Abstract
Purpose:
Perfluorohexyloctane ophthalmical solution (PFHO) forms an anti-evaporative layer at the air–tear interface and is indicated for treatment of the signs and symptoms of dry eye disease (DED). This study evaluated the ocular pharmacokinetics and biodistribution of PFHO in rabbits.
Methods:
Radiolabeled PFHO was administered to female Dutch Belted rabbits as single (35 µL to each eye) or multiple (twice daily for 5 days) topical ocular doses. Animals were euthanized at designated timepoints. Tears (antemortem), ocular tissues, and blood were collected for pharmacokinetic analysis; heads and carcasses were collected for autoradiographic analysis. Concentrations were measured using liquid scintillation counting.
Results:
After multiple doses, maximum concentration (Cmax) and area under the concentration-time curve were highest in tears (2330 µg/g, 3720 µg•h/g) and Meibomian glands (222 µg/g, 1440 µg•h/g), followed by other anterior tissues (cornea, 27.6 µg/g, 463 µg•h/g; palpebral conjunctiva, 14.0 µg/g, 136 µg•h/g). PFHO was measurable in tears for 8 h and in Meibomian glands for ≥24 h. Distribution to the posterior ocular segment was minimal, and plasma concentrations were low (single-dose Cmax, 0.97 µg/g; multiple-dose Cmax, 3.2 µg/g). In non-ocular tissues, PFHO was confined primarily to nasal tissues and gastrointestinal tract contents; exposure to other systemic tissues was negligible.
Conclusions:
Exposure of PFHO was highest in tears, consistent with its anti-evaporative mode of action, followed by the Meibomian glands. PFHO exposure was very low in posterior ocular tissues and negligible in systemic circulation, consistent with the clinical safety profile.
Introduction
Dry eye disease (DED) is a common disease of the ocular surface.1,2 Dry eye symptoms are estimated to affect 38 million adults in the United States (US), 3 of whom approximately 18 million have been diagnosed with DED. 4 DED can be categorized as aqueous-deficient, evaporative, or mixed, 1 with the majority of cases having an evaporative etiology.5,6 Normally, the lipid layer of the tear film provides a protective barrier for the ocular surface and prevents evaporation of the underlying aqueous layer.7,8 The Meibomian glands are the primary source of the lipids that compose the lipid layer of the tear film, and Meibomian gland dysfunction (MGD) causes alterations in the tear film lipid layer that compromise its anti-evaporative properties,9,10 leading to DED.1,11
Perfluorohexyloctane ophthalmical solution (PFHO, previously known as NOV03) is a single-ingredient topical ophthalmical drop indicated for the treatment of signs and symptoms of DED (MIEBO®,
The efficacy and safety of PFHO have been demonstrated in 4 randomized controlled trials15–18 and a long-term extension study 19 in patients with DED and clinical signs of MGD. The aim of the preclinical study reported here was to assess the ocular pharmacokinetics (PK) and biodistribution of PFHO after topical ocular administration to female Dutch Belted rabbits. Results are expected to help further elucidate the mechanism of action of PFHO and improve understanding of the distribution of the molecule in the body.
Methods
This study was conducted by Covance (Madison, WI, USA) in accordance with Covance’s Standard Operating Procedures and guidelines for animal care and use. All procedures complied with applicable regulations and ethical standards for the care and use of laboratory animals. This study adhered to the Association for Research in Vision and Ophthalmology (ARVO) statement for the Use of Animals in Ophthalmical and Vision Research. Experimental and animal handling procedures were performed in accordance with the US Animal Welfare Act Regulations (9 CFR 3). The study complied with local regulations regarding radiation protection.
Animals
A total of 45 female Dutch Belted rabbits were used in the study. At dosing, the rabbits were 6 months of age, with body weights ranging from 1.5 kg to 2.1 kg. Animals were housed individually in a room under controlled temperature (16°C to 22°C) and relative humidity (50% ± 20%), with a standard 12-h light/dark cycle. Animals were maintained on a standard rabbit diet and provided fresh water daily, ad libitum. Animals were acclimated to study conditions for ≥8 days before dosing.
Preparation of test material
Radiolabeled PFHO (14C-PFHO; Selcia Limited) and non-radiolabeled PFHO (
Administration
Rabbits were assigned to 1 of 4 groups and received single-dose (groups A, C, and D) or multiple-dose (group B) administration of 14C-PFHO (Table 1). The target dose was 35 µL (45.6 mg) of PFHO (25 µCi); doses were administered to the right eye and then the left eye (total bilateral volume, 70 µL). Each topical dose was administered to the central or superior part of the cornea using a positive displacement micropipette and permitted to spread across the ocular surface. Animals were restrained for approximately 1 min after dose administration to prevent rubbing of the eyes.
Doses Administered and Sample Collection
2 animals per timepoint.
1 animal per timepoint.
Sample collection
Tears were collected from 2 animals per timepoint (in group A and group B) at each of the following predetermined timepoints after single-dose (group A) or final dose (group B) administration: 0.25, 0.5, 1, 2, 4, 8, and 24 h. One dye-free TearFlo™ Test (TFT; HUB Pharmaceuticals, Scottsdale, AZ, USA) strip was used for each eye at each timepoint. The TFT strip was weighed before and after tear collection; in addition, the length of wetting on the TFT was measured.
Immediately after tear collection, animals were euthanized with sodium pentobarbital and blood samples were collected by cardiac puncture from 2 animals per group per timepoint. Approximately 5 mL of blood was collected into tubes containing dipotassium ethylenediaminetetraacetic acid. Samples were stored on wet ice until all scheduled aliquots were taken for radioanalysis, and the remaining blood sample was centrifuged to obtain plasma.
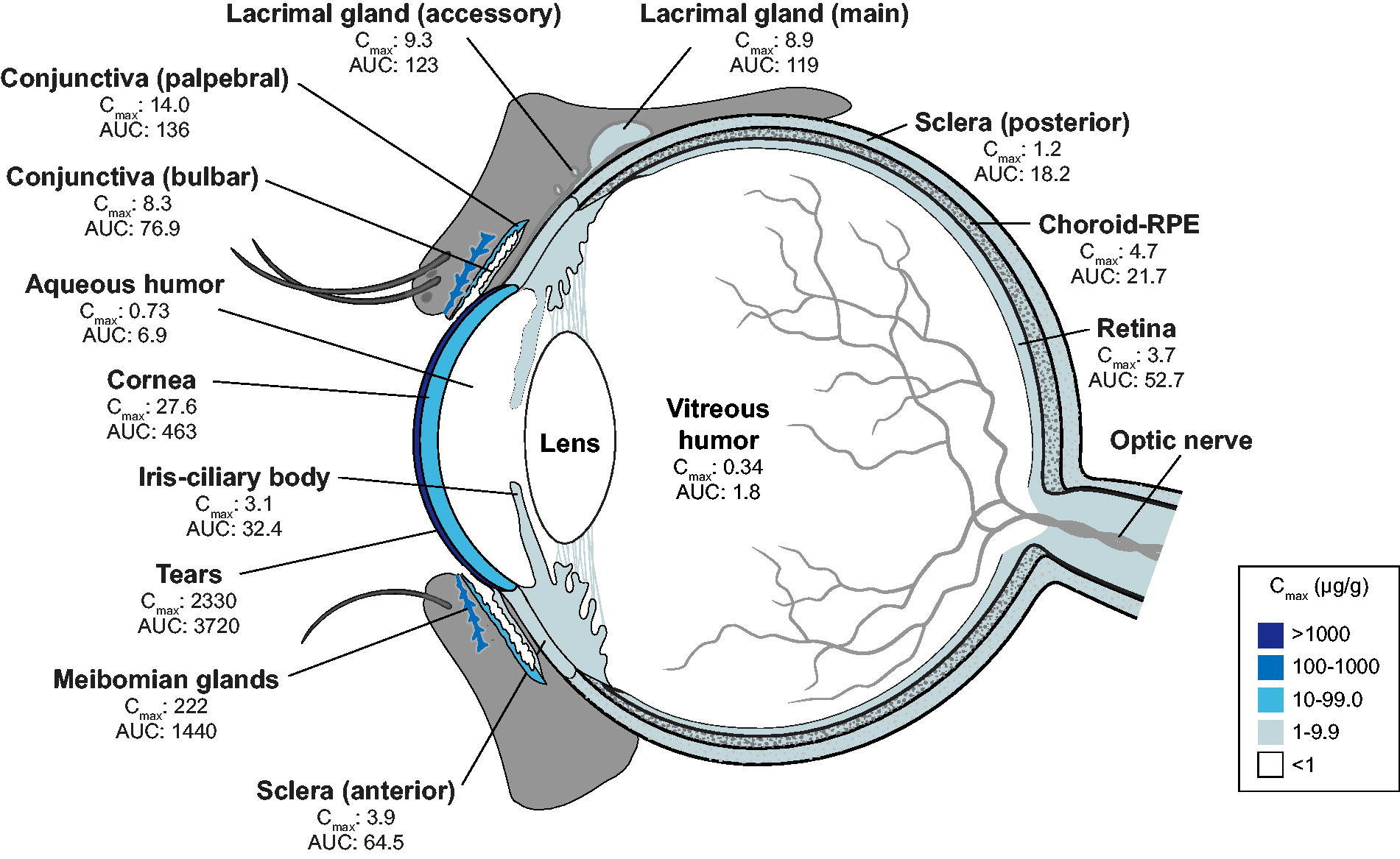
Ocular samples were collected at the time the animals were sacrificed. Both eyes were enucleated, selected tissues were collected, and the remainder of the eye was then flash-frozen in liquid nitrogen for 15 to 20 s. Fresh tissues collected included aqueous humor, conjunctiva (bulbar and palpebral), lacrimal gland (accessory and main), and Meibomian glands (tarsal plate). Within 5 days, the remainder of the frozen eyes were dissected further to isolate the choroid-retinal pigmented epithelium (choroid-RPE), cornea, iris-ciliary body, retina (collected using filter paper), sclera (anterior and posterior), and vitreous humor.
Heads (group C) and whole bodies (group D) were obtained immediately following euthanasia and blood collection. Shaved carcasses were frozen in a hexane/dry ice bath for approximately 1 h, stored at approximately –70°C for ≥12 h, and then stored at –20°C. Frozen heads or carcasses were embedded in chilled carboxymethylcellulose and frozen into blocks, which were then stored at –20°C in preparation for autoradiographic analysis. Sections (40 µm in thickness) were collected at 6 levels of interest in the transverse plane for the head and the sagittal plane for the carcass.
Radioanalysis and autoradiography
Ocular tissue and blood samples were analyzed for radioactivity in a Model 2900TR liquid scintillation counter (Packard Instrument Company) for ≥5 min or 100,000 counts, using automated data capture (Debra™ [LabLogic Systems, Ltd]; and QuantaSmart™). Ocular tissues were homogenized before radioanalysis, and all samples were analyzed in duplicate (if sample size allowed). If the results of the sample analysis were below twice the background value (based on comparison of disintegrations per minute values), the result was reported as below the limit of quantitation and included as 0 in calculations of mean and variance. The lower limit of quantitation, in µg equivalents/g, was 3.17 for the tears, 0.14 for the Meibomian glands, 0.27 and 0.47 for the bulbar and palpebral conjunctiva, 0.19 and 0.47 for the main and accessory lacrimal glands, 0.28 for the cornea, 0.049 for the aqueous humor, 1.33 for the iris-ciliary body, 0.27 and 0.12 for the anterior and posterior sclera, 0.13 for the vitreous humor, 3.31 for the choroid-RPE, and 0.53 for the retina.
PK parameters for radioactivity were determined in ocular tissues, blood, and plasma using Phoenix WinNonlin™ (Version 6.4 or higher; Pharsight Corporation). PK parameters included maximum concentration (Cmax), time to maximum concentration (Tmax), and area under the concentration-time curve from time 0 to the last measurable timepoint (AUC0-t).
For head and whole-body autoradiography, mounted sections were exposed on phosphorimaging screens and scanned (Typhoon™ scanner; Cytiva Life Sciences). Autoradiographic standard image data were sampled via MCID™ analysis software (InterFocus Imaging Ltd.) to create calibrated standard curves, and tissue concentrations were interpolated.
Results
Test article and dose formulation analysis
The mean radiopurity of 14C-PFHO was 99.5% prior to dose preparation. Mean radiopurity of predose and postdose aliquots of the dose preparation was >99% on both day 1 and day 5, confirming the stability of the test article under the conditions of the study.
Pharmacokinetic analysis (groups A and B)
The PK parameters for PFHO in ocular tissues (i.e., tears, Meibomian glands, cornea, conjunctiva, lacrimal glands, sclera, retina, iris-ciliary body, aqueous and vitreous humor, and choroid-RPE) and blood (i.e., whole blood and plasma) after topical administration of 14C-PFHO are shown in Table 2. After single-dose or multiple-dose topical administration, PFHO was found primarily in tears and Meibomian glands (Figs. 1A and 1B). The concentration in tears and Meibomian glands peaked 15 min after administration, and PFHO was present in tears for up to 8 h and in Meibomian glands for at least 24 h after single-dose administration (Fig. 1A). Substantial concentrations were also found in the lacrimal glands and other anterior tissues (i.e., cornea, conjunctiva, and sclera) (Table 2, Fig. 2). Limited distribution into posterior segment tissues was observed (Table 2). In all of these tissues, maximum concentrations of PFHO after multiple-dose administration were similar to those observed after a single administration, suggesting that little accumulation had occurred.
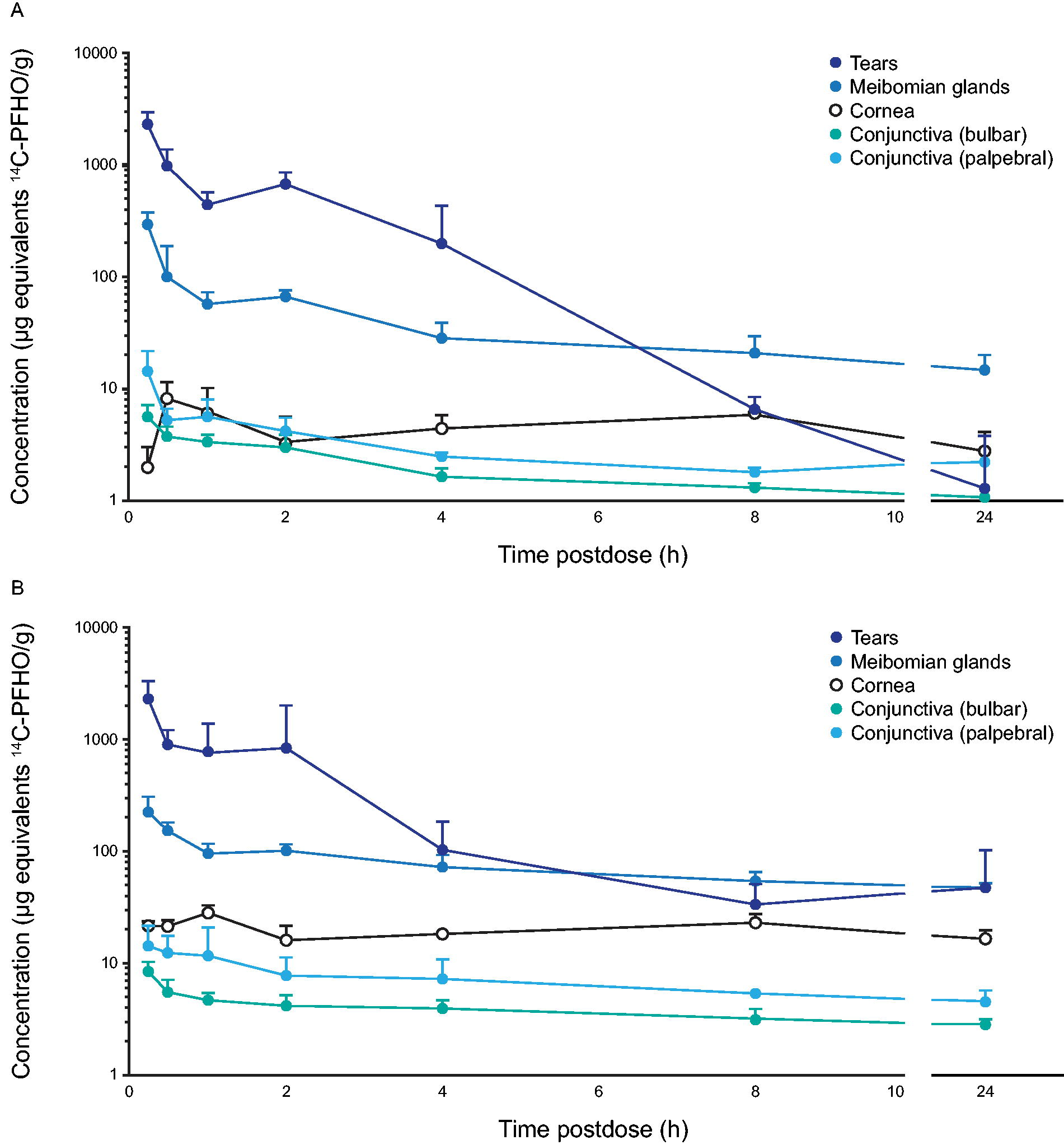
Concentrations of PFHO in tears, Meibomian glands, and anterior segment tissues after single-dose
Ocular distribution of PFHO after repeated topical administration. Samples were taken from 2 animals (4 eyes) at each timepoint on day 5 (0.25, 0.5, 1, 2, 4, 8, and 24 h). Data represent mean values. AUC, area under the concentration time curve from time 0 to the last measurable timepoint, measured in µg•h/g; Cmax, maximum concentration, measured in µg/g.
Pharmacokinetic Parameters for PFHO in Ocular Tissues, Blood, and Plasma
Samples were taken from 2 animals (4 eyes) at each timepoint on day 1 and day 5 (0.25, 0.5, 1, 2, 4, 8, and 24 h). Data represent mean values. Data listed in order of decreasing day 1 Cmax values for tissues within each ocular segment.
AUC0-t, area under the concentration-time curve from time 0 to the last measurable timepoint; Cmax, maximum concentration; ND, not detected; Tmax, time to maximum concentration; RPE, retinal pigment epithelium.
Very low levels of radioactivity were present in whole blood and plasma whether radiolabeled PFHO was administered as a single dose or twice daily for 5 days (Table 2). After peaking at approximately 4 h post-administration, concentrations of radioactivity in blood and plasma declined thereafter and were below the limit of quantitation by 96 h after single-dose administration. In the group administered multiple doses, concentrations in blood and plasma were assessed only through 24 h after the final dose.
Autoradiography (groups C and D)
Quantitative whole-head autoradiography confirmed the ocular tissue distribution of 14C-PFHO observed in the PK analysis. From 15 min through 24 h postdose, concentrations were generally highest in the eyelid margin, internal eyelid, external eyelid, and cornea; PFHO was also detected in the nasal turbinates and nasolacrimal duct (Fig. 3). Autoradiography of the whole body (from 0.5 h through 168 h postdose) showed that, apart from ocular and nasal tissues, radioactivity was primarily confined to the gastrointestinal tract and its contents (Fig. 4). In other tissues, such as brain, liver, kidney, heart, and fatty tissues, either no radioactivity was detected or the concentration marginally exceeded the limit of quantification at isolated timepoints.
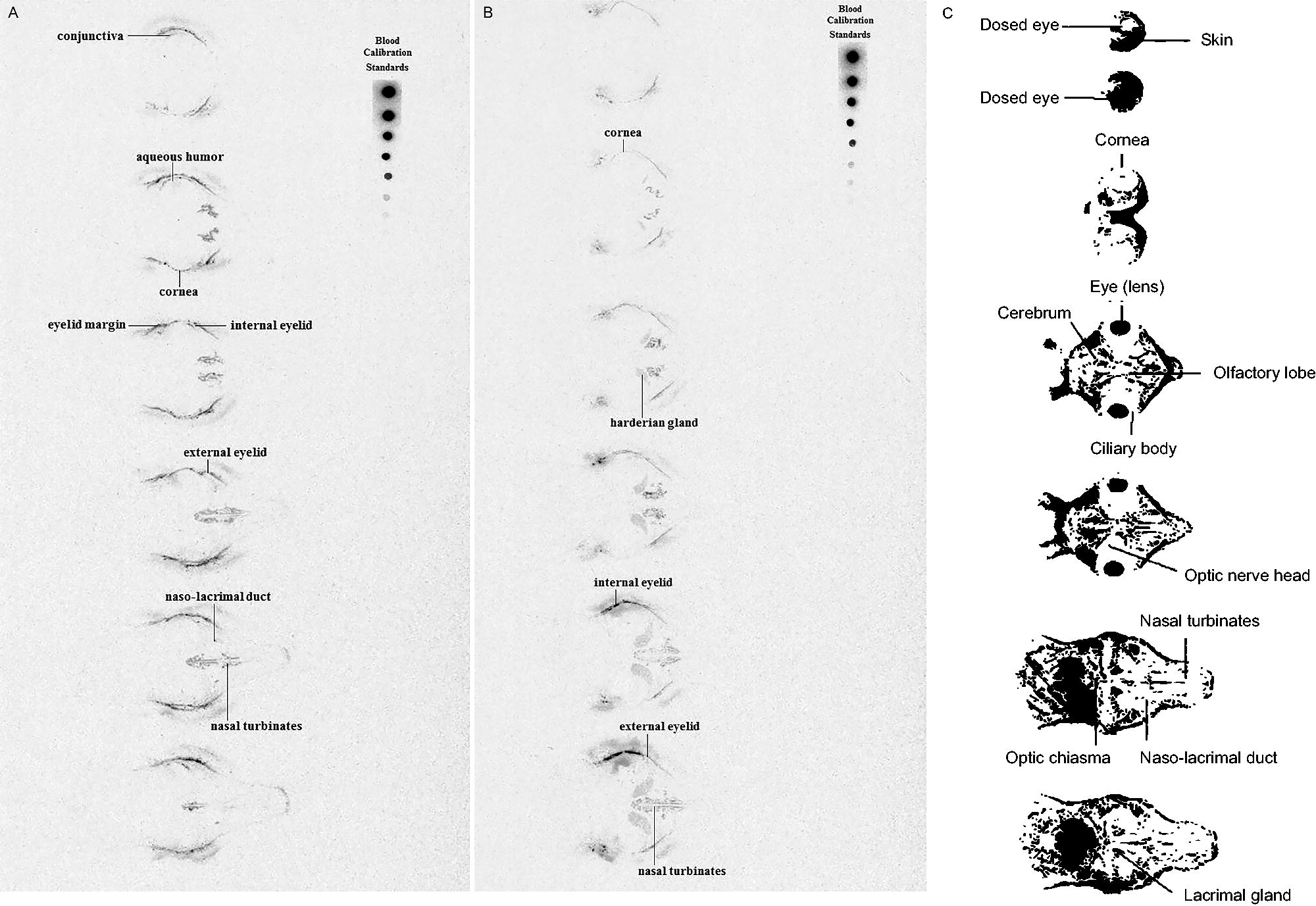
Autoradiography of the heads of Dutch Belted rabbits 0.25 h
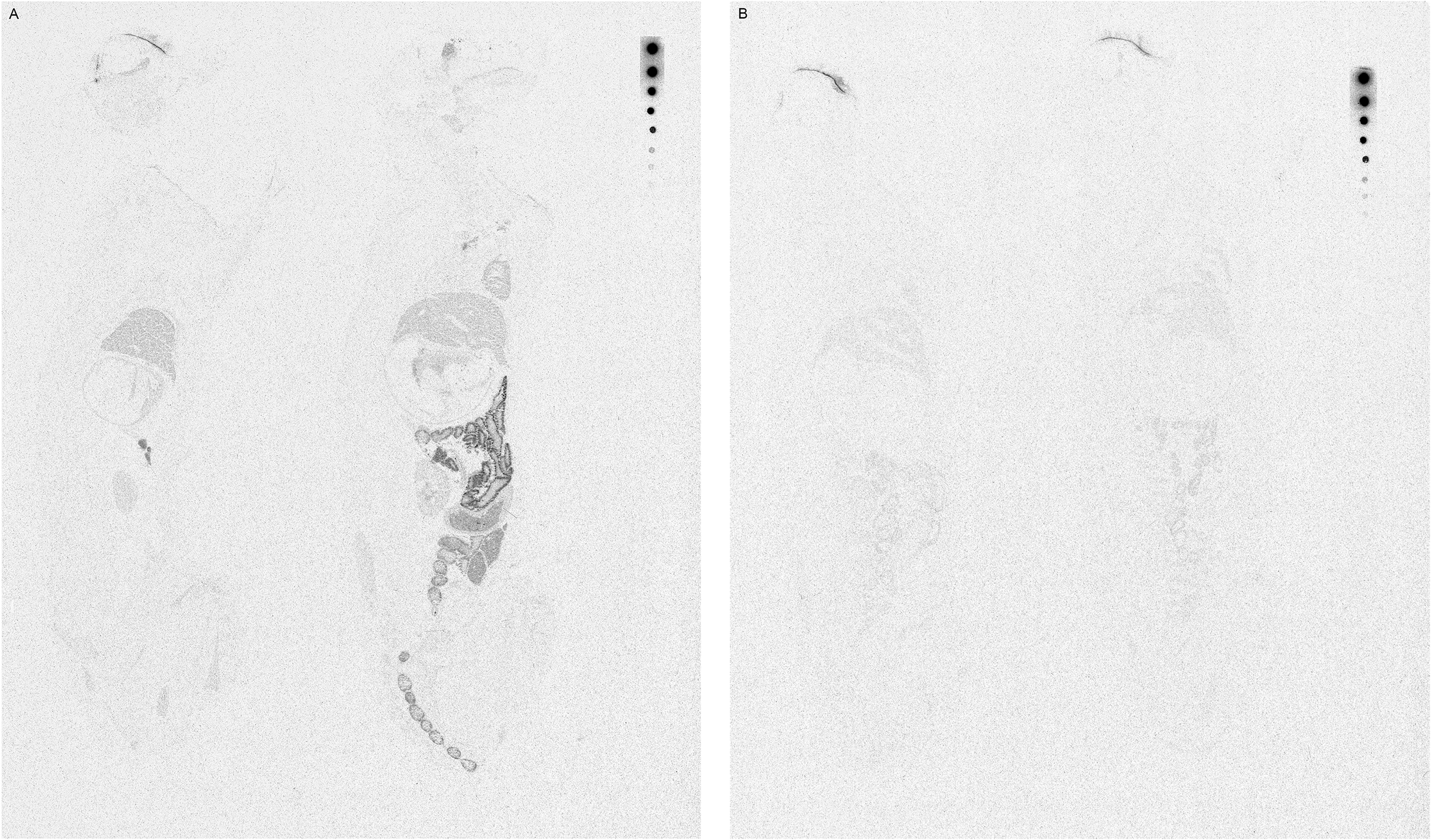
Whole-body autoradiography 8 h
Discussion
After single and repeated topical ocular administration in Dutch Belted rabbits, 14C-PFHO distribution and exposure were greatest in tears and Meibomian glands, consistent with the clinically intended use and the ocular surface being the direct application site for the eye drop. Penetration of cellular membranes is not needed to reach the tears and Meibomian glands. Given its lipophilicity and low surface tension, 20 PFHO is presumed to reach the Meibomian glands via the orifices.
The residence time of PFHO was up to 8 h in tears and 24 h (the last evaluation timepoint) in Meibomian glands. By contrast, aqueous eye drops typically have a short ocular retention time, with expulsion of excess fluid via reflex blinking.21,22 Over-the-counter lubricating eye drops (aqueous- and lipid-based formulations) have a brief ocular surface retention time of up to 20 to 40 min in patients with DED.23–25 The long ocular residence time of PFHO is consistent with its mechanism of action: PFHO forms a monolayer at the air–tear film interface to inhibit evaporation of the aqueous component of the tear film. 12 In patients with evaporative DED, PFHO is considered a functional substitute for the deficient tear film lipid layer. 26 The long residence time of PFHO in Meibomian glands suggests that they may serve as a depot. In addition, findings from other studies suggest that PFHO might also interact with meibum, thus leading to increased thickness of the tear film lipid layer.14,27 The PK profile observed in this study, with long residence times in tears and Meibomian glands, supports the QID (four times daily) dosing schedule used in the phase 3 studies of PFHO in patients with DED16,17 and recommended in the approved product label. 12
The PK data from the present study are consistent with clinical efficacy findings and support the notion that PFHO provides protection against evaporation for an extended period. The anti-evaporative effects of PFHO have been demonstrated in vitro, 26 and randomized controlled trials have consistently demonstrated that PFHO improves both the signs (e.g., corneal fluorescein staining) and symptoms (e.g., eye dryness, burning/stinging) of DED.15–17
Much lower concentrations of 14C-PFHO were observed in posterior segment ocular tissues, indicating that PFHO distribution to the back of the eye is limited. Concentrations in systemic tissues were negligible after topical ocular administration of 14C-PFHO. Minimal concentrations were observed in plasma and in blood, but these declined after approximately 4 h and were below the limit of quantitation by 96 h postdose.
Evaporation of PFHO from the ocular surface is likely a main route of elimination, based on findings from an in vitro evaluation, 26 an ex vivo model, 20 and demonstrated low or negligible absorption into the majority of the ocular tissues and systemic circulation. 28 Indeed, nearly 50% of a 50-µL drop of PFHO had evaporated from the surface of a porcine eye at 24 h post-instillation. 20 The presence of radioactivity in the rabbit nasolacrimal ducts and gastrointestinal tract, observed after topical administration in this study, shows that a proportion of PFHO entered the gastrointestinal tract via nasolacrimal drainage. This PFHO portion remains unabsorbed in the gastrointestinal tract and is then excreted unchanged in feces. 29 This finding is consistent with the physiochemical properties of the molecule, including the very low water solubility, the absence of functional groups, and the perfluorinated carbon chain, which in sum limit membrane penetration. 30
The current study did not include mass balance determinations, and the relative amounts of PFHO that evaporated from the ocular surface into the air or drained into the nasolacrimal ducts were not evaluated, as this was not in the scope of this topical ocular study. Following oral administration of PFHO in a preclinical rodent study, the majority of the administered dose was excreted in feces; biliary excretion was not a route of elimination. 29 PFHO is both chemically and biologically inert, 28 and in vitro microsome studies indicate that it is unlikely to be metabolized in the human body. 12 The negligible systemic absorption of PFHO observed after topical ocular administration to rabbits in this preclinical study is consistent with the PK profile of PFHO ophthalmical drop in a clinical study of patients with DED, which found that most blood samples (>70%) had no measurable PFHO and that the remaining samples had levels just above the limit of quantification (1 ng/mL). 15 These findings are consistent with the favorable systemic safety profile of PFHO observed across clinical trials in patients with DED.15–17
The current study had some limitations. Because drug concentration measurements required postmortem tissue sampling, data reflect a limited number of animals for each assessment, taking ethical and animal welfare considerations into account. The study duration was relatively short; the maximum duration of administration of PFHO was twice daily for 5 days. The volume administered to rabbits in this study (35 µL per eye) was used to ensure assay sensitivity but is approximately 3 times the volume of a dispensed drop (11 µL) of the product approved for use in patients with DED.12,26
Conclusion
Following ocular administration in rabbits, the highest concentrations of PFHO were found in tears and Meibomian glands, with measurable levels detected through 8 h and 24 h, respectively. These study findings support the mechanism of action of PFHO—formation of a monolayer at the air-liquid interface to inhibit evaporation—in patients with DED. The PK profile of PFHO in this study, notably the very low distribution to the posterior segment, negligible systemic tissue exposure, and lack of accumulation after multiple administrations, is consistent with the favorable clinical safety profile in patients with DED.
Footnotes
Acknowledgments
Editorial and medical writing assistance was provided under the direction of the authors by Nancy Holland, PhD, Synchrony Medical Communications, LLC, West Chester, PA, and funded by
Authors’ Contributions
S.K.: Supervision, conceptualization, data interpretation; writing—review and editing. R.G.: Resources, writing—review and editing. K.E.: Project administration, writing—review and editing. J.K.: Project administration; writing—review and editing. M.E.C.: Data interpretation; writing—review and editing. F.S.M.: Data interpretation; writing—review and editing.
Author Disclosure Statement
S.K., R.G., K.E., and J.K. are employees of Novaliq. M.E.C. is an employee of
Funding Information
This study was sponsored by Novaliq GmbH.
