Abstract
Purpose:
Dry eye disease (DED) is classified as aqueous deficient, evaporative, or mixed. We investigated the therapeutic effect of the novel anti-inflammatory drug phosphosulindac (PS) in rabbit models of DED encompassing its pathogenesis, and its transition to chronicity.
Methods:
We treated three rabbit models of DED with PS (hydrogel formulation) or vehicle topically applied 1 × /day. We induced aqueous-deficient DED (acute and chronic) by injecting Concanavalin A into lacrimal glands; evaporative DED by injecting into the upper eyelid inactivated Mycobacterium tuberculosis in complete Freund's adjuvant; and mixed DED through desiccative stress, induced by holding open the eye for 3 h. We determined corneal sensitivity, tear break-up time (TBUT), Schirmer's tear test (STT), tear osmolality, and fluorescein staining of the ocular surface.
Results:
PS reversed all abnormal DED parameters. In acute DED, PS dose dependently normalized corneal sensitivity and tear osmolality; and improved TBUT, STT, and fluorescein staining. PS normalized corneal sensitivity and improved all other parameters in chronic aqueous-deficient DED. In evaporative DED, PS normalized corneal sensitivity and improved TBUT and fluorescein staining (osmolality and STT were not significantly changed in this model). In the desiccative stress model, PS improved TBUT and fluorescein staining but had no effect on STT or tear osmolality.
Conclusions:
PS rapidly reversed almost all DED parameters in its three subtypes. The normalization of the suppressed corneal sensitivity suggests the possibility of marked symptomatic relief by PS. The hydrogel formulation allows once-daily dosing. PS merits further development as a potential treatment for DED.
Introduction
Dry eye disease (DED) is a prevalent multifactorial disease of the ocular surface characterized by loss of homeostasis of the tear film, and accompanied by ocular symptoms (reviewed in Clayton 1 ). DED is classified into the aqueous-deficient and evaporative subtypes, whereas a third sizable subgroup shares features of both and is classified as mixed.
Several etiologies of DED promote an unstable and hyperosmolar tear film leading the vicious cycle of DED (reviewed in Wei and Asbell 2 and Pflugfelde and de Paiva 3 ). The key participants in this vicious cycle include ocular surface desiccation stress (DS); tear hyperosmolarity (HO); ocular surface inflammation; and barrier disruption by lysis of epithelial tight junctions, accelerating apoptotic cell death. Ample evidence has substantiated this vicious cycle (reviewed in Pflugfelde and de Paiva 3 ).
Stress signaling triggered by DS and HO leads to the nonspecific innate immune response, followed by the slower, but more specific adaptive immune response, in which CD4+ T cells damage the ocular surface. These epithelial changes further destabilize the tear film, amplify inflammation, and propagate the vicious cycle of DED. To “break the cycle” has been proposed as a means of controlling DED. 4
Aside from artificial tears, which only provide brief symptomatic relief, cyclosporine and lifitegrast dominate the treatment of DED. Both have significant limitations, including side effects, lack of an analgesic effect, and high cost.5–7 We are developing the anti-inflammatory phosphosulindac as a therapeutic agent for DED. An initial study using a rabbit model of aqueous-deficient DED indicated the therapeutic potential of this small molecule administered three times daily formulated in nanoparticles at concentrations of 2.2%–2.6%. 8
The development of a successful treatment for DED should address the spectrum of its clinical subtypes, use the lowest possible drug concentrations, and have a convenient dosing schedule. We studied, therefore, the efficacy of phosphosulindac (PS) in evaporative, desiccative, and hyperosmolar rabbit models of DED. We also used a novel formulation of PS allowing once-a-day dosing, and a drug concentration <10% of those previously used.
The rabbit was selected because its eye globe size and its ocular surface carboxylesterase levels are close to those of the human eye 9 ; PS is hydrolyzed by esterases, and rabbit eyes do not significantly inactivate it. 10 We employed the following models of DED: An aqueous-deficient model induced by injecting concanavalin A into the periorbital lacrimal glands 11 ; a Meibomian gland dysfunction (MGD) model induced by injecting into the eyelids complete Freund's adjuvant (CFA) 12 ; and an acute desiccative stress model, which permits the study of the acute stages of evaporative DED. These models encompass the evolution of the disease from its earliest acute manifestations on the ocular surface to chronic DED. Thus, collectively they provided the opportunity to assess the effect of PS on the entire pathophysiological spectrum of DED.
Our data demonstrate a significant therapeutic effect of PS in DED, encompassing its main subtypes, acute and chronic disease, as well as both the signs and symptoms of this diverse and prevalent clinical entity.
Methods
Reagents and study drug
Concanavalin A, fluorescein, and most routine chemicals were purchased from Sigma-Aldrich (St. Louis, MO). CFA containing heat-inactivated Mycobacterium tuberculosis, strain H37Ra was purchased from Difco Laboratories, (Detroit, MI).
PS (OXT-328) was a gift from Medicon Pharmaceuticals, Inc., (Setauket, NY). PS was formulated as a hydrogel following standard protocols. Briefly, Carbopol 980 was dissolved into water and the pH was adjusted to about 7.0 to allow gelation. PS and the other excipients (polyvinyl alcohol, Vitamin E TPGS, hydoxypropyl-β-cyclodextrin, Tween 80, and mannitol) were then added. The pH was adjusted to 7.4 ± 0.2 and the osmolality to 280–320 mOsm/kg H2O. The final concentration of PS was 0.2% (w/v), except for the dose–response studies in which several concentrations of PS were used (0.05%, 0.1%, 0.2%, and 0.4%).
Animal studies
All animal studies were approved by the appropriate Institutional Animal Care and Use Committee of Stony Brook University and followed the guidelines of the Association for Research in Vision and Ophthalmology Statement for the Use of Animals in Ophthalmic and Vision Research.
Animals
We used New Zealand White (NZW; Charles River Laboratories, Kingston, NY) and Dutch Belted Black (DBB; Convance Inc., New York, NY) rabbits, about 10 months of age, without ocular diseases. Rabbits, singly housed in a room whose temperature (70 ± 5°F) and humidity (40% ± 5%) were strictly controlled, were acclimated for a minimum of 2 weeks before study. The rabbits had unlimited access to water and standard rabbit chow. The nictitating membranes of the rabbits were removed at least 2 weeks before experiments. After sedation with acepromazine (1 mg/kg), the nictitating membrane was infiltrated with 1% lidocaine with 1:100,000 epinephrine and cut off with scissors close to its base.13,14
Induction of DED
We employed the three rabbit models of DED, summarized below.
Dacryoadenitis model
DED was induced by injecting ConA under ultrasound guidance into all periorbital lacrimal glands of rabbits anesthetized with inhaled isoflurane. 11 Acute DED was induced with a single injection of ConA on day 1; for chronic DED, ConA injections were repeated on days 7 and 14. Both eyes of each rabbit were topically treated with a single 40-μL drop of PS hydrogel or vehicle at the time of injection and once per day thereafter for up to 5 (acute model) or 18 (chronic model) days.
Evaporative model
Eight microliters of complete Freud's adjuvant (CFA) were injected into the nasal, central, and temporal thirds of the upper eyelid margin of both eyes of NZW rabbits anesthetized with inhaled isoflurane. 12 After the injections, rabbits were treated daily with PS or vehicle as above for 14 consecutive days.
Desiccative stress model
These studies were performed in a room maintained at 22–25°C and 45% ± 10% humidity. 15 DBB rabbits, anesthetized by a combination of ketamine and xylazine, were subjected to desiccative stress by holding both their eyes open with a speculum for 3 h. A single 50-μL drop of 0.2% PS hydrogel or vehicle was applied topically to each eye 5 min before initiation of desiccative stress. The speculum was removed, and the eyes were blinked three to five times. Following that, parameters of DED was determined in each eye.
DED parameters
Following already reported methodology,8,9 we determined, in all animal models, the following parameters at baseline and as indicated in Results. Animals were randomized into study groups using a random number generator producing similar baseline levels of each parameter outlined below. The investigators conducting the study were blinded to the identity of each group.
Tear osmolarity
Tear osmolarity was measured with the TearLab. Osmolarity test (TearLab, Escondido, CA) in tears was sampled at the lower junction of the palpebral and bulbar conjunctiva following the manufacturer's instructions.
The tear break-up time
The tear break-up time (TBUT) was determined by applying a 25 μL drop of 0.2% fluorescein over the eye and observing the precorneal tear film under blue light. The time taken to develop black dots, lines, or obvious disruption of the fluorescein film was measured for up to 1 min. If the break-up was not seen by 1 min, the observation was halted and tear break-up time (TBUT) was recorded as 60 s, even if actually longer.
Fluorescein staining of the ocular surface space
Fluorescein staining was performed by instilling 20 μL 2% fluorescein solution on the ocular surface. 16 After removing excess fluorescein, the cornea and the superior bulbar conjunctiva were photographed with a digital camera under blue light. The extent of fluorescein staining of the cornea and superior conjunctiva was scored using a modified NEI scoring method. The cornea was divided into five sections, with the superior conjunctiva regarded as one section. Each section was scored from 0 (absent) to 3 (severe) based on the amount, size, and punctate epithelial erosions.17,18
The Schirmer's tear test
The Schirmer's tear test was performed in eyes anesthetized with 1% preservative-free lidocaine. A Weck-Cel® surgical sponge (Beaver-Visitec International, Waltham, MA) was placed into the lower fornix to dry the eye. Color Bar Schirmer strips (Eagle Vision, Memphis, TN) were placed in the midportion of the lower lid, and tear production was recorded as the length of moistened strip at 5 min.
Corneal sensitivity
Corneal sensitivity was measured with the Cochet–Bonnet Esthesiometer (Luneau, France) in a quiet examination room and without anesthesia. 19 For esthesiometry,8,20,21 each rabbit was placed in a restraining bag 16 h after the last dose of PS and before any sedation. The nylon filament was applied to the central cornea, its most sensitive area. Corneal touch threshold testing started at the full filament length of 6 cm, and the filament was incrementally retracted by 0.5 cm until a positive response (full blink) was noted in 3–5 attempts. The length of filament simulating a positive response was the corneal sensitivity score.
Statistical analyses
Data were examined for normality using normal probability plots and Kolmogorov test statistics. Except for ocular surface staining, outcomes were sufficiently normal for parametric analysis with residual plots from each analysis confirming model fit. Dose-dependent effects of PS on acute DED were compared between groups using one-way analysis of variance (ANOVA). Because the primary comparisons of interest were each dose versus vehicle controls, Dunnett's multiple comparisons procedure 22 was employed. The acute and chronic effects of PS on tear osmolality in DED was analyzed using repeated measures ANOVA, with time as a within-subjects factor, followed by Tukey's multiple comparisons procedure. 23 In these models, the best-fitting variance–covariance structure was selected based on information criteria.
The effect of PS on evaporative DED and desiccative stress was analyzed using one-way ANOVA followed by Tukey's procedure for multiple comparisons. Owing to its distribution, all comparisons of ocular surface staining were made using the nonparametric Wilcoxon rank-sum test with each pairwise comparison adjusted for Type I error using Hochberg's multiple comparisons procedure. 24 All data were analyzed using SAS, version 9.4 (Cary, NC).
Results
The effect of PS on acute and chronic aqueous-deficient DED
To study aqueous-deficient DED, we used the ConA-based rabbit model by titrating the number of ConA injection, and we generated either acute or chronic DED. 11 The former captures the early stages of the disease while the latter recapitulates key features of its routine clinical presentation when patients seek medical attention. We have recently documented the similarity between this model and human DED.
Initially, we evaluated the dose-dependent effect of PS on acute DED. Compared with vehicle controls, PS 0.05% and 0.1% had no statistically significant effect on any of the DED parameters (Figs. 1 and 2 and Table 1). An effect was clearly established at PS 0.2% where it appeared to plateau.
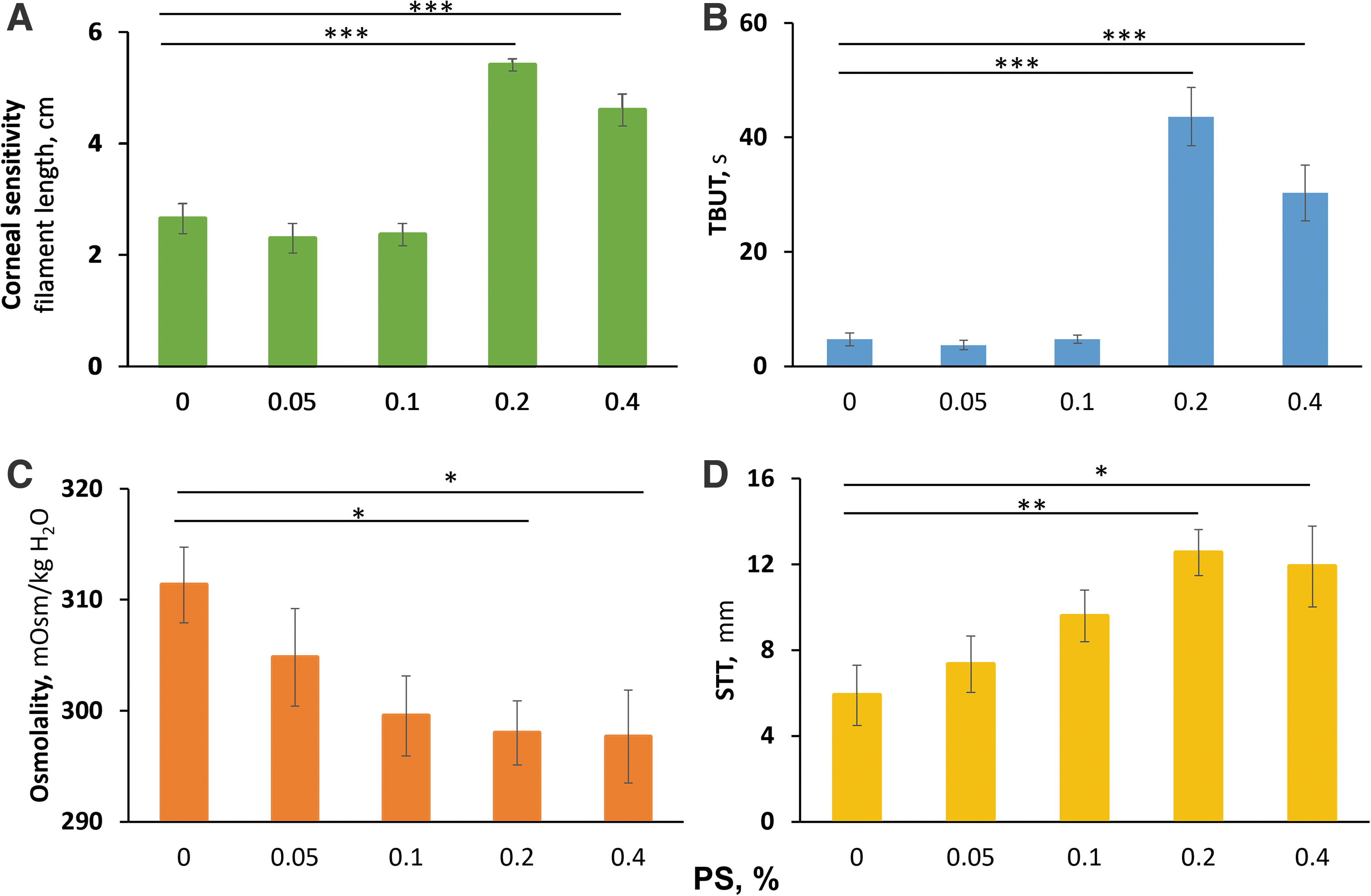
The dose-dependent effects of PS in acute DED. DED was induced in NZW rabbits by a single injection of ConA to the periorbital lacrimal glands. The DED parameters shown here
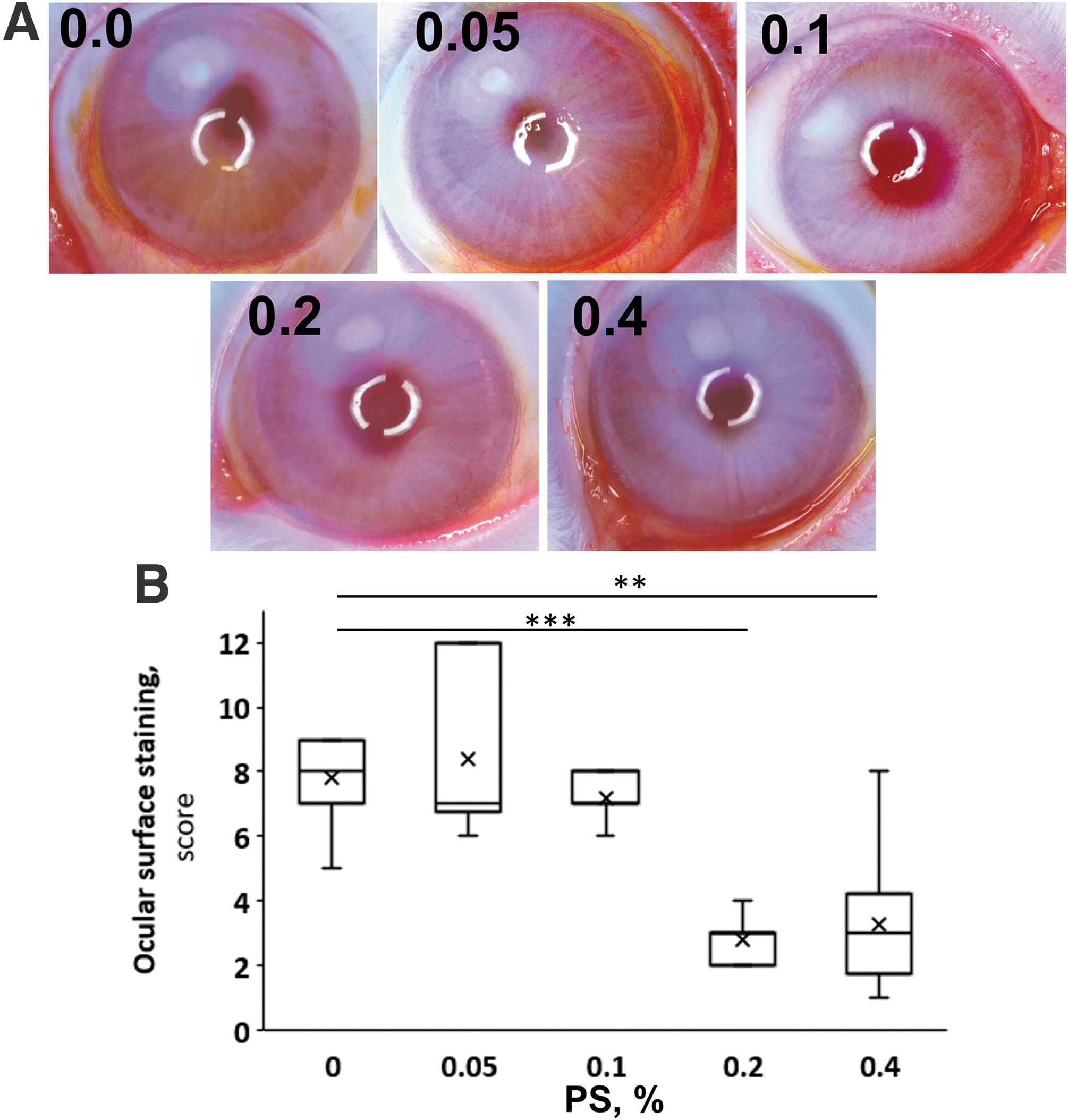
The dose-dependent effect of PS on ocular surface staining in acute DED. The rabbits reported in this figure are the same as in Fig. 1. The ocular surface was stained with fluorescein and scored as in Methods.
The Effect of Phosphosulindac on Dry Eye Disease Parameters in Acute Dry Eye Disease
The following comparisons to the corresponding baseline were statistically significant: f versus a, P < 0.001; j versus a, P < 0.001; g versus b, P < 001; l versus b P < 0.001; k versus c, P < 0.05; p versus c, P < 0.05; h versus d, P < 0.01; m versus d, P < 0.001; i versus e, P < 0.001; n versus e, P < 0.01.
PS, phosphosulindac; SEM, standard error of the mean; STT, Schirmer's tear test; TBUT, tear break-up time.
The 0.2% PS concentration was uniformly efficacious significantly improving all five parameters as follows: TBUT: 4.7 ± 1.1 versus 43.6 ± 5.1 s (vehicle vs. PS 0.2%; mean ± standard error of the mean for these and all subsequent values), representing a 9.3-fold increase; osmolality: 311.3 ± 3.4 versus 298.0 ± 2.9 mOsm/kg H2O, normalizing it; Schirmer's tear test (STT): 5.9 ± 1.4 versus 12.5 ± 1.1 mm, doubling it; corneal sensitivity: 2.7 ± 0.3 versus 5.4 ± 0.1 cm, nearly normalizing it (normal value = 6 filament cm); and ocular surface staining: 7.8 ± 0.4 versus 2.8 ± 0.3, also normalizing it (normal value = 2.5 ± 0.3). Remarkably, as can be seen in the figures and the table, doubling the PS concentration to 0.4% brought no additional improvement to these values. Consistent with previous experience with this compound, there was a sharp transition from no or minimal efficacy to maximal efficacy.
Hyperosmolality plays a key role in DED.25,26 Therefore, we monitored tear osmolality frequently before and at 2, 4, 6, and 24 h and on day 4 (Fig. 3A). From the normal baseline value of 278.8 ± 2.8 mOsm/kgH2O, tear osmolality increased rapidly reaching its peak value at 4 h (321.25 ± 15.3 mOsm/kg H2O). By 6 h the rabbit eyes became edematous, probably accounting for the decreased osmolality for the next 20 h. As the edema of the eye resolved, tear osmolality increased again, being 319.25 ± 9.6 mOsm/kgH2O on day 4. Compared with the vehicle group, PS 0.2% significantly decreased tear osmolality on day 4, but not at 4 h (Fig. 3B).
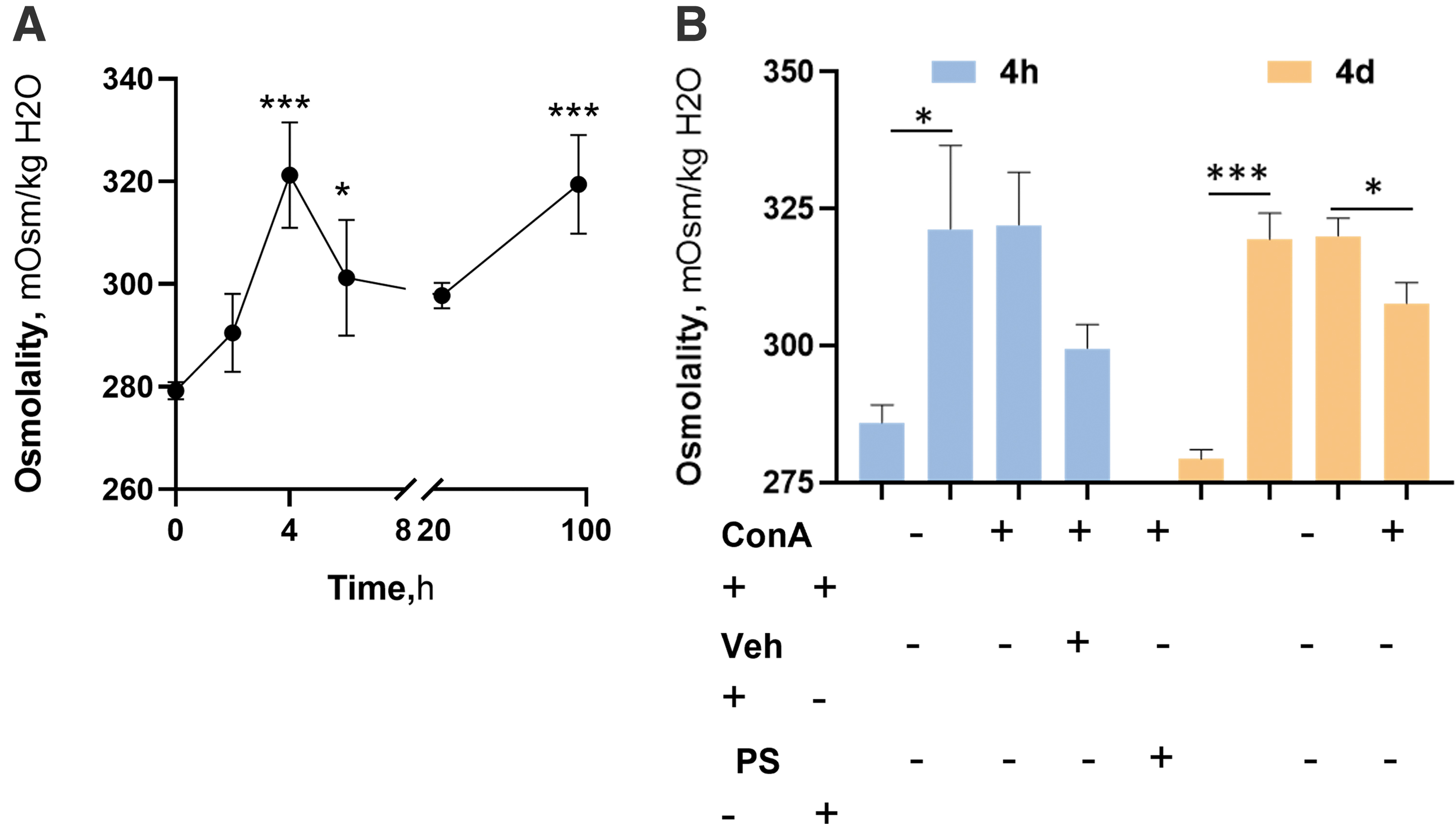
The effect of 0.2% PS on tear osmolality in acute DED. The rabbits reported in this figure are the same as in Fig. 1. Tear osmolality was determined at the indicated time points. Most rabbits showed chemosis starting at 6 h and continuing to day 2, likely in response to the ConA injection.
To evaluate the effect of PS on the clinically more relevant chronic DED, we used the rabbit model that is based on repeated ConA injections. PS 0.2% had its stronger effects on corneal sensitivity and TBUT, normalizing the former (Fig. 4 and Table 2). In particular, as expected, DED markedly suppressed corneal sensitivity decreasing it by 52% and 38% on days 5 and 17, respectively. In contrast to vehicle treatment that had no appreciable effect on corneal sensitivity, PS 0.2% markedly improved it by day 5 and normalized it by day 17.
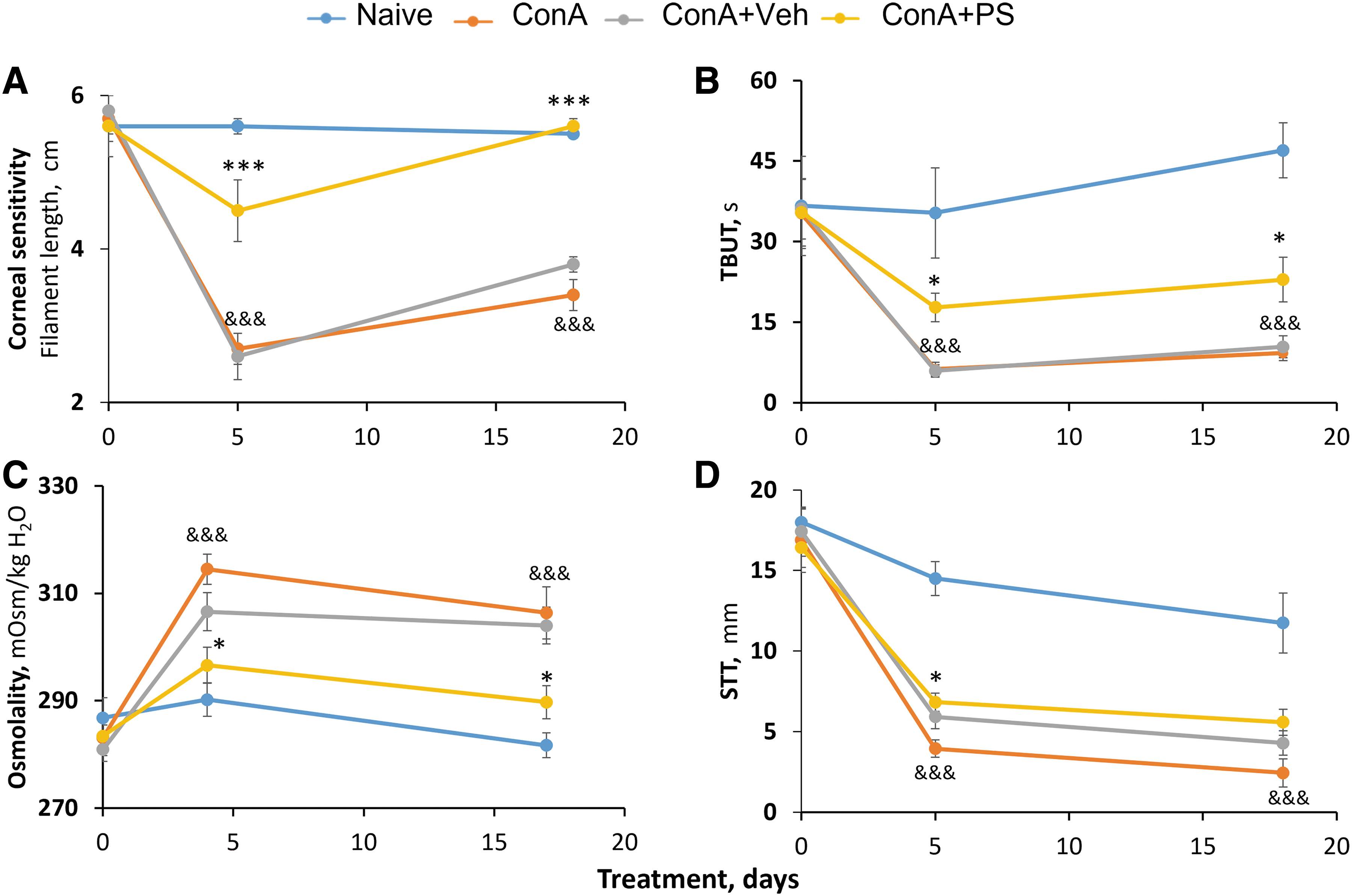
The effect of 0.2% PS on chronic DED. DED was induced in NZW rabbits by 3 weekly injections of ConA to the periorbital lacrimal glands; the first was on day 0. The DED parameters shown here
The Effect of 0.2% Phosphosulindac on Dry Eye Disease Parameters in Chronic Dry Eye Disease
The following comparisons to the corresponding vehicle group were statistically significant: h versus a, P < 0.001; i versus b, P < 0.001; j versus c, P < 0.05; k versus d, P < 0.05; l versus e, P < 0.05; p versus f, P < 0.05; m versus g, P < 0.05.
DED, dry eye disease.
A similar, but quantitatively less pronounced effect was noted on TBUT and tear osmolality that were suppressed by DED compared with naive control animals. PS 0.2% improved tear osmolality and TBUT to nearly the same degree. Tear osmolality, on day 4 decreased from 306.6 ± 3.5 mOsm/kg H2O to 296.6 ± 3.3 mOsm/kg H2O (P < 0.05) and on day 17 from 304.0 ± 3.4 mOsm/kg H2O to 289.8 ± 3.1 mOsm/kg H2O (P < 0.05). TBUT on day 5 nearly tripled from 6.3 ± 1.3 s to 17.8 ± 4.2 s, (P < 0.05) and on day 18 it more than doubled from 9.3 ± 1.4 s to 22.9 ± 4.2 s, (P < 0.05) In addition, PS 0.2% markedly improved STT to 6.8 ± 0.6 mm on day 5, while vehicle treatment had no significant effect on it.
The effect of PS on evaporative DED
In this model, following the injection of Complete Freud's Adjuvant, the injected upper eyelids become edematous by day 2, peaking at about day 7 and significantly subsiding by day 14. DED is evident on day 7 by reduced TBUT and increased corneal staining compared with naive animals, although corneal sensitivity remained intact (osmolality and STT were difficult to determine).
Corneal sensitivity was suppressed in CFA-treated animals compared with naive. On day 14, PS 0.2% restored it to normal while vehicle had no significant effect on it (Figs. 5 and 6 and Table 3). At the same time, TBUT was markedly (86%) suppressed in CFA-treated rabbits compared with naive, and treatment with PS 0.2% improved it significantly, more than doubling it (from 3.3 ± 1.3 s to 15.8 ± 3.1 s; P < 0.01), whereas vehicle had no effect on it.
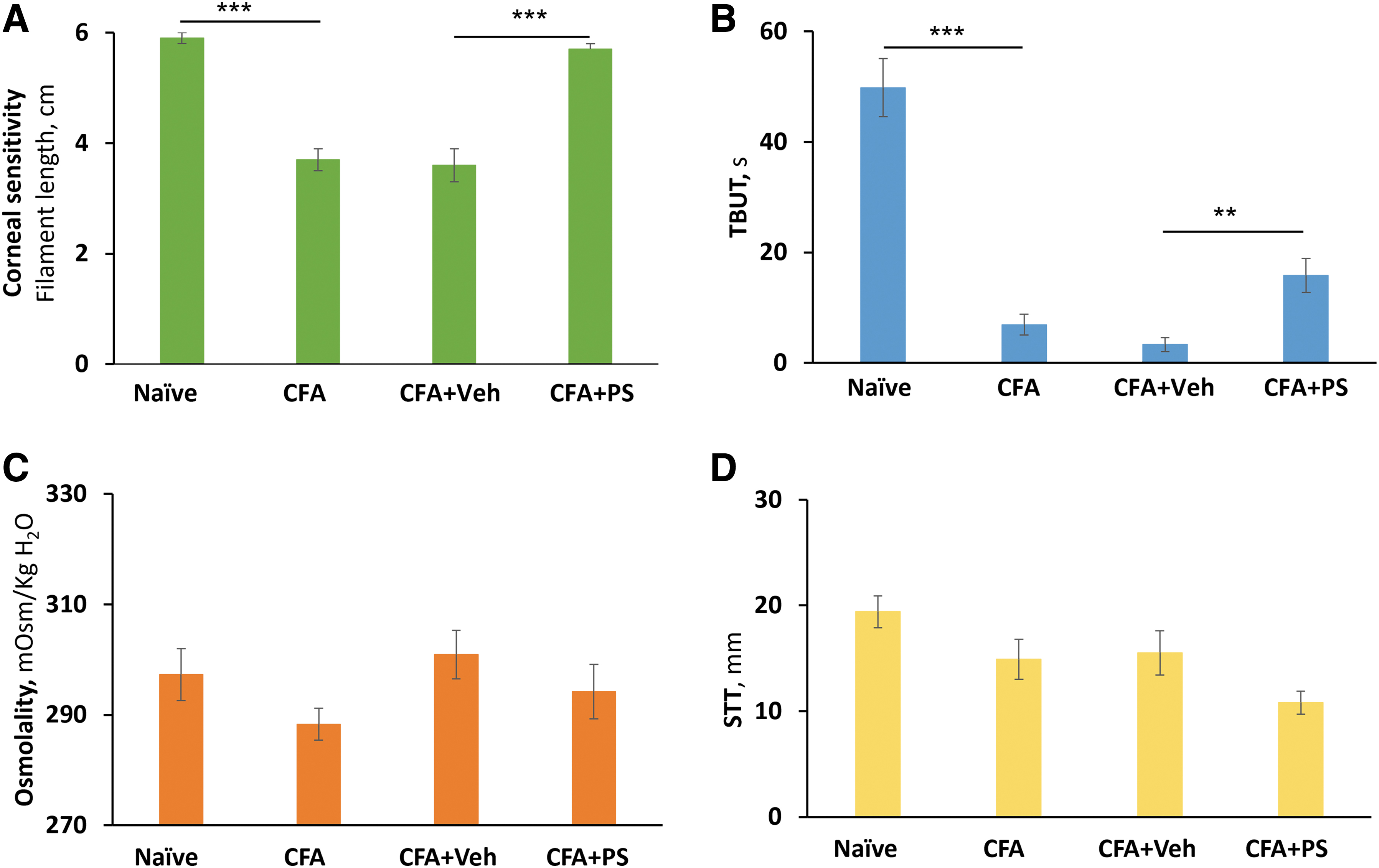
The effect of 0.2% PS on evaporative DED. DED was induced in NZW rabbits by CFA 3 injections to the upper eyelid on day 0. The DED parameters shown here
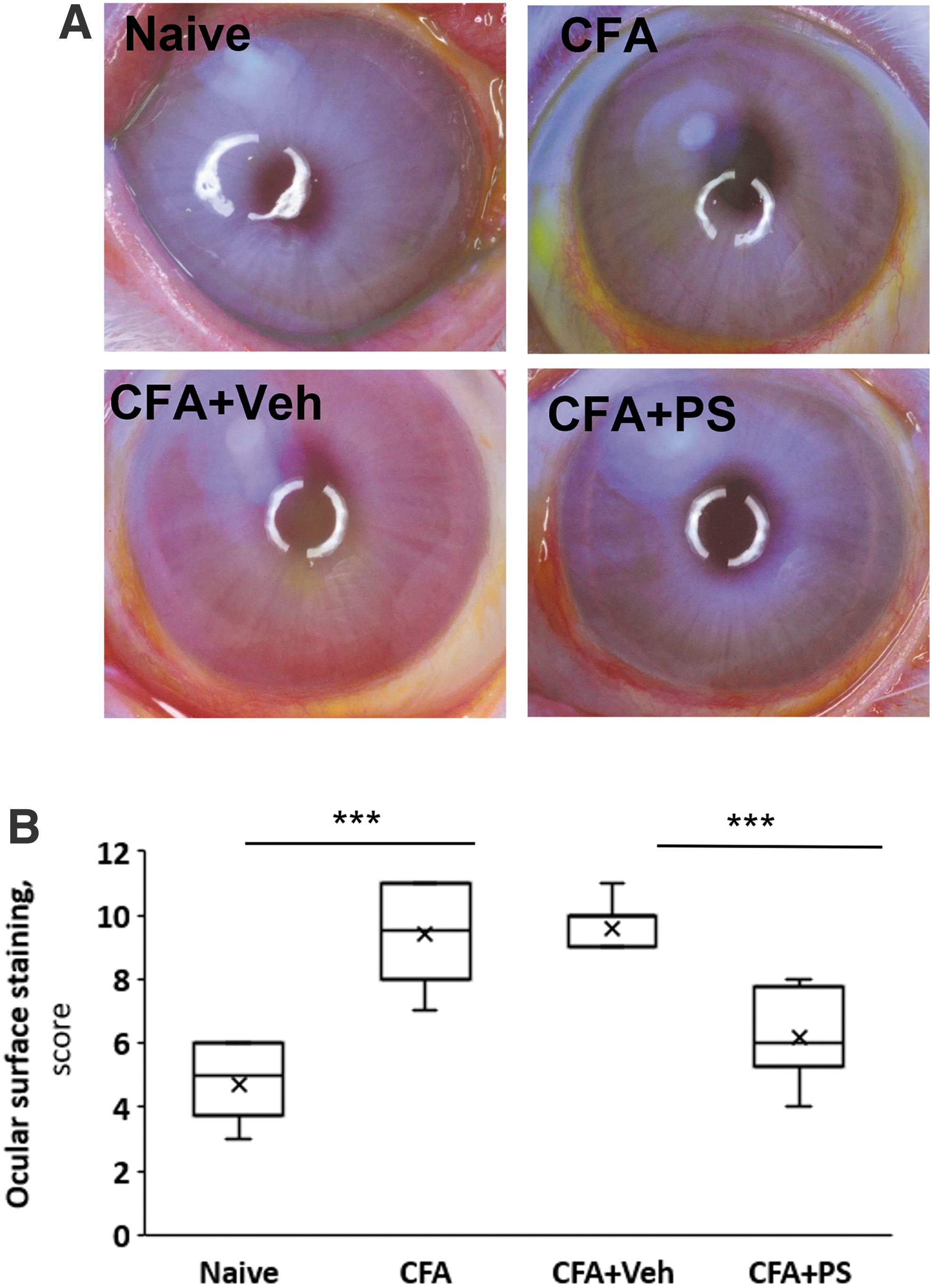
The effect of 0.2% PS on ocular surface staining in evaporative DED. The rabbits reported in this figure are the same as in Fig. 5. The ocular surface was stained with fluorescein and scored as in Methods.
The Effect of 0.2% Phosphosulindac on Dry Eye Disease Parameters in Evaporative Dry Eye Disease and in Desiccation
The following comparisons to the corresponding vehicle group were statistically significant: a versus e, P < 0.001; f versus b, P < 0.01; g versus c, P < 0.001; h versus g, P < 0.05; i versus k, P < 0.05.
The tear osmolality and STT remained essentially unaffected by any of the treatments, including the injection of CFA. CFA-treatment doubled corneal staining from 4.7 ± 0.4 to 9.4 ± 0.5 ocular surface staining score (P < 0.001). Treatment with PS 0.2% markedly decreased ocular surface staining from a score of 9.4 ± 0.5 to 6.2 ± 0.4, P < 0.001, a change that was significantly compared with both the CFA and the vehicle (9.6 ± 0.4) groups (P < 0.001 for both).
The effect of PS on the ocular effects of DS
Desiccation is considered one of the initiating factors of DED in its vicious cycle. 3 To assess whether PS inhibits the contribution of ocular surface desiccation to this disease, we used an animal model that captures the early stages of desiccation. Thus, rabbits were pretreated with PS 0.2% applied topically to the eyes as eye drops 5 min before the initiation of desiccation and their response to PS was assessed 3 h later.
Within 3 h, desiccation induced two profound changes to the eye, compared with baseline values (determined before the application of PS): it markedly increased the ocular staining score (fivefold), indicating significant damage to the surface epithelia, and decreased TBUT by almost 99% indicating major changes in the stability of the tear film. The suppression of STT was also significant, but less pronounced. Interestingly, the tear osmolality was increase (from 286.6 ± 2.9 to 304.5 ± 9.0 mOsm/kg H2O) but this change did not reach statistical significance.
Pretreatment of these rabbits with PS 0.2% 5 min before the initiation of desiccation had a strong effect on two out of the four parameters that we determined (Figs. 7 and 8 and Table 3). Of note, corneal sensitivity could not be reliably assayed in these rabbits as they were under deep anesthesia. Specifically, compared with baseline TBUT improved, increasing over sevenfold (from 1.6 ± 0.4 s to 7.2 ± 2.1 s; P < 0.05), while ocular surface staining was reduced by 36% (from 12.5 ± 0.7 to 9.6 ± 0.7; P < 0.05); in both cases, the effect of PS differed significantly from the effect of vehicle. PS 0.2% had no effect on STT or osmolality, being essentially identical to the effect of vehicle for both.
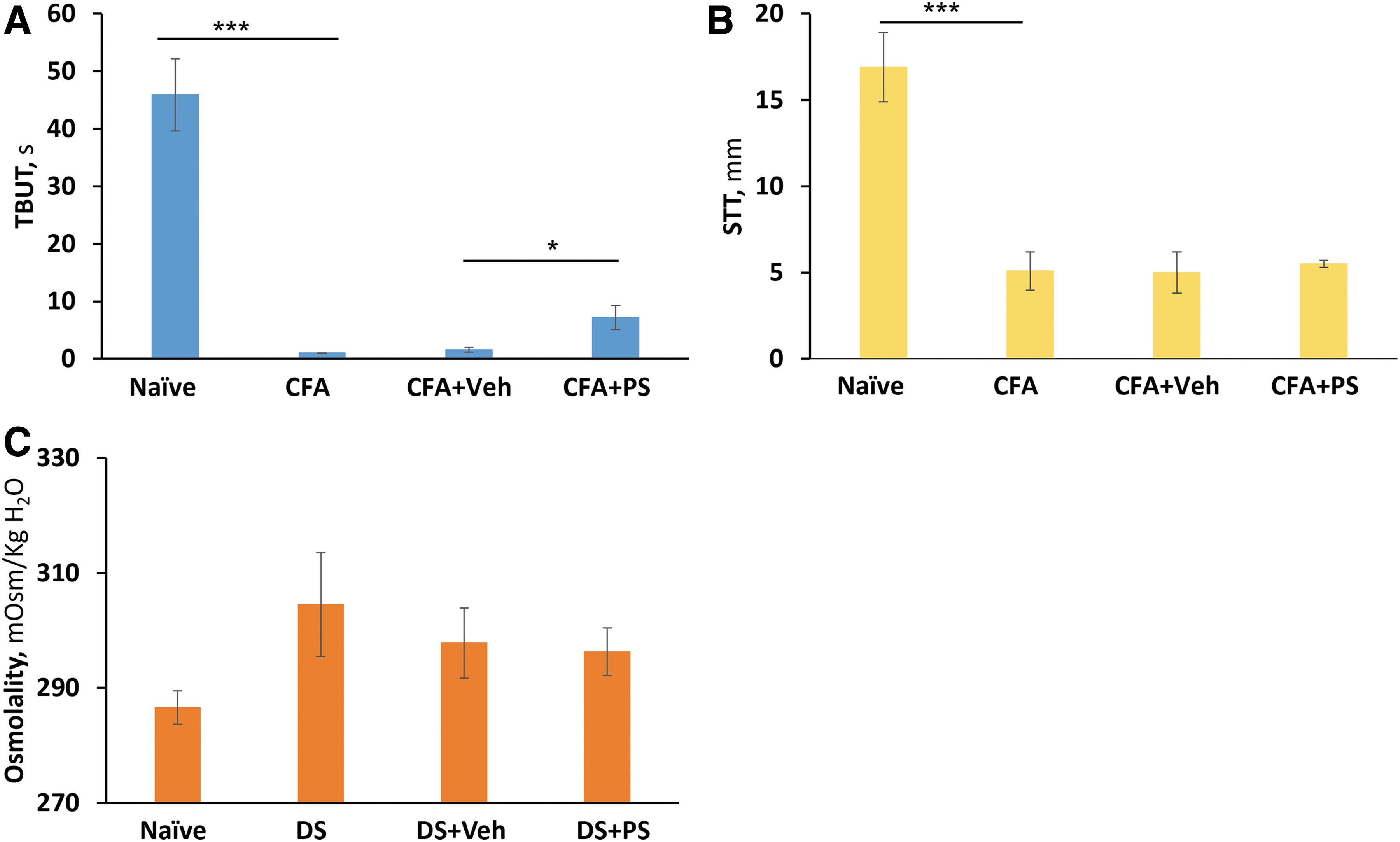
The effect of 0.2% PS on desiccative stress. DS was induced in anesthetized DBB rabbits by holding open both their eyes with a speculum for 3 h. Rabbits were pretreated with PS 0.2% applied topically 5 min before initiation of desiccative stress. DED parameters
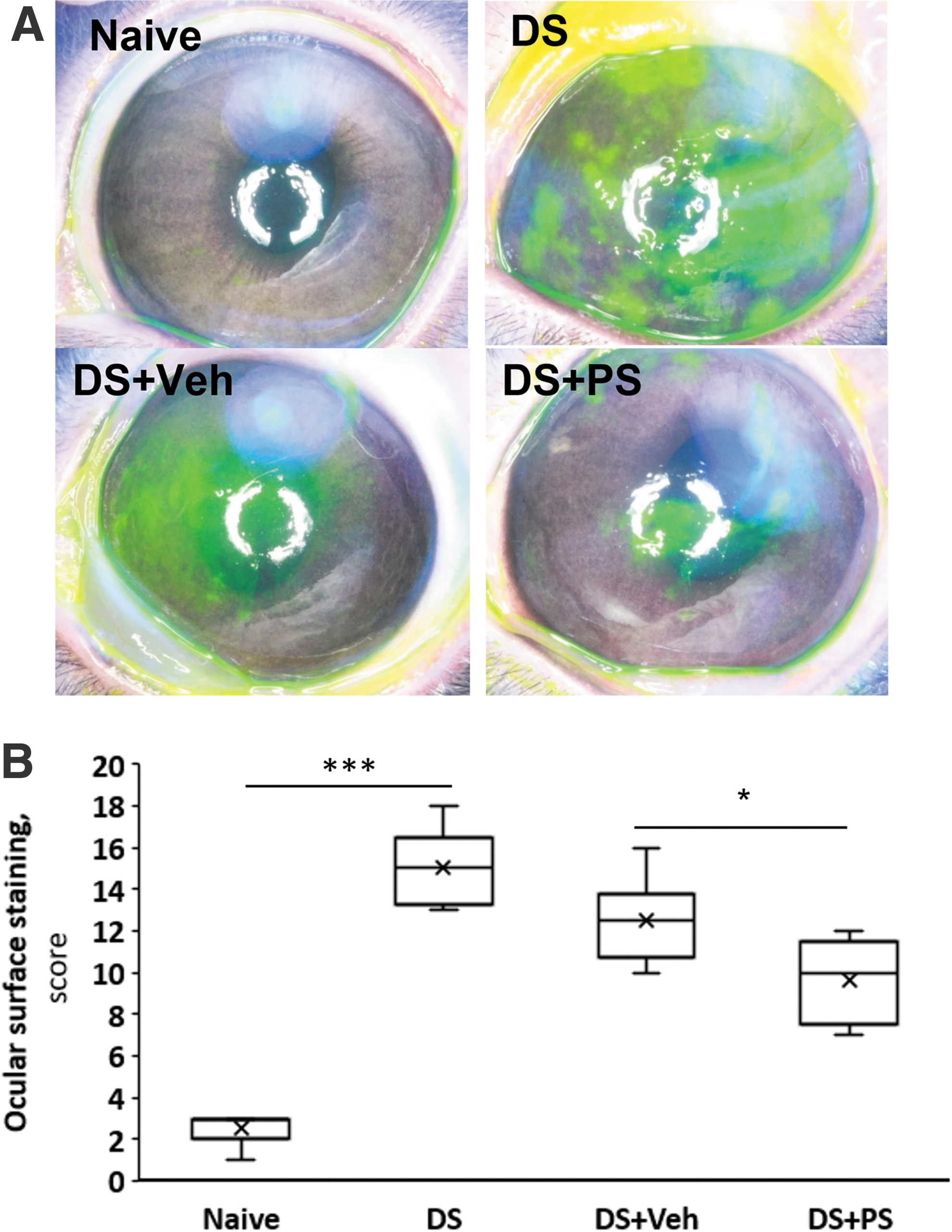
The effect of 0.2% PS on ocular surface staining in desiccative stress. The rabbits reported in this figure are the same as in Fig. 7. The ocular surface was stained with fluorescein and scored as in Methods.
The safety of PS
No side effects, local or systemic, were noted during or after the administration of PS to rabbits in these studies.
Discussion
Our results demonstrate that PS is efficacious in DED when administered topically as eye drops in a novel hydrogel formulation that allows once-a-day dosing. These studies were performed in three rabbit models of DED that recapitulate key features of its pathophysiology. PS was efficacious in all, improving both signs and symptoms of the disease.
DED presents clinically with a varied combination of signs and symptoms, with a minority of patients having signs of the disease but no symptoms or the reverse.1,27 Corneal sensitivity, suppressed in DED, 27 is the only equivalent of an ocular symptom that can be readily determined in experimental animals. In our studies, PS consistently normalized it in both acute and chronic DED. Of the DED signs, PS always improved TBUT, a measure of tear stability, and corneal staining, a measure of the structural integrity of the ocular surface. STT and tear osmolality had varied responses to PS, depending on the model used.
DED patients do not routinely present with DED that is “acute” or of recent onset. Regardless, our model of acute DED, pathophysiologically important, can be very informative for the pharmacology of our agent.8,11 Extensive transcriptomic studies revealing significant correlation between human and rabbit DED transcriptomes, established it as a robust animal model of DED with the potential of guiding the development of successful therapeutic strategies.28–32
The most interesting findings concerning the effect of PS in this acute model of DED are its clear dose dependence and the rapid and substantial improvement of all five parameters that were evaluated. The normalization within days of corneal sensitivity, TBUT, and tear osmolality indicate PS's strong pharmacological effect. The effect of PS on tear osmolality is impressive, given its dramatic increase on day 4; it remained high during the period of observation despite its modest blunting associated with chemosis, likely secondary to the ConA injection to the palpebral superior lacrimal gland.
All five DED parameters (corneal sensitivity, TBUT, tear osmolality, STT, and surface staining), show a sharp transition between the PS concentrations of 0.1% (no efficacy) and 0.2% (maximal efficacy). It is unclear what underlies this rather unusual behavior of PS, which we have observed in other systems as well.
The progression of DED to chronicity reflects the transition of the immune response of the ocular surface from innate to adoptive immunity, accompanied by changes in multiple signaling cascades.3,33,34 On day 17, when DED was chronic, PS 0.2% normalized corneal sensitivity and significantly improved TBUT and tear osmolality compared with vehicle, while leaving STT unaffected. The latter, different from the improvement by PS in the acute model, is most likely due to the fibrotic stage of the lacrimal glands after repeated ConA injections, 8 rendering them unable to respond to PS with increased tear production.
PS proved also efficacious in the treatment of the evaporative model of DED that is distinct from the previous one as it primarily affects the Meibomian glands instead of the lacrimal glands. Despite this important difference, its response to PS is similar to that of the ConA model. Again, the most prominent effect is the normalization of corneal sensitivity, followed by improvements in ocular surface staining and TBUT, compared with vehicle treatment; tear osmolality and STT were not changed.
Our model of evaporative DED is based on the rabbit model of MGD. 12 In the original description of this model, however, it was not determined if this also led to evaporative DED. In this study, we demonstrated for the first time that MGD at 14 days resulted in ocular findings consistent with an evaporative phenotype with decreased TBUT and corneal sensitivity and increased fluorescein staining but without a significant effect on STT. This is not surprising because STT, best used to document severe aqueous deficiency, 35 may be maintained early in MGD by compensatory lacrimal flow.36,37
It is interesting that tear osmolality did not change in this model, because early MGD is considered a normal-to-high volume hyperosmolar state 38 and increased evaporation should increase tear film osmolarity. 39 A possible explanation for the lack of hyperosmolality is that mildly elevated localized evaporation and focal tear film HO cause osmotic flow out of superficial corneal cells normalizing tear osmolality.40–42
Desiccation of the ocular surface is an early contributor to DED pathogenesis. In the DED vicious cycle, inflammation and other important changes in the ocular surface epithelium lie downstream of it. Desiccating stress is linked to inflammation by triggering immune mechanisms 43 and rapidly activating inflammatory molecules in corneal epithelium.44,45 Desiccative stress is also associated with transient abnormalities in lacrimal gland function, and decreased STT.46,47 The lack of a significant increase in tear osmolality in our model may be due to reflex tear production, which could maintain tear osmolality values normal. 48 Of note, the usefulness, sensitivity, and specificity of tear osmolality as a marker of DED was recently questioned. 49
Our study assessed the effect of PS on the contribution of desiccation to DED. Compared with vehicle, PS improved both TBUT and ocular surface staining, both of which (especially TBUT) were severely and rapidly affected by desiccation. Although the improvements brought about by PS were modest in magnitude, they are considered impressive given the severity of the acute ocular damage in this model. Of note, pharmacokinetic studies (unpublished data) indicate that the corneal and conjunctival Tmax of PS after its topical application is 30 min.
Corneal tactile sensitivity, inversely related to symptomatology,50,51 may be a very important variable to monitor for agents that promote symptomatic relief in DED. Neurosensory alterations in DED may occur by direct effects on corneal nerves or indirectly through effects on corneal epithelial cells or dendritic cells. 52 Tear film instability 53 and increased tear film osmolality may mediate such neurosensory changes,44,54 although the latter was inferred from instillation of hyperosmotic solutions rather than by direct measurement.44,55
Our studies, showing restoration of corneal sensitivity in the presence of normal tear osmolality suggest a more complex process. This may be also true for the suspected role of inflammation in corneal sensitivity; decreasing inflammation may allow for nerve regeneration (increased corneal sensitivity) and decreased neurotransmitter release (decreased symptoms).27,52
On the other hand, the recently reported correlation with nociceptive and inverse correlation antinociceptive soluble mediators in the tears and symptoms 56 suggest another possible locus of effect of PS. The improvement of corneal sensitivity by PS in all models, one of its most prominent effects, underscores the therapeutic potential of PS, but could also offer insight into the pathogenesis of DED. This is reminiscent of a similar effect in humans displayed for example by cyclosporine, which, however, required several weeks of treatment and was of much lower magnitude. 57
The efficacy of PS in all three models suggests that PS may be of benefit in DED across multiple clinical presentations and etiologies, because each model targets a different anatomic structure and is theorized to represent a different cause of tear film abnormality; the ConA model an aqueous-deficient type, the MGD model, a primary evaporative phenotype, and the desiccative model a mixed type resulting in severe epithelial damage. 43 This study also demonstrates that PS is efficacious during all stages of the evolution of DED from the initiating acute phase to its chronicity. The clinical potential of PS is underscored by the fact that it was assessed with the commonly used diagnostic panel; in rabbits, as in patients, no single diagnostic test abnormality can define DED.58,59
Finally, our results document the superior performance of a novel formulation of PS that allows once-a-day dosing. Our unpublished data suggest that this specific hydrogel ensures a prolonged residence time of the drug on the ocular surface that may enhance its uptake and thereby its efficacy. Confirmed in humans, these findings suggest that in such a formulation, PS could increase patient compliance through simplified dosing.
In conclusion, our studies demonstrate the efficacy of PS in several distinct animal models representing a broad spectrum of DED subtypes. PS demonstrated consistently strong efficacy on parameters reflecting both signs and symptoms of DED. These preclinical findings suggest that PS merits further evaluation as a candidate agent for the treatment of DED.
Footnotes
Author Disclosure Statement
The authors declare no competing interests, except for B.R. who has an equity position in Medicon Pharmaceuticals, Inc. and Apis Therapeutics, LLC; and L.H., who has served as an employee of Medicon Pharmaceuticals, Inc. is currently an employee with an equity position in Apis Therapeutics, LLC.
Funding Information
Financial support was received from Institutional funds, Stony Brook University. Medicon Pharmaceuticals, Inc., provided test compounds. Dr. Ernest Natke helped with the preparation of the article.
