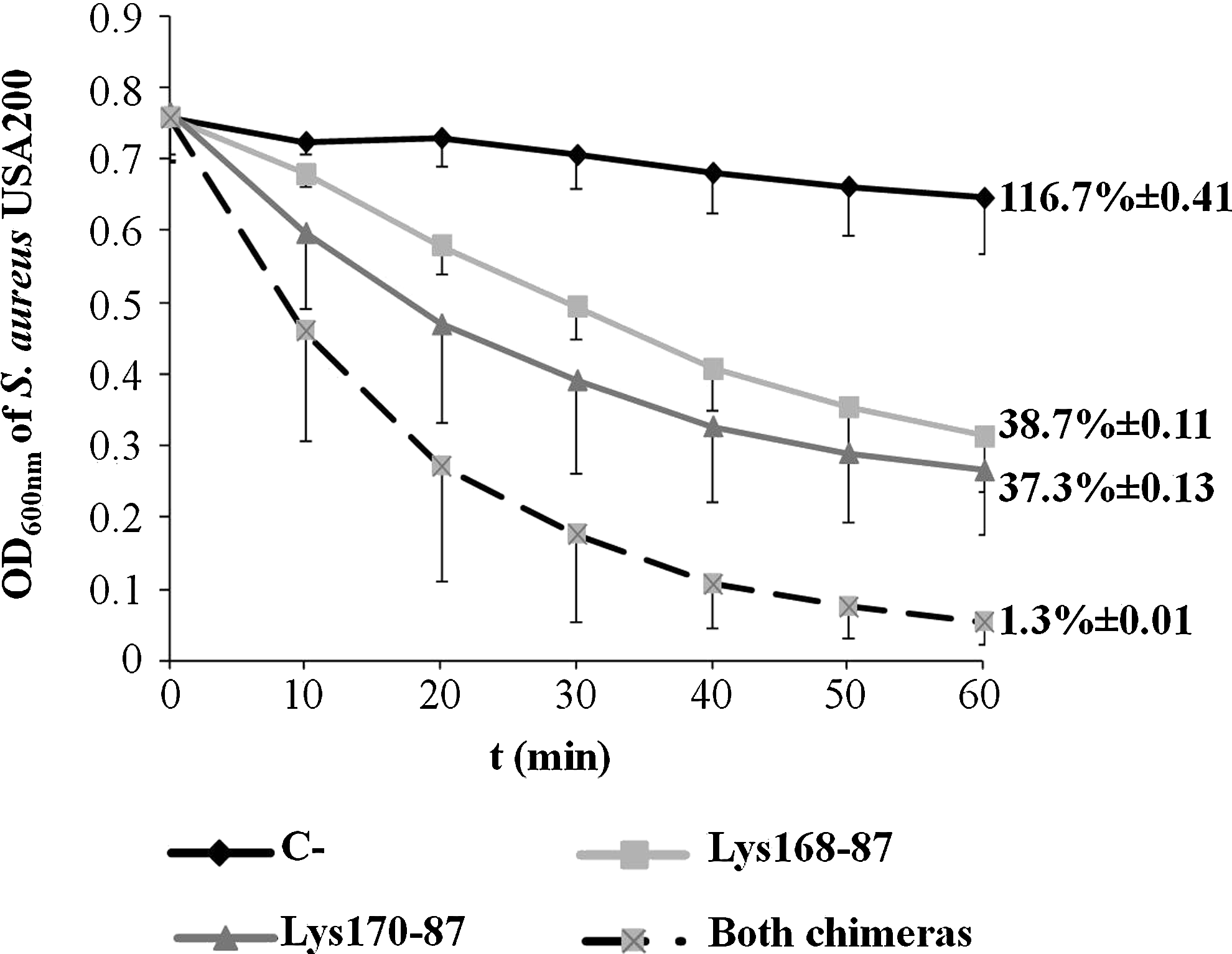
Lytic action of Lys168-87 and Lys170-87 against S. aureus strain USA200 in liquid media. The graphic shows the change of cell suspensions OD600 over time, after adding each chimera separately (10 μg/ml) or in combination (5 μg/ml each). Endolysin buffer was added to the negative control (C−) instead of the enzymes. Values are the means of three independent experiments with indication of standard deviation. The values on the right side indicate the percentage of the initial CFU/ml after 60 min of enzymes' action and the corresponding standard deviation. OD, optical density.
Activity of the chimerical endolysins against other Gram-positive pathogenic bacteria
As mentioned before, the parental enterococcal endolysins Lys168 and Lys170 showed a clear preference for E. faecalis when tested against a representative group of typed strains of this species and of E. faecium. When comparing their lytic action, Lys170 showed to be more effective than Lys168. 62 By replacing their putative CWBD by that of the staphylococcal endolysin Lys87 we could efficiently retarget their activity toward S. aureus, as initially envisaged (see above). Next, we studied how the chimeras acted on Gram-positive pathogenic bacteria other than S. aureus (Supplementary Table S4). When tested against the indicated E. faecalis and E. faecium clinical isolates, the chimerical enzymes essentially reproduced the lytic profile displayed by the parental enterococcal endolysins, 62 with Lys170-87 producing larger and clearer lysis halos compared with Lys168-87 (Fig. 5 and Supplementary Table S5). Note that the E. faecalis and E. faecium isolates tested here were not genetically characterized and the vast majority of them have been previously shown to be susceptible to both Lys168 and Lys170. 62
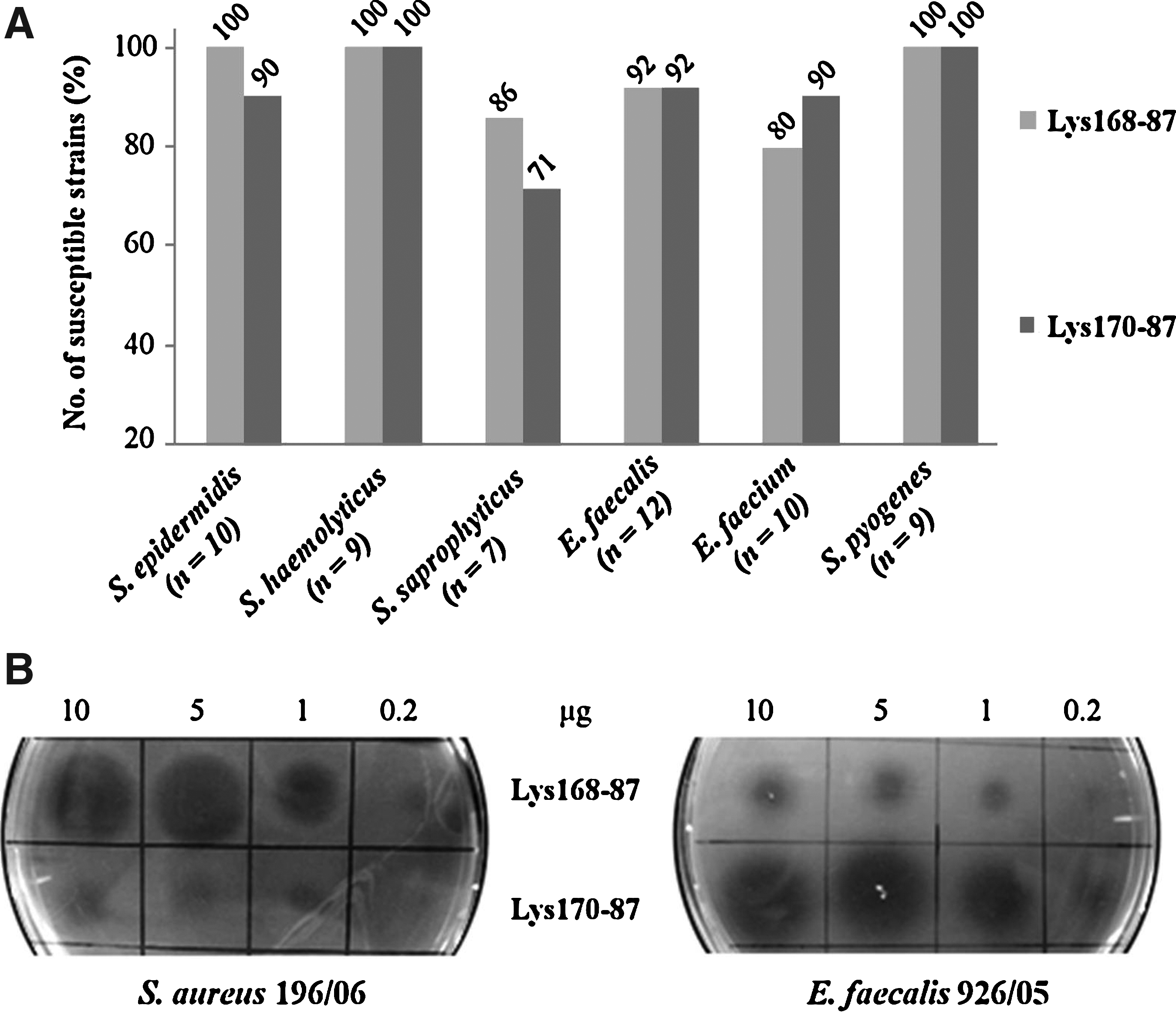
Lytic action of Lys168-87 and Lys170-87 in other Gram-positive bacterial pathogens. (
Interestingly, and in contrast to the parental enterococcal endolysins, the chimeras were also able to cause lysis of clinical isolates of other staphylococcal pathogens and Group A streptococci (Fig. 5A and Supplementary Table S5). Qualitatively, Lys168-87 demonstrated equal or superior lytic action than Lys170-87 against all tested bacteria, except for the enterococcal isolates, where Lys170-87 showed to be more powerful (Fig. 5B and Supplementary Table S5).
Discussion
A survey of the literature indicates that researchers frequently have to deal with the poor solubility of different endolysins, including those of S. aureus phages, when aiming their production and purification.9,31,51,58,74,80 This is also a recurrent problem in our lab and generally the available technical approaches to increase solubility, or to refold proteins recovered from inclusions bodies, are time consuming and poorly effective.
We propose here that construction of chimeras, where we take advantage of the heterologous, highly soluble endolysin functional domains, may represent a solution to obtain large amounts of active lytic enzymes with retargeted and/or extended lytic spectrum relative to parental endolysins. As far as we know, this is the first time that such approach is employed with the specific objective of obtaining highly soluble lytic enzymes targeting S. aureus. There is though, at least one report where a similar strategy was attempted to solve the insolubility problem of an S. aureus phage endolysin. In this case the putative CD of the endolysin P16 from phage P68 was fused to the inferred CWBD of the virion-associated lysin P17, also from the same phage. However, the resulting chimera remained insoluble and accumulated as inclusion bodies. 51
The construction of chimerical enzymes as a mean to improve the lytic efficacy or change the lytic spectrum, both in vitro and in vivo, toward relevant pathogens has been previously described. Recently, a chimerical endolysin (ClyS) assembling a CD and a CWBD from different S. aureus phages, and in which the latter displayed Staphylococcus-specific binding, was shown to lyse different S. aureus MRSA and MSSA strains and several coagulase-negative Staphylococcus species. 17 This chimera revealed to be more effective than mupirocin for skin decolonization of MRSA and MSSA. 61 In another example of homologous fusion, the CHAP domain of LysK, the endolysin from S. aureus phage K, was fused to the CWBD of the bacteriocin lysostaphin from Staphylococcus simulans. 38 The recombinant endolysin PRF-119 exhibited antimicrobial activity against a great number of MSSA and MRSA clinical isolates, with an average MIC90 of 0.391 μg/ml.
Heterologous fusion constructs where the native CWBD of the streptococcal LambdaSa2 endolysin Cpl-7 was replaced by that of either lysostaphin or LysK resulted in a approximately fivefold increase in staphylolytic activity mediated by the Cpl-7 CD, while maintaining significant streptolytic activity. 8 The results suggested that the CWBD of S. aureus lytic enzymes may not always be staphylococcal-specific. Yet in another example, chimeras composed of the streptococcal phage endolysin B30, or of its CHAP CD only, fused to the mature form of lysostaphin were shown to display lytic specificity for streptococcal pathogens and S. aureus. 22
In the work reported here, we have fused the CD of two highly soluble E. faecalis phage endolysins, Lys168 and Lys170, to the CWBD of the staphylococcal phage endolysin Lys87, which showed high propensity to precipitate in the form of inclusion bodies. Our rational was that the CD of the enterococcal enzymes would confer high solubility to the resulting chimerical endolysins. In addition, the fact that Lys168 and Lys170 CDs were of the CHAP and Amidase-2 families, respectively, increased the chances of these domains to act on the S. aureus cell wall, if targeted by the Lys87 CWBD. Actually, the Lys168 CHAP domain, as deduced by Pfam analysis, shares 98% identity with that of the endolysin of S. aureus phage SAP6. 62 Lys170 Amidase-2 CD should confer N-acetylmuramoyl-L-alanine amidase activity as this was the activity experimentally determined for ORF9, an endolysin that is virtually identical to Lys170.62,76 Amidases cleave the amide bond that links the N-acetyl muramic acid of glycan strands to the L-alanine residue of peptide stems. This bond and linked residues are common to the vast majority of bacterial cell wall peptidoglycans, including that of S. aureus. 73
The chimeras thus constructed proved to efficiently lyse S. aureus clinical strains. When tested at their highest concentration each recombinant endolysin was able to induce lysis in more than 96% of 143 S. aureus clinical isolates. The lytic efficiency of each chimera varied among the different isolates, as judged by the relative size and transparency of the lysis halos (Supplementary Tables S2 and S3). Remarkably, all the 30 typed strains composing a panel representative of the most relevant MRSA epidemic clones (Table 1) showed to be susceptible to both chimerical enzymes. Very few anti-staphylococcal endolysins have been tested in such a representative group of MRSA typed strains.
Also interesting was the fact that these chimerical endolysins not only essentially maintained the lytic performance of the parental enterococcal endolysins when tested against Enterococcus, 62 but were also extended in their lytic spectrum being active against other Staphylococcus species and Streptococcus pyogenes. This suggests that the Lys87 CWBD may target a bacterial cell wall epitope that is common to several Gram-positive bacterial species, while that of the parental enterococcal endolysins binds to a ligand predominantly found on enterococcal cell wall. This apparent wider target range of the CWBD of anti-staphylococcal lytic enzymes was also observed for lysostaphin and LysK, as mentioned above. Thus, Lys168-87 and Lys170-87 can be added to the small list of broadly active, phage-derived lytic enzymes.8,22,80
A curious difference was observed when we compared the lytic efficiency of Lys168 and Lys170 with that of the derived chimeras Lys168-87 and Lys170-87. When tested against their natural target, E. faecalis, Lys170 generally produced larger and more transparent lysis halos than Lys168. However, this scenario was inverted when Lys168-87 and Lys170-87 were assayed against S. aureus (see illustrative example in Fig. 5B). This appears to indicate that the lytic performance of a given endolysin is not a mere function of the affinity and binding of the CWBD to the cell wall. In fact, other parameters such as the affinity of the CD to its substrate may influence lytic activity. 49
Lys168-87 and Lys170-87 could also lyse liquid cell suspensions of viable S. aureus. In addition, they showed a clear synergistic effect when simultaneously added to cells (Fig. 4). To the best of our knowledge, such synergy between phage-derived hydrolytic enzymes in the effective lysis and killing of S. aureus has never been reported. Several studies though have demonstrated the occurrence of synergy between S. aureus phage endolysins and bacteriocins such as nisin 31 or lysostaphin 9 or between endolysins and conventional antibiotics.17,20,51,63
In conclusion, our results indicate that the engineering of chimerical endolysins from heterologous functional domains can be a good strategy to obtain large quantities of soluble and highly effective peptidoglycan hydrolases, either with narrow or broad lytic spectrum. The efficacy of the chimeras Lys168-87 and Lys170-87 is currently under study in animal models of S. aureus infection.
Footnotes
Acknowledgments
D. Proença and C. Cantante work has been supported through Ph.D. fellowships SFRH/BD/64177/2009 and SFRH/BD/48598/2008, respectively from Fundação para a Ciência e a Tecnologia (FCT, MCTES, Portugal). C. Milheiriço was supported by Postdoctoral fellowship SFRH/BPD/63992/2009 from FCT.
Author Disclosure Statement
Technophage has proprietary rights over E. faecalis phages F168/08, F170/08, and S. aureus phage F87s/06, their encoded endolysins Lys168, Lys170, and Lys87, and the derived chimerical enzymes Lys168-87 and Lys170-87. All authors reviewed and approved the present article before submission. All authors declare that there are no conflicts of interest. All authors contributed to the elaboration of the article. S.F., D.P. and C.C. were responsible for endolysin cloning, production, and purification; SF executed all experiments evaluating lytic action of chimerical endolysins; F.A.S., C.L., C.C., and S.L. were responsible for phage isolation, purification, and extraction of phage DNA; C.L. and S.L. performed phage genomic analysis; C.M. was responsible for handling and molecular typing of S. aureus typed clinical strains. H.deL., P.C.-S., M.G., M.P., and C.S.-J. were responsible for the experimental design and work supervision; C.S.-J. coordinated the collaborative work of the involved laboratories.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
