Abstract
Purpose:
To evaluate the efficacy of topical lotilaner ophthalmic solution, 0.25%, in patients with Demodex blepharitis.
Methods:
Eighteen adults with Demodex blepharitis, defined as >10 collarettes on the upper lid and/or mite density of ≥1.5 mites per lash (upper and lower), were treated bid for 42 days with the topical lotilaner ophthalmic solution, 0.25%. Contact lens wear, artificial eyelashes, and lid structural abnormalities were among the exclusion criteria. No other antibacterial, antiparasitic, or anti-inflammatory treatment or lid hygiene products were permitted. One eye of each patient was selected for analysis and assessed on day 7, 14, 28, and 42. Collarettes were graded at each visit, and mite density was evaluated by microscopy at each visit except day 7. Outcome measures were collarette elimination (≤2 lashes with collarettes) and mite eradication (0 mites). Drop tolerability, adverse events, visual acuity, and slit-lamp biomicroscopy were assessed.
Results:
Collarette elimination was achieved in 13/18 participants (72.2%) by day 42. Mean collarette grade (upper lid) declined from 3.56 ± 0.17 to 0.28 ± 0.11. Mite eradication was achieved in 14/18 participants (77.8%) by day 42. Mean mite density decreased from 2.63 ± 0.39 to 0.12 ± 0.08 mites/lash. Participants reported good tolerability. Adverse events were mild and transient and did not result in treatment discontinuation.
Conclusion:
Six weeks of at-home topical therapy with the lotilaner ophthalmic solution, 0.25%, was effective in eliminating the most common objective signs of Demodex blepharitis, with a collarette elimination rate of 72% and mite eradication in 78% of eyes by day 42. ISRCTN registration #: 24398865
Introduction
Demodex blepharitis is a chronic progressive condition characterized by inflammation, ocular irritation, and eyelid erythema. It is caused by an overpopulation or infestation of Demodex mites, with the likelihood of infestation increasing with age.1–4
In patients with Demodex blepharitis, collarettes (cylindrical dandruff), comprising partially digested epithelial cells, mite waste, and eggs, form at the base of the lashes. These collarettes are now recognized as pathognomonic for Demodex blepharitis.4–6 Currently, there are no FDA-approved treatments for Demodex blepharitis.
Lotilaner is a veterinary medication approved for use in several countries, including the United States and the European Union. It is a member of the isoxazoline class, which inhibits the γ-aminobutyric acid (GABA)-gated chloride channels (GABACls) of arthropods. 7 Ectoparasites exposed to isoxazolines exhibit a spastic paralysis leading to their starvation and death. 8 Demodex mites are invertebrate ectoparasites. Isoxazolines such as lotilaner are selective for the invertebrate nervous system rather than the mammalian nervous system and, as such, have been considered safe in mammals.8,9 Preclinical ex vivo testing demonstrated that lotilaner, 0.25%, killed >95% of Demodex mites within 24 h (Vehige, et al. American Academy of Optometry, 2019).
Lotilaner has been formulated as an eye drop (TP-03; Tarsus Pharmaceuticals, Inc., Irvine, CA). It has a long half-life and is highly lipophilic, which promotes the absorption of the drop into the lash follicles and meibomian glands.
We recently reported on the first clinical trial (“Mars”) in humans to evaluate the safety and efficacy of TP-03 for the treatment of Demodex blepharitis. 10 In that single-arm pilot study, participants applied the TP-03 lotilaner ophthalmic solution, 0.25%, twice a day bilaterally for 28 days. No treatment-related adverse events were reported and no clinically significant changes observed in corrected distance visual acuity (CDVA), intraocular pressure, or slit-lamp biomicroscopy findings. No patients discontinued treatment due to any medication intolerance. Treatment with lotilaner ophthalmic solution, 0.25%, significantly improved the collarette grade in both the upper (P = 0.0034) and lower (P = 0.0122) lids of the analysis eye by day 14; the effects were maintained through day 90. Demodex mite density for the analysis eye was also statistically significantly improved (P < 0.05) from baseline at all study visits.
In a second study (TRS-004, “Jupiter”), 11 we evaluated lotilaner in a randomized-controlled trial, comparing the active ingredient with vehicle, with similar results.
The present study (“Io”) builds on these earlier studies, with a longer duration of treatment (42 days). In addition to the longer duration of treatment, in the present study we focused on more stringent outcome criteria, evaluating the proportion of subjects with elimination of collarettes and complete eradication of mites, rather than merely improvements in these clinical signs of Demodex blepharitis.
Methods
This was a single-arm, open-label, Phase 2a treatment study. The study was approved by the Asociación para Evitar la Ceguera (APEC) Committee on Ethics in Investigation and conducted in accordance with the tenets of the Declaration of Helsinki. A total of 27 participants with blepharitis due to Demodex infestation who had previously been assigned to the control arm of TRS-004 (an earlier Phase 2a study called “Jupiter”) and who had successfully completed that study were eligible for enrollment in the present nonrandomized, single-arm treatment study (Fig. 1).
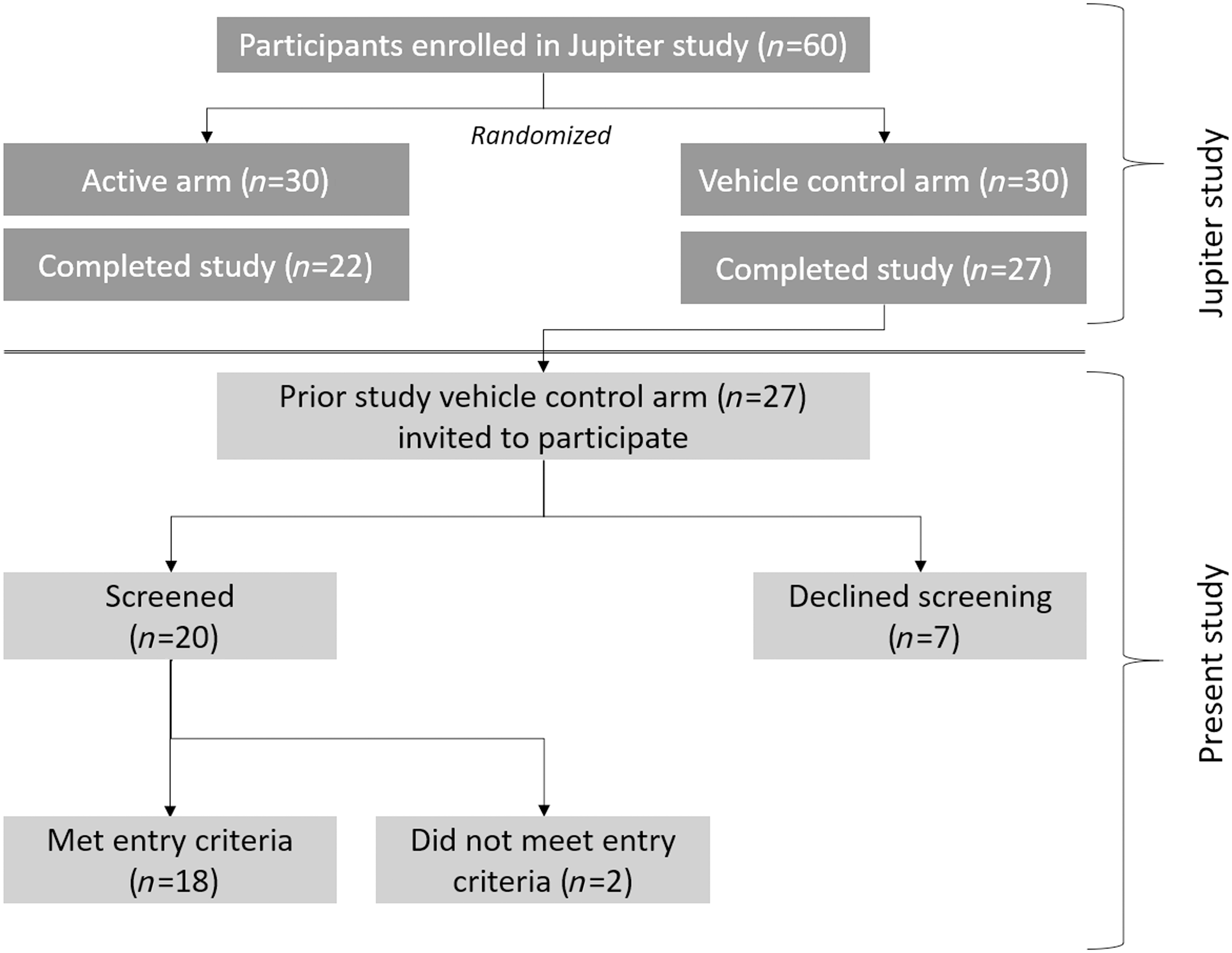
Schematic showing how participants in this clinical study were recruited and screened.
To be eligible, patients had to be at least 18 years old, able to provide informed consent, and deemed capable of complying with the study protocol requirements. For inclusion in the study, participants had to have >10 lashes with collarettes present on the upper lid and/or have a Demodex mite density of ≥1.5 mites per lash (upper and lower) in at least 1 eye.
Patients who were wearing or had recently used contact lenses, artificial eyelashes, or eyelash extensions were excluded from participation. No systemic or topical antibacterial, antiparasitic, or anti-inflammatory steroid treatments; topical tea tree oil (TTO) or hypochlorous acid; or other lid hygiene products were permitted during the treatment period or for 14 days before the screening visit. Patients were excluded if they had used a topical prostaglandin analogue (PGA) to promote eyelash growth, but were allowed to use PGA treatment for medical reasons as long as they had not initiated treatment recently (within the past 30 days) and planned to be on a stable dose during the study. Other exclusion criteria included acute ocular infection; active inflammation other than blepharitis; and pregnancy.
Written informed consent was obtained before administration of any study medication or testing. At the screening visit, potential participants were evaluated for eligibility, including confirmation of Demodex infestation. Collarettes were assessed, mites counted, and slit-lamp photographs of the upper and lower lid margins were obtained. A pregnancy test was administered to females of childbearing age. For those who qualified to participate, the first dose and additional day 1 testing could take place on the same day as the screening visit or at another visit within 14 days.
The initial dose of lotilaner ophthalmic solution, 0.25% (TP-03; Tarsus Pharmaceuticals, Inc.), was administered under supervision in the clinic at the APEC, in Mexico City, Mexico. After instilling the drop, participants were instructed to close their eyes and rub their finger across the upper lid, applying gentle pressure, for 2–3 s, to express the medication across the upper and lower lid margins. Drops were then dispensed in a preserved, multidose drop bottle for home use for the remainder of the study. Participants were instructed to instill 1 drop in each eye, twice daily for 42 days.
Participants were assessed on day 7 (±2 days), 14 (±3 days), 28 (±4 days), and 42 (±5 days). At each visit, CDVA was assessed and slit-lamp biomicroscopy was performed. Collarettes were evaluated on a non-linear scale and assigned a grade from 0 to 4 (0 = 0–2 lashes with collarettes per eyelid, 1 = 3–10 lashes with collarettes per eyelid; 2 = >10 but <1/3 of the lashes with collarettes; 3 = ≥1/3 but <2/3 of lashes; and 4 = ≥2/3 of the lashes). The upper lid has approximately 150–200 lashes. Magnified images focused on the lashes, and lid margins were obtained at day 1 and 42.
Demodex density was evaluated at baseline, day 14, and every visit thereafter. Two or more lashes were removed from the upper and lower eyelids (selecting those with collarettes if present). Lashes were placed on glass slides, covered with an artificial tear with an emulsifier, placed under a cover slip, and viewed with a microscope so that the mites could be seen and counted.
One eye of each participant was selected as the analysis eye. The analysis eye was the eye that met all the inclusion criteria. If both eyes met all the inclusion criteria, then the analysis eye was the eye with the highest Demodex density at the screening visit or, if both eyes had equal Demodex density, the right eye.
Efficacy outcome measures included collarette cure, defined as Grade 0 or ≤2 collarettes on the upper lid of the analysis eye, and mite eradication, defined as 0 mites per lash on the analysis eye at day 42. On day 42, participants were asked to rate drop comfort as “very comfortable,” “somewhat comfortable,” “neither comfortable nor uncomfortable,” “slightly uncomfortable,” or “very uncomfortable.” Safety was determined by assessing adverse effects related to the treatment, as well as evaluating any changes in CDVA or slit-lamp biomicroscopy findings.
This is a single-arm study designed to provide those participants who received the vehicle control in a previously completed randomized pilot study the opportunity to receive the active treatment. The maximum sample size is based on the maximum number of participants randomized to the vehicle control in the previous pilot study (named “Jupiter”). The sample size for that study was based on clinical and practical considerations, as there were no available data on the clinical effects of the treatment. Efficacy results for proportions were summarized using the 95% confidence interval. Changes from baseline were analyzed using paired statistics, paired t-test, or Wilcoxon signed-rank test as appropriate. Comparisons were 1-sided using an α of 0.05. No adjustment was made for multiple comparisons. Ocular safety parameters were summarized separately for the right and left eyes using descriptive statistics.
Results
Twenty potential participants from the previous controlled trial were screened. Of these, 18 met the study criteria and were enrolled. All 18 completed the study, which was conducted between November 11, 2019, and January 20, 2020. Participants ranged in age from 46 to 81 years (mean 69.7 ± 2.3). Demographic and baseline characteristics are shown in Table 1.
Demographics and Baseline Characteristics
SEM, standard error of the mean.
Elimination of collarettes (Grade 0 or ≤2 collarettes on the upper lid of the analysis eye) was achieved in 12 of 17 participants (70.6%) at day 28, and 13 of 18 participants (72.2%) at day 42 (Fig. 2). The mean collarette grade for the upper lid declined over time from 3.56 ± 0.17 at baseline to 0.28 ± 0.11 at day 42 (Fig. 3). Similar results were observed for the lower eyelid. The upper eyelids of all eyes had at least a 1-grade reduction in collarettes, and 94.1% had at least a 2-grade reduction in collarettes by day 14. By day 42, 100% of eyes, both upper and lower lids, had a 2-grade or greater reduction in collarettes. Figure 4 shows the change in collarettes in a representative eye from baseline to day 42.
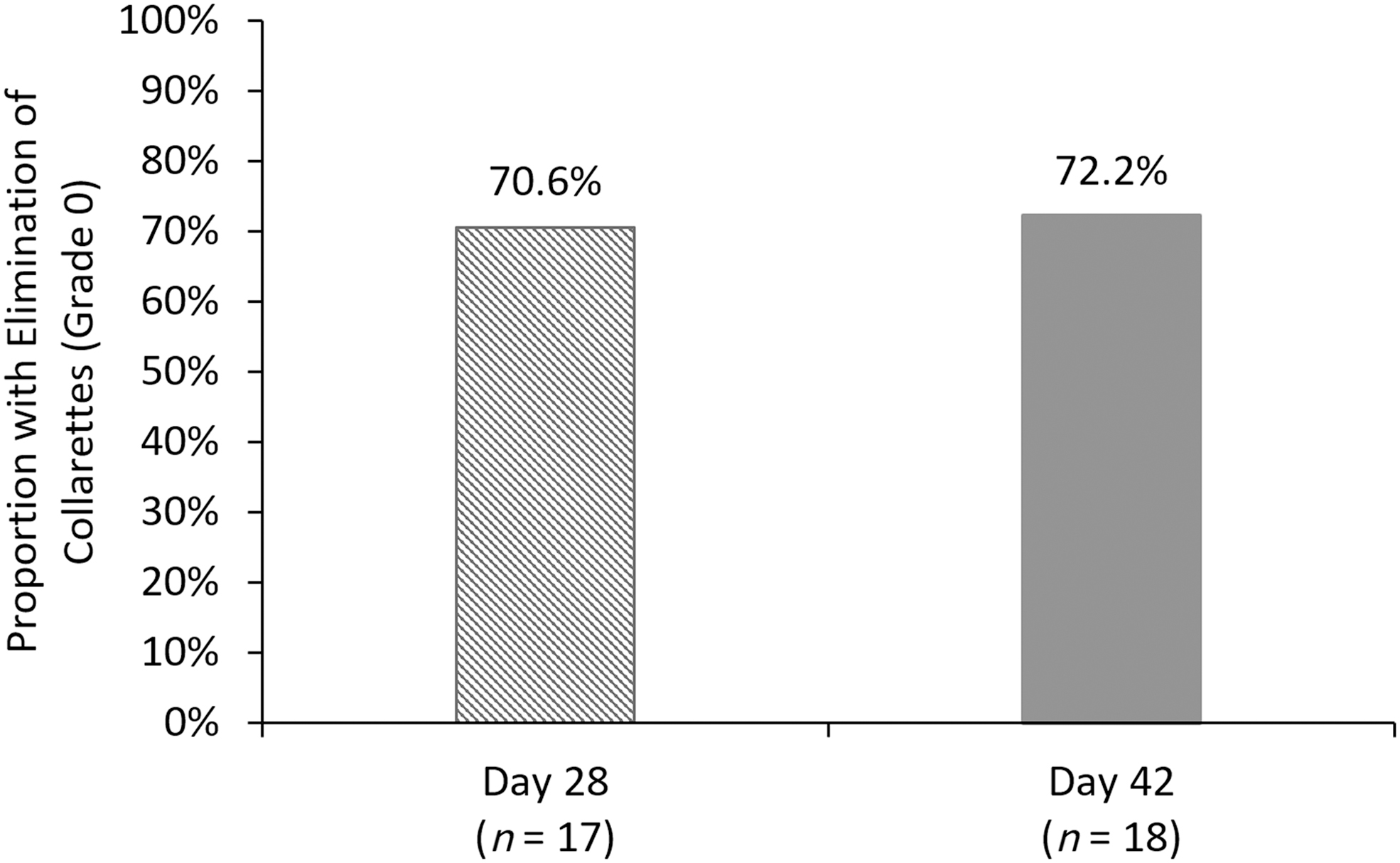
Proportion of participants with elimination of collarettes (Grade 0) for the upper eyelid of the analysis eye.
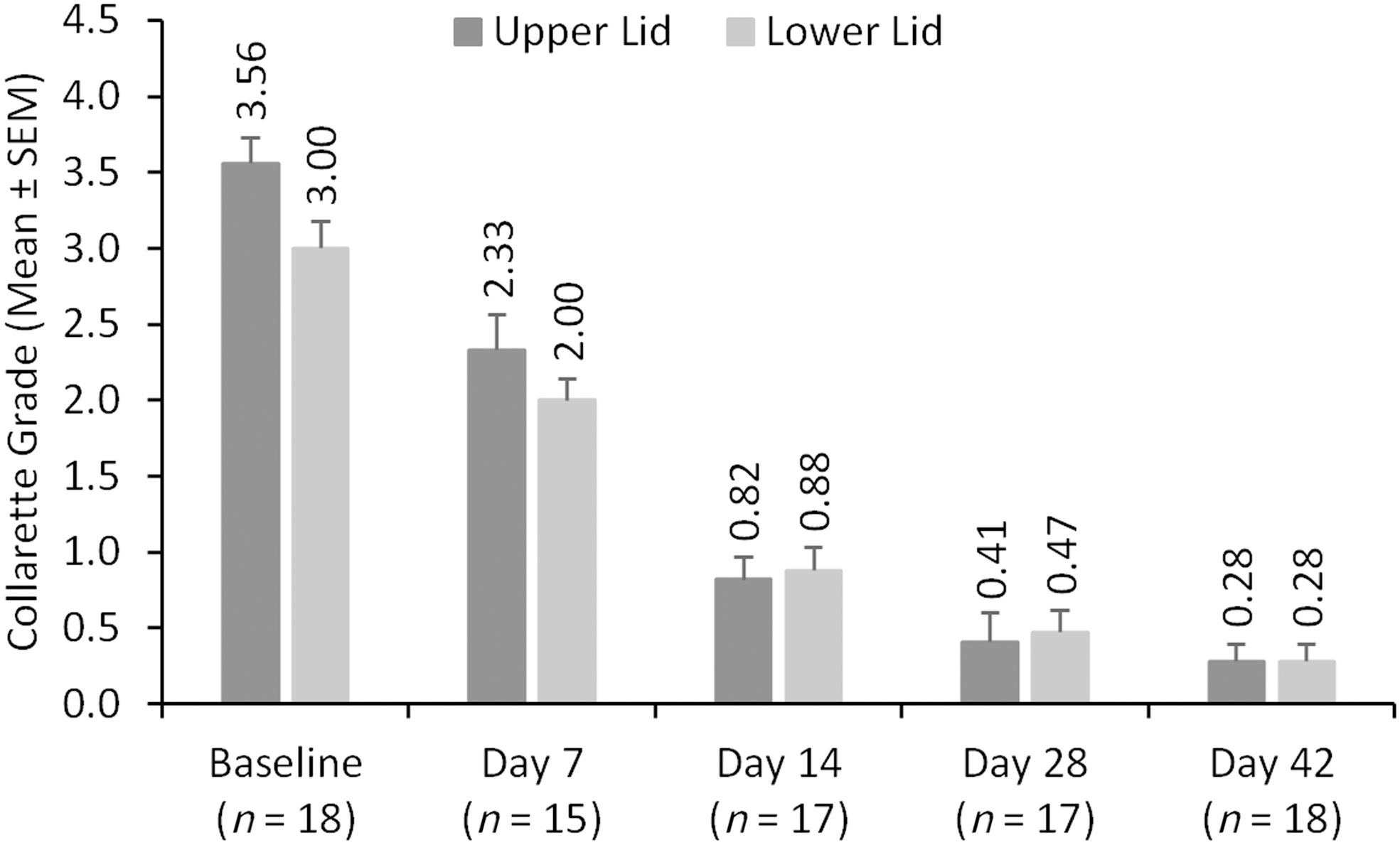
Mean collarette grade at each visit for the upper and lower eyelids of the analysis eye. SEM, standard error of the mean.
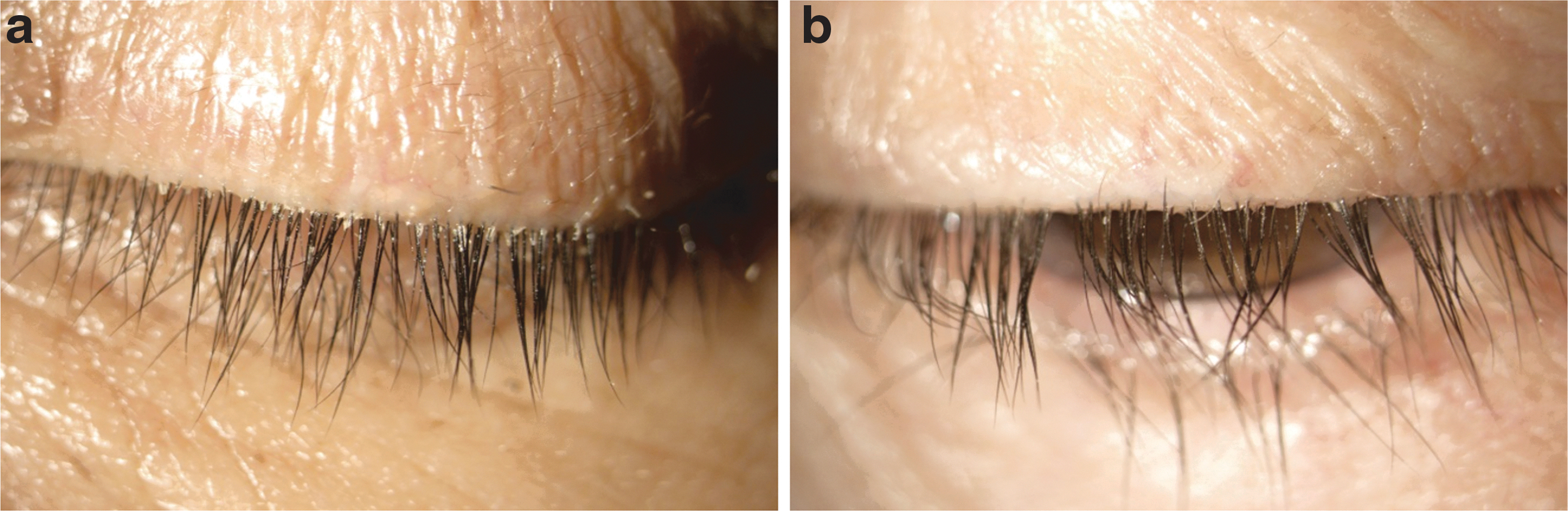
Left eye of participant 001–249. The baseline image of the lashes
The proportion of eyes with Demodex mite eradication (mite density of 0) increased over time. By day 42, total mite eradication was achieved in 14 of 18 participants (77.8%, Fig. 5) and near or total eradication (≤0.5 mites per lash) was achieved in 94.4% of eyes. Mean mite density in the analysis eye decreased from 2.63 ± 0.39 mites/lash at baseline to 0.12 ± 0.08 mites/lash at the conclusion of the study on day 42 (Fig. 6), a >30-fold reduction in mite density.
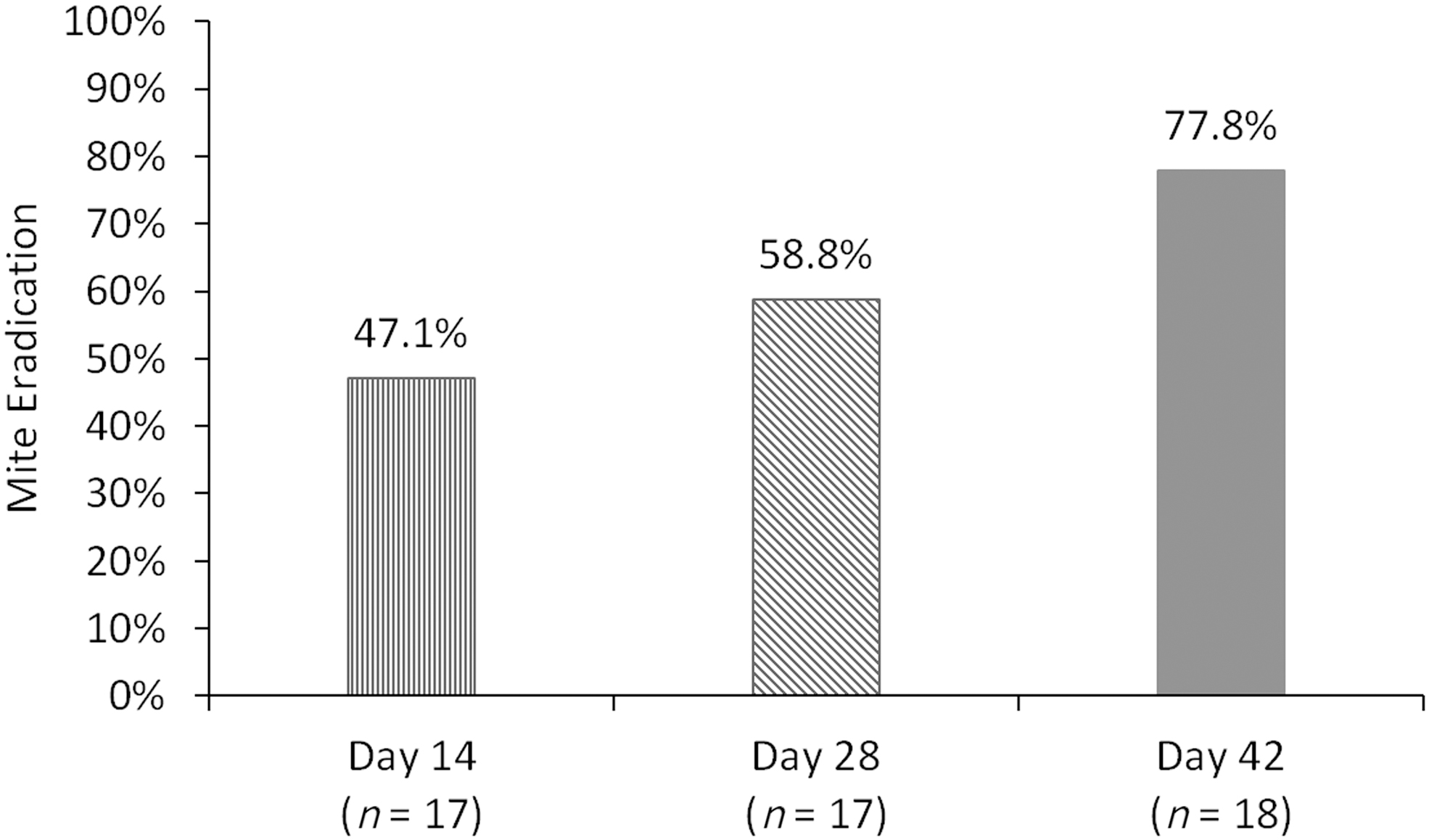
Proportion of participants with mite eradication (mite density of 0) at various time points.
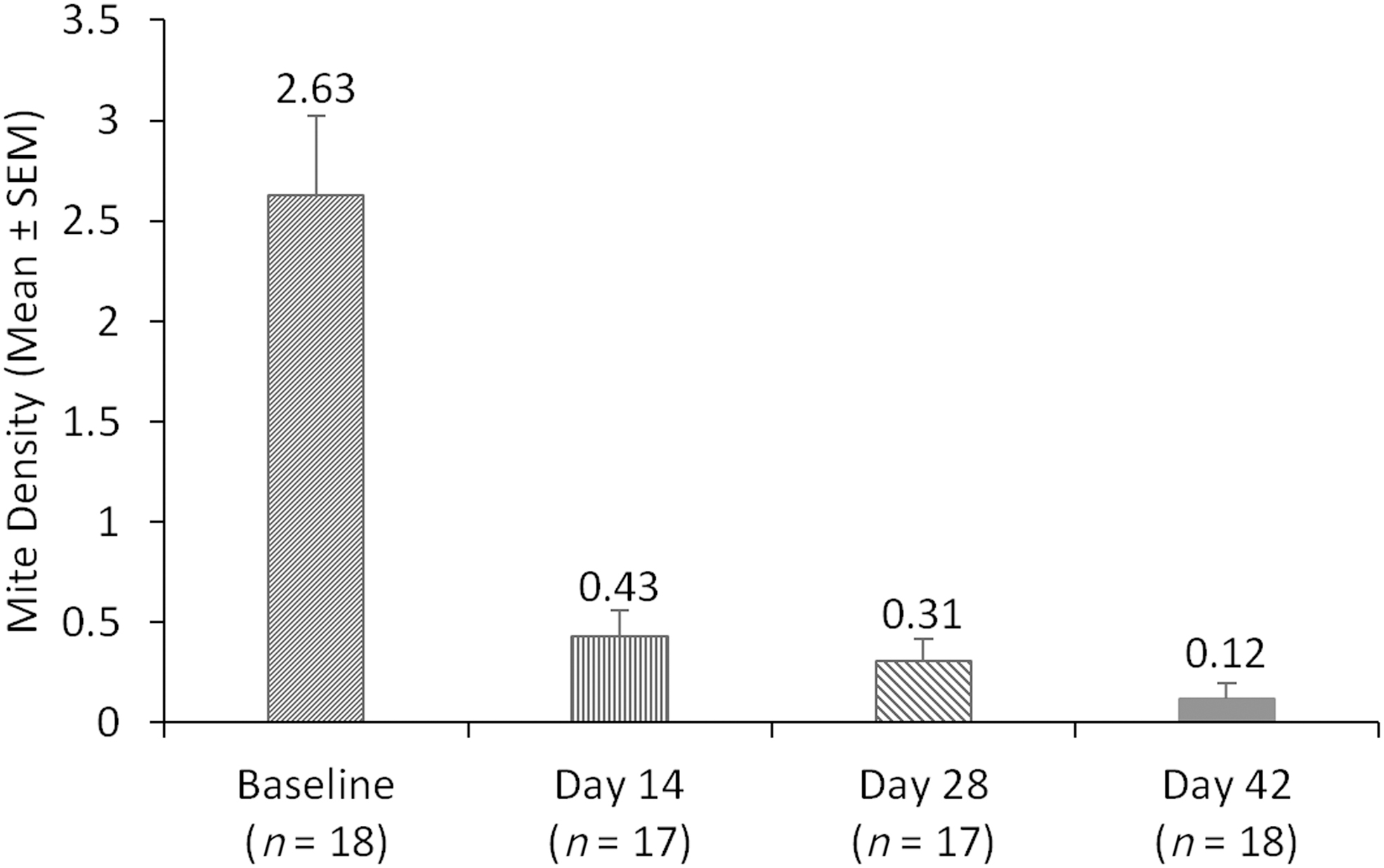
Mean mite density over time for the analysis eye.
At day 42, participants reported good tolerability of the drop, with 12 of 18 (66.7%) rating it somewhat or very comfortable and the rest neutral (Fig. 7).
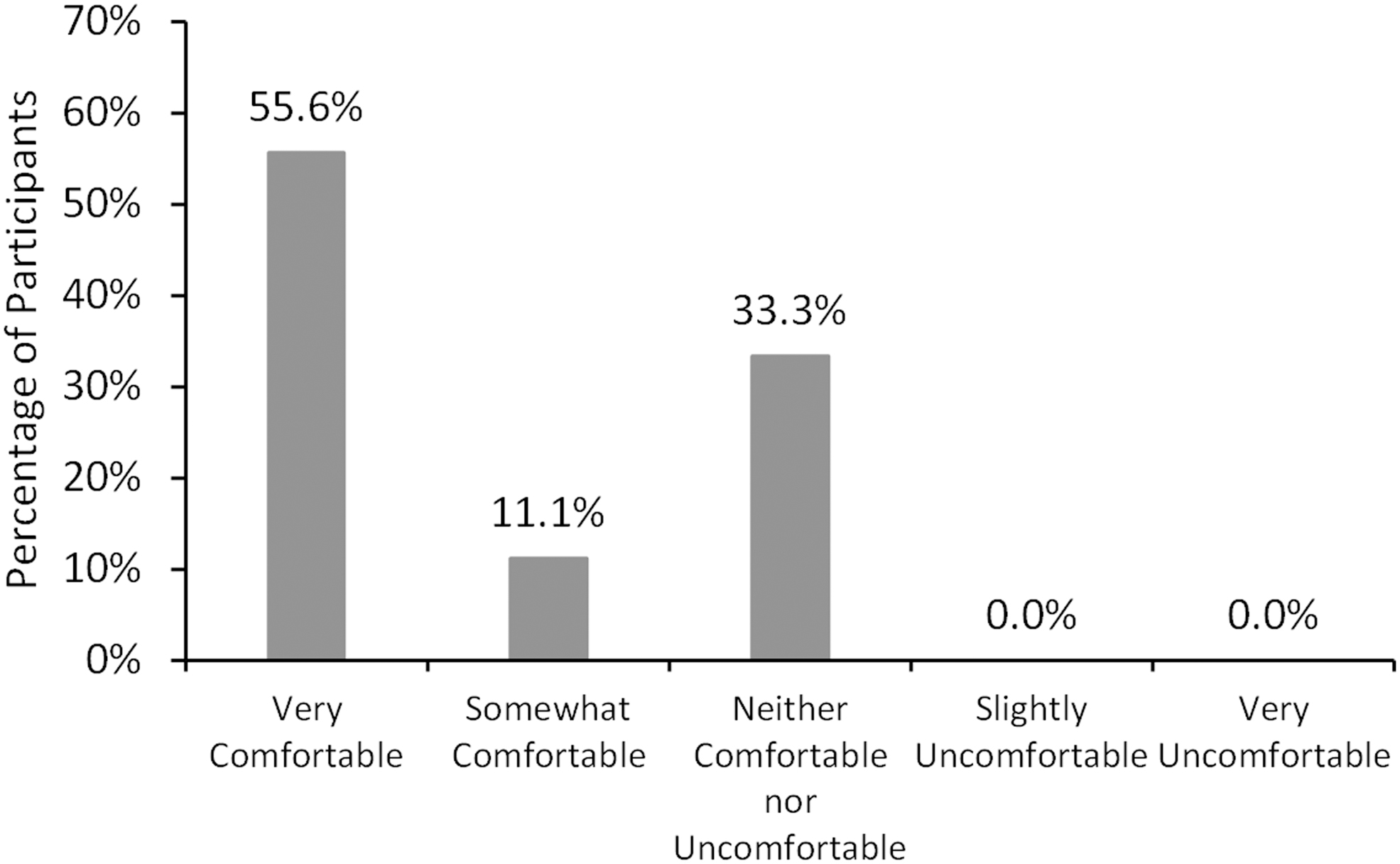
Patient-reported comfort of the study drug at day 42 (n = 18).
Five participants reported adverse events that were related or possibly related to the study drug.
One participant reported mild blurriness following instillation that lasted for 2–3 h. The investigator noted that the participant's preexisting dry eye disease may have contributed to this occurrence. Four participants reported mild burning after drop instillation that lasted <10 min. None of these adverse events resulted in discontinuing treatment with the study drug.
There were no increases or decreases in CDVA greater than 2 lines (0.2 logMAR) during the study, and no clinically significant changes in slit-lamp biomicroscopy findings of the eyelids (other than collarettes), cornea, or conjunctiva. Three eyes of 2 participants had increases in corneal staining that improved at subsequent visits.
Discussion
This study builds on our previous, unpublished pilot study of lotilaner ophthalmic solution, 0.25%, for the treatment of Demodex blepharitis.
The treatment phase in the current study was extended to 42 days (6 weeks). The Demodex mite life cycle, from egg through adult stage, is ∼3 weeks,3,6,12 and so, it is possible that a 6-week treatment would be more effective at mite eradication by killing mites over 2 full life cycles. And, in fact, the rate of mite eradication improved from 57.1% in our previous pilot study with 28 days of lotilaner 0.25% treatment to 77.8% with 42 days of treatment in the present study.
Among TTO and other lid hygiene products that are designed for at-home use by patients, no single treatment has been able to fully eradicate mites at 4 weeks. 13 Koo et al. reported an overall Demodex mite eradication rate of 23.6% in patients treated with TTO for 1 month. 14 However, in this study, patients underwent weekly in-office lid scrubs with high-concentration TTO in addition to twice-daily at-home lid scrubs, and so, the treatment regimen was quite arduous in comparison with a topical drop.
Compared with our previous clinical studies of lotilaner ophthalmic solution, 0.25%, in which improvements in clinical signs were evaluated, a major focus of this study was clinical cure of blepharitis, as defined by eradication of mites and elimination of collarettes. The debris found at the base of the lashes is pathognomonic for Demodex blepharitis and is relatively easy to identify and evaluate during a standard slit-lamp examination. At baseline, the participants had quite severe blepharitis, as evidenced by average collarette grades of 3.56 (upper lid) and 3.00 (lower lid), which are equivalent to >1/3 and in many cases >2/3 of the lashes having collarettes. By day 42, 72.2% had achieved collarette Grade 0, or ≤2 lashes per lid with collarettes.
The excellent safety and good ratings for comfort of the drop are encouraging. Currently, the most common management of Demodex blepharitis involves the use of scrubs, wipes, or gels containing TTO or its major acaricidal component, T4O. A recent Cochrane review found that the efficacy of TTO products is uncertain. 15 TTO lid hygiene is known to cause ocular irritation14,16–18 and contact dermatitis. 19 It can be toxic to epithelial cells and fibroblasts20,21 and has recently been shown to be harmful to human meibomian gland epithelial cells in vitro. 22 In future controlled studies of lotilaner in a larger number of eyes, it will be important to continue to evaluate the tolerability of this agent.
This was a small pilot study. A Phase 3 randomized-controlled trial with the same treatment duration and same endpoints in a larger study population is underway. Additional work is needed to fully understand the duration of effect following 6 weeks of treatment. Longer term follow-up in future studies would be helpful to learn if and when collarettes and mites recur.
In conclusion, treatment for 6 weeks with lotilaner ophthalmic solution, 0.25%, was effective in eliminating the most common objective signs of Demodex blepharitis, with a collarette elimination rate of 72% and mite eradication in 78% of eyes by study day 42. To our knowledge, this is the first study to report the clinical cure of Demodex blepharitis in terms of both collarette elimination and mite eradication. Lotilaner ophthalmic solution, 0.25%, is an easy-to-use, at-home topical therapy that was well tolerated and caused no serious adverse events.
Footnotes
Authors' Contributions
Jan Beiting (Wordsmith Consulting, Cary, North Carolina) and Raman Bedi, MD (IrisARC—Analytics, Research & Consulting, Chandigarh, India), provided editorial assistance in the preparation of this article.
Author Disclosure Statement
R.G.S. reports personal fees from Tarsus Pharmaceuticals, Kedalion Therapeutics, LayerBio, and Laboratorios Sanfer, outside the submitted work. None of the previous disclosures conflict with the present work. E.Y. is a consultant to Tarsus Pharmaceuticals. M.H. and S.N.B. are employees of Tarsus Pharmaceuticals. The rest of the authors report no competing financial interests related to this work.
Funding Information
Financial support for article preparation was provided by Tarsus Pharmaceuticals.
