Abstract
Introduction:
The lens’s metabolic demands are met through a continuous circulation of aqueous humor, encompassing a spectrum of components such as organic and inorganic ions, carbohydrates, glutathione, urea, amino acids, proteins, oxygen, carbon dioxide, and water. Metabolomics is a pivotal tool, offering an initial insight into the complexities of integrated metabolism. In this investigative study, we systematically scrutinize the composition of intraocular fluid in individuals afflicted with cataracts.
Methods:
The investigation involved a comprehensive analysis of aqueous humor samples from a cohort comprising 192 patients. These individuals were stratified by utilizing the SPONCS classification system, delineating distinct groups characterized by the hardness of cataracts. The analytical approach employed targeted quantitative metabolite analysis using HILIC-based liquid chromatography coupled with high-resolution mass spectrometric detection. The metabolomics data analysis was performed with MetaboAnalyst 5.0.
Results:
The results of the enrichment analysis have facilitated the inference that the discerned disparities among groups arise from disruptions in taurine and hypotaurine metabolism, variations in tryptophan metabolism, and modifications in mitochondrial beta-oxidation of short-chain saturated fatty acids and pyrimidine metabolism.
Conclusion:
A decline in taurine concentration precipitates diminished glutathione activity, prompting an elevated requirement for NAD+ and instigating tryptophan metabolism along the kynurenine pathway. Activation of this pathway is additionally prompted by interferon-gamma and UV radiation, leading to the induction of IDO. Concurrently, heightened mitochondrial beta-oxidation signifies a distinctive scenario in translocating fatty acids into the mitochondria, enhancing energy production.
Introduction
Approximately 95 million individuals across the globe are afflicted by cataracts. 1 The prevalence and absolute numerical count of individuals affected by blindness attributed to cataracts persist at elevated levels, notwithstanding a decreasing trend in incidence observed across various geographical regions worldwide. 2 The exclusive remedial approach for cataracts is surgical intervention, making it one of the most prevalent surgical procedures globally. 3 Comprehending the structural underpinnings of protein stability and interactions within the unimpaired ocular lens constitutes the pivotal path toward addressing the substantial medical and economic challenges of cataracts. 4
The lens is an intricately structured, transparent optical syncytial tissue whose primary function is to converge incoming light onto the retinal surface. Its transparency is a consequence of the composition of its constituent fiber cells, which consist of densely concentrated proteins. 5 A monolayer of epithelial cells is located in immediate proximity to the anterior lens capsule. These cells exhibit heightened metabolic activity and engage in the full spectrum of typical cellular functions, encompassing DNA, RNA, protein, and lipid biosynthesis. The mammalian lens is composed of three distinct categories of crystallin proteins, namely alpha (α), beta (β), and gamma (γ), each composed of several subunits. 6
The lens is characterized by the absence of both vascularization and innervation. The lens has developed an internal ion circulation mechanism intricately linked to fluid motion, establishing an internal microcirculatory system. This adaptation compensates for the absence of vascularization within the lens. 7 Consequently, the metabolic requirements of the lens are sustained through a continuous circulation of aqueous humor, which functions as a medium for eliminating metabolic waste products and transporting essential nutrients. 8
The aqueous humor is produced by the ciliary epithelium through the active transport of solutes, primarily sodium chloride, from the stroma to the posterior chamber of the eye, with the passive movement of water accompanying this process. 9 Aqueous humor formation involves three distinct mechanisms: diffusion, ultrafiltration, and active secretion. 8 The energy needed for active transport is generated by the hydrolysis of adenosine triphosphate to adenosine diphosphate. 10 This process is facilitated by the enzyme Na-K-ATPase, which is present in both pigmented and nonpigmented ciliary epithelial cells. Of notable pharmacological significance, Na-K-ATPase can be inhibited by various molecules, including cardiac glycosides, dinitrophenol, and acetazolamide. 11 The primary constituents of the aqueous humor include organic and inorganic ions, carbohydrates, glutathione, urea, amino acids, proteins, oxygen, carbon dioxide, and water. 12
While metabolic diseases are frequently associated with their characteristic metabolites (such as diabetes with glucose and cardiovascular disease with cholesterol), it is essential to recognize that biochemical pathways do not operate in isolation; instead, they are interconnected and influence each other. 13 Advancements in analytical technologies have reached a stage where most known endogenous metabolites can be quantified using one of several existing analytical platforms. 14 These technologies are integral to the swiftly evolving field of metabolomics, offering researchers an initial glimpse into the intricacies of integrated metabolism and, consequently, the profound impact of integrated metabolism on human health. To elucidate deviations in the levels of specific metabolites from the normal range and, more significantly, to forecast how to modify inputs, such as diet, drugs, and lifestyle, to achieve a desired metabolic alteration, an in-depth understanding of the functionality of pathways and their associated reactions is imperative. 15
Metabolomics leverages advanced analytical chemistry methodologies to facilitate the high-throughput characterization of metabolites from cells, organs, tissues, or biofluids. 16 The rapid expansion of metabolomics research has spurred a renewed fascination with metabolism and the pivotal roles that small-molecule metabolites assume in many biological processes. Consequently, conventional perspectives regarding metabolites, previously regarded as mere constituents of cellular structure or as cellular energy sources, are undergoing a profound transformation. Indeed, metabolites are increasingly recognized for their diverse and pivotal functions as signaling molecules, immune system regulators, endogenous toxins, and sensors responsive to environmental cues. 17
Not only are alterations in metabolism observable but also the tools to interpret these changes within the context of specific metabolic pathways are readily available. This is primarily because understanding biochemical pathways has been a central research objective in the life sciences for well over a century. 15
Methods
Study design
The investigation entailed the examination of aqueous humor specimens derived from a cohort comprising 192 patients who underwent cataract surgery at Pauls Stradins Clinical University Hospital. These patients were categorized into two groups based on the SPONCS 18 classification system, which differentiated between individuals with hard and softer cataracts. Patients with softer cataracts were categorized within stages 1 to 3, encompassing subcapsular cataracts with a clear nucleus, mild hardness, and moderate hardness. In contrast, harder cataracts were classified within stages 4–5, including advanced hardness and hypermature/Morgagnian cataracts. According to this approach, the study groups were categorized into 130 cases with soft cataracts and 62 with hard cataracts. Among the chosen participants, 70 were men, while 122 were women. The average age of individuals diagnosed with cataracts was 73.7 years, with a standard deviation of ± 9.29. The age range varied from a minimum of 50 years to a maximum of 94 years. It is noteworthy that patients with concomitant ocular pathologies, such as glaucoma, age-related macular degeneration, or diabetes, were not excluded from the study (Table 1).
Summary of Demographics and Ophthalmic Comorbidities Among Patients Included in the Study
The t-test was used for comparing age distributions, while the chi-squared test was employed for analyzing associations among categorical variables.
Before the commencement of the surgical procedure, a paracentesis was performed, followed by a biopsy of the aqueous humor employing a 27G needle. Depending on the quantity of available fluid, between 50 and 120 µL of aqueous humor were aspirated and subsequently transferred to Eppendorf tubes. These samples were then promptly frozen and transported to the Faculty of Materials Science and Applied Chemistry of Riga Technical University for storage and subsequent analysis.
The research conducted adhered to the Declaration of Helsinki. Notably, the study’s ethical considerations underwent meticulous scrutiny and obtained approval from the Medical Ethics Committee of Rigas Stradins University, as indicated by decision number 2-PEK-4/307/2023. Furthermore, the study received the necessary approval from the Pauls Stradins Clinical University Hospital.
Metabolite analysis
Metabolites were extracted using a methanol-based extraction protocol. Around 10 µL of the aqueous humor sample was transferred to an empty Eppendorf tube and mixed with 80 µL methanol and 10 µL isotopically labeled internal standard. Each sample was vortexed for 15 s and then centrifuged for 10 min at 10.00 RPM. The supernatant was transferred into an HPLC glass vial.
Targeted quantitative metabolite analysis was conducted using HILIC-based liquid chromatography combined with mass spectrometric detection employing a Thermo Orbitrap Exploris 120 mass spectrometer. An ACQUITY UPLC BEH Amide 1.7 μm 2.1 × 100 mm analytical column (Waters) was employed for chromatographic separation. The gradient elution was carried out using 0.15% formic acid and 10 mM ammonium formate in water as mobile phase A and a solution of 0.15% formic acid and 10 mM ammonium formate in 85% acetonitrile as mobile phase B. The initial conditions were set to 100% in mobile phase B. After 6 min, a 0.1 min gradient (6.0–6.1 min) was started, and the mobile phase B level was reduced to 94.1%. From 6.1 to 10 min, mobile phase B was set to 82.4%, and from 10 to 12 min mobile phase B was set to 70.6%. The column was then equilibrated for 6 min at initial conditions. The total analysis time was 18 min. The mobile phase flow rate was 0.4 mL/min, the injection volume was 2 μL, and the column temperature was 40°C. For MS detection, an Orbitrap Exploris 120 (Thermos Fisher Scientific) mass spectrometer was used. The MS analysis was performed in ESI positive and ESI negative modes using full scan detection; the scan range was set from 50 to 600 m/z, and the mass resolution was set to 60000. The ESI spray voltage was set to 3.5 kV in positive mode and 2.5 kV in negative mode; the gas heater temperature was set to 400°C; the capillary temperature was set to 350°C; the auxiliary gas flow rate was set to 12 arbitrary units; and the nebulizing gas flow rate was set to 50 arbitrary units. For quantitative analysis, seven-point calibration curves with internal standardization were used. Tracefinder 51.1 General Quan (Thermo Fisher Scientific) software was used for LC-MS data processing and quantification. Every reported metabolite was identified at level A 19 by using an authentic standard compound previously mapped to the analytical system. Detailed information about metabolite identification (RT, m/z, HMDB IDs) is provided in the Supplementary Table S1.
Metabolite data analysis
The metabolomics data analysis was performed with MetaboAnalyst 5.0. 20 Data normalization was implemented to ensure that metabolites of different magnitudes were reasonably analyzed. Specifically, the metabolite levels with different ranges were normalized by the median with a log (base 10) transformation. The metabolite levels were scaled by mean-centered and divided by the square root of the standard deviation of each variable. The statistical analysis was conducted with the normalized concentrations of each metabolite. The two-group comparison data were analyzed by an unpaired t-test using parametric tests. A one-way analysis of variance (ANOVA) followed by a Tukey’s comparison was used for statistical analysis. Enrichment analysis was applied based on SMPDB (the Small Molecule Pathway Database).
Results
The outcomes of the enrichment analysis have enabled us to infer that the observed distinctions among groups emanate from perturbations in taurine and hypotaurine metabolism, alterations in tryptophan metabolism, and mitochondrial beta-oxidation of short-chain saturated fatty acids.
By employing a targeted metabolomics approach, more than 100 metabolites were measured, with 54 of these compounds identified in the aqueous humor samples shown in Figure 1A. Noteworthy constituents encompassed amino acids, organic acids, acylcarnitines, nucleotides, and glucose. A discernible disparity in metabolite profiles was noted between soft and hard cataracts. Amino acids within aqueous humor samples obtained from individuals with hard cataracts exhibited reduced levels compared with those observed in the soft cataract cohorts. Conversely, constituents of the carnitine family, particularly octanoyl-carnitine, demonstrated heightened concentrations, as shown in Figure 1B. Notably, amino-n-butyric acid displayed a notable increase in concentration in hard cataracts (5.08 µM) relative to soft cataracts (1.01 µM), yielding a fold change of 7.82 and a highly significant p-value (P = 0.0003). Similarly, kynurenine and members of the carnitine family shown in Figure 1C, including octanoylcarnitine and butyrylcarnitine, exhibited significant elevations in hard cataracts, with P-values of 0.0036 and 0.0066, respectively. The fold change for octanoylcarnitine was particularly pronounced at 29.21, indicative of a substantial metabolic alteration associated with cataract hardness. Although the fold change for tryptophan was modest (FC = 0.40), its levels still manifested a statistically significant difference (P = 0.0456), albeit with a smaller effect size.
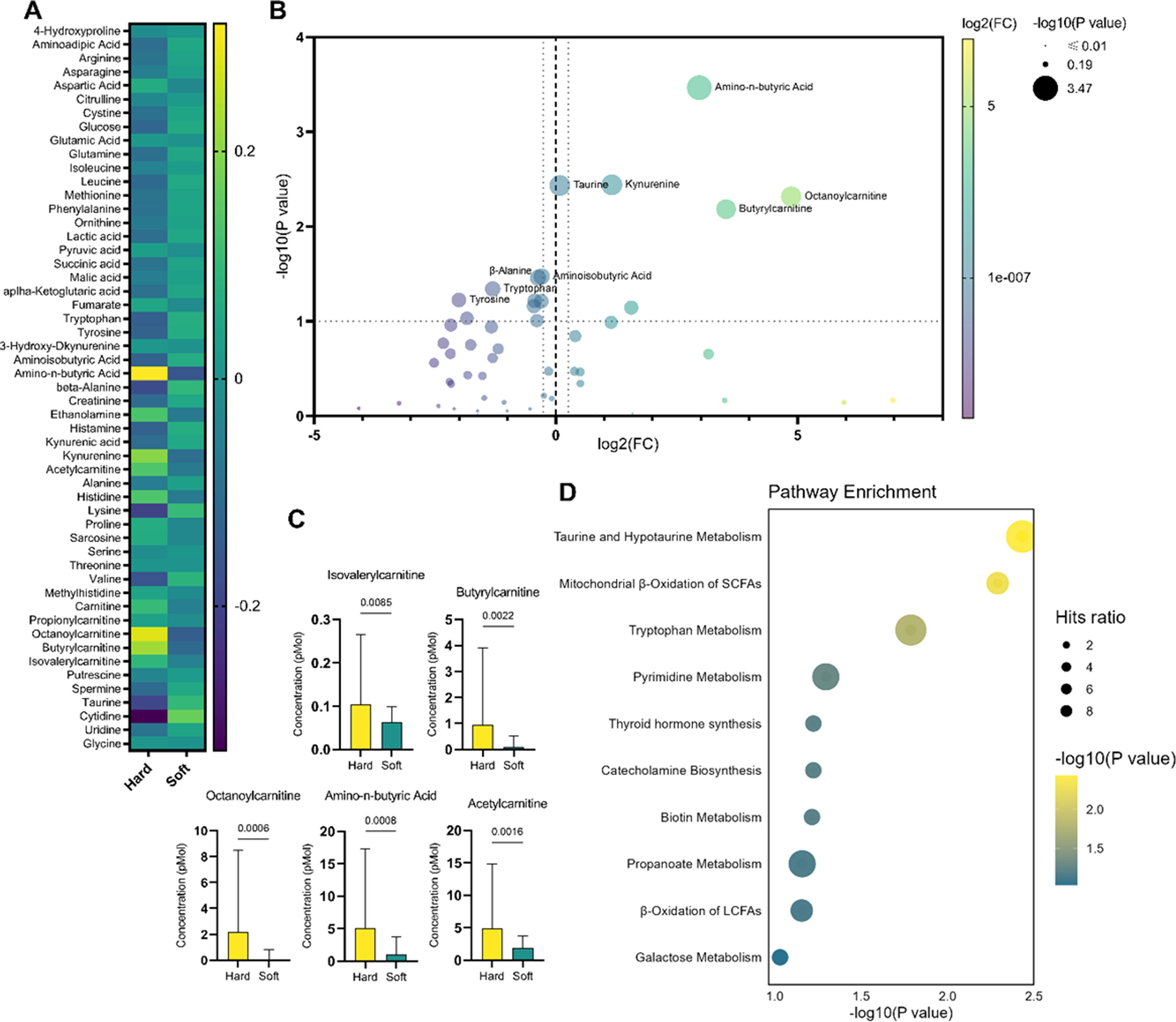
Metabolomic analysis of differences between aqueous humor samples from soft and hard cataracts.
Pathway enrichment analysis, a computational technique in molecular biology, was applied to discern the most significantly altered metabolic pathways. This method identifies biological functions overrepresented in a group of metabolites, surpassing what would be expected by chance alone. The subsequent ranking of these functions is based on their relevance within the analyzed metabolite set. The outcome of the pathway enrichment analysis unveiled notable impacts on taurine metabolism, fatty acid β-oxidation, and pyrimidine metabolism, as shown in Figure 1D.
One hit (taurine) out of 14 compounds was identified in the context of taurine and hypotaurine metabolism. For mitochondrial beta-oxidation of short-chain saturated fatty acids, two hits (carnitine and octanoylcarnitine) were detected out of 27 total compounds. Five hits shown in Figure 2A were identified out of 59 compounds in tryptophan metabolism. These results further contribute to understanding the nuanced alterations in metabolic pathways associated with the observed distinctions between hard and soft cataracts.
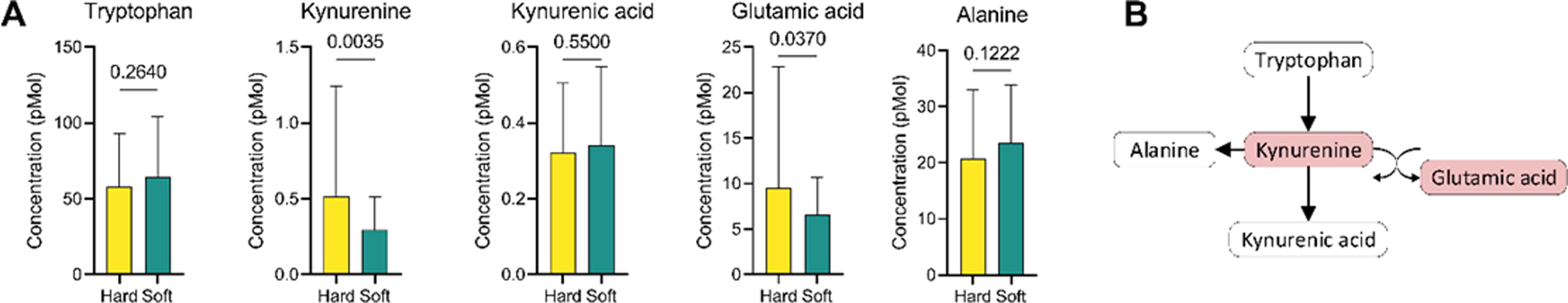
Tryptophan metabolism pathway alteration.
In the tryptophan metabolism pathway, an oxoglutaric acid gains an amino group from kynurenine to form a glutamic acid, as shown in Figure 2B. The lower level of kynurenine may result in a lower level of glutamic acid in the soft cataract group.
Discussion
Osmolytes constitute a category of low-molecular-weight organic compounds that notably influence the characteristics of biological fluids. This class of organic molecules is crucial in modulating osmotic pressure and upholding cellular homeostasis across diverse organisms, particularly in response to environmental stressors. 21 Osmolytes also play a role in impeding the accumulation of reactive oxygen species (ROS), mitigating DNA and RNA damage, and inhibiting transcription processes. Numerous osmolytes, including sugars, amino acids, and polyols, enhance protein stability by facilitating their compaction into native structures. This mechanism rescues defective proteins, averting intracellular toxicity. 22
Taurine, a pivotal organic osmolyte within mammalian cells, undergoes notable dynamics in response to osmotic cell swelling. 23 Specifically, there is an augmentation in taurine release facilitated through a volume-sensitive taurine efflux pathway, concomitant with a reduction in active taurine uptake mediated by the taurine-specific transporter TauT. The intricacies of cellular signaling cascades, the profile of second messengers, the activation patterns of specific transporters, and the subsequent temporal dynamics governing the readjustment of osmolyte content and cellular volume exhibit considerable variability across distinct cell types. 24
Taurine manifests diverse physiological functions, encompassing conjugation with bile acids, osmoregulation, antioxidation, facilitation of neuronal development, and membrane stabilization. 25 The antioxidative efficacy of taurine hinges on its ability to mitigate mitochondrial oxidative stress. 26 When examining the clinical complications associated with diabetes, a nexus becomes apparent between perturbed taurine metabolism and the onset of cellular dysfunctions. These dysfunctions contribute significantly to the clinical complications observed in diabetes. 27
Numerous investigations have been conducted to elucidate the beneficial ramifications of taurine supplementation and its facilitation of intricate physiological processes. Taurine is observed to exert a stimulatory effect on both protein synthesis and the proliferation of C2C12 myoblast cells, a phenomenon mediated through the PI3K-ARID4B-mTOR pathway. This delineation provides a nuanced comprehension of the regulatory mechanisms by which taurine modulates anabolic processes within muscle cells. 28 Taurine demonstrates inhibitory effects on methamphetamine-induced autophagic processes by activating mTOR. In the context of METH-induced damage to PC12 cells, taurine protects against ROS production, apoptosis, and autophagy. This protective effect is, at least in part, mediated through the mTOR signaling pathway. 29 The attenuation of acrylamide-induced apoptosis in vivo by taurine through a PI3K/AKT-dependent pathway offers novel insights into the molecular mechanisms underlying taurine was observed to diminish phosphorylation levels of ERK1/2 without affecting the phosphorylation of adenosine monophosphate-activated protein kinase or ribosomal protein S6 kinase associated with the MTORC1 pathway. These findings imply that taurine may facilitate the nuclear translocation of transcription factor EB through ERK1/2 activation, thereby expediting the autophagic process. 30
The central significance of glutathione (GSH) within the crystalline lens is underscored by its pivotal role as an antioxidant, protecting against the oxidation of lens proteins. 31 Noteworthy is the observed progressive reduction in GSH levels within normal lenses as age advances, diminishing from approximately 5 μmol per gram of lens to 3 μmol per gram of lens. 32 The lens epithelium and outer cortex are believed to play a crucial role in furnishing the predominant share of lens GSH through de novo synthesis, facilitated by a continuous influx of constituent amino acids from the aqueous humor. GSH is a tripeptide composed of cysteine, glutamate, and glycine. 33 In addition, extracellular GSH recycling from the gamma-glutamyl cycle contributes to this GSH pool. However, under impaired de novo synthesis conditions, such as in the aging human lens with reduced GSH levels, compensatory mechanisms come into play, indicating the lens’s capacity to uptake GSH from the adjacent ocular tissues. 34 A deficiency in GSH elicits an AMPK (AMP-activated protein kinase)-independent induction of autophagy in germ cells, functioning as an adaptive stress response. 35 The depletion of GSH initiates a cascade of events that may ultimately culminate in cellular demise. 36 Taurine facilitates the uptake of GSH in lens epithelial cells. 37
Age-related nuclear cataract is associated with a decline in GSH levels, specifically within the central region of the lens. 38 Taurine, a vital antioxidant in lens tissue, experiences a reduction in concentration as individuals age, notwithstanding its initial abundance in lenses during early life. The depletion of GSH and the dietary supplementation of taurine emerge as potentially significant factors in impeding the progression of cataract formation. 39 Epidemiological data indicate an association between elevated levels of taurine and a reduced incidence of specific types of cataracts. 40 Mechanistically, taurine demonstrates a multifaceted impact on cellular processes, including the reduction of cellular senescence, protection against telomerase deficiency, suppression of mitochondrial dysfunction, mitigation of DNA damage, and attenuating inflammation. Consequently, taurine deficiency is posited as a potential contributor to the aging process, as its reversal has been shown to extend the health span of worms, rodents, and primates. 41
The detoxification of free radicals necessitates reduced GSH, a process contingent upon the availability of nicotinamide adenine dinucleotide phosphate (NADPH). 42 Substantially diminished nicotinamide adenine dinucleotide (NAD+) is concurrent with aging. Augmenting NAD+ levels has been demonstrated to enhance health and lifespan in various animal models. The biosynthesis of NAD+ emerges as a prospective target for the prevention and treatment of age-associated diseases. 43 NAD+ is an indispensable cofactor in numerous enzyme-catalyzed reactions, encompassing alcohol, lactate, and amino acid metabolism. In addition, NAD+ is pivotal as an electron transporter, driving oxidative phosphorylation and facilitating ATP production. 44 The traditional function of NAD+ involves serving as a coenzyme that catalyzes cellular redox reactions, transitioning into its reduced form, NADH. This pivotal role manifests in fundamental metabolic processes, including glycolysis, fatty acid beta-oxidation, and the tricarboxylic acid cycle. 45 NAD kinase facilitates the phosphorylation of NAD+ to generate NADPH, employing adenosine triphosphate as the phosphoryl donor. 46
There is a growing consensus that the levels of NAD+ experience a decline at the cellular, tissue/organ, and organismal levels throughout aging. This reduction in NAD+ levels influences the activities of enzymes that consume NAD+, contributing to a diverse array of age-associated pathophysiologies. 47
As the aging process ensues and concentrations of NAD+ decline, there is an increased activation of de novo synthesis. NAD+ can be synthesized de novo through two primary pathways: the kynurenine pathway, starting from
The accumulation of kynurenine with age has been observed in various organs and tissues. Increased levels of kynurenine relative to tryptophan are correlated with frailty and heightened age-related mortality. Moreover, the kynurenine pathway is implicated in age-associated diseases, including inflammation, neurodegeneration, and immunosenescence. 52 The kynurenine pathway is involved in acute and chronic skin damage induced by ultraviolet radiation. 53 Elevated levels of kynurenine and neopterin have been associated with more advanced cataracts. 54 Reduced levels of tryptophan and kynurenine have been identified in the plasma of individuals with Parkinson’s disease. 55 The heightened levels of kynurenine noted during the aging process may play a role in the observed bone fragility among older people. 52
Interferon-gamma (IFN-y) and UV (ultraviolet) radiation stimulate the expression of indoleamine 2,3-dioxygenase (IDO), an enzyme crucial in the catabolism of tryptophan, by releasing various cytokines, including interleukin 1. IDO converts tryptophan and other indole derivatives into kynurenine. 56 The Kynurenine–Tryptophan (Kyn/Trp) ratio serves as an index for assessing the activity of IDO. 57 IDO-mediated catabolism of tryptophan not only inhibits the immunoregulatory kinases, mTOR, and PKC-Θ, but also induces autophagy. 58
Dysregulated tryptophan catabolism and subsequent depletion of NAD+ contribute to increased fatty acid oxidation by IFN-y in coronary endothelial cells. 59 IDO contributes to cellular survival and proliferation by augmenting free fatty acid oxidation, supplying the necessary energy for these processes. 60 Anoxia, followed by subsequent reoxygenation, induces the upregulation of IDO. Consequently, increased IOD activity culminates in tryptophan depletion and triggers the activation of the GCN2 (general control nonderepressible 2) kinase pathway. 61 GCN2 functions as a molecular sensor in T cells, enabling them to detect and respond to the conditions established by IDO. 62 Amino acids and insulin collaboratively influence translation processes by activating mTOR, AMPK, and GCN2 transduction pathways. 63
Acylcarnitines are emerging as a pivotal group of biomolecules in metabolomics, given the distinctive acylcarnitine profiles observed in a growing spectrum of diseases and nutritional conditions. 64 Long-chain acylcarnitines play dual roles, functioning as transporters in oxidative catabolism and as modulators of various physiological and pathophysiological processes. Their involvement extends to crucial aspects such as cardiac electrophysiology, insulin signaling, cellular stress responses, and regulating inflammatory pathways. 65
The most significant metabolites are those from the carnitine family involved in carnitine metabolism. Octanoylcarnitine, butyrylcarnitine, propionylcarnitine, amino-n-butyric acid, and isovalerylcarnitine are all upregulated in hard cataracts, indicating a different transportation situation of fatty acids into the mitochondria for energy production. The lens of the eye is avascular; energy requirements are primarily met through anaerobic glycolysis. 66 Elevated levels of octanoyl-carnitine have been observed to be increased in patients’ plasma with pseudoexfoliation syndrome. 67 In patients with age-related macular degeneration, there is an observed enrichment in the metabolic pathway associated with the oxidation of branched-chain fatty acids and the synthesis of carnitine. 68 At the same time, carnitine has antioxidant properties, and any disruption in its metabolism may affect the lens’s ability to defend against oxidative damage. 69 This may also play a role in the cataract process. The regulation of ROS will become fragile and sensitive due to the anaerobic environment. Such regulation is also easily destroyed in the case of abnormal fatty acid oxidation. 70
Conclusion
While investigations into intraocular fluid metabolomics are still in their nascent stages, it is evident that they hold the promise of providing valuable insights into cataract progression. Our study provides evidence of specific metabolic distinctions in aqueous humor samples from soft and hard cataracts. We identified the significant role of taurine and hypotaurine metabolism, tryptophan metabolism, and mitochondrial beta-oxidation of short-chain saturated fatty acids in the development of cataracts. Considering these findings, the primary objective of intervention should center on taurine supplementation, aiming to impede the advancement of the disease. Although a definitive method for reversing the effects of cataracts remains elusive, arresting the disease’s progression promises to enable individuals to lead lives with their natural lenses intact. Considering the progression of the disease, additional supplementation with NAD+ precursors should be contemplated for patients aiming to arrest its advancement. This could lower an increased tryptophan metabolism through the kynurenine pathway. Furthermore, the activation of this pathway is prompted by interferon-gamma and UV radiation, resulting in the induction of IDO. A reduced energy demand would diminish the heightened mitochondrial beta-oxidation, which signifies a distinctive process in the translocation of fatty acids into the mitochondria, ultimately contributing to the augmentation of the energy production process. Such an attenuation would halt the progression of this sequence of events, ultimately preventing the culmination of mTOR inhibition and the activation of the Sir2 pathway. Commercially available and ready-to-use eye drops, formulated with taurine and NAD+ precursors, provide a convenient option for individuals seeking to incorporate these beneficial compounds for ocular health.
We clearly demonstrated that aqueous humor analysis is an emerging method for unraveling the processes within the eye’s anterior chamber. This approach extends beyond cataracts, potentially enhancing our understanding of disease progression in conditions such as glaucoma, pseudoexfoliation, endothelial dystrophies, and numerous others. The findings of this study lay the foundation for a more efficient pharmacological target for preventing and treating cataracts. The potential of metabolomics to leverage the accumulated knowledge of biochemistry toward achieving personalized and preventive healthcare is undeniably profound.
Footnotes
Authors’ Contributions
A.Z.: Conceptualization (equal), Formal analysis (equal), Investigation (equal), Methodology(equal), Project administration (equal), Data collection (equal), and Writing—review & editing (equal). J.V.: Conceptualization (equal), Data collection (equal), Formal analysis (equal), Investigation (equal), and Review & editing (equal). K.K.: Formal analysis (equal), Investigation (equal), Data collection (equal), Resources (equal), and Review & editing (equal). J.F.: Data collection (equal), Formal analysis (equal), and Review & editing (equal). G.L.: Conceptualization (equal), Formal analysis (equal), Investigation (equal), Methodology (equal), Project administration (equal), Data collection (equal), Resources (equal), and Review & editing (equal).
Data Availability Statement
We acknowledge and appreciate the Journal of Ocular Pharmacology and Therapeutics’ commitment to promoting data sharing and transparency in research. The data supporting the results presented in our article have been deposited in an appropriate repository. A comprehensive data accessibility statement, along with the corresponding repository link, is provided to facilitate access and verification of the findings. This ensures compliance with the Journal of Ocular Pharmacology and Therapeutics’ guidelines, and we encourage readers and fellow researchers to utilize and explore the available data for a deeper understanding of the study outcomes.
Author Disclosure Statement
All authors declare no conflicts of interest in relation to this work.
Funding Information
No funding was received for this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
