Abstract
Purpose:
Corneal endothelial dystrophies are characterized by endothelial cell loss and dysfunction. Recent evidence suggests that corneal endothelial cells (CECs) can regenerate although they do not do so under normal conditions. This work sought to test whether CECs can be stimulated to proliferate in organ culture by wounding and/or by treatment with the engineered human fibroblast growth factor 1 (FGF1) derivative TTHX1114.
Methods:
Human donor corneas obtained from eye banks were maintained in organ culture in the presence or absence of TTHX1114. Wounds in the corneas were created by quartering the corneas. The CEC monolayer was identified as a regular layer by Hoechst staining of the nuclear DNA with cell outlines delineated by immunohistochemical identification of ZO-1. Nuclei and nuclei incorporating 5-ethynyl-2′-deoxyuridine (EdU) were counted using ImageJ.
Results:
CECs in normal corneas in undisturbed monolayers had low, but measurable, rates of proliferation. CECs at the edge of a wound had higher rates of proliferation, probably due to the release of contact inhibition. TTHX1114 increased proliferation at wound edges. After 7 days of culture, proliferating CECs formed contiguous groups of labeled cells that did not migrate away from one another. TTHX1114-treated cells, including the EdU labeled proliferating cells, retained normal morphology, including cell/cell junction ZO-1 staining.
Conclusions:
Proliferation of CECs in organ-cultured corneas is low, but can be stimulated by wounding or by the administration of TTHX1114 with the effects of each being additive. The CEC monolayer appears to have a population of progenitor cells that are susceptible to stimulation.
Introduction
Corneal endothelial dystrophies, including Fuchs endothelial corneal dystrophy (FECD), are characterized by a progressive loss of corneal endothelial cells (CECs).1–3 Corneas that have lost sufficient CECs lose the ability to pump fluid out of the cornea leading to edema, corneal opacity, and loss of vision. The only therapeutic option for such patients is the transplantation of healthy endothelial cells from a normal donor, and tens of thousands of such surgeries are performed every year.4, 5 A treatment that stimulated the proliferation of CECs in these visually compromised corneas would have the potential to treat FECD and other endothelial dystrophies.
While CECs do not seem to proliferate in vivo to any significant extent, there are several lines of evidence arguing that they retain the capacity to do so (reviewed in Van den Bogerd et al. 6 ). When removed from patients and cultured as dissociated cells in vitro, CECs can proliferate7–9 and retain function as shown by their ability to restore normal endothelial function when transplanted back into FECD patients. 10 CECs in intact corneas express proliferative markers such as Ki-67, although in a small subpopulation of cells. 11 CECs at the edge of a wounded area of a cornea in organ culture have increased Ki-67 expression,12,13 incorporate 3 H-thymidine, and display mitotic figures, 14 suggesting that wounding of the endothelial layer can stimulate proliferation.
Pharmacological stimulation of CEC proliferation would provide the equivalent of transplantation or cell therapy without the potential for rejection/surgical mishaps or the high cost and production issues of cell therapy.15,16 Fibroblast growth factors (FGFs) are endogenous stimulators of CECs that are used as a standard component of culture media and are both protective 17 and potently mitogenic for CECs.18–23 FGFs stimulate CEC migration and corneal wound healing in vitro and can accelerate the recovery of the endothelial layer from wounding in vivo,24,25 thus making them candidates for regenerative therapy. Different FGFs interact with different subsets of FGF receptors (FGFRs), with fibroblast growth factor 1 (FGF1) interacting with the full spectrum of tyrosine kinase containing FGFRs and other FGFs such as FGF2, interacting with various subsets.26,27 FGF1 has specifically been shown to be proliferative for CECs and to stimulate endothelial recovery.19,28
Dissociated cell culture and animal models, while useful, are imperfect reflections of human responses in vivo. Primary or immortalized endothelial cell cultures may not accurately reflect the environment in the intact cornea, where cell/cell contact and the influence of the extracellular matrix, Descemet's membrane, and stroma, among other agents and elements, may be important regulators of CEC growth.29,30 Culturing of CECs has been shown to change their protein expression profile, including that of growth factors and their receptors.31–33 CECs grow readily in dissociated cell culture 6 but do not proliferate meaningfully in organ culture, 34 demonstrating that the regulation of proliferation in these 2 environments is substantially different. Corneas in organ culture retain function and can be successfully used as donor tissue for transplantation, 35 and their CECs retain functionality, including intact barrier and pump functions as reflected by maintained corneal fluid balance 34 and appropriate ZO-1 and Na,K ATPase staining. 36 The differences between dissociated cell culture and organ culture make it important to investigate the regulation of proliferation and functional integrity of CECs in organ culture, which may be more representative of the in vivo condition.
This study sought to explore stimulation of CEC proliferation in the human organ culture system using both wounding and pharmacological stimulation. The latter was tested using TTHX1114, an engineered version of human FGF1 that has been shown to stimulate rabbit corneal epithelial cells and protect them from chemical damage, 37 and is a close relative of FGFs that accelerate dermal wound healing in diabetic animals.38,39 The utility of FGFs is limited by their instability and short half-life in biological systems, consistent with their natural role as signals intended to be limited in time and space. TTHX1114 incorporates several amino acid substitutions to the FGF1 protein known to make the molecule more stable in biological systems.40,41 Since corneal dystrophies such as FECD are driven by loss of endothelial cells, a drug able to stimulate the proliferation of CECs such as TTHX1114 would have potential utility in the treatment of endothelial dystrophies, including FECD.
Methods
Tissue preparation and reagents
Human corneas were obtained from the Lions Eye Institute for Transplant and Research, Tampa, FL. This work used existing specimens without identification of subjects and is exempt from IRB approval under 45 CFR 46.101(b)(4). Donor ages, CEC densities, and time to culture are shown in Table 1. Corneas were stored in Optisol at 2°C–6°C between the time of harvest and use.
Donor Information
Corneas were removed from Optisol, rinsed in phosphate-buffered saline (PBS) and the sclera trimmed to leave a 1–2 mm scleral ring. Whole corneas were then incubated at 37°C in 5% CO2 for 24 h in Opti-MEM (Invitrogen, Carlsbad, CA) supplemented with 1 × insulin, transferrin, and selenium (Corning, Ithaca, NY), 1 × antibiotic/antimycotic (Thermo Scientific, Waltham, MA), 0.02 mg/mL CaCl2 (Amresco, Solon, OH), and 0.2 mg/mL ascorbic acid (Sigma, St. Louis, MO) with 8% heat-inactivated fetal bovine serum (FBS; Thermo Scientific). After 24 h of incubation, corneas were cut into quarters with a scalpel; the point of the quarter is thus the center of the cornea and the curved edge opposite the point is the scleral rim. Corneal quarters were incubated in the above medium with 0.4% FBS containing 10 μM 5-ethynyl-2′-deoxyuridine (EdU; Invitrogen) with or without 100 ng/mL TTHX1114 for 2–7 days. The medium was changed every other day.
TTHX1114 (human N-methionyl-FGF1, 141 amino acid form, containing the substitutions Cys16Ser, Ala66Cys, and Cys117Val using the 140 aa numbering system (see Eveleth et al. 37 and Xia et al. 39 ) was prepared by the Frederick National Laboratory for Cancer Research, Biopharmaceutical Development Program.
Fixation and staining
After incubation, cornea quarters were fixed in methanol chilled to −20 for 30 min, rinsed at room temperature in PBS containing 0.05% Tween-20 (Sigma), and blocked for 30 min at 37°C in PBS containing 2% bovine serum albumin (Fisher Scientific, Pittsburgh, PA) and 2% goat serum (Sigma, St. Louis, MO) (blocking solution). Quarters were subsequently incubated in 2.5 μg/mL mouse anti-ZO1 (clone 1A12; Thermo Scientific) in the above blocking solution for 60 min at 37°C, rinsed 3 times in PBS containing 0.05% Tween-20, and fixed at room temperature for 30 min in 0.5% paraformaldehyde (Electron Microscopy Sciences, Hatfield, PA) in PBS. The quarters were then rinsed again in PBS containing 0.05% Tween-20 and permeabilized at room temperature for 5 min in PBS containing 1% Triton X-100 (MP Biomedicals, Santa Ana, CA) followed by 3 rinses of 5 min each in PBS.
The EdU click-it reaction with Alexa Fluor 488 and subsequent staining with Hoechst 33342 was performed as described previously. 37 Next, quarters were rinsed again in PBS containing 0.05% Tween-20 for 30 min and incubated in blocking solution containing Alexa Fluor 555-labeled goat anti-mouse IgG (Thermo Scientific) for 60 min at 37°C and rinsed again 3 times in PBS containing 0.05% Tween-20 for 30 min.
Microscopy
Corneal quarters after fixation and staining were flat mounted on slides with the endothelial side up in VECTASHIELD H-1000 (Vector Laboratories, Burlingame, CA). For quantification of proliferating cells, a Nikon E400 epifluorescence microscope was used to capture 3 nonadjacent 20 × images of the mid zone and 3 of the area at the edge or within 3 field diameters of the edge for a total of 6 fields per condition per cornea. The total number of cells and the number of EdU-incorporating cells in each field were counted using ImageJ (National Institutes of Health, Bethesda, MD). The % of CECs incorporating EdU for the images were averaged. High-quality images were captured using a Zeiss 710 confocal microscope. The image in Fig. 3 was taken using a Nikon A1 confocal microscope by capturing a series of 20 × images of a single field with a corneal edge at varying depths of focus. The stack of images was compiled using the Bio-Formats plug-in in ImageJ, and animated on Photoshop (Adobe, San Jose, CA) to show the CECs migrating down the cut edge.
Results
To test the potential for human CEC proliferation, whole corneas cut into quarters were cultured and the proliferation of CECs was measured by counting the proportion of cells incorporating EdU. The endothelial layer was easily visualized under the microscope and differentiated from the stroma since the endothelial cells are arranged in a monolayer with regularly shaped nuclei and clear ZO-1 immunoreactive borders.
To enable comparisons within a cornea, some quarters from each cornea were incubated with TTHX1114, whereas others served as controls. EdU incorporation was quantitated separately in the mid zone where the endothelial layer is undisturbed by the quartering and at the edge of the wound created by quartering (Fig. 1A).
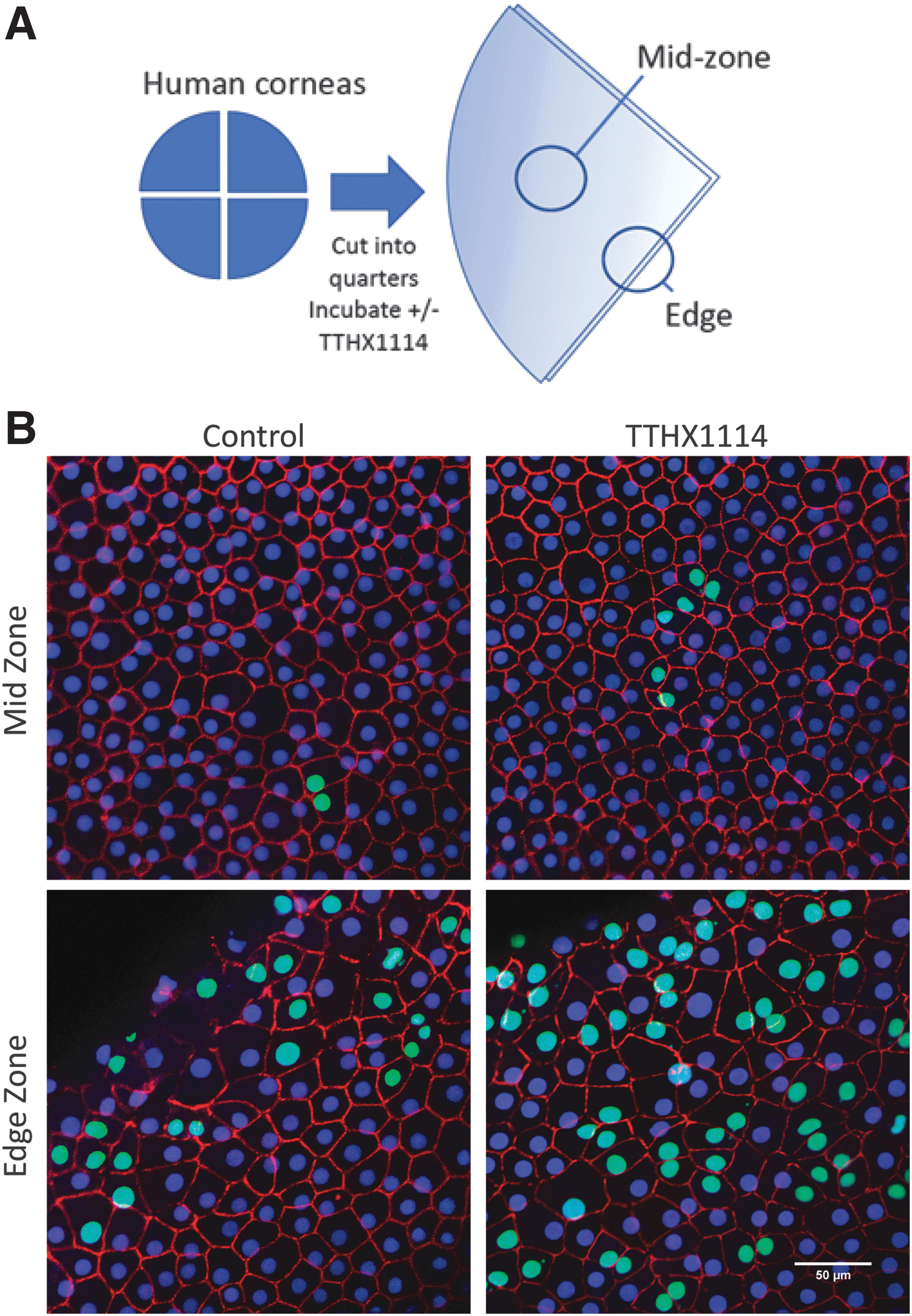
Proliferation of CECs in normal corneas in organ culture over 2 days.
In normal control (unstimulated) corneas, the level of proliferation in the mid zone was generally very low to unmeasurable over 48 h with less than 2% of cells incorporating EdU (Figs. 1B and 2). The corneal endothelial layer retained its structure with ZO-1-rich cell/cell junctions and regularly spaced endothelial cells. Treatment with TTHX1114 for 48 h increased proliferation in the mid zone in 7/10 of these corneas (Figs. 1B and 2), but the overall EdU incorporation rate remained less than a mean of 5% even with treatment. The morphology of the CECs did not appear to be affected by TTHX1114 treatment. Only 2 normal corneas, both from the same individual, showed greater than 5% EdU incorporation in the mid zone at 48 h regardless of treatment with TTHX1114.
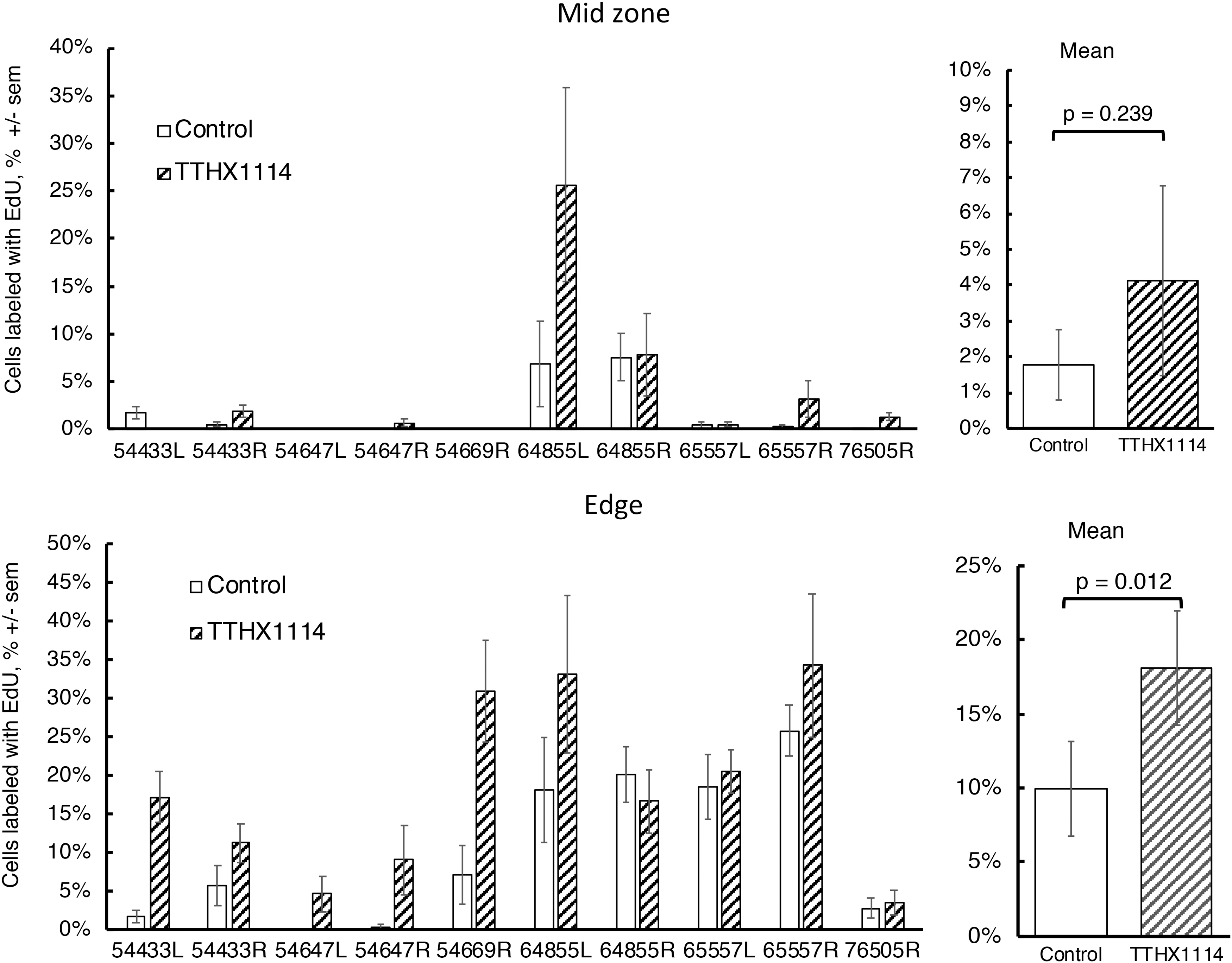
Quantitative analysis of CEC proliferation in normal corneas and stimulation by TTHX1114 after 48 h. Two quarters from each quartered cornea were incubated in media only and the other 2 in TTHX1114 (100 ng/mL). For each quarter, 3 fields were imaged and counted at edge and in the mid zone; each bar represents mean ± sem of 6 fields. Right panels, comparison of the mean of the TTHX1114-treated versus control using paired t-test.
At the wound edge central to the limbus, where the cornea was cut to separate the quarters, EdU incorporation of the CECs was generally higher than in the mid zone in both control and TTHX1114-treated quarters. The pattern of ZO-1 staining showed that the cell boundaries at the edge were less regular and the cell areas appeared increased. Treatment with TTHX1114 increased EdU incorporation in 9/10 corneas near the wound edge.
After 48 h of incubation, the average % of cells incorporating EdU in the control (untreated) mid zone was 1.8 ± 0.98 (mean ± sem) and in the TTHX1114-treated group the average was 4.12 ± 2.64 (P = 0.239, paired t-test). In the control edge zone, the average % of cells incorporating EdU was 10 ± 3.2 and this increases to 18 ± 3.8 in the TTHX1114-treated group (P = 0.012). In both the mid zone and edge, the level of proliferation was highly variable between corneas in both the unstimulated and stimulated conditions.
The presence of clear cell boundaries with high ZO-1 staining right up to and over the cut edge suggests that the endothelial layer may be migrating over the cut edge toward the anterior cornea while retaining the differentiated phenotype of a barrier layer as has been observed in rabbit corneas.42,43 Figure 3 shows an example of this in a TTHX1114-treated cornea, with the endothelial cell layer extending down the cut edge toward the anterior cornea. The retention of the ZO-1-positive cell/cell contacts can be seen along with an increase in the area of the CECs.
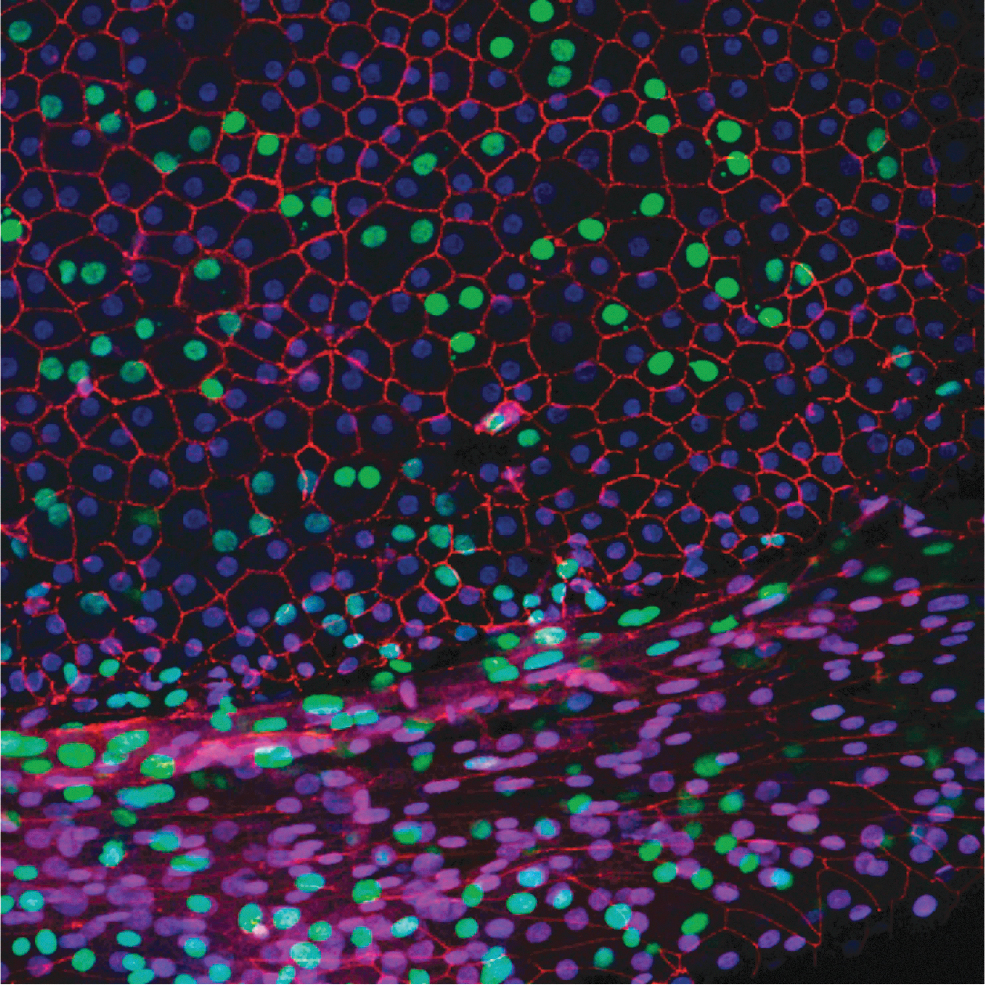
Confocal Z-stack illustrating the endothelial layer at the wound edge migrating down the wound surface toward the anterior corneal surface. The posterior (endothelial) corneal surface is at the upper left. The corneal quarter (from cornea 0170R) was incubated with TTHX1114 (100 ng/mL) for 48 h and stained for nuclear DNA (blue), EdU (green), and ZO-1 (red) as described in materials and methods.
At 48 h, labeled cells were seen as single cells or often as a pair of cells next to one another, suggesting that these pairs may be daughter cells. When corneas were incubated for 7 days rather than 48 h with EdU and TTHX1114, a larger number of cells became labeled (Fig. 4).
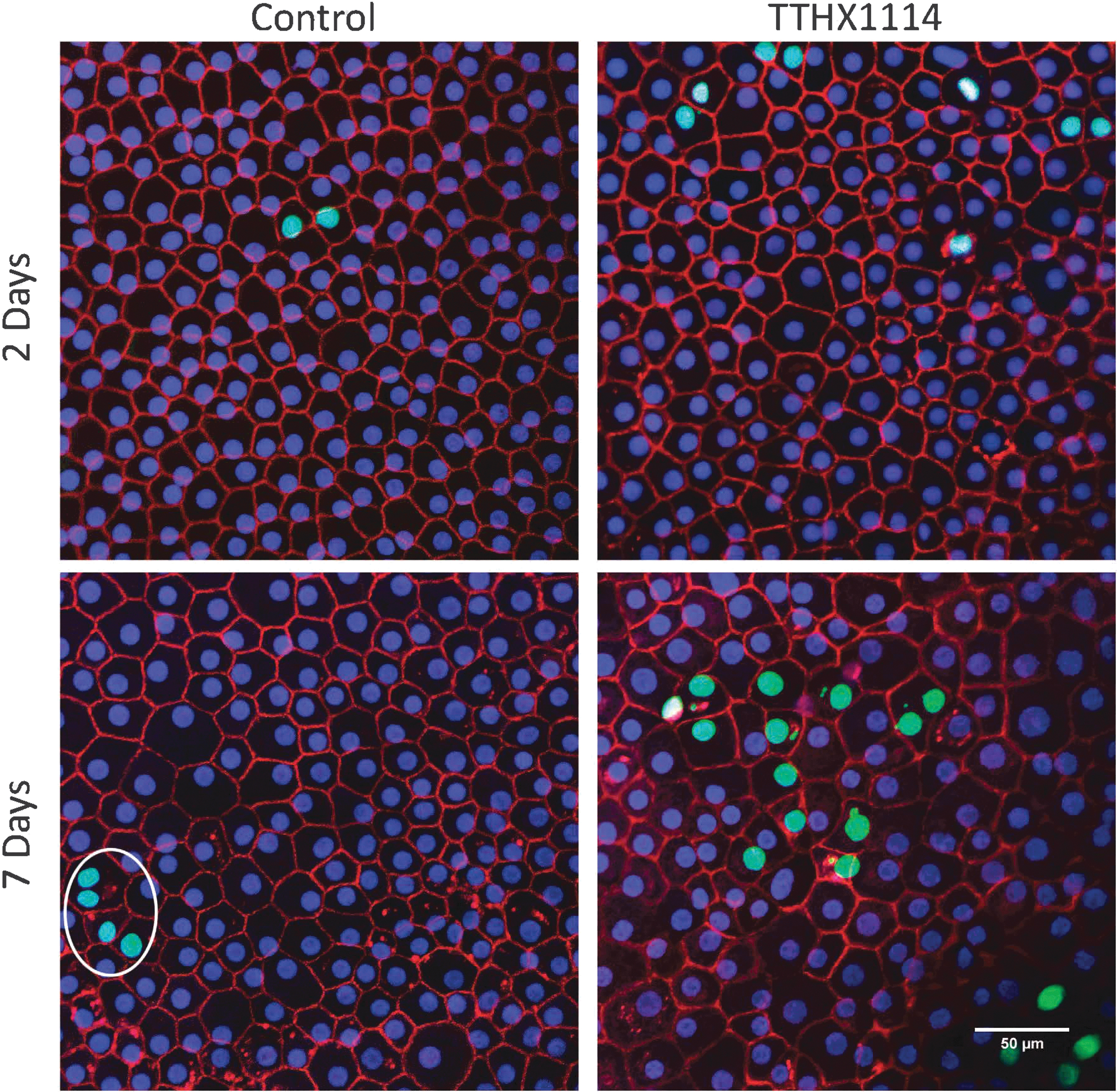
Proliferating CECs in the mid zone form clusters over time. Photomicrographs of corneas maintained in organ culture for 2 (top) and 7 (bottom) days in the continuous presence of EdU with (right) or without (left) TTHX1114 (100 ng/mL). An example of a cluster is circled. Images in the figure were of corneas 0206R (2-day Mid control), 0170R (2-day Mid TTHX), 0170L (7-day Mid control), and 0170L (7-day Mid TTHX).
In the mid zone after 7 days, small clusters of labeled cells, defined as 3 or more cells bordering one another, were observed as well as single and paired cells. The majority of cells in the mid zone remained unlabeled.
At both the 2 and 7 day time points, the endothelial cells in the mid zone continued to display a normal CEC morphology, remaining in a monolayer as a hexagonal matrix with well-defined ZO-1-rich cell boundaries. The size of the cells did not appear to change, and they expressed the functional ZO-1 marker in the same subcellular localization as untreated cells. The proliferating (EdU incorporating) cells had similar morphology to the nonproliferating cells.
At the wound edge, increased number of cells were labeled at 7 days (Fig. 5). Like the mid zone, proliferating cells formed clusters, and the clusters appeared to become larger with longer incubation times. Single and paired labeled cells were still observed along with the clusters.
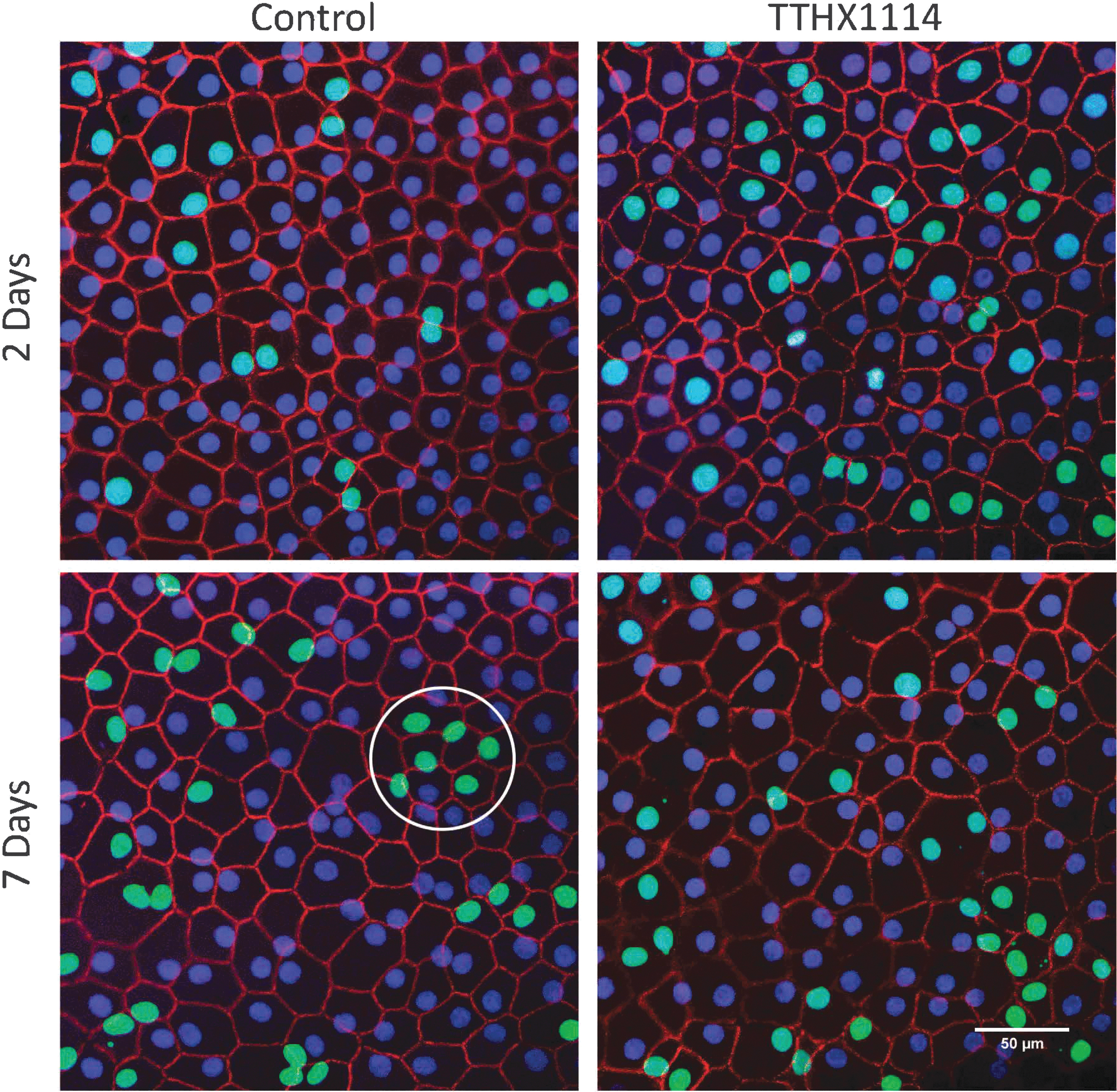
Proliferating CECs at the edge form clusters over time. Photomicrographs of corneas maintained in organ culture for 2 (top) and 7 (bottom) days in the continuous presence of EdU with (right) or without (left) TTHX1114 (100 ng/mL). An example of a cluster is circled. The wound edge is outside the image area for all 4 images. Images in the figure were of corneas 0206R (2-day Edge control), 0170L (2-day Edge TTHX), 0206R (7-day Edge control), and 0206L (7-day Edge TTHX).
Both in the mid zone and at the wound edge, clusters were observed to form contiguous areas in the monolayer in some cases. Distinct, separated clusters as large as 8 cells were observed at 7 days.
Quantification of the number of cells incorporating EdU showed that in both mid zone and edge at 7 days, the number of cells incorporating EdU at 7 days in control corneas remained small and did not appreciably increase from 2 to 7 days (mean ± sem of 6.84 ± 3.45 at 2 days vs. 7.51 ± 3.45 at 7 days, P = 0.73 by paired t-test) while treatment with TTHX1114 resulted in an increase from 2 to 7 days that was not statistically significant (mean ± sem of 8.61 ± 4.19 at 2 days vs. 14.53 ± 3.81 at 7 days, P = 0.066 by paired t-test) (Fig. 6). Even in corneas displaying no incorporation of EdU at 2 days, TTHX1114 treatment resulted in some labeling at 7 days.
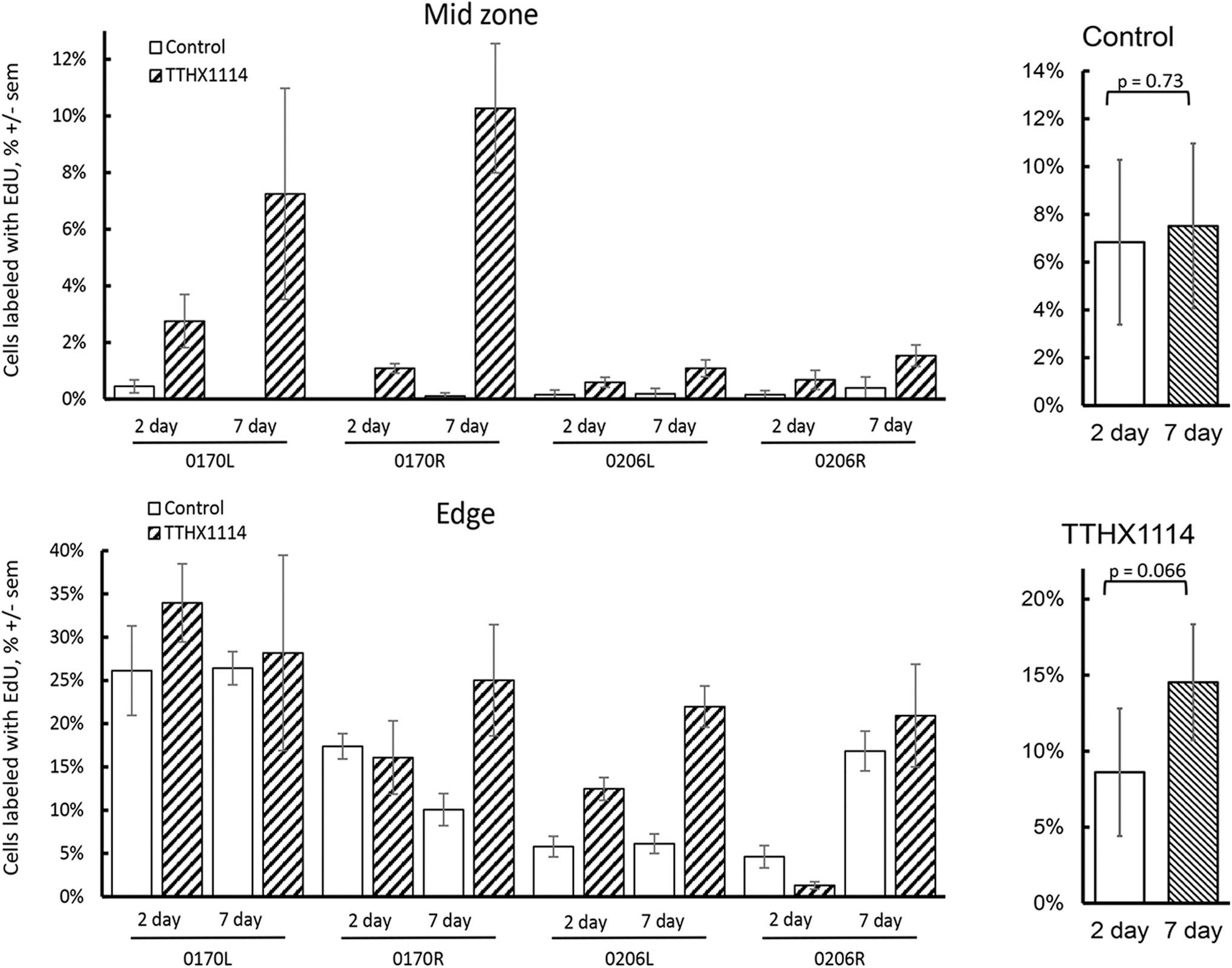
EdU incorporation with 2 versus 7 days incubation. Corneas were cut into quarters with 2 quarters incubated for 2 days and 2 for 7 days in the presence of both EdU and TTHX1114. Right panels, comparison of the mean of the 2 and 7 day % EdU incorporation in TTHX1114 treated versus control (P values using paired t-test).
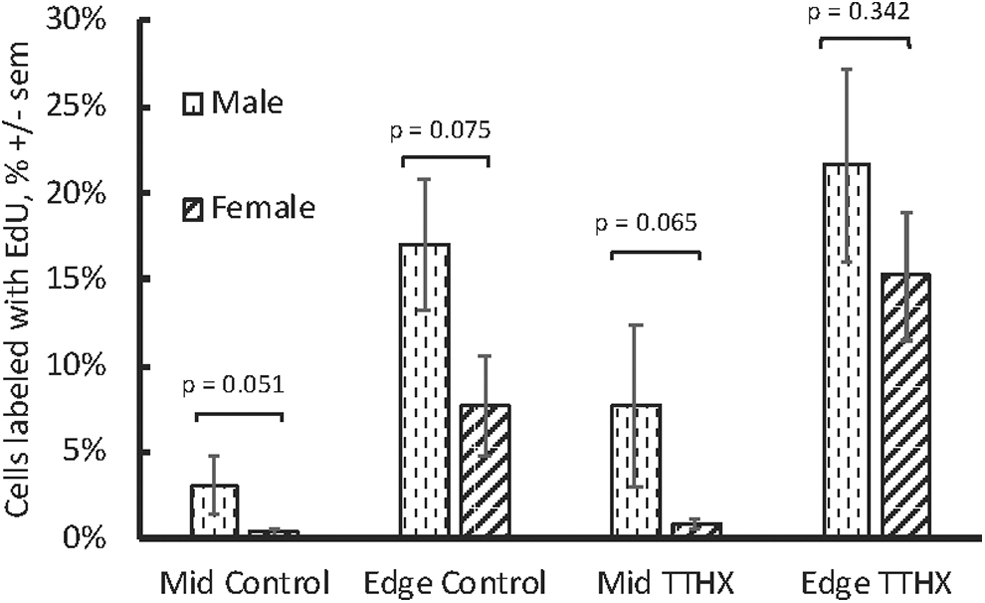
The rate of proliferation and the degree of stimulation by TTHX1114 was examined for possible correlations to donor age, baseline CEC density, sex, and time from death to culture (duration of storage in Optisol). There was no statistically significant correlation with any variable. Proliferation was greater in corneas from male donors than female donors generally (Fig. 7), but these differences were not statistically significant, nor was there a statistically significant difference in the response to TTHX1114 stimulation.
Discussion
The goal of these experiments was to ascertain whether CECs from normal human corneas can be stimulated to proliferate in organ culture where the low basal proliferation may better reflect the in vivo situation 34 compared with dissociated culture systems that produce high levels of proliferation of normal and dystrophic/FECD CECs.7,8,44–46
In this human corneal organ culture model, unstimulated CECs in the mid zone had very few cells incorporating EdU, indicating that this model is more reflective of the in vivo situation where the number of cells changes only slowly.
In the absence of TTHX1114, CECs at the wound edge had higher rates of EdU incorporation than in the mid zone. The CECs at the wound edge were also more susceptible to stimulation by TTHX1114, consistent with stimulation of the wound healing response. In some corneas, the response to wounding was as large as the response to TTHX1114, but in most, the response to TTHX1114 was greater than the response to wounding. In a few corneas with robust wound responses, TTHX1114 did not increase EdU incorporation beyond wounding. This is consistent with other studies of FGFs, where maximal responses are seen in the context of cellular stress or damage.
EdU incorporation per se does not measure proliferation but rather DNA synthesis and the progression of the cell through S phase, and the cells could arrest in G2. Given the small proportion of cells that do incorporate EdU, quantitation of proliferation by cell counting in this short time frame is challenging. In dissociated culture, incorporation of BrDU by CECs is reflective of proliferation. 13 TTHX1114 stimulation of CECs increases EdU incorporation and cell numbers in parallel, although it should be noted that in the absence of contact inhibition, a far greater proportion of the CECs incorporate EdU and proliferate (data not shown). A number of studies have shown that CECs are arrested in G147–49 although there may also be a population of very slow cycling cells not likely to be seen here. 50 Based on these observations the increased EdU incorporation likely indicates proliferation.
TTHX1114-treated cells regardless of proliferative responses or location in either the mid zone or the edge, retained the phenotype of endothelial cells, including remaining in a monolayer in a hexagonal array with the cell/cell junctions staining for ZO-1. No differences between drug-treated and untreated cells were observed in the pattern of ZO-1 staining or cell hexagonality. This suggests that under these organ culture conditions the endothelial cells retain functional integrity and that treatment with TTHX1114 does not impact function at least over these short time frames.
Epithelial cells on the anterior side of these corneas would be expected to migrate down the wound edges and could complicate this analysis. In similar organ cultures, it has been shown that the epithelial and endothelial layers meet and that both cell types appear not to invade the other's territory. 42 Epithelial cells are distinguishable from endothelial cells by the pattern of ZO-1 staining51,52 and while these cells are occasionally observed they were not counted in our analysis.
Since both wounding and treatment with TTHX1114 appeared to stimulate proliferation and since the effects appear to be independent and additive, they may operate through independent mechanisms. Release from contact inhibition may play a role in the stimulation of CECs at the wound edge.12,13,53 Studies of transplantation with fragments of Descemet's54,55 with wound edges similar to those in this study indicate that CECs can migrate from the wound edge to seal wounds and cover relatively large areas of cornea. In this study, no relationship was found between the CEC density at the time of tissue donation/harvesting by the eye bank and proliferation, suggesting that the release from contact inhibition requires large changes in cell density such as those found at a wound edge. The stimulation by TTHX1114 likely operates through stimulation of FGF receptors on the CECs. It is possible that the 2 mechanisms interact, as wounding may alter the expression of FGF receptors.
After 7 days of stimulation there remains a residual population of cells that have not responded. Responsiveness to wounding or TTHX1114 may be (1) random at the cellular level or (2) revealing a pre-existing proliferation-competent CEC population. These 2 models can be differentiated by the pattern of labeled CECs after allowing sufficient time for several rounds of division stimulated by TTHX1114. In a random model, the expectation after prolonged stimulation would be that the labeled cells would occur in pairs, while in the proliferation-competent model, the expectation would be that the labeled cells would occur in contiguous groups as the stimulated proliferation-competent cells undergo repeated rounds of division. The presence of clusters of labeled cells at longer incubation times supports the proliferation-competent model. The potential that the CEC layer contains a population of proliferation-competent or progenitor cells has been suggested by several lines of evidence, including the existence of stem or proliferative markers in the CEC layer and the ability to generate proliferative colonies of CECs in dissociated cell culture with appropriate stimuli.51,56–59 It has been suggested that progenitor cells may occupy a niche in the trabecular meshwork (TM) or in the transition zone between the TM and the endothelial monolayer.60,61 The observation here of clusters of EdU-incorporating cells in the mid and central cornea away from both the TM and the wound edge suggests that proliferation-competent cells exist distributed across the CEC layer and are not localized to a niche at the periphery of the cornea. Increased proliferation at the wound edge indicates that release from contact inhibition induces proliferation competence.
The largest distinct cell cluster size at 7 days was 8 cells suggesting that the responsive cells in the clusters undergo a complete cell cycle in about 2 days. This is consistent with the cycle time observed in dissociated cell culture. 8 The consistent appearance of smaller clusters, including pairs of cells at all time points, suggests that either some cells have a much longer cell cycle or that cells can enter and exit the cell cycle under these conditions. Pulse-chase experiments in the mouse have confirmed the presence of slow cycling cells, but it is not known if these are present in the human. 50
An independent modulator of the proliferation and the response to stimulation in these corneas may be the sex of the donor. Proliferation rates were quite variable between corneas, but consistently greater in corneas from male donors under all conditions. FECD is more common in females than males. 62 However, donor sex does not appear to affect transplant success rates63–65 or CEC density after transplant. 66 This observation needs to be explored further.
A significant limitation of this study is the variability between corneas in the basal level of proliferation of the CECs. Variability between left and right corneas from the same individual was less than variability between individuals. The observation that the level of proliferation is correlated between corneas from the same individual suggests that the mechanisms regulating CEC proliferation in vivo are tissue specific and remain operative in organ culture. The process of tissue collection, storage, and retrieval may contribute to the observed variability. 67 While corneas for transplant have high levels of viability and transplants are generally successful after similar storage times, 68 little data exist on the ability of corneas rejected for transplant for various reasons to withstand the process. CECs undergo apoptosis on storage and CECs from FECD patients are hypersensitive to a variety of stressors.69–71 Examination of factors related to the eye banking process showed that there was a trend toward lower proliferation, as the corneas were stored longer before culture began, but this trend was not statistically significant. It seems most likely that the observed variation is an inherent variability between corneas.
Stimulation of proliferation of endothelial cells has potential utility in the treatment of endothelial dystrophies, including FECD. FECD is characterized by a loss of endothelial cells and the goal of the current therapy, transplantation, is to restore a higher endothelial cell count. Therefore, a drug that increased endothelial cell numbers could have utility in FECD. As a protein, the delivery of TTHX1114 to the target tissue may not be feasible as an eye drop due to limited penetration of the cornea and the binding of the drug to the heparan sulfate proteoglycans (HSPGs) and ECM components in the corneal stroma. For this reason, intracameral injection is the intended clinical route, providing direct contact of the endothelial cells with the drug. HSPGs are upregulated in FECD 72 potentially providing a depot effect that is enhanced in diseased corneas.
These experiments show that CECs from normal corneas can be stimulated to proliferate by wounding or treatment with TTHX1114, and that the 2 operate independently and additively to drive proliferation.
Footnotes
Acknowledgments
The authors would like to thank Steve Crockett for statistical advice, Tony Wong for histopathology advice and services, and the SDSU EM-Imaging Facility for use of their confocal microscope. In addition, the authors would like to thank the donors of eyes and the Lions Eye Institute for Transplantation and Research for providing corneas.
Author Disclosure Statement
D.E. reports personal fees from Trefoil Therapeutics, during the conduct of the study; personal fees from Trefoil Therapeutics, outside the submitted work. In addition, D.E. has a patent 15/024,824 pending and D.E. holds stock in Trefoil Therapeutics, Inc. S.P., J.W., and J.J.-E. report personal fees from Trefoil Therapeutics, during the conduct of the study; personal fees from Trefoil Therapeutics, outside the submitted work. R.A.B. reports personal fees from Trefoil Therapeutics, during the conduct of the study; personal fees from Trefoil Therapeutics, outside the submitted work; In addition, R.A.B. holds stock in Trefoil Therapeutics, Inc.
Funding Information
Support: National Institutes of Health, National Center for Advancing Translational Sciences, Therapeutics for Rare and Neglected Diseases CRADA# 2016-04.
