Abstract
Purpose:
To evaluate the durability of thermomechanical device treatment effect in patients with meibomian gland dysfunction (MGD) at 6 months post-treatment.
Methods:
This was an extension of an initial 3-month, prospective, controlled, randomized, masked, multicenter pivotal study, in which subjects with MGD were randomized to thermomechanical device treatment (3 sessions, 2 weeks apart) or a single control treatment. The extension study was a single-arm, observational study in the same 5 sites of the pivotal study. A subset of subjects from the thermomechanical device group with an increase in tear break-up time (TBUT) of 2.5 s or greater in at least 1 eye at 1- or 3-month follow-up and able to attend the 6-month follow-up were included. Effectiveness endpoints included changes in TBUT, Meibomian gland score (MGS), and Ocular Surface Disease Index (OSDI) from baseline to 6 months. Device-related adverse events (AEs) were also assessed.
Results:
At 6 months post-treatment, 21 subjects (42 eyes) demonstrated significant improvements from baseline in mean TBUT (5.2 ± 3.8 s; P < 0.001), mean MGS (18.2 ± 10.9; P < 0.0001), and mean OSDI (−24.3 ± 26.5; P = 0.0004). Improvements in corneal staining scores were also observed. No ocular AEs were reported.
Conclusions:
The findings of the extension study demonstrate that the clinical benefit of the thermomechanical device, evaluated by TBUT, MGS, and OSDI, can be maintained out to 6 months and that the device is safe and effective in improving the signs and symptoms of evaporative dry eye disease in MGD.
Introduction
Dry eye disease (DED) is characterized by the loss of homeostasis of the tear film.1,2 Evaporative DED is identified as the most common subtype and is associated with meibomian gland dysfunction (MGD), occurring in over 75% of patients with dry eye.3,4 MGD has been defined as “a chronic, diffuse abnormality of the meibomian glands, commonly characterized by terminal duct obstruction and/or qualitative/quantitative changes in the glandular secretion.” 5 Meibomian glands, located in the tarsal plate of the eyelids, secrete meibum that contributes to the superficial lipid layer of the tear film. The lipid layer is essential in reducing the rate of tear evaporation and enhancing tear film stability.5,6 MGD can lead to increased tear evaporation, tear film instability, eye irritation, inflammation, and ocular surface disease.5,7,8 The reported rates of MGD prevalence based on clinical signs range from 38% to 68% in populations over the age of 40 years. 3
To soften or liquefy the meibum and facilitate its outflow in an effort to improve lipid profile and allow a more uniform tear dispersion, conventional MGD treatment comprises lid hygiene, eyelid warming, and meibum expression.9–11 Other treatments depending on severity and indications can include artificial tears including lipid-containing eye drops for symptomatic relief, supplementation with omega-3 fatty acids, topical antibiotics, oral tetracyclines, and anti-inflammatory and immunomodulatory agents.9,10
The shortcomings of warm compress therapy include the challenge in maintaining adequate temperature and the need for continuing therapy on a daily basis, which can impact patient compliance.12,13 With advancement in medical devices, in-office devices have been used for effective treatment of MGD, including those that deliver controlled localized heat and pressure.14–17 Tixel device (Novoxel, Netanya, Israel) is a device based on thermomechanical action with a very brief exposure time; the Tixel device has been widely used in the treatment of skin indications.18–24 An observation of improved DED symptoms following fractional skin treatment for periorbital wrinkles with Tixel in older patients prompted pilot studies to characterize the effect of Tixel in improving signs and symptoms of DED with encouraging results.25,26
Recently, the safety and effectiveness of the Tixel device in the management of MGD were demonstrated in a randomized controlled multicenter pivotal trial of subjects with MGD, assigned to treatment with either Tixel or LipiFlow Thermal Pulsation System (Johnson & Johnson Vision, Milpitas, CA). 27 The results at 1- and 3-month follow-up showed comparable safety and effectiveness to LipiFlow with noninferiority of change in TBUT and significant improvement in key clinical parameters from baseline. Following the study, the Tixel i® device has been 510(k)-cleared by the U.S. Food and Drug Administration (FDA) for the treatment of evaporative dry eye due to MGD. An observational extension study was planned in Tixel subjects to assess the longer term durability of treatment.
The objective of the extension study was to evaluate the durability of the clinical benefit out to 6 months in a subgroup of Tixel subjects who had shown an improvement in tear break-up time (TBUT) in at least 1 eye at either 1- or 3-month follow-up. The assessments of key clinical signs and symptoms included TBUT, meibomian gland score (MGS), Ocular Surface Disease Index (OSDI), and rate of adverse events (AEs).
Methods
Study design
This prospective, observational, single-arm study was a 3-month extension of an initial 3-month, prospective, randomized, masked, controlled, multicenter clinical trial whose results were previously reported. 27 The extension study was performed between April 2023 and September 2023. A subgroup of Tixel subjects from the initial study was enrolled in this extension study and was evaluated for the durability of the clinical benefit out to 6 months. The study involved 6-month follow-up of the initial Tixel treatment; there was no retreatment in the study.
All of the evaluations were performed at the same 5 clinical sites who took part in the initial pivotal study. This extension study adhered to the tenets of the Declaration of Helsinki and was approved by Advarra (Columbia, MD, USA), a central Institutional Review Board. Informed consent was obtained from all participants prior to study initiation, and Health Insurance Portability and Accountability Act regulations were followed. The study was registered at the U.S. clinical trials website (Clinicaltrials.gov Identifier NCT05162261).
Subjects
Subjects were originally screened for eligibility for inclusion in the main pivotal study based on the following criteria: aged 22 years or older with dry eye symptoms for the previous 3 or more months, reported use of lubricants for the previous 1 or more months, OSDI score between 23 and 79, TBUT <10 s in each eye, MGS ≤12 in each eye, and at least 15 glands in each lower eyelid that were expressible during slit lamp examination. The key criteria for exclusion from participation in the main pivotal study were the use of dry eye treatments (other than lubricants), contact lens wear within a prespecified period prior to the study and history of major dermatologic, systemic or ocular conditions, tattoos, permanent makeup, or irritated skin in the treatment area. Among the subjects who were assigned to treatment with Tixel in the randomized pivotal study, subjects were included in this extension study if they met the inclusion criteria of improvement in TBUT of 2.5 s or greater from baseline in at least 1 eye at 1- or 3-month follow-up visits and were available and willing to attend the 6-month follow-up visit.
Tixel device
Although there was no retreatment involved in the extension study, a brief description of the device and method used in the initial pivotal study is provided below. The Tixel device has a small tip with an area of 0.30 cm2 consisting of an array of 24 (6 × 4) evenly spaced pyramids. Each pyramid was 1.25 mm tall with a blunt apex of approximately 0.01 mm2 (Fig. 1). The parameters in the pivotal study included a single pulse duration, that is, time of contact between heated tip and skin, of 6 ms and tip protrusion distance of 400 µm.
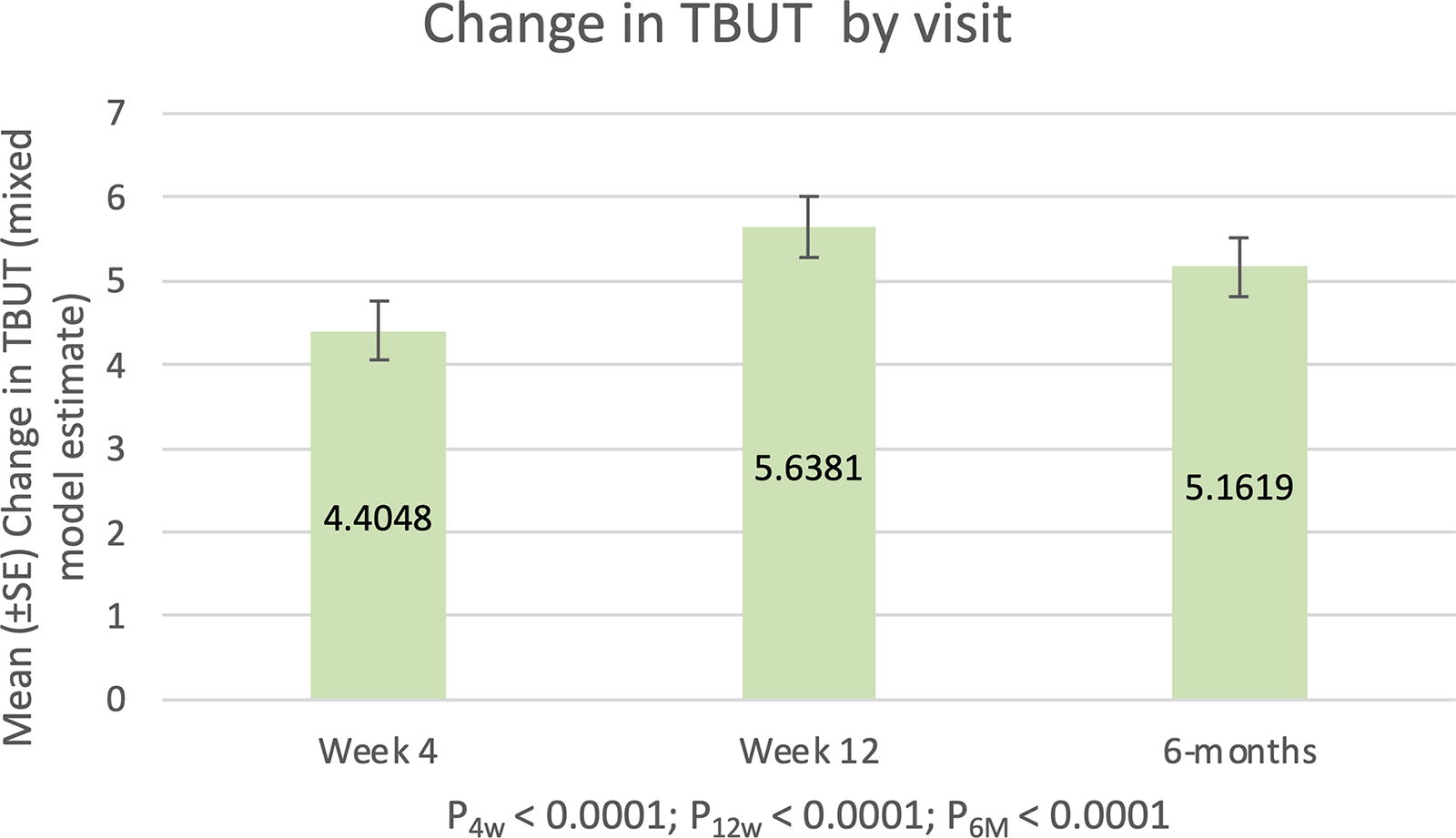
Change from baseline in LS mean TBUT from pre-treatment baseline through 6 months post-treatment with Tixel (N = 42 eyes of 21 subjects). These were estimated from a linear mixed-effects model, with subject as random effect, visit as repeated measure, and baseline score as covariate. FU, follow-up; LS, least-squares; SE, standard error; TBUT, tear break-up time.
The Tixel procedure involved 3 bilateral sessions at 2-week intervals. After both upper and lower eyelids were cleaned and anesthetized, the clinician delivered a row of 5 pulses near each lid margin in the nasal, medial, and temporal regions. A second row of 5 pulses was delivered to each eyelid, adjacent to the first row for a total of 20 pulses applied to the upper and lower eyelids. This was repeated for the contralateral eyelids.
Assessments
In the extension study, subjects were required to complete a 6-month follow-up visit in addition to the visits completed in the pivotal study, namely, baseline, Tixel treatment consisting of 3 sessions, and 1-month and 3-month follow-up post last treatment session. The assessments at the extension 6-month follow-up visit comprised all the assessments using the same methodology that were performed in the pivotal study at the 1- and 3-month follow-up visits. For the effectiveness measurements by examiner, 3 fluorescein TBUT measures were averaged, and MGS was assessed using the Meibomian Gland Evaluator (Johnson & Johnson Vision) to evaluate the quality of meibomian gland secretions on the lower eyelids. Five consecutive glands located in each of the temporal, central, and nasal regions along the lower eyelid margin were graded on a scale of 0–3 for the characteristics of the expressed gland contents, where 0 = no secretion, 1 = inspissated/filamentary secretion, 2 = cloudy liquid secretion, and 3 = clear liquid secretion. 28 The sum of the grades for all 15 glands determined the total MGS score (range 0–45); as such, the MGS score represents both the number of secreting meibomian glands and the quality of those secretions. In order to avoid iatrogenic ocular surface staining confounding any results, the evaluations were conducted in the following sequence: OSDI, refraction, Best Corrected Visual Acuity (BCVA) assessment, keratometry, slit lamp examination of anterior segment, lid margin abnormalities, eyelid margin assessment, TBUT, corneal fluorescein staining, conjunctival lissamine green staining, MGS, and intraocular pressure (IOP) measurement. Ocular AEs were to be recorded at each study visit.
Study endpoints
The effectiveness endpoints in the extension study were the change from baseline in TBUT, MGS, and subjective symptoms using the OSDI at the 6-month follow-up. Additionally, the change between observed TBUT, MGS, and OSDI at 3 months and 6 months is also evaluated. Safety was assessed via the recording of AEs, grading of ocular surface staining, and changes in IOP and BCVA.
Statistical analyses
Statistical analysis was performed using SAS version 9.4 (SAS Institute Inc., NC, USA). No sample size or power calculations were conducted due to the exploratory nature of this extension study.
For differences from baseline (Day 0), the P-values were the result of separate tests of the null hypothesis of mean difference = 0 for each follow-up of 4-week, 12-week, and 6-month visits. There were no corrections for multiplicity. For eye-level measurements (TBUT, MGS, corneal staining, and conjunctival staining), the P-values were from simple mixed-effects linear models (with baseline as covariate) that allow for within-subject correlation. For subject level measurements (OSDI scores), the P-values were from a paired t test. A P-value of 0.05 was the threshold for determining statistical significance.
Results
Subject disposition
Twenty-one subjects who were available to attend the 6-month follow-up following Tixel treatment in the pivotal study and met the inclusion criteria were enrolled in the extension study. The data analysis included 42 eyes of 21 subjects. The demographic and baseline characteristics are presented in Table 1. The age range of the subjects at baseline evaluation was 39–81 years, with a mean of 66.5 ± 9.9 years.
Demographics and Baseline Characteristics of the Study Population
MGD, meibomian gland dysfunction; MGS, meibomian gland score; OSDI, ocular surface disease index; SD, standard deviation; TBUT, tear break-up time.
Clinical outcomes
Table 2 displays the mean changes from baseline for effectiveness parameters of TBUT, MGS, and OSDI at post-treatment time points of 1 month, 3 months, and 6 months. Changes in safety parameters of ocular surface staining are also shown in Table 2.
Observed Changes from Baseline in Dry Eye Disease Parameters and Symptoms After Tixel Treatment at 1-, 3-, and 6-Month Follow-Ups
BL, baseline; CI, confidence interval; CFS, corneal fluorescein staining; CLS, conjunctival lissamine green staining; M, month; MGS, meibomian gland score; OSDI, Ocular Surface Disease Index; SD, standard deviation; TBUT, tear break-up time.
Effectiveness
There was significant improvement from baseline in TBUT after the Tixel treatment at the 6-month follow-up visit (P < 0.001). TBUT improved from 4.0 ± 1.5 s at baseline to 8.4 ± 3.2 s at the 1-month follow-up and 9.6 ± 4.9 s at the 3-month follow-up. At the 6-month follow-up, the mean TBUT was 9.2 ± 4.0 s, and the improvement in TBUT was retained with a significant mean change from baseline of 5.2 ± 3.8 s (Table 2). The mean TBUT change from baseline through 6 months post-treatment following the mixed-effects linear model is presented in Fig. 1.
Similar results of significant improvement from baseline were observed for MGS after the Tixel treatment at the 6-month follow-up visit (P < 0.001). Mean MGS of 6.6 ± 2.3 at baseline increased to 15.9 ± 9.0 at 1-month follow-up and to 22.7 ± 11.2 at the 3-month follow-up. At the 6-month follow-up, MGS continued to improve with a mean MGS of 24.8 ± 10.9, and a mean change from baseline of 18.2 ± 10.9 (Table 2). The mean values of MGS change from baseline through 6 months post-treatment following the mixed-effects linear model are displayed in Fig. 2.
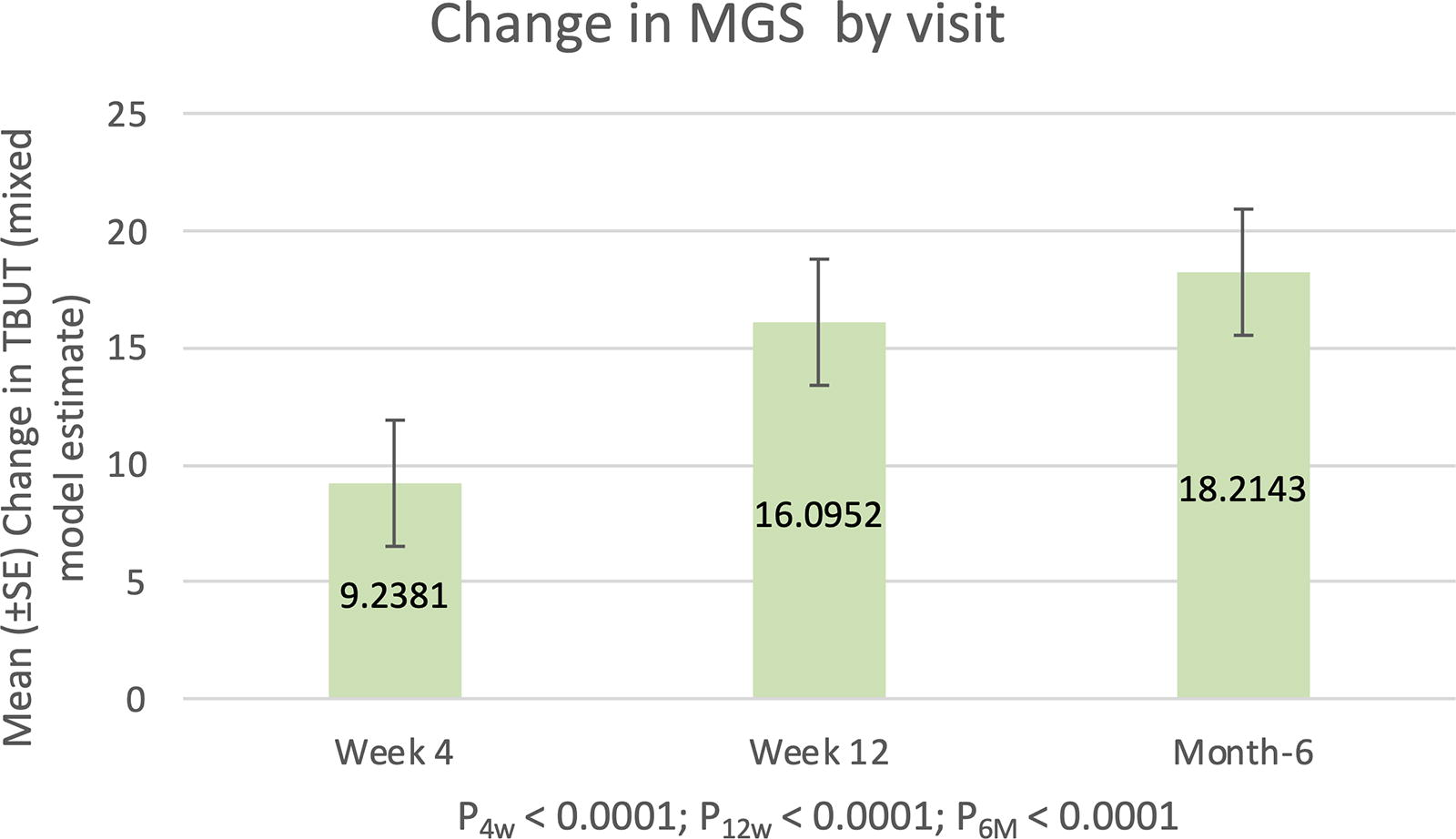
Change from baseline in LS mean MGS from pretreatment baseline through 6 months post-treatment with Tixel (N = 42 eyes of 21 subjects). These were estimated from a linear mixed-effects model, with subject as random effect, visit as repeated measure and baseline score as covariate. MGS, meibomian gland score.
Symptomatic improvement was also observed, as assessed by the OSDI questionnaire at baseline and at post-treatment follow-up visits. With a decrease in OSDI scores indicating improvement in symptoms, the OSDI score decreased from 46.2 ± 19.7 at baseline to 21.4 ± 18.8 at the 1-month and 19.3 ± 15.5 at the 3-month follow-up. At the 6-month follow-up, OSDI remained improved with a mean score of 21.9 ± 19.4, indicating a significant mean change from baseline of −24.3 ± 26.5 (P = 0.0004; Table 2). The mean OSDI scores from baseline through 6 months post-treatment are displayed in Fig. 3.
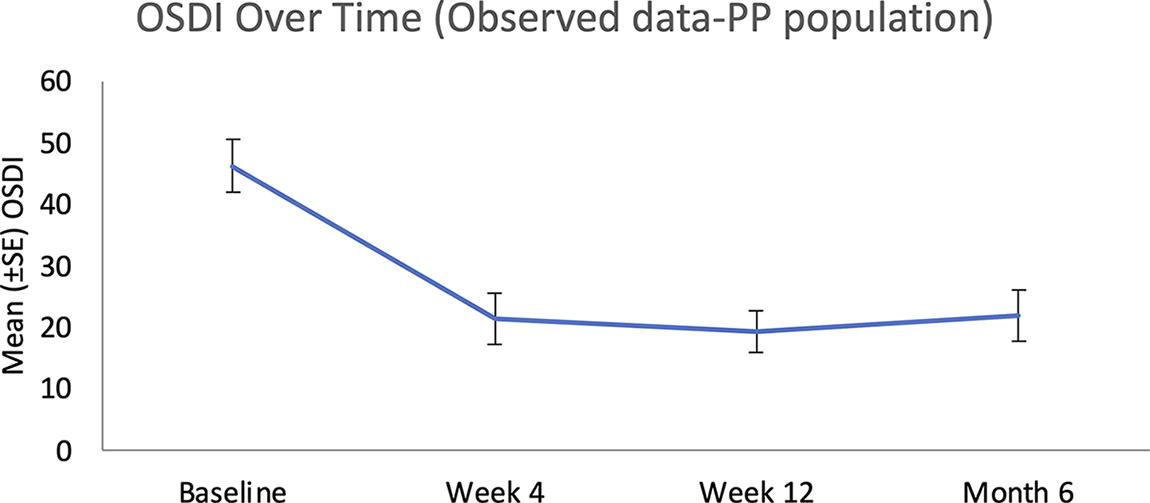
Mean (±SE) OSDI over time from pretreatment baseline through 6 months post-treatment with Tixel (N = 21 subjects). OSDI, Ocular Surface Disease Index.
There were no statistically significant changes between 3 and 6 months for TBUT, MGS, and OSDI (all P > 0.05).
Safety
No ocular AEs were reported during the extension study. Additionally, there was no significant change from baseline IOP at the 6-month follow-up visit. BCVA was stable, with 20/32 or better acuity in 95% of eyes at baseline (40 of 42 eyes) and 93% eyes at month 6 (39 of 42 eyes). No remarkable changes were noted from baseline in lid margin abnormality score, keratometry, or eyelid margin assessment.
Corneal and conjunctival staining
At baseline, mean corneal and conjunctival staining scores were 2.1 ± 1.8 and 2.0 ± 2.2, respectively. At the 1-month and 3-month follow-up visits, corneal staining averaged 0.6 ± 0.9 and 0.4 ± 0.6, respectively. At the 6-month follow-up visit, corneal staining scores remained low with an average of 0.6 ± 1.1 and a significant mean change from baseline of −1.5 ± 1.5 (P < 0.0001; Table 2). Conjunctival staining averaged 0.9 ± 1.2 and 0.7 ± 1.1 at the 1-month and 3-month follow-up visits, respectively. At 6-month follow-up, conjunctival staining increased to an average of 2.2 ± 3.2, which was not significantly different from baseline (P = 0.7470).
Discussion
The initial randomized controlled pivotal clinical trial demonstrated that the Tixel procedure was safe and effective through 3 months post last session of treatment. The primary purpose of this extension study was to evaluate the durability of the effect of Tixel treatment for MGD through a longer follow-up timepoint of 6 months post-Tixel treatment in a subcohort of subjects from the initial study. This was an observational study with no retreatment involved. The assessments in the extension study were the same as those in the initial study. The effectiveness assessments comprised of TBUT, MGS, and OSDI, whereas safety assessments included monitoring of related AEs, corneal staining, conjunctival staining, slit lamp biomicroscopy, IOP, and BCVA.
The results of the extension study showed that the significant improvements from baseline in TBUT, MGS, and OSDI in the initial study were retained at 6 months. Additionally, there was no significant difference between 3 and 6 months in these parameters, which further confirms the durability of treatment at 6 months. There were no related AEs and no other significant safety findings at 6 months.
The difference from extension studies of other in-office thermal based devices such as TearCare (Sight Sciences Inc, Menlo Park, CA, USA) and LipiFlow Thermal Pulsation System was in the observed maintenance of treatment effect to 6 months with Tixel for all parameters of TBUT, OSDI, and MGS in the subcohort of treated subjects. In the TearCare extension study, TBUT was the first measure to dip toward baseline values as early as 3 months post-treatment and continued to show a further drop by 6 months, initiating evaluation of retreatment at 7 months. 29 MGS and OSDI also showed a notable trend toward baseline at 6 months. 29 In the long-term evaluations of LipiFlow, similar to TearCare, peak improvement at 1 month and a general trend toward baseline at subsequent evaluations were observed for TBUT and OSDI. MGS continued to retain improvement with LipiFlow to 9 months.15,30 In this Tixel extension study, an improvement and maintenance of treatment effect was observed for TBUT and OSDI symptom score through 6 months post-treatment, whereas a continuing upward trend was noted for MGS at 6 months (Fig. 2), although not statistically significantly different from 3 months. The timepoints beyond 6 months that continue to demonstrate sustained treatment effect of Tixel or a significant dip toward baseline, as evaluated by objective and subjective measures of TBUT, MGS, and OSDI, would require investigation with a longer term study.
DED, including evaporative DED resulting from MGD, is an important public health concern, which impacts the patient’s ability to perform routine daily activities and reduces the quality of life. 3 Although warm compress is considered the first line of treatment, compliance issues of regular warm compress and the chronic nature of MGD add to the challenges of effectiveness and efficiency in achieving the desired treatment effect. In-office devices have demonstrated significantly greater effectiveness compared with conventional warm compress.14,31 Having multiple alternatives of in-office devices equips clinicians with options to tailor the treatment that offers not only demonstrable short-term improvement but also longer term durability. Given the increasing prevalence with age for DED, the treatment options that provide ease of use, greater duration of effect, and require lesser frequency of re-treatments are valuable in delivering effective treatment and better convenience for optimal quality of patient care.
Available in-office devices differ in aspects such as concurrent treatment of 1 or both eyelids, use of in-eye applicator, and duration of procedure. The Tixel treatment, which uses a very brief pulse duration for rapid heat transfer, is delivered in 3 sessions to both upper and lower eyelids. The procedure takes 2 min per session, does not involve contact with the ocular surface, and is not limited by the size of palpebral fornices.
The initial pivotal study upon which this extension study is based was designed as a randomized controlled trial with outcomes evaluated by masked assessors in a multicenter trial. The strength of the initial study in gathering data from multiple centers was retained in this study. The limitations of this extension study include a small sample size, a single-arm study, and the consequent use of unmasked assessor. Longer-term comparative studies with a larger sample size in a diverse population can contribute to further evidence of treatment response in different cohorts, taking into account the variability in natural disease progression. Longer-term data will also help to evaluate the durability of treatment effects beyond 6 months and assess need for repeat or alternate treatments. We acknowledge that the initial study and its extension have been funded by the manufacturer, as is the case for any new eyecare product. As the device has been 510(k)-cleared by the FDA for the treatment of evaporative dry eye due to MGD, future research initiated by independent users (clinicians) can be expected that will add to the current knowledge base.
Conclusion
The Tixel device, a thermomechanical system that has previously demonstrated safety and effectiveness to 3 months post-treatment in eyes with evaporative DED due to MGD, showed durability of treatment effectiveness to 6 months in a subset of eyes. The device safely and effectively maintains improvement in clinical signs of tear film stability, quality of glandular secretions, and ocular symptoms through 6 months and offers a valuable addition to the currently available treatment options for this indication.
Footnotes
Acknowledgments
The authors thank all participants in the study. Writing assistance in the preparation of this article was provided by Sandhya Subramaniam, MS, FAAO, from Regulatory Pathways Group, Inc. Support for this assistance was funded by Novoxel Ltd.
Authorship
All named authors meet the criteria for authorship recommended by the International Committee of Medical Journal Editors (ICMJE).
Authors’ Contributions
Concept and design: I.K.; Data collection: E.S., A.V., G.D.P., J.E., and G.J.B.; Writing—original draft preparation: I.K. and Y.A.G.; Writing—review and editing: E.S., A.V., G.D.P., J.E., and G.J.B.
Author Disclosure Statement
I.K. and Y.A.G. are employees of Novoxel. E.S., A.V., G.D.P., J.E., and G.J.B. have no competing interests.
Funding Information
The study and Rapid Service Fee were funded by Novoxel Ltd.
