Abstract
Lantibiotics are a unique group within the antimicrobial peptides characterized by the presence of thioether amino acids (lanthionine and methyllanthionine). These peptides are produced by and primarily act on Gram-positive bacteria exerting multiple activities at the cytoplasmic membrane of susceptible strains. Previously, the cell wall precursor lipid II was identified as the molecular target for the prototype lantibiotic nisin. Binding and sequestration of lipid II blocks the incorporation of the central cell wall precursor into the growing peptidoglycan network, thereby inhibiting the formation of a functional cell wall. Additionally, nisin combines this activity with a unique target-mediated pore formation, using lipid II as a docking molecule. The interaction with the pyrophosphate moiety of lipid II is crucial for nisin binding. We show that, besides binding to lipid II, nisin interacts with the lipid intermediates lipid III (undecaprenol-pyrophosphate-
Introduction
The first discovered and most extensively studied lantibiotic is nisin.
24
It is produced by some
Early experiments demonstrated that, due to their strong amphiphilic nature, nisin and related lantibiotics impair membranes causing dissipation of the membrane potential, rapid efflux of small metabolites, and cessation of cellular biosynthetic processes.27,29,30 Subsequently, nisin was shown to destabilize model membranes at concentrations in the micromolar range15,19 and to bind and sequester the peptidoglycan precursor lipid II, which enables the formation of specific pores in the nanomolar range.8,10,38
Lipid II represents the central cell wall building block of peptidoglycan biosynthesis that is structurally conserved among eubacteria. The precursor consists of the bactoprenol carrier lipid (C55-P), which is linked to the disaccharide unit
This dual mode of action, in which inhibition of peptidoglycan synthesis and pore formation are most efficiently combined, makes nisin a very potent antimicrobial agent and impedes the emergence of resistant strains. The binding motif of nisin is conserved among a number of lantibiotics targeting lipid II, for example, subtilin, epidermin, gallidermin, plantaricin C, and mutacin 1140.
40
The interaction primarily involves the formation of five hydrogen bonds between the pyrophosphate moiety of lipid II and the amide backbone of the
Besides lipid II, nisin and related lantibiotics were also shown to interact with lipid I
In
In this study we report the
Materials and Methods
Chemicals and reagents
All chemicals were of analytical grade or better. 1,2-Dioleoyl-
Bacterial strains and growth conditions
Cloning of WTA biosynthesis enzymes
Standard procedures for molecular cloning and transformation were used.14,31 Genes encoding enzymes involved in WTA biosynthesis were amplified from
Restriction sites are shown by underline.
Expression and purification of cytoplasmic proteins
Expression and purification of the membrane-bound protein TagO
Purification of recombinant TagO-His6 (Llm) enzyme was performed as described previously with minor modifications.
26
In short,
Nisin purification
Nisin was obtained and purified from culture supernatants of
Antibiotic susceptibility testing
Minimal inhibitory concentrations (MICs) were determined by standard broth microdilution, according to the Clinical and Laboratory Standards Institute guidelines, in polypropylene microtiter plates (Nunc brand) using cation-adjusted Mueller-Hinton broth (Oxoid).
Antagonization assays
Antagonization of the antibiotic activity of nisin and gallidermin by potential target molecules was performed by an MIC-based setup in microtiter plates. Nisin (0.2 μg/ml) and gallidermin (0.04 μg/ml) (each corresponding to 8×the MIC) were mixed with potential HPLC-purified antagonists (C55-P, C55-PP, lipid I, lipid II, lipid III, and lipid IV) in a 10-fold molar excess with respect to the antibiotic.
In vitro WTA synthesis reactions using purified proteins and substrates
The enzymatic activity of the TagO-catalyzed lipid III (undecaprenol-pyrophosphate-
Nisin and gallidermin were added in a 2:1 molar ratio with respect to the amount of purified C55-P, lipid III, and lipid IV, respectively, in all
In vitro lipid I/II synthesis and purification
Large scale synthesis and purification of the peptidoglycan precursors lipid I and II were performed as described previously. 34
Mass spectrometry
Mass spectrometry analysis of cell wall precursors was performed using a matrix assisted laser desorption/ionization-time of flight (MALDI-TOF) mass spectrometer (Bruker Biflex; Bruker Daltonics) working in a linear negative mode. Samples were mixed with 6-aza-2-thiothymine (in 50% [v/v] ethanol and 20 mM ammonium citrate) as matrix material and spotted onto the MALDI target. Mass spectra were measured in the range of 1,000–1,200 and 1,300–1,350 Da, respectively, and analyzed by Flexanalysis 2.0 (Bruker Daltonics).
Complex formation of nisin with cell wall precursors
Binding of nisin to C55-P, C55-PP, lipid II, and lipid III was analyzed by incubating 5 nmol of each purified cell wall precursor with 0, 10, or 20 nmol nisin in 50 mM Tris-HCl (pH 7.5). Complex formation was analyzed by incubating the lipid precursors with the respective amount of nisin. Free precursors were extracted with n-butanol/pyridine acetate (pH 4.2) (2:1; vol/vol) and analyzed by TLC. Lipid-containing precursors were detected by phosphomolybdic acid staining.
Quartz crystal microbalance
Quartz crystals were prepared and cleaned as described previously. 12 Supported bilayers were completed by transferring a monolayer consisting of DOPC±0.1 mol% lipid intermediate onto a covalently fixed C16H33SH monolayer using the Langmuir-Blodgett technique. Fitting the quartz crystals into the flow chamber was performed under water to guarantee the integrity of the bilayer. Nisin was investigated at 300 μM. A LiquiLab21 quartz crystal microbalance (ifake.V.) was employed, which enables the detection of frequency in real time. Measurements and calculation of the kinetic binding constants were performed as described.13,36
CF efflux experiments
Large unilamellar vesicles for CF efflux experiments were prepared by the extrusion technique as described previously. 39 Vesicles were made of DOPC doped with 0.1 mol% of the respective cell wall precursors (C55-P, C55-PP, lipid I, lipid II, lipid III, or lipid IV; referring to the total amount of phospholipid). CF-loaded vesicles were prepared with 50 mM CF and then diluted in 2 ml of TBS buffer (10 mM Tris-HCl [pH 7.2]; 0.85% NaCl [w/v]) at a final concentration of 25 μM phospholipid on a phosphorous base. Nisin was added in a final concentration of 50 nM, with or without pre-incubation of liposomes with 50 nM of gallidermin for 60 seconds, respectively. The increase in fluorescence intensity was measured at 520 nm (excitation at 492 nm) on an RF-5301 spectrophotometer (Shimadzu) at room temperature. The peptide-induced CF leakage was expressed relative to the total amount of CF released after lysis of the vesicles by addition of 20 μl of 20% Triton X-100.
Results
In vitro synthesis of WTA precursors lipid III and lipid IV
Undecaprenylphosphate represents the central-membrane-bound lipid carrier involved in the synthesis of diverse bacterial cell envelope polymers and polysaccharides, including peptidoglycan and WTA biosynthesis.
We used the well-characterized
Purified, recombinant MraY and MurG synthesize the peptidoglycan precursors lipid I and lipid II
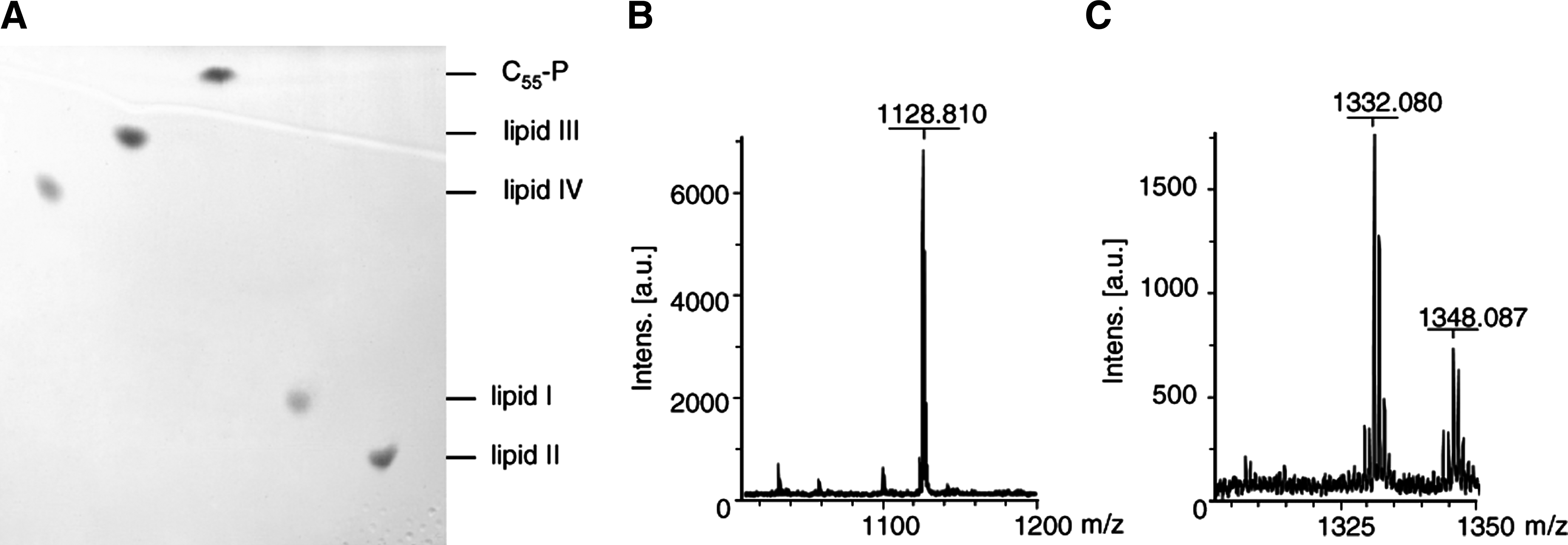
Thin-layer chromatography of purified wall teichoic acid (WTA) (lipid III and lipid IV) and peptidoglycan (lipid I and lipid II) precursors
Altered migration was also observed for the TagO reaction product lipid III (undecaprenyl-PP-GlcNAc) (lane 2). The TagO-catalyzed addition of
Interaction of nisin and gallidermin with WTA precursors
Binding of nisin to lipid II mainly involves the pyrophosphate and the MurNAc moiety as revealed by structural analysis of the nisin:lipid II complex, 20 while the second sugar (GlcNAc) does not appear to significantly contribute to binding.
As lipid III, the first lipid intermediate of the WTA biosynthesis pathway, has a similar pyrophosphate sugar linkage moiety, we investigated the impact of nisin on the lipid III synthesis reaction using purified TagO, C55-P, and [ 14 C]-UDP-GlcNAc. In the positive control, a radiolabeled lipid band was detected, corresponding to lipid III (Fig. 2, lane 2; Rf=0.89), whereas no synthesis product was observed when TagO was omitted (lane 1).
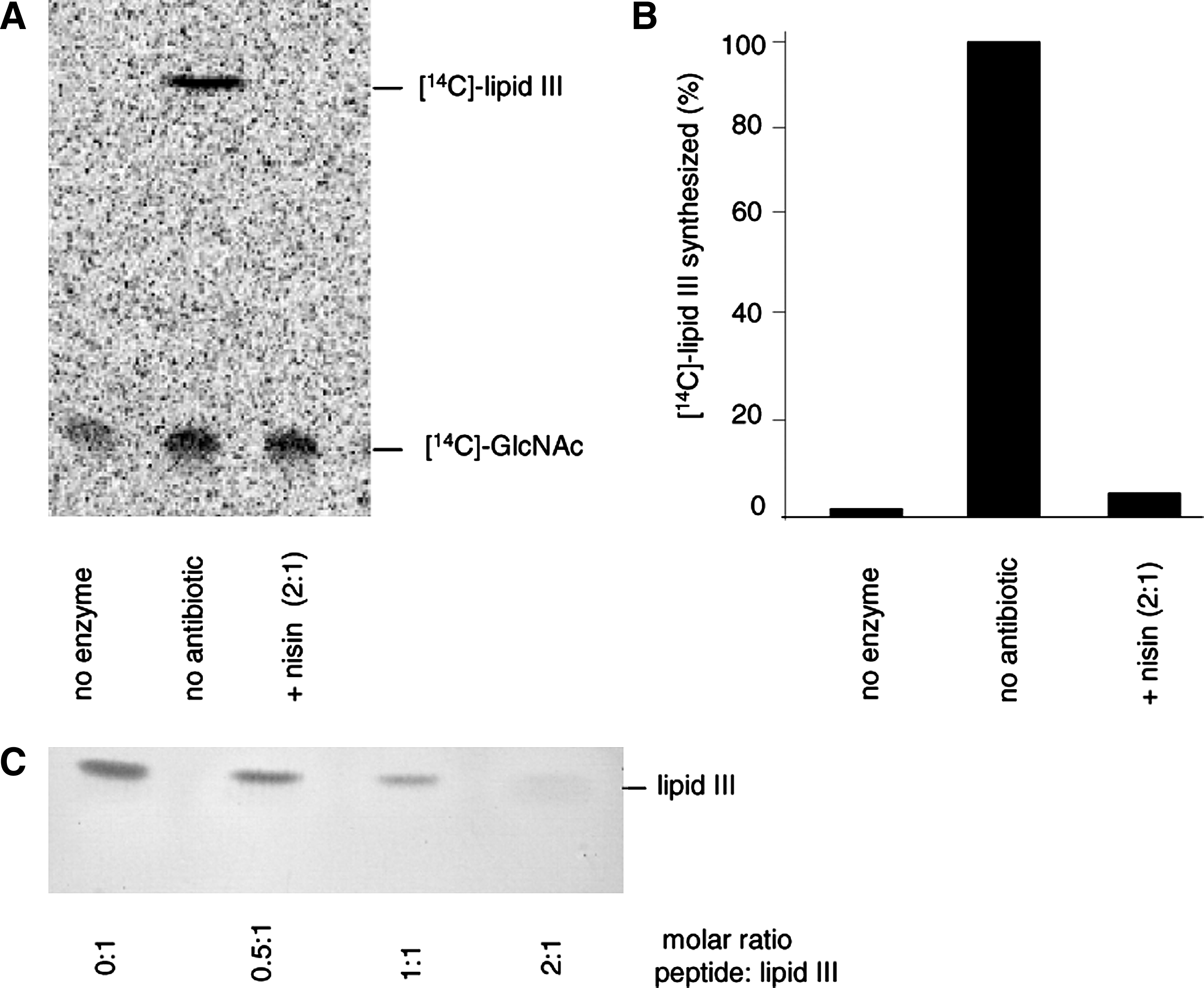
Nisin forms a stoichiometric complex with the WTA precursor lipid III.
No synthesis product could be detected when nisin was added at a twofold molar excess with respect to C55-P (lane 3), suggesting that nisin forms a stable complex with lipid III, which escapes extraction of the lipid intermediate from the synthesis mixture (lane 3), as described for the nisin:lipid II complex. 39
To further verify the formation of a nisin:lipid III complex, purified lipid III was incubated with defined amounts of nisin, followed by extraction and subsequent TLC analysis.
In accordance with the results obtained in the TagO synthesis assay, increasing nisin concentrations gradually diminished the amount of lipid III visible on the TLC plate and almost complete sequestration of lipid III was observed at a twofold molar excess of nisin (Fig. 2C). Similar results were obtained with gallidermin (data not shown).
Quantitative analysis of the TagA-catalyzed lipid IV synthesis reaction using radiolabeled UDP-GlcNAc revealed dose-dependent inhibition by nisin and gallidermin (Fig. 3). Added in equimolar concentrations, nisin and gallidermin inhibited the formation of lipid IV, compared with a positive control—where no antibiotic was added—to about 70% and 50%, respectively. At a molar ratio of 2:1 (nisin:lipid), nisin inhibited the reaction to about 20% and gallidermin prevented the synthesis of radiolabeled lipid IV almost completely. Again, with this experimental setup, it was not possible to differentiate whether the lantibiotics inhibit the synthesis of lipid IV by binding to the precursor lipid III, or whether they block the extraction of the reaction product by complexation. To obtain further information on the nature of the inhibition, we performed antagonization assays with nisin and gallidermin. We used conventional microbroth dilution for MIC determination and added the lipid intermediates in a 10-fold molar excess. Lipid III and lipid IV, as much as lipid I and lipid II, completely antagonized the activity of both lantibiotics (Table 2).
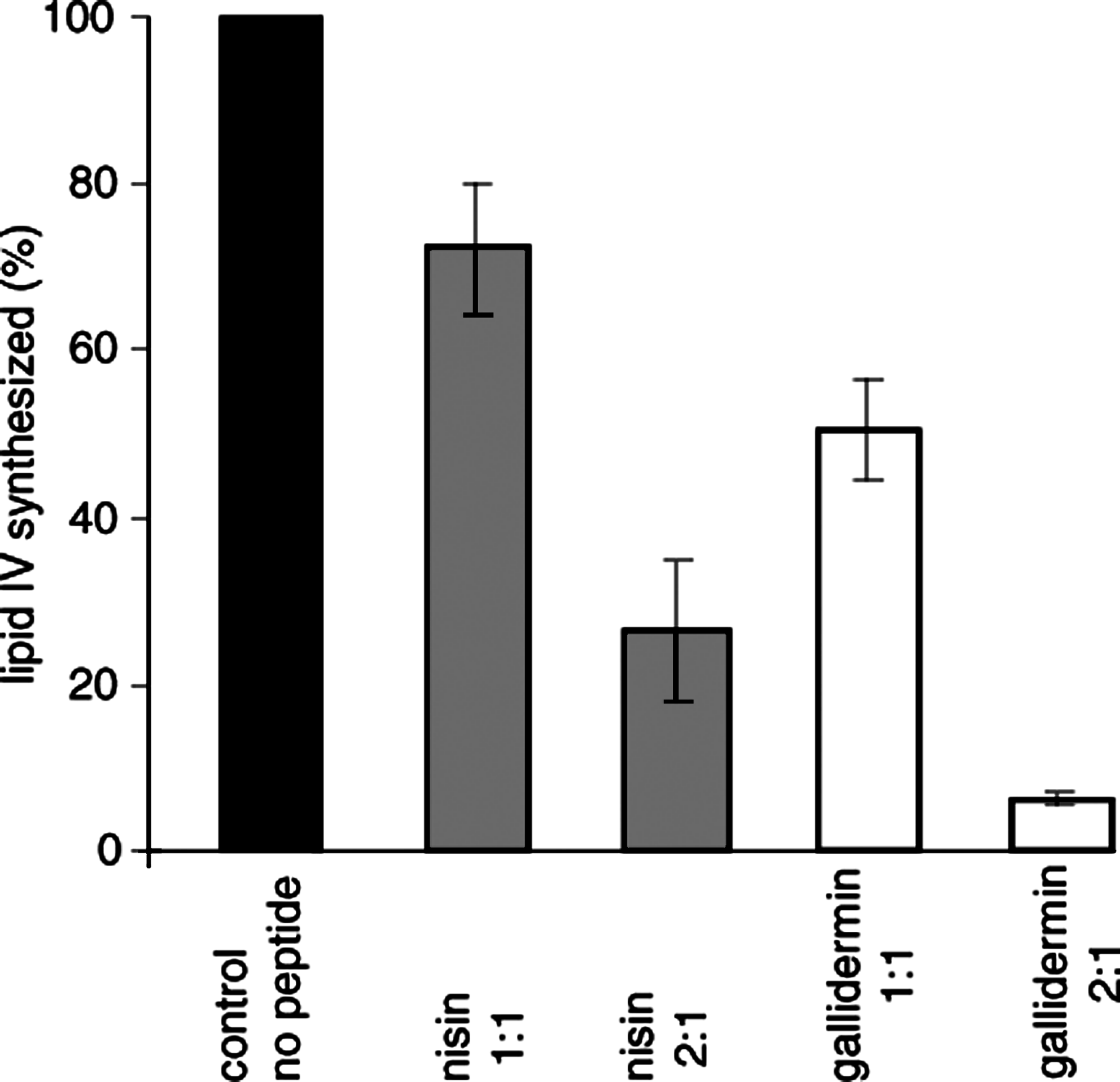
Impact of nisin (gray) and gallidermin (white) on the TagA-catalyzed lipid IV synthesis. TagA was incubated with purified lipid III and [14C]-UDP-GlcNAc. The reaction was initiated by the addition of 1.1 μg MnaA and TagA and incubated for 4 hours at 30°C. Lantibiotics were added at molar ratios of 1 and 2 with respect to the substrate lipid III. Reaction products were extracted with BuOH/PyrAc (2:1) and separated by TLC. Detection and quantification was carried out using a Storm Phosphoimager.
MICs, minimal inhibitory concentrations.
C55-P, in contrast, had no impact, even when added at higher concentrations (25-fold excess; data not shown). This molecule lacks the pyrophosphate moiety, which is essential for the interaction with the nisin-binding motif. Nisin further trapped C55-PP in a stable complex that resided in the aqueous phase after extraction (Fig. 4). An almost complete sequestration of the lipid carrier C55-PP was observed when nisin was added in a twofold molar excess, pointing to a complex stoichiometry of 2:1, as seen with the other WTA and peptidoglycan intermediates.
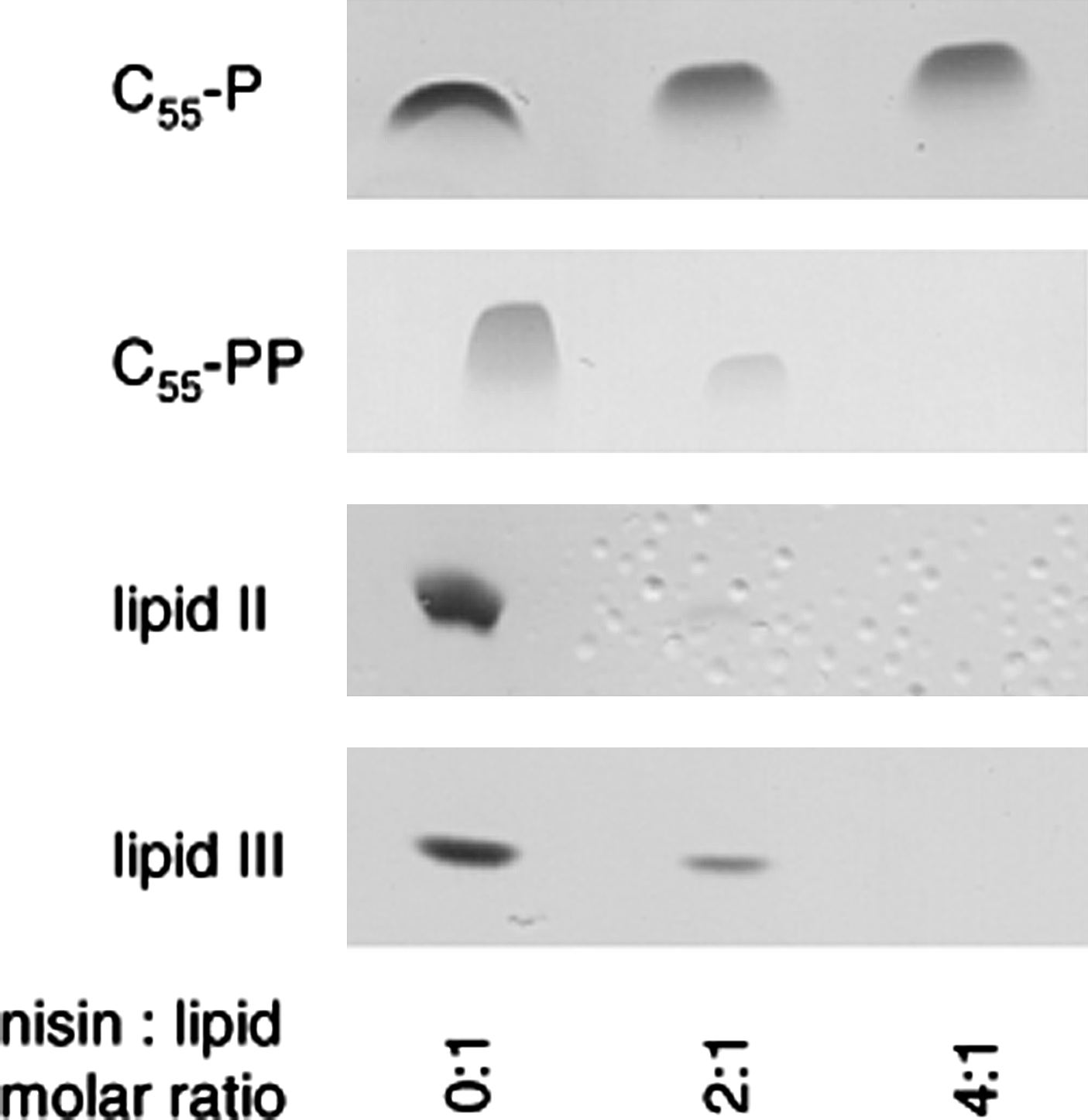
Complex formation of nisin with various cell wall precursors. Cell wall precursors C55-P, C55-PP, lipid II, and lipid III were incubated with increasing concentrations of nisin in molar ratios of 0, 2, and 4 with respect to the respective precursor. Reaction mixtures were extracted and unbound lipids were analyzed by TLC.
Determination of binding parameters
To determine the binding parameters of nisin for phospholipid bilayers doped with various lipid intermediates, we used the quartz crystal microbalance (QCM) biosensor technique.
13
Kinetic binding constants, such as
Association rate (
In line with previous observations and the results obtained from the antagonization assays, binding to C55-P was not detectable. On the contrary, C55-PP-binding affinity is comparable to that of bactoprenol-bound lipid intermediates. However, nisin binding to C55-PP was distinguished by a less-pronounced association and a considerably slower dissociation compared to lipid II, lipid III, and lipid IV.
Nisin-induced CF efflux from lipid-III- and lipid-IV-doped liposomes
To investigate whether the interaction of nisin with lipid intermediates of WTA biosynthesis, that is, lipid III and lipid IV, facilitates the formation of target-specific pores, we prepared CF-loaded DOPC liposomes doped with 0.1 mol% of lipid III and lipid IV and monitored the efflux of the fluorescent dye after addition of nisin. As observed with lipid-I- and lipid-II-doped control vesicles, nisin induced an immediate and rapid CF release from liposomes doped with either lipid III or lipid IV (Fig. 5), reaching comparable values, that is, 50% CF release at 50 nM of nisin. As described for nisin and lipid II, 38 pore formation in lipid-III-doped vesicles was dependent on the concentration of both, peptide and lipid intermediate (data not shown).
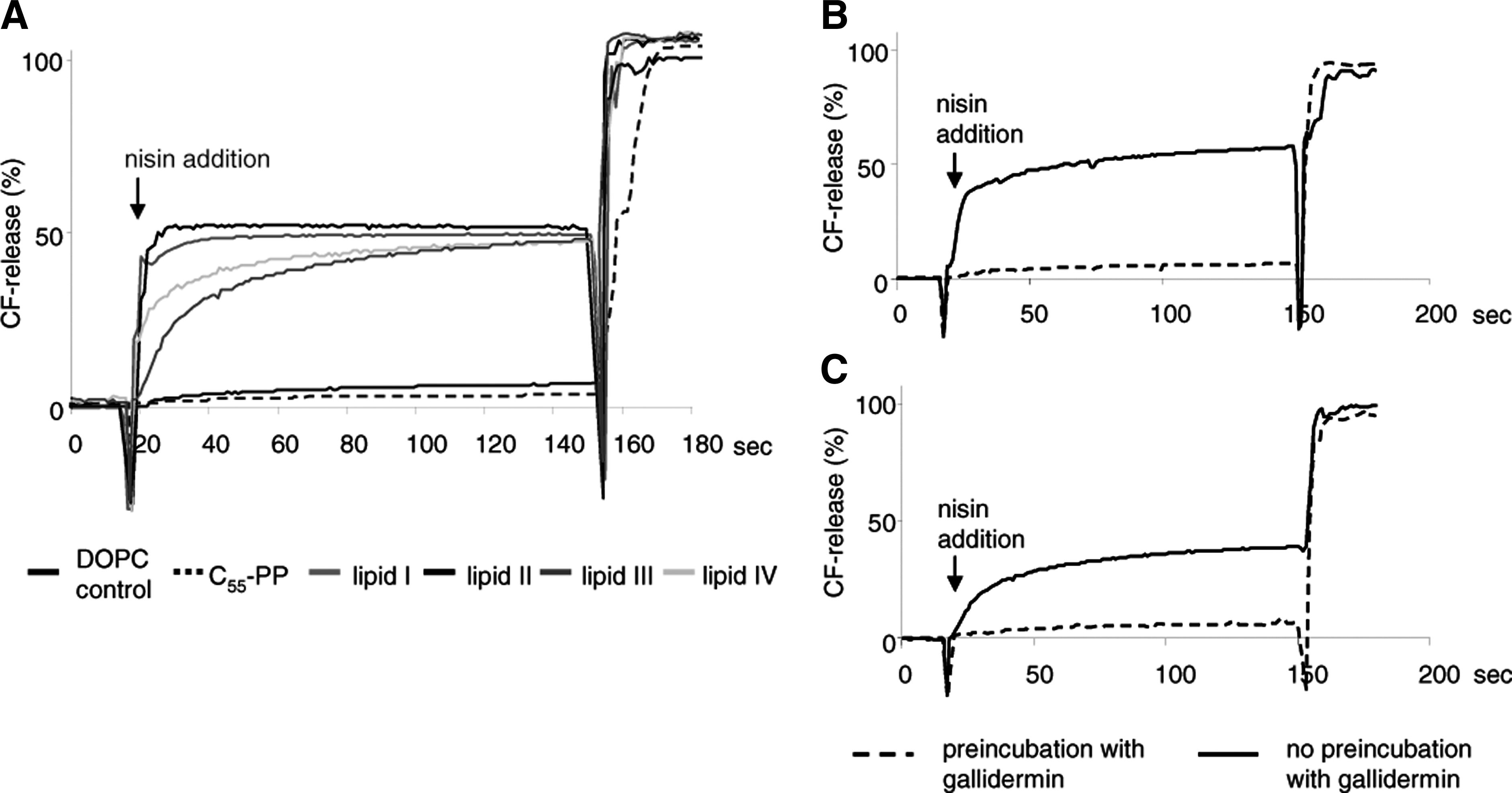
Nisin-induced pore formation. Nisin-induced carboxyfluorescein (CF) release from liposomes made of 1,2-dioleoyl-
Only minor marker release was observed with pure DOPC vesicles or vesicles doped with C55-PP. In the experimental system used here, gallidermin is unable to form pores, but was found to prevent nisin-induced pore formation by shielding lipid II. We therefore preincubated lipid-III- and lipid-IV-doped liposomes with gallidermin before nisin addition and found that its pore formation capacity was completely blocked.
The results provide evidence that WTA precursors can function as docking molecules for targeted pore formation
Discussion
Lantibiotics are active against Gram-positive bacteria and most of them target components of the bacterial cytoplasmic membrane, with the majority of the known compounds exerting their antimicrobial activity by binding to lipid II. 2
Type B lantibiotics, that is, mersacidin, selectively inhibit peptidoglycan biosynthesis by targeting lipid II. 10 In contrast to this, type A lantibiotics, such as nisin, combine a multiplicity of activities in one molecule. The interaction with the cell wall precursor lipid II most efficiently blocks the assembly of a functional cell wall and may promote, dependent on the overall size of the individual lantibiotic, efficient target-mediated pore formation.8,10,38 Moreover, sequestration of lipid II profoundly impacts on the entire cell wall synthesis machinery, resulting in delocalization of the precursor from the septum, aberrant septum formation, and most likely the disorganization of multimeric protein complexes. 17
The various mechanisms combined in one lantibiotic molecule may potentiate the antibiotic activity and explain MIC values in the nanomolar range, as observed with some very sensitive bacteria, such as lactococci. 18 Moreover, development of resistance is gravely hampered, as different essential bacterial components are simultaneously targeted. 1
In this study we report the
Lipid II and the WTA intermediates differ in the identity of the first sugar being MurNAc or GlcNAc, respectively. Moreover, lipid II is characterized by a pentapeptide side chain attached to the first monosaccharide. Antimicrobial activity of nisin against VRSA (VanA type), already suggested that the terminal D-Ala-D-Ala terminus, is not involved in the interaction with nisin.
8
This hypothesis was further substantiated by nuclear magnetic resonance studies in which the solution structure of the nisin:lipid II complex was described.
20
Hsu
Although C55-PP probably does not serve as docking molecule for the formation of a stable pore, it might contribute to nisin activity
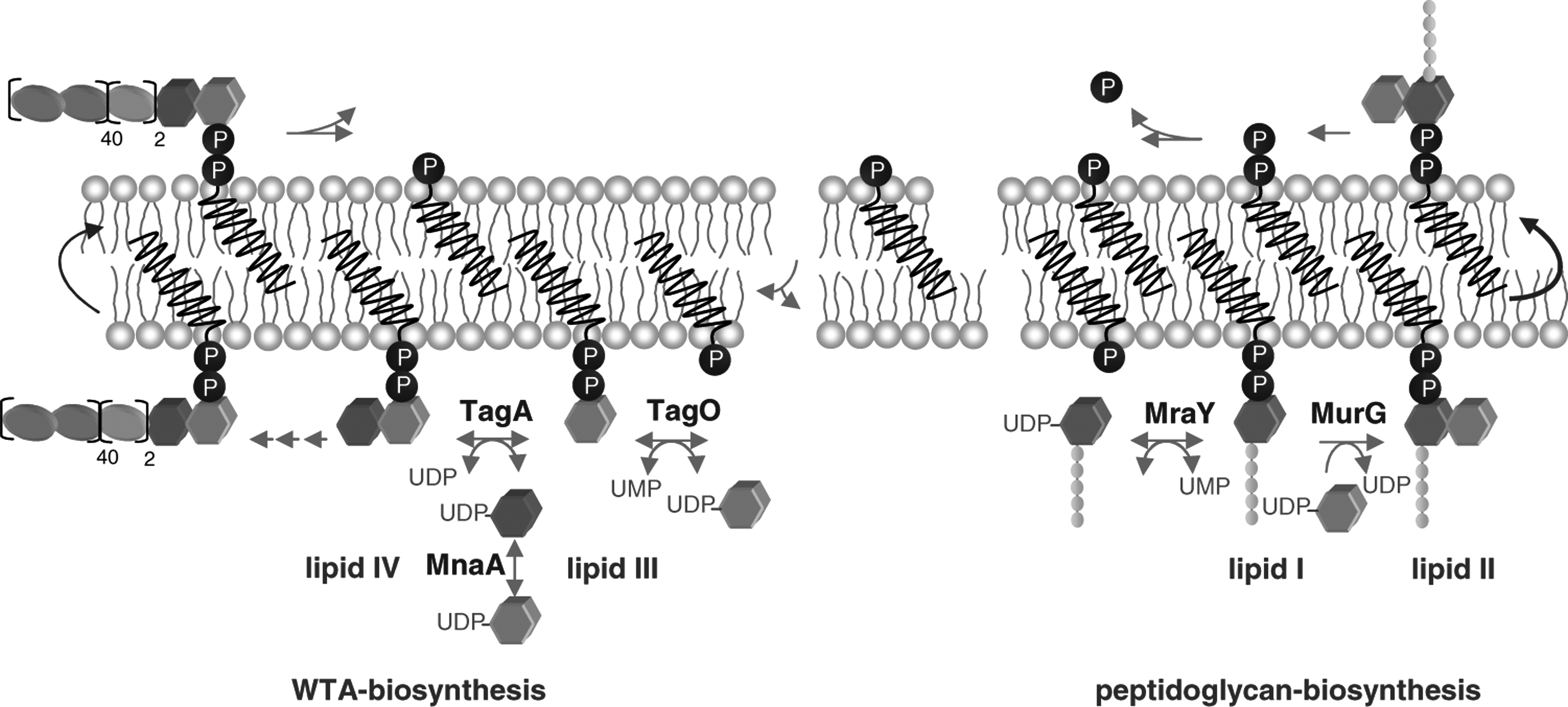
Schematic representation of peptidoglycan and WTA biosynthesis.
The accessibility of WTA precursors must be taken into account to evaluate their importance as targets
Footnotes
Acknowledgment
The authors thank Michaele Josten for mass spectrometry analysis and nisin purification. This work was supported by the German Research Foundation (DFG; SA292/15-1, SCHN 1284/1-2, WI 1912/2-1) and the BONFOR Program of the Medical Faculty, University of Bonn.
Disclosure Statement
None of the authors has a financial relationship with a commercial entity that has an interest in the subject of this article.
