Abstract
Abstract
Purpose:
A new gel formulation containing loteprednol etabonate (LE), a C-20 ester corticosteroid used to treat ocular inflammation, was developed to provide increased retention on the ocular surface for improved drug delivery to intraocular tissues. This investigation evaluated concentrations of LE in tear fluid following topical instillation of LE gel to humans and the ocular and systemic pharmacokinetics of LE following administration to rabbits.
Methods:
LE ophthalmic gel 0.5% was administered as a single topical dose to human volunteers (n=12) and Dutch Belted rabbits (n=40). In the human study, tear sampling was performed at 6, 9, 12, and 24 h after instillation. In the rabbit study, tears and ocular tissues were collected from 5 min through 24 h postdose. Serial blood samples were collected from one cohort of rabbits for plasma analysis. Concentrations of LE were determined by high performance liquid chromatography tandem mass spectrometry.
Results:
In humans, LE was detected in tears at all the time points assessed with mean concentrations of 114 μg/g at 6 h declining to 2.41 μg/g at 24 h postdose. In rabbits, LE was detected in all ocular tissues within 5 min after dosing. Maximum concentrations of LE were achieved within 0.5 h and were highest in tear fluid (1560 μg/g), followed by bulbar conjunctiva (4.03 μg/g), cornea, (2.18 μg/g), iris/ciliary body (0.162 μg/g), and aqueous humor (0.0138 μg/mL). LE remained measurable in all ocular tissues through 24 h with the exception of aqueous humor. In contrast, plasma levels of LE were low with no detectable levels after 4 h.
Conclusions:
The gel formulation of LE provided prolonged exposure to LE on the ocular surface, with measurable levels in tears through 24 h in both humans and rabbits, for delivery of LE to anterior segment tissues, as evidenced by sustained levels of LE in rabbit conjunctiva, cornea, and iris/ciliary body.
Introduction
I
Upon instillation, a drug immediately mixes with the tear fluid and is diluted. The average tear volume in humans is ∼7 μL,4,7,8 but the eye is capable of accommodating up to 30 μL in the cul-de-sac for a short period of time.3,8,9 Under normal conditions, tear fluid is eliminated and replaced at a rate of ∼1 μL/min (16% min−1).7–12 However, following drug administration, the elimination rate may increase substantially.8,13–15 Most commercial eyedrop bottles dispense between 30 and 50 μL of fluid per drop.6,8,16–18 Since the amount of fluid administered generally exceeds the volume that can be retained in the cul-de-sac, a large portion of the dose is eliminated from the eye by spontaneous blinking4,19 and nasolacrimal drainage or by clearance via the capillaries in the conjunctiva.15,18,20 Furthermore, if the drug is irritating to the cornea or conjunctiva, the trigeminal nerve stimulates reflex tearing, resulting in additional dilution and increased elimination.21,22 As a result of these clearance mechanisms and the physical barriers provided by the cornea, the amount of drug that reaches intraocular tissues following topical dosing is limited. It is estimated that typically <5% of the administered dose reaches its intended target. 23 For this reason, optimizing drug delivery continues to be a significant focus of research.
Loteprednol etabonate (LE) is a potent corticosteroid that was developed using a retrometabolic design in which an inactive metabolite of a known drug is chemically altered to create a new biologically active molecule. Under physiological conditions, the new drug is converted to an inactive metabolite(s) after it produces a local therapeutic effect.18,24,25 LE is derived from Δ 1 -cortienic acid, an inactive metabolite of prednisolone, and unlike other corticosteroids, it has a chloromethyl ester at the carbon-20 position instead of a ketone group (Fig. 1). Following ocular administration, any LE that is not bound to the glucocorticoid receptor is quickly metabolized to the inactive metabolites, Δ 1 -cortienic acid etabonate and Δ 1 -cortienic acid, by ester hydrolysis, thereby minimizing the potential for elevating intraocular pressure (IOP), a common and often limiting side effect of topical corticosteroids.18,26 Several studies have shown that compared to C-20 ketone corticosteroids, LE has a much lower propensity to induce IOP elevation.27–29 In addition, the C-20 ketone group, present in all other topical ophthalmic corticosteroids, has been reported to form adducts with lysine residues in lens protein30,31 which is believed to be a mechanism for the formation of cataracts, another common side effect of corticosteroid use. 26 Since LE lacks this functional group, cataract formation by this mechanism is not possible, although other mechanisms cannot be ruled out.
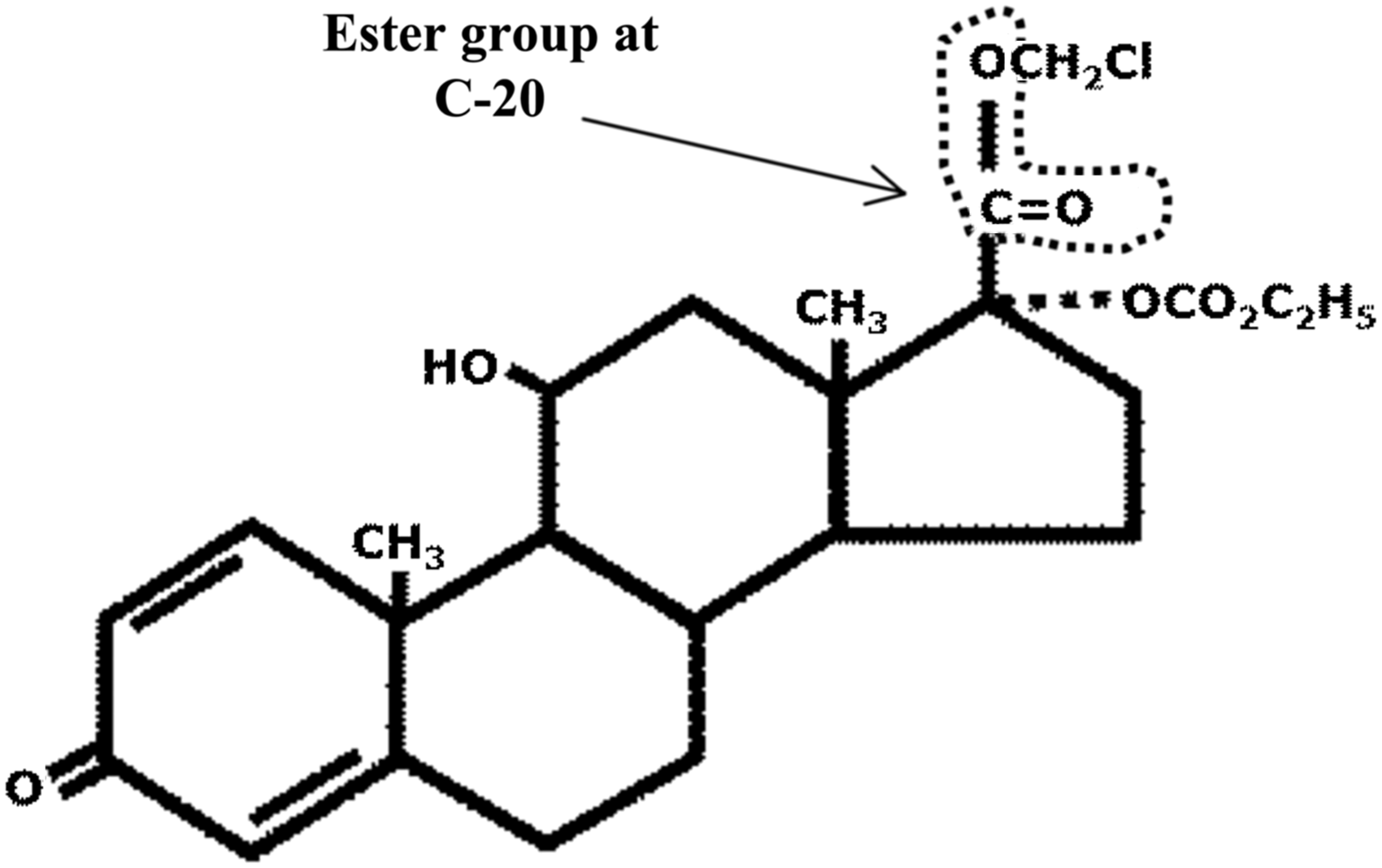
Two-dimensional chemical structure of loteprednol etabonate. Unlike other corticosteroids, loteprednol etabonate has an ester function at the carbon-20 position (arrow) instead of a ketone group.
Previously marketed in the United States in a variety of suspension formulations (Lotemax®, Alrex®, and Zylet®) and in an ointment formulation (Lotemax ointment), a novel gel formulation of LE, Lotemax gel (LE ophthalmic gel, 0.5%) was recently approved by the Food and Drug Administration for the treatment of postoperative pain and inflammation following ocular surgery. 32 Designed to adhere to the ocular surface to maximize exposure to LE in the anterior segment tissues, the new gel formulation contains polycarbophil, a mucoadhesive gel-forming polymer. 33 The gel formulation is nonsettling and delivers a consistent amount of drug with each dose without the need to shake the bottle before instillation. 33 Additionally, formulation attributes, such as a low concentration of benzalkonium chloride (BAK), an osmolality and pH that are close to that of normal tears, and the inclusion of known demulcents, are thought to improve patient comfort. The efficacy and safety of LE ophthalmic gel 0.5% were demonstrated in 2 randomized, double-masked, vehicle-controlled studies in patients following uncomplicated cataract surgery.34,35 In each study, patients self-administered either LE gel or vehicle 4 times per day for 14 days following surgery. At postoperative Day 8 and 15, a significantly greater proportion of patients treated with LE gel were pain free and had complete resolution of anterior chamber cells compared to patients treated with vehicle (P<0.001). More than 85% of patients reported no drop sensation and ≤1% of patients reported symptoms of ocular discomfort (eg, dryness, itching, discharge) or drug-related blurring following instillation of either LE gel or vehicle, confirming that the new formulation is well tolerated.
The purpose of this investigation was to evaluate the pharmacokinetic (PK) properties associated with the LE gel formulation. To do this, concentrations of LE in tear fluid were evaluated following topical instillation of LE ophthalmic gel 0.5% to healthy volunteers. Additionally, the ocular and systemic PK of LE were characterized following a single topical ocular administration of LE ophthalmic gel 0.5% to rabbits.
Methods
Materials
Human subjects received commercially available Lotemax (LE ophthalmic gel, 0.5%) manufactured by Bausch & Lomb, Inc. The formulation used in the rabbit study was identical to the commercial LE gel formulation but was from a premarketing lot. All other materials were purchased from commercial sources.
Tear concentrations in human volunteers
Exposure to LE in human tear fluid was assessed as part of a single-center open-label study conducted at the Bausch & Lomb Research Clinic (Bausch & Lomb, Inc.) in accordance with Good Clinical Practices, the International Conference on Harmonization guidelines, the Declaration of Helsinki, and the Health Insurance Portability and Accountability Act guidelines. The study was approved by the Institutional Review Board before initiation, and each subject provided informed consent before enrollment. Subjects were at least 18 years of age with best-corrected visual acuity (VA) of 20/20 and normal IOP in both eyes with no active ocular disease or allergic conjunctivitis. Subjects were instructed not to use any topical ocular medications or systemic medications other than acetaminophen during the study. Eligible subjects had to have a Schirmer test (without anesthesia) of ≥10 mm within 5 min at screening.
The study duration ranged from 3 to 9 days from screening until study exit. Subjects were screened at Visit 1 (Day-7 to Day-1), which included VA, biomicroscopy, and Schirmer assessments. On Visit 2 (Day 1), subjects who met eligibility requirements received a single topical drop (∼40 μL) of LE ophthalmic gel 0.5% directly from the bottle into the conjunctival sac of either the right eye or left eye according to a randomization schedule, and tear samples were collected from the study eye of each subject on a Schirmer strip at 6 h (±20 min), 9 h (±20 min), and 12 h (±30 min). Since the objective was to determine if LE ophthalmic gel provides sustained ocular surface exposure to LE, sample collection was not initiated until several hours after dosing. Also, a limited sampling regimen was used to allow tear fluid volumes to return to normal between serial sample collections. Schirmer strips (TearFlo™ Sterile Schirmer strips; Eagle Vision) were inserted into the lower conjunctival sac over the junction of the temporal and central one third of the lower eyelid margin until tear fluid wet 10 mm of the strip in a maximum of 5 min. On Visit 3 (Day 2), subjects returned to the clinic and tear samples were collected on a Schirmer strip at 24 h (±30 min) after the Visit 2 gel instillation. The weight of each tear sample was determined by weighing the Schirmer strip and collection tube before and after sample collection, and tear fluid samples were stored frozen at −80°C until analysis. Subjects were exited following Visit 3.
No formal sample size calculation was performed a priori. It was assumed that 12 subjects would provide sufficient data to assess the tear levels of LE following instillation of LE ophthalmic gel, 0.5%.
Ocular and systemic PKs in rabbits
Male Dutch Belted (pigmented) rabbits (Covance Research Products) ∼4 months of age and weighing between 1.7 and 2.1 kg were used in the rabbit PK study. All in-life procedures were conducted at PharmOptima. Rabbits were individually housed under controlled environmental conditions and offered food and water ad libitum. The study was conducted in accordance with the Association for Research in Vision and Ophthalmology statement for the Use of Animals in Ophthalmic and Vision Research, and the Institute of Laboratory Animal Resources Guide for the Care and Use of Laboratory Animals. The protocol was reviewed and approved by the Institutional Animal Care and Use Committee of the test facility before the start of the study.
A comprehensive health assessment, including an examination of the anterior segment of the eye, was performed on all rabbits before inclusion on the study. The ophthalmic evaluation was conducted on both eyes using slit-lamp biomicroscopy and indirect ophthalmoscopy to confirm that there were no preexisting abnormalities of the cornea, conjunctiva, episcleral vessels, anterior chamber, pupil, or lens that would compromise the results of the study. Observations were recorded according to the McDonald–Shadduck Scoring system. 36 Only rabbits that were confirmed to be in good physical condition and free from any overt ocular defects were used in the study.
The rabbits (n=40) were weighed and randomized to one of 10 sampling subgroups. Using a positive-displacement pipette, a single 35-μL topical instillation of LE ophthalmic gel, 0.5% was administered to both eyes of each animal while fully awake to provide a target dose of 175 μg of LE per eye. Immediately after dosing, the eyelids were gently held closed for several seconds to facilitate even distribution of the test substance over the surface of the eye and minimize runoff. At predetermined time intervals (0.0833, 0.25, 0.5, 1, 2, 4, 6, 8, 12, and 24 h) after dosing, the rabbits (n=4 per collection time) were euthanized by intravenous overdose of sodium pentobarbital (Sleepaway®; Fort Dodge Animal Health). Tear fluid, aqueous humor, and bulbar conjunctiva samples were collected from each eye in situ before enucleation. The tear fluid was collected immediately following euthanasia by inserting a Schirmer test strip (Schirmer Tear Test Standardized Sterile Strips; Intervet) into the lower conjunctival sac of each eye for 30 s. The amount of tear fluid collected was determined by subtracting the weight of the Schirmer strip before collection from the weight of the strip after collection. Next, the surface of the eyes was thoroughly rinsed with saline to wash away any residual test material. Aqueous humor was removed from each eye via paracentesis using a 0.5-cc syringe equipped with a 28-gauge needle, and a representative sample of bulbar conjunctiva was collected. Both eyes were then enucleated and flash-frozen in liquid nitrogen. The length of time from insertion of the Schirmer test strips to the time that both eyes were completely frozen was typically less than 9 min. Once frozen, the cornea and iris/ciliary body were dissected away from the globe, collecting each tissue in its entirety. All tissues were rinsed with saline and gently blotted with gauze before being placed into pretared vials, weighed, and stored frozen at −80°C until analysis.
To determine systemic exposure, serial blood samples (∼1 mL each) were collected through an indwelling catheter placed into a central ear artery of rabbits in the 24-h sampling subgroup. Blood samples were collected into syringes pretreated with K3 EDTA, transferred to microcentrifuge tubes, and stored on wet ice before being centrifuged to obtain plasma. Following centrifugation, the entire plasma volume was pipetted into prelabeled tubes and stored at −80°C until analysis.
Bioanalysis
The human tear fluid samples were analyzed using a validated high performance liquid chromatography tandem mass spectrometry (HPLC/MS/MS) method. Briefly, LE was extracted from the tear fluid on Schirmer strips using 1:1 acetonitrile: water (v/v). The volume of solvent added was adjusted for each individual sample to ensure all samples were extracted at a ratio of 10 mg of tear fluid to 1 mL of 1:1 acetonitrile: water. Following extraction, an aliquot of the sample was added to a 96-well plate along with the 2 H5-LE internal standard in the extraction solvent. The 96-well plate was vortexed for 30 s and then analyzed by HPLC/MS/MS. Sample extracts were analyzed against a standard curve prepared with 1:1 acetonitrile: water. Quality control samples were prepared using human tear fluid. The HPLC system consisted of LC-10AD pumps and a SCL-10A system controller, both from Shimadzu Scientific Instruments, Inc., along with a CTC PAL autosampler from LEAP Technologies with a 20-μL PEEK loop. The chromatography was conducted using a Thermo Hypersil Gold C18 (2.1×50 mm, 5 μm) column from Thermo Scientific. The triple quadrupole mass spectrometer used was an AB Sciex API 3000 with a TurboIonSpray® source in Selective Reaction Monitoring (SRM) mode. The lower limit of quantitation was 0.0200 μg/g.
Human tear concentration data were summarized using descriptive statistics only. Arithmetic means, SD, median, min, and max values were determined for each collection time point. Samples were considered outliers and excluded from the final summary if concentration values were collected outside the corresponding time window or if sample concentration values were below the limit of quantitation and at least 10-fold below the median, or if sample concentration values were more than 10-fold above the median concentration.
The rabbit ocular tissues and plasma were also analyzed by a HPLC/MS/MS method. All samples were thawed to room temperature before extraction. Tear fluid, bulbar conjunctiva, cornea, and iris/ciliary body samples were processed by adding a variable amount of 1:1 acetonitrile: water (v:v) mixture to each vial using a Tecan Freedom EVO 150 from Tecan Group Ltd. The volume of acetonitrile: water was adjusted for each sample based on the individual sample weight to ensure a constant matrix concentration for all samples, standards, and quality control samples. All samples were sonicated and vortexed. An aliquot of the extracted sample was added to a 96-well plate along with internal standard for analysis. Acetonitrile: water was not added to the aqueous humor or plasma samples. Aqueous humor and plasma samples were subject to protein precipitation by the addition of acetonitrile to a 96-well plate and internal standard. Calibration standards were prepared at a minimum of 8 concentration levels by spiking known amounts of each analyte into the appropriate blank pigmented rabbit tissue extract. Quality control samples were prepared in a like manner at 4 concentration levels. Blank pigmented rabbit plasma was used to prepare the calibration standards and quality control samples for plasma analysis. The lower limit of quantitation for ocular tissues was between 0.000100 μg/mL (aqueous humor) and 0.0200 μg/g (tear fluid) based on the average weight of each tissue. The lower limit of quantitation for plasma was 0.100 ng/mL. The HPLC system consisted of Shimadzu LC-20AD pumps and a CBM-20A controller, both from Shimadzu Scientific Instruments, Inc. The autosampler used was a CTC PAL from LEAP Technologies. The chromatography was conducted on a Gemini C6-phenyl (4.6×150 mm, 5 μm) column from Phenomenex. Two triple quadrupole mass spectrometers (both from AB Sciex) with a TurboIonSpray source in SRM mode were used: an API 4000 QTRAP for tear fluid, bulbar conjunctiva, cornea, and iris/ciliary body samples, and an API 5500 for aqueous humor and plasma samples. 2 H5-LE was also used as the internal standard for the analysis of the rabbit ocular tissues and plasma.
PK analysis of rabbit data
PK analysis of rabbit ocular tissue and plasma samples was performed using noncompartmental methods in WinNonlin Professional® (version 5.3; Pharsight Corporation) using nominal sample collection times. Since it is not possible to collect more than one sample of each tissue per eye, the mean concentration of LE at each time point was calculated for each ocular tissue. From these data, a mean composite concentration versus time profile was generated and subsequently used for PK analysis. Individual ocular tissue samples with a measured concentration that was more than 10-fold higher or lower than the median concentration in the respective sample pool was considered an outlier and not included in any calculations. For plasma, individual concentration versus time data from serial sampling was used for PK analysis.
The maximum concentration of LE (Cmax) and the time at which the maximum concentration was observed (Tmax) were determined directly from the concentration versus time profiles. The area under the concentration vs. time curve (AUC0–t), where t represents the time of the last measurable concentration and mean residence time (MRT), were calculated using the log/linear trapezoid method in WinNonlin. The concentration of LE in tear fluid at time zero was determined by log-linear regression analysis of the first 2 data points and extrapolation to time zero (intravenous bolus model). Concentrations of LE in the remaining ocular tissues and plasma at time zero were assumed to be zero (extravascular model).
Results
LE concentrations in human tear fluid
Seventeen subjects were screened and 12 subjects were randomized. All 12 randomized subjects completed the study. The mean (SD) age of subjects was 45.6 (11.4) years, and all but one was male. One subject was Asian while the rest were Caucasian. All subjects had tear samples collected within the specified time windows.
Figure 2 shows the mean LE concentrations in human tear fluid after a single topical instillation of LE ophthalmic gel 0.5% to healthy volunteers in comparison to LE concentrations observed in rabbits. Excluding outliers, mean (SD) LE levels in human volunteers were 114 (116) μg/g at 6 h, 19.7 (18.2) μg/g at 9 h, 26.9 (29.6) μg/g at 12 h, and 2.41 (3.36) μg/g at 24 h. LE tear concentrations from 2 subjects taken at the 9 h time point and 1 subject taken at the 24 h time point were determined to be outliers because the values were >10-fold above the median concentration. LE tear concentrations were similar when outliers were included in mean calculations.
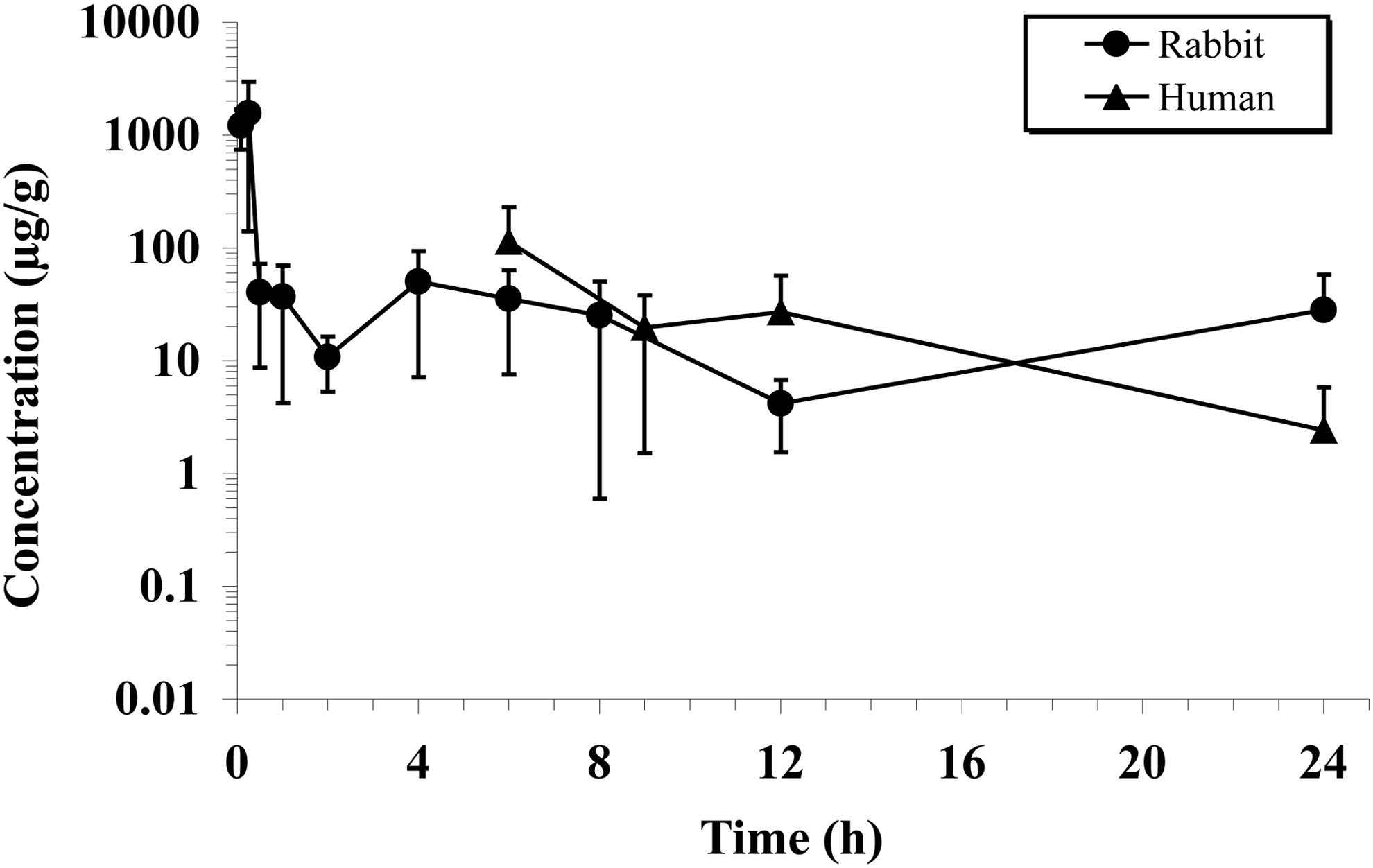
Loteprednol etabonate (LE) concentration versus time profiles through 24 h in the tear fluid of rabbits and humans following topical ocular administration of LE gel, 0.5%. A single tear fluid sample was collected from each rabbit eye beginning 5 min after dosing. A total of 4 tear fluid samples were collected from the treated eye of all human subjects beginning 6 h after dosing. Data represent mean (±SD) concentrations from 8 rabbit eyes and 10–12 human eyes per collection time, excluding outliers.
Ocular and systemic PKs in rabbits
Ocular and systemic PK parameters for LE following topical ocular administration of LE ophthalmic gel, 0.5% to rabbits are presented in Table 1. Based on the aforementioned exclusion criteria, concentrations of LE in 1 cornea, 1 conjunctiva, and 2 aqueous humor samples were determined to be outliers and not included in the PK analysis. Although concentrations of LE in 4 tear fluid samples also met the outlier criteria, there was considerable variability in the data set, and the observed coefficient of variation (CV%) at each collection time was ∼80%, on average. Therefore, these data points were included in the PK analysis.
Units for aqueous humor are μg/mL and μg·h/mL for Cmax and AUC(0–24h), respectively.
Units for plasma are ng/mL and ng·h/mL for Cmax and AUC(0–24h), respectively.
Cmax, AUC(0–24h), and MRT values are presented as mean (±SD) for plasma (n=4).
Median Tmax reported for plasma (n=4).
MRT, mean residence time.
LE was detected in all ocular tissues within 5 min of dosing. Based on Cmax and AUC(0–24h) values, exposure to LE was the highest in tear fluid, followed by bulbar conjunctiva, cornea, iris/ciliary body, and aqueous humor. In all of the tissues examined, maximum concentrations of LE were achieved within 30 min of dosing. As expected, LE levels in the tear fluid decreased substantially (∼97% of Cmax) within the first 30 min of dosing but remained relatively constant over the next several hours (Fig. 2). At 8 h postdose, concentrations of LE were ∼30% lower than the levels observed at 30 min. Although the concentration of LE at 24 h appeared nearly identical to the concentration at 8 h, there was considerable interanimal variability at the 24-h time point, with the observed CV% in excess of 100%. Measurable concentrations of LE were also present in the bulbar conjunctiva, cornea, and iris/ciliary body throughout the 24 h sampling period, whereas levels in the aqueous humor were detected through 12 h after dosing (Fig. 3). MRTs ranged from 1.9 h in aqueous humor to 9.4 h in the cornea.
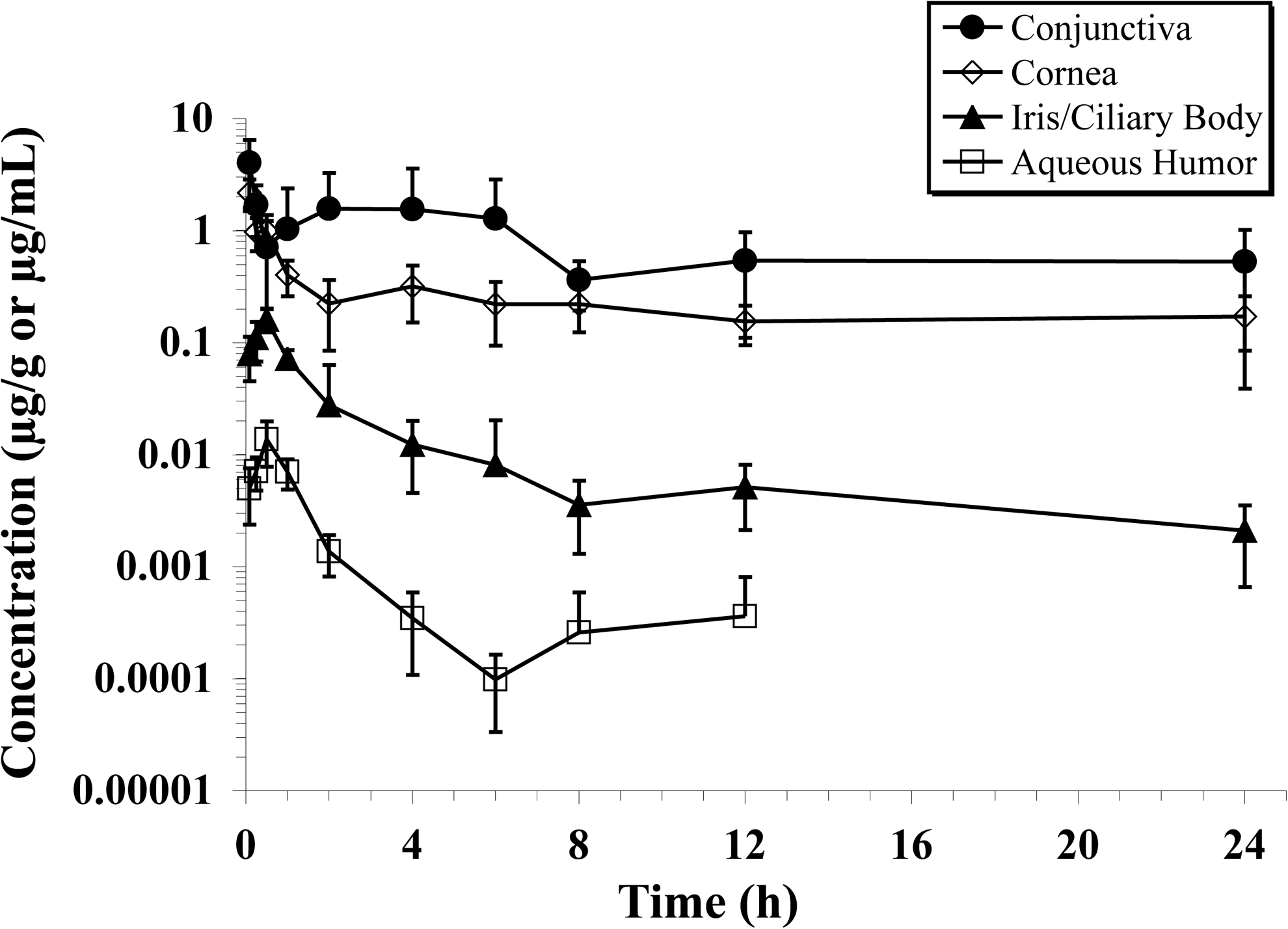
Loteprednol etabonate concentration versus time profiles in the bulbar conjunctiva, cornea, aqueous humor, and iris/ciliary body of rabbits following a single 35 μL topical ocular administration of LE gel, 0.5% (175 μg) to each eye. Data represent mean (±SD) concentrations from 6 to 8 eyes per collection time, excluding outliers. Concentrations of loteprednol etabonate in the aqueous humor were below the limit of quantitation (LLQ=0.100 ng/mL) in all animals by 24 h postdose (data not shown).
LE was detected in plasma through 4 h after a single topical administration, with peak concentrations achieved within 1.5 h of dosing. As depicted in Fig. 4, exposure to LE in plasma was low but highly variable, with maximum concentrations between 2 and 17 ng/mL. For one animal, LE concentrations within the first 15 min of dosing were at least 15-fold higher than the LE concentrations observed in the other animals during this time frame. Furthermore, by 30 min postdose, the measured concentration of LE in this animal had decreased to less than 4% of the concentration observed at 15 min. The reason for this variability is unclear.
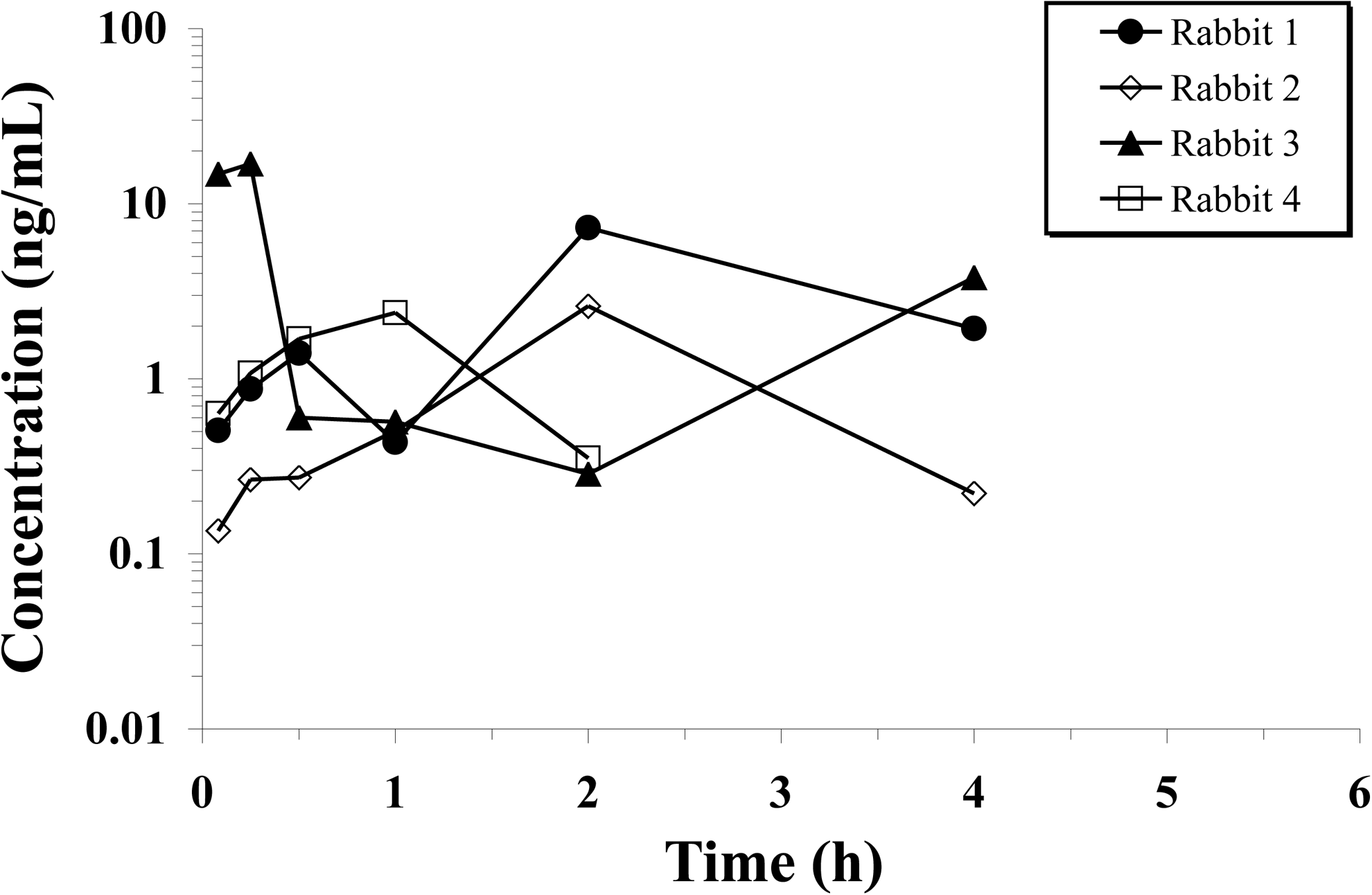
Loteprednol etabonate concentration versus time profiles in plasma from 4 rabbits following a single 35 μL topical ocular administration of LE gel, 0.5% (175 μg) to each eye. Samples were collected over a 24-h period starting 5 min after dosing. Data represent individual concentrations at each collection time. Concentrations of loteprednol etabonate were below the lower limit of quantitation (LLQ=0.100 ng/mL) in all animals by 6 h postdose (data not shown).
Discussion
Topical ocular dosing is the preferred method of drug administration for the treatment of anterior segment diseases of the eye but is often hampered by the eye's protective barriers and clearance mechanisms. To overcome these obstacles, innovative approaches to drug delivery are being investigated on an ongoing basis. The new gel formulation of LE is a polycarbophil-based formulation that is nonsettling and was engineered to provide sustained ocular surface exposure for improved drug delivery to anterior segment tissues. Although a gel in the bottle, the formulation transitions to a shear-thinning, moderately viscous, mucoadhesive suspension upon instillation in the eye, and mixing with tears. 33 The objective of this present investigation was to evaluate the ocular PK of LE following instillation of LE ophthalmic gel, 0.5%. Results in healthy human volunteers showed that LE was present in the tear fluid for 24 h following a single topical dose of LE gel. Results in rabbits were consistent with those from human subjects with significant tear levels likewise present through 24 h postdose. Furthermore, sustained exposure to LE was observed in rabbit anterior segment tissues, including the bulbar conjunctiva, cornea, and iris/ciliary body from 5 min postdose through 24 h postdose. Levels of LE in the aqueous humor were lower and shorter-lived than in the other tissues evaluated, likely due to the rapid ester hydrolysis of LE to inactive metabolites in this compartment, which was not unexpected. Systemic exposure following topical instillation of LE gel was minimal, with no detectable levels in plasma after 4 h.
Results from these studies confirm that LE gel provided prolonged ocular surface exposure, potentially improving bioavailability. In rabbits, maximum concentrations of LE in the tear fluid (1560 μg/g) were achieved 15 min postdose. Based on an average tear volume of 7 μL in rabbits,7,13 the theoretical concentration of LE at time zero following a 35-μL instillation of LE ophthalmic gel, 0.5% is ∼4170 μg/g. Therefore, 37% of the theoretical concentration at time zero was present on the ocular surface after 15 min. Based on the reported tear turnover rate of ∼7% per minute in rabbits, 12 one can estimate that without the mucoadhesive vehicle used in LE gel, namely polycarbophil, most LE would have been eliminated from the rabbit eye after about 15 min. The sustained retention of LE in tear fluid observed with LE gel during this time frame suggests that the polycarbophil-based formulation is adhering to the ocular surface for continuous drug delivery to anterior segment tissues. Tear levels of LE declined after the 15-min postdose time point likely due to precorneal clearance factors and distribution of drug into ocular tissues. The latter premise is supported by measurable levels of LE observed in the conjunctiva, cornea, and iris/ciliary body through 24 h. Anterior segment tissue levels of LE observed following instillation of LE gel appear higher than those reported previously for the suspension formulation of LE in rabbits, albeit from a different PK model. Glogowski and Proksch studied the ocular tissue penetration of LE following instillation of a single 50-μL instillation of LE ophthalmic suspension, 0.5% in Fauve de Bourgogne rabbits with corneal inflammation. 37 Although concentrations of LE in the tear fluid and iris/ciliary body were not evaluated in that study, the mean (SD) maximal concentration of LE in the conjunctiva and cornea was 3.62 (5.47) and 1.40 (1.45) μg/g, respectively, whereas the AUC(0–24h) was 6.10 and 3.30 μg·h/g, respectively. In the current study, which used a smaller dose volume (35 μL), the maximum concentration of LE in the conjunctiva and cornea was 4.03 (2.42) and 2.18 (0.685) μg/g, respectively, whereas the AUC(0–24h) was 18.2 and 5.44 μg·h/g. Thus, it appears that the gel formulation increased maximum concentrations and total exposure in the conjunctiva and cornea, respectively. However, head-to-head studies comparing the LE gel formulation with the LE suspension formulation using the same study design are needed to confirm these PK differences.
The finding of similar concentrations of LE in human tear fluid compared to rabbit tear fluid is interesting given the anatomical and physiological differences between these species that could impact bioavailability. For instance, the surface area of the cornea is ∼50% larger in rabbits than in humans,4,38 and the rabbit cornea is about 20% thinner and the epithelium more permeable than the human cornea. 7 These differences might lead to greater drug absorption from tears in the rabbit eye. In addition, the human cornea has been reported to be more sensitive to mechanical stimuli than the rabbit cornea, 7 and therefore, subject to greater reflex tearing upon drug instillation, hastening dilution, and elimination. Under steady state, the tear turnover rate in humans is faster than in rabbits (16% min−1 vs. 7% min−1, respectively) 39 as is blink rate (360 to 900 times h−1 vs. 4 to 5 times h−1, respectively). 38 Although blinking mixes the drug with tear fluid and distributes it over the surface of the eye potentiating absorption, it also facilitates drug clearance through the nasolacrimal drainage. In addition to these anatomical and physiological variations between rabbits and humans, the different sampling regimens used in each study may have also influenced LE concentrations measured in tears. In the rabbit study, only a single sample was collected from each eye, whereas in the human study, tear fluid was collected from each subject at 4 time points. Thus, it is possible that the repeated sampling regimen used in the human study contributed to a reduction in LE levels in tear fluid over time. The polycarbophil-based vehicle used in the LE gel formulation appears to have attenuated the impact of all of these factors, resulting in similarly prolonged retention of LE on the surface of the eye in both humans and rabbits.
Results from our studies are consistent with previous studies demonstrating that polycarbophil improves drug retention on the ocular surface and increases intraocular penetration compared with more conventional topical dosage forms. Akpek et al. compared the ocular PK of azithromycin in rabbits following a single topical administration of a formulation that contained polycarbophil and one that did not. 40 Based on AUC(0–144h) values, exposure to azithromycin in the polycarbophil-based formulation was greater by 19.4-fold in the tear fluid, 9.3-fold in the conjunctiva, 4.3-fold in the cornea, and 2.8-fold in the aqueous humor compared to the polycarbophil-free formulation. These differences were statistically significant (P<0.05). Si et al. evaluated the ocular PK of bromfenac (0.045% and 0.09%) in DuraSite compared to Xibrom™ (bromfenac ophthalmic solution, 0.09%; Ista Pharmaceutical Co., Ltd.) in rabbits. 41 Durasite® (InSite Vision, Inc.) is a patented vehicle comprised of polycarbophil, edetate sodium dihydrate, and sodium chloride. After a single topical ocular dose of the 0.045% or 0.09% bromfenac in DuraSite formulations, exposure in aqueous humor was ∼2 to 4-fold greater than that with Xibrom, based on AUC(0–24h) values. Lehr et al. evaluated the effect of polycarbophil on the ocular penetration of gentamicin in rabbits after a single topical dose. 42 In this study, 2 polycarbophil-based formulations at different pHs (2.5 and 7.5) were compared to an aqueous formulation in saline. Based on AUC(0–4h) values, exposure to gentamicin with both polycarbophil-based formulations was higher in the conjunctiva (∼2.1- to 2.2-fold), cornea (∼2.0- to 2.7-fold), and sclera (1.1- to 2.2-fold) compared to the aqueous formulation. In the aqueous humor, exposure to gentamicin was ∼5.6-fold lower with the polycarbophil formulation at pH 7.5, but ∼7.8-fold higher with the polycarbophil formulation at pH 2.5.
Although not the focus of the human study, safety findings were unremarkable with only one adverse event reported (headache), which was not considered treatment-related. No ocular adverse events or significant changes in VA were observed in any subject. These results are consistent with results from clinical studies with LE ophthalmic gel, 0.5% for the treatment of patients with postcataract pain and inflammation, which reported very few adverse events, mostly related to the surgical procedure itself.34,35
Most topical steroids are formulated as suspensions, which require vigorous shaking before administration to resuspend the drug. This may be problematic for patients who forget to shake the bottle or do not shake it well enough. Inadequate shaking, in turn, may result in treatment failure if the patient does not receive a full dose. The level of polycarbophil in LE ophthalmic gel, 0.5% mitigates this issue by keeping drug particles homogeneously dispersed within the gel matrix so there is no need to shake the bottle to resuspend the drug before dosing. 33 At low shear stress (ie, in the bottle), the formulation is a viscous gel and therefore the drug particles do not settle. However, under high shear stress (ie, when the formulation is squeezed through the dropper tip), the formulation is converted to less viscous mucoadhesive suspension, allowing the drug to be administered easily. 33 In addition, the new LE gel formulation has an osmolality and pH that are close to normal tears, a low level of preservative, and contains glycerin and propylene glycol, 2 known demulcents. The osmolality of the gel formulation is 280 to 300 mOsm, which is consistent with normal tear fluid that has an osmolality of 300 to 350 mOsm. 33 The pH of the gel formulation is 6.5. Under normal conditions, the pH of tear fluid is between 7.4 and 7.7. Ophthalmic preparations with a pH between 6 and 9 are generally easily buffered by the tear fluid, and therefore, well tolerated.6,43 In some individuals, BAK can cause allergic reaction or irritation. 44 The concentration of BAK in the new gel formulation has been reduced from 0.01% in LE suspension formulations to 0.003%. This was possible by the addition of boric acid and EDTA to the formulation, both of which enhance the preservative efficacy of BAK. 33 Taken together, these formulation attributes may provide greater comfort to patients. Indeed, as indicated previously, more than 85% of patients in clinical studies reported no discomfort on drop instillation, and the majority of patients experienced no dryness, itching, or discharge.
In conclusion, the results from these studies demonstrate that the new polycarbophil-based gel formulation of LE provided sustained ocular surface exposure to LE following a single topical ocular administration for delivery to anterior segment tissues. Along with consistent dose uniformity provided by this nonsettling formulation, 33 the use of LE ophthalmic gel, 0.5% provides sustained exposure at the intended target tissues for treating patients with postoperative inflammation and pain.
Footnotes
Acknowledgments
The authors thank Sherwin Jiang for the bioanalysis of LE levels in samples from the nonclinical studies, Christopher DiSalvo for his help in summarizing the bioanalytical methods, and Heleen DeCory for editorial assistance.
Author Disclosure Statement
All authors are employees of Bausch & Lomb, Inc.
