Abstract
Abstract
Purpose:
We have manufactured a novel human foldable capsular vitreous body (FCVB). In this article, we determine whether the human FCVB releases levofloxacin in vitro and evaluate the release characteristics.
Methods:
Levofloxacin at dosages of 100, 250, and 500 μg/mL in 4.0 mL H2O was injected into the novel human FCVB; then, the capsules were immersed in cups of modified Franz diffusion cells. Two hundred microliters of liquid was aspirated at intervals of 10, 20, 40, 60, 120, 180, 240, 300, and 360 min. The levofloxacin concentrations in the cups were detected using the liquid chromatographic-tandem mass spectrometry (HPLC-MS/MS) method. Five hundred μg/mL levofloxacin liquor at volumes of 4.0, 5.0, and 6.0 mL was injected into the novel human FCVB, and the release characteristics were detected for each. The capsules of the FCVB were observed under scanning electron microscopy before and after the release study.
Results:
The novel human FCVB released levofloxacin in a time-dependent manner under 100, 250, and 500 μg/mL dosages from 10 to 360 min. Particularly in the 250 μg/mL levofloxacin group, the concentration (Q) had a good linear relationship with time (t1/2), Q=1.3381t1/2+10.2818 (r=0.9954, Higuchi equation). The human FCVB released levofloxacin in a dose-dependent manner under 100, 250, and 500 μg/mL dosages from 10 to 360 min in the total tendency, but the 500 μg/mL group released less levofloxacin than the 250 group at 300 and 360 min. The human FCVB did not release levofloxacin in a volume-dependent manner with 4.0, 5.0, and 6.0 mL volume. The 4.0-mL volume group released more levofloxacin than the other 2 volume groups. Numerous 300-nm-mili apertures were observed in the capsule of the FCVB at the beginning and end of this observation.
Conclusions:
A human FCVB can sustainably, in vitro, mechanically release levofloxacin in both a time-dependent and dose-dependent manner, but not in a volume-dependent manner. This study sets forth a novel combined research and therapeutic strategy as a vitreous substitute and intravitreal drug delivery system for the treatment of bacterial endophthalmitis.
Introduction
Intravitreal first-line drug therapy is recommended: vancomycin (1.0 mg/0.1 mL) to cover Gram-positive microorganisms, and ceftazidime (2.25 mg/0.1 mL) to cover Gram-negative microorganisms. 5 However, ceftazidime is physically incompatible with vancomycin, and it is necessary to inject the 2 antibiotics from separate syringes to avoid precipitation.6,7 Recently, there has been a drive in ophthalmology to explore the clinical potential of fluoroquinolones. 8 Levofloxacin is a third-generation fluoroquinolone and is the antibacterially active L-isomer of the racemate, ofloxacin. Levofloxacin has activity against both Gram-positive and Gram-negative bacteria. 9 Studies have been carried out to determine the safety and efficacy of levofloxacin as a treatment in conjunctivitis and keratitis,10,11 and of intravitreal levofloxacin in Staphylococcus epidermidis endophthalmitis.8,12
As an alternate route of administration, systemic antibiotics have been used concurrently for bacterial endophthalmitis. 13 However, the blood-ocular barrier prevents entry and subsequent activity of most systemic antimicrobial and anti-inflammatory drugs. 3 Recommended management of bacterial endophthalmitis includes direct injection of antibiotics into the vitreous. 2 This method of treatment, however, is fraught with risks. Direct intravitreal injection increases the risk of retinal detachment, cataracts, vitreous hemorrhage, and endophthalmitis. 14 Photoreceptors and other cells of the retina are especially sensitive to antimicrobial agents, and the biochemical pathways necessary for vision may potentially be disrupted. 3 An intravitreal drug delivery system (DDS) is a feasible solution to this unique problem.15,16
Our previous studies propose a new strategy to replace the natural vitreous body using a novel human foldable capsular vitreous body (FCVB).17–21 This novel FCVB consists of a thin, vitreous-shaped capsule with a tube-valve system created using computer and industrial technology. After foldable installation into the eye, a balanced salt solution is injected into the capsule and inflated to support the retina and to control intraocular pressure through the tube-valve system. 17 Interestingly, the FCVB changes refraction very little compared with silicone oil and heavy silicone oil, based on the Gullstrand-Emsley and Liou-Brennan schematic eyes. 18 Reports from the Chinese State Food and Drug Administration (No. G20080656) and additional tests conducted in accordance with International Standardization Organization guidelines show that the FCVB has good mechanical, optical, and biocompatible properties. 19 An exploratory clinical trial conducted at Zhongshan Ophthalmic Center in China showed that human FCVB is a flexible, effective, and safe vitreous substitute over a 3-month implantation period in human eyes. 20 Moreover, rabbit FCVB can sustainably, mechanically release dexamethasone sodium phosphate (DexP), and may be used as an intravitreal DDS for DexP. 21 The purpose of the current study is to evaluate levofloxacin release characteristics from human FCVB and to provide a combined therapeutic strategy as a vitreous substitute and DDS for the treatment of bacterial endophthalmitis.
Methods
Basic material and fabrication of the novel human FCVB
The human FCVB was fabricated with tailor-made modified liquid silicone rubber. The basic material, Dow Corning Class VI elastomers (Dow Corning Company), is a liquid silicone rubber, gelatinous at room temperature; vulcanized at 160°C, with a heating duration of 200 s, the material became semisolid elastic rubber.
The FCVB was fabricated via injection-forming technology in a specially designed mirror steel mold. The core of the mold precisely mimics the shape of the human vitreous body. The gaps in the mold control the thickness of the capsular film, which can be as thin as 30 μm. 19
Levofloxacin release with varied dosages
Levofloxacin, at dosages of 100, 250, and 500 μg/mL in H2O, were injected into the capsules of the human FCVB; the capsules were then immersed in cups of modified Franz diffusion cells, as shown in Fig. 1.
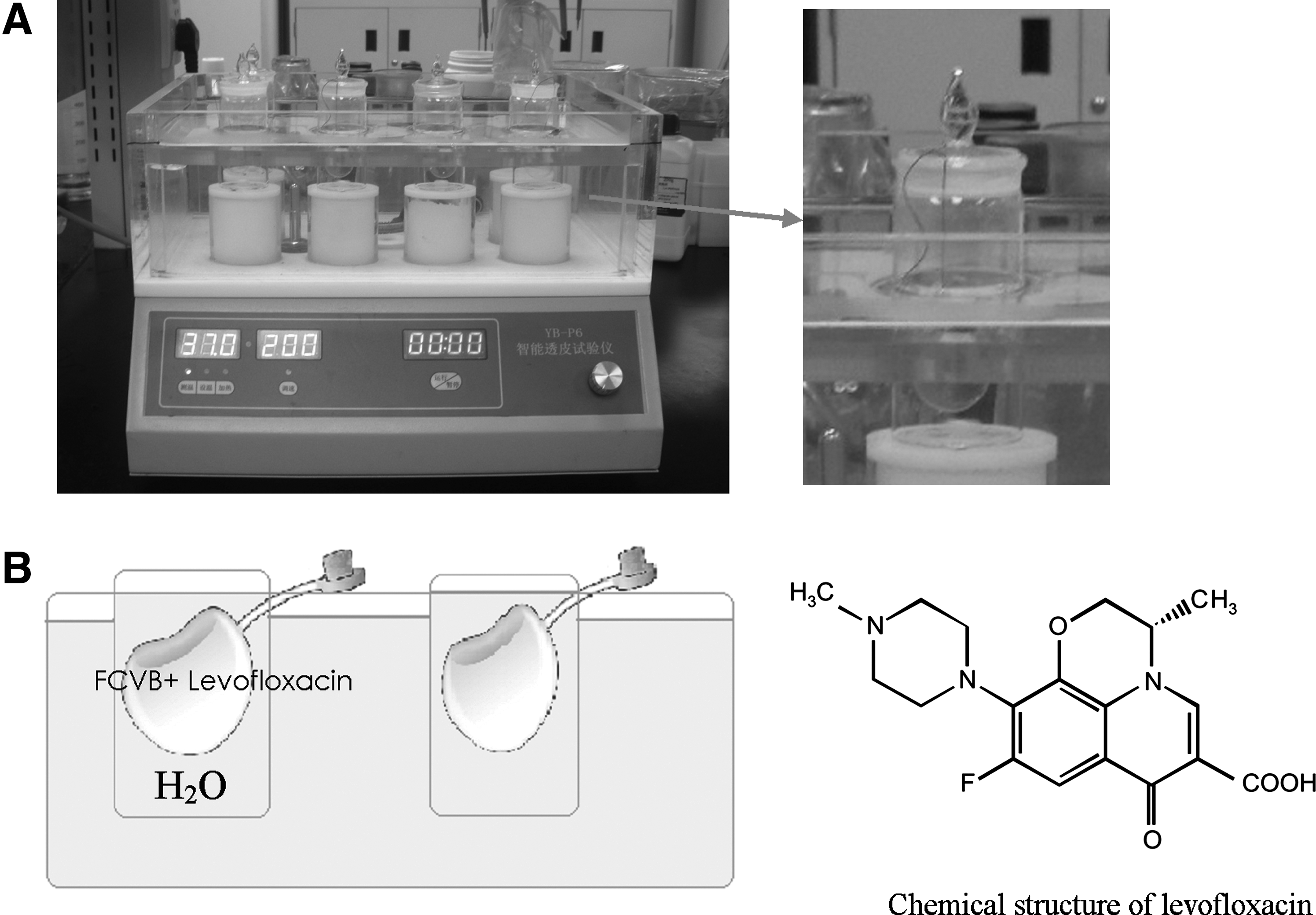
Levofloxacin sustained-release studies in vitro.
Two hundred microliters of liquid in each cup was aspirated for measurement at 10, 20, 40, 60, 120, 180, 240, 300, and 360 min. The levofloxacin content in the liquid was then detected via a sensitive liquid chromatographic-tandem mass spectrometry (HPLC-MS/MS) method.
Levofloxacin release with varied levofloxacin liquor volumes
Levofloxacin liquor, at volumes of 4.0, 5.0, and 6.0 mL, were injected into the capsules of the human FCVB. The internal pressure of the FCVB at different volumes was tested using a Pneumatic Transducer Tester (Fluke). The capsules were immersed in cups; 200 μL of liquid was aspirated at the 9 time points just listed and detected via HPLC-MS/MS. The concentrations of levofloxacin were all 500 μg/mL.
Levofloxacin sample assay
The HPLC-MS/MS system consisted of a Surveyor MS pump, a Surveyor autosampler (Thermo-Finnigan), and a ThermoFinnigan TSQ Quantum triple quadrupole mass spectrometer equipped with an ESI source. A reversed-phase column Waters XTerra C18 (3×150 mm, 3.5 μm; Waters) was used for all chromatographic separation at room temperature (20°C). The mobile phase (1% formic acid in water and acetonitrile, 40:60, v/v) was pumped at a flow rate of 0.3 mL/min. The mass spectrometer was operated in the positive electrospray ionization mode. Quantification was performed using selected reaction monitoring (SRM) in the positive mode. The ion transition of the mass-to-charge ratio (m/z) 361.7→318.0 for levofloxacin was monitored, with a collision-induced energy of 18 eV.
The reference formulation of levofloxacin (lot number 30537-200301) was obtained from the National Institute for the Control of Pharmaceutical and Biological Products (China). Acetonitrile and methanol of HPLC grade were obtained from SK Chemicals, Korea. Formic acid of HPLC grade was obtained from MERCK (State Food and Drug Administration of China).
The stock solutions were prepared in methanol at a concentration of 20 μg/mL in 100-mL glass vials, and serially diluted with methanol to prepare standard working solutions at the desired concentrations: 5, 10, 50, 100, 500, 800, and 1,000 ng/mL. Thirty microliters samples were transferred into 2.0-mL Eppendorf tubes. After addition of 30 μL of Cip solution (IS), 30 μL of methanol, and 300 μL of mobile phase, the mixture was vortexed for 1 min and centrifuged at 12,000 rpm for 5 min. Approximately 0.1 mL of the supernatant was transferred into autosampler vials, then 10 μL was injected into the HPLC column. Data acquisition was performed using Xcalibur 1.3 software (ThermoFinnigan). Peak integration and calibration were performed using LCQuan software (ThermoFinnigan).
Scanning electron microscopy of the human FCVB
The capsule of the human FCVB was cut into the appropriate size, and the sample was cleaned, coated with gold, and fixed to a specimen stub. An image of the specimen surface was captured on a scanning electron microscope. The capsules of the 4.0 and 6.0 mL groups were observed before and at the end of this release study.
Results
The standard weight of the human FCVB was 0.33±0.005 g; the standard volume of the human FCVB was 3.8±0.5 mL. The FCVB is transparent, with high light transmittance of 92%. With volumes of 4.0, 5.0, and 6.0 mL, the internal pressures of the FCVB were 23, 55, and 68 mmHg, respectively.
The representative mass spectra of levofloxacin, HPLC-MS/MS chromatograms, and accuracy and precision of the quality control (QC) samples are shown in Figs. 2 and 3, and Table 1. The model fitting results for levofloxacin released from the FCVB are shown in Table 2. Levofloxacin released from the human FCVB, with varied dosages and varied volumes, are set forth in Figs. 4–6. Scanning electron microscopic images of the capsule of the human FCVB are shown in Fig. 7.
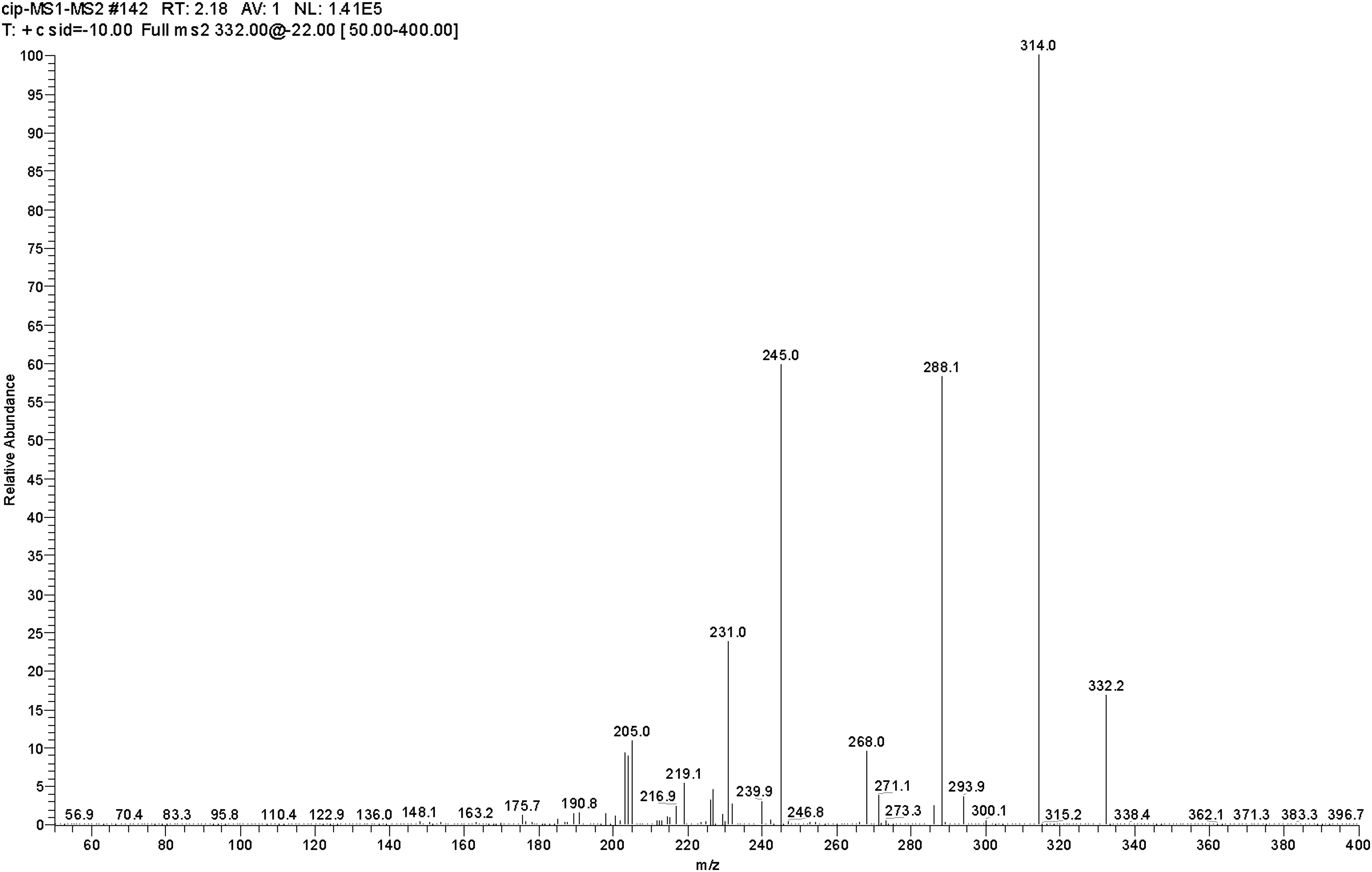
Production mass spectra of [M+H]+ for levofloxacin. The fragment ions of m/z 361.7 were chosen as production for monitoring levofloxacin.
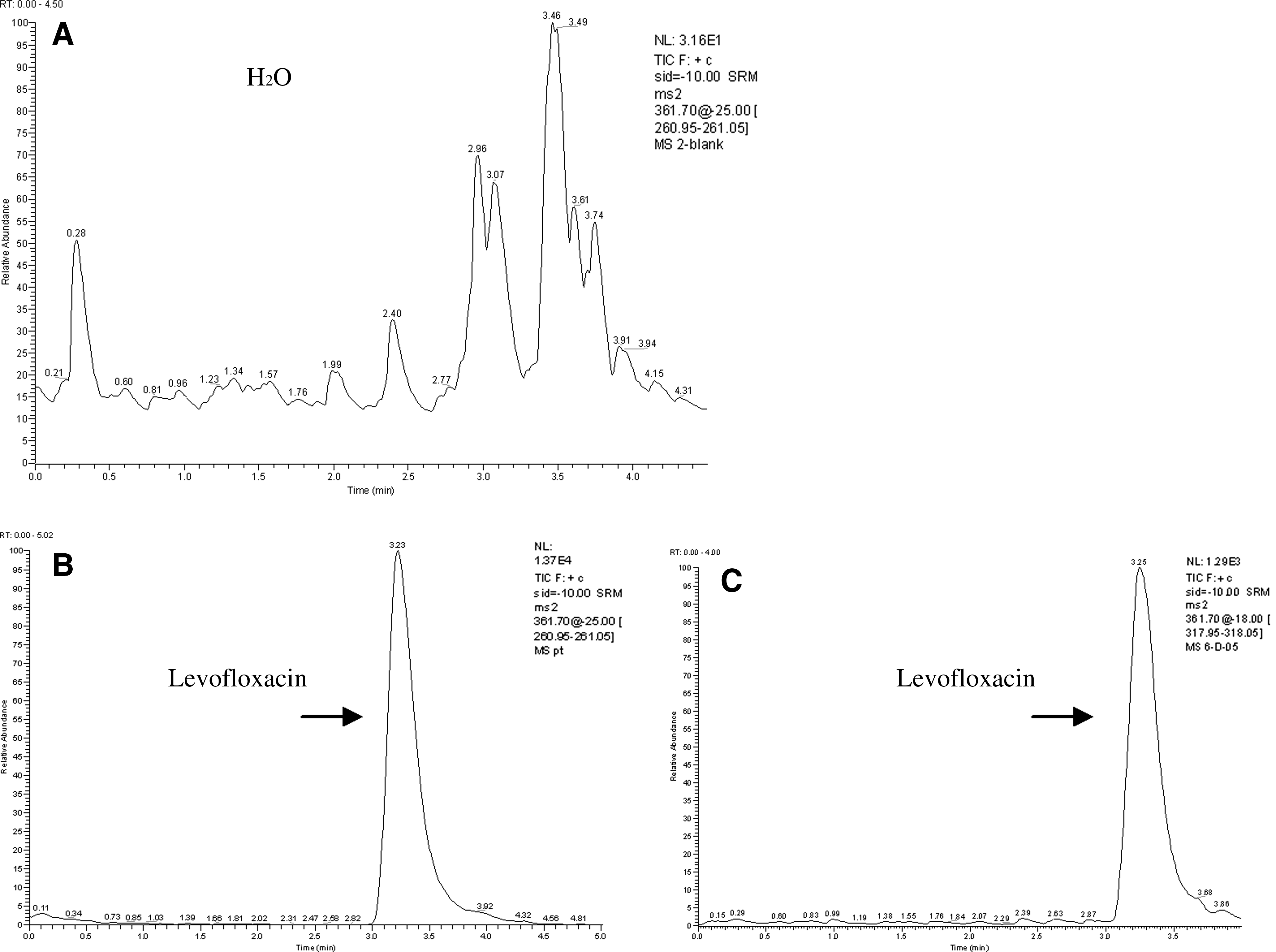
Representative liquid chromatographic-tandem mass spectrometry (HPLC-MS/MS) chromatograms of levofloxacin.
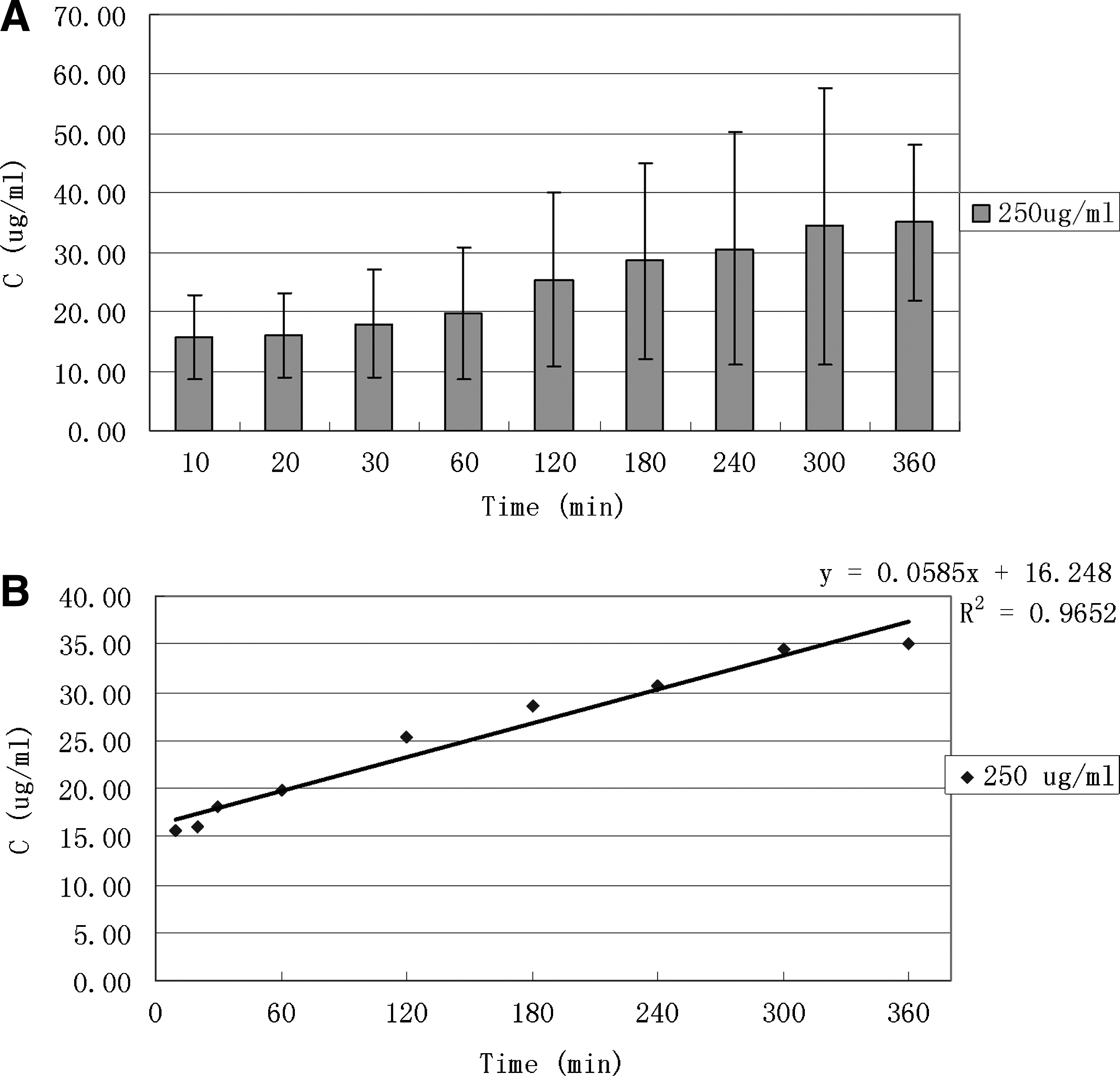
Levofloxacin released from human FCVB at dosage of 250 μg/mL in time-dependent manner.
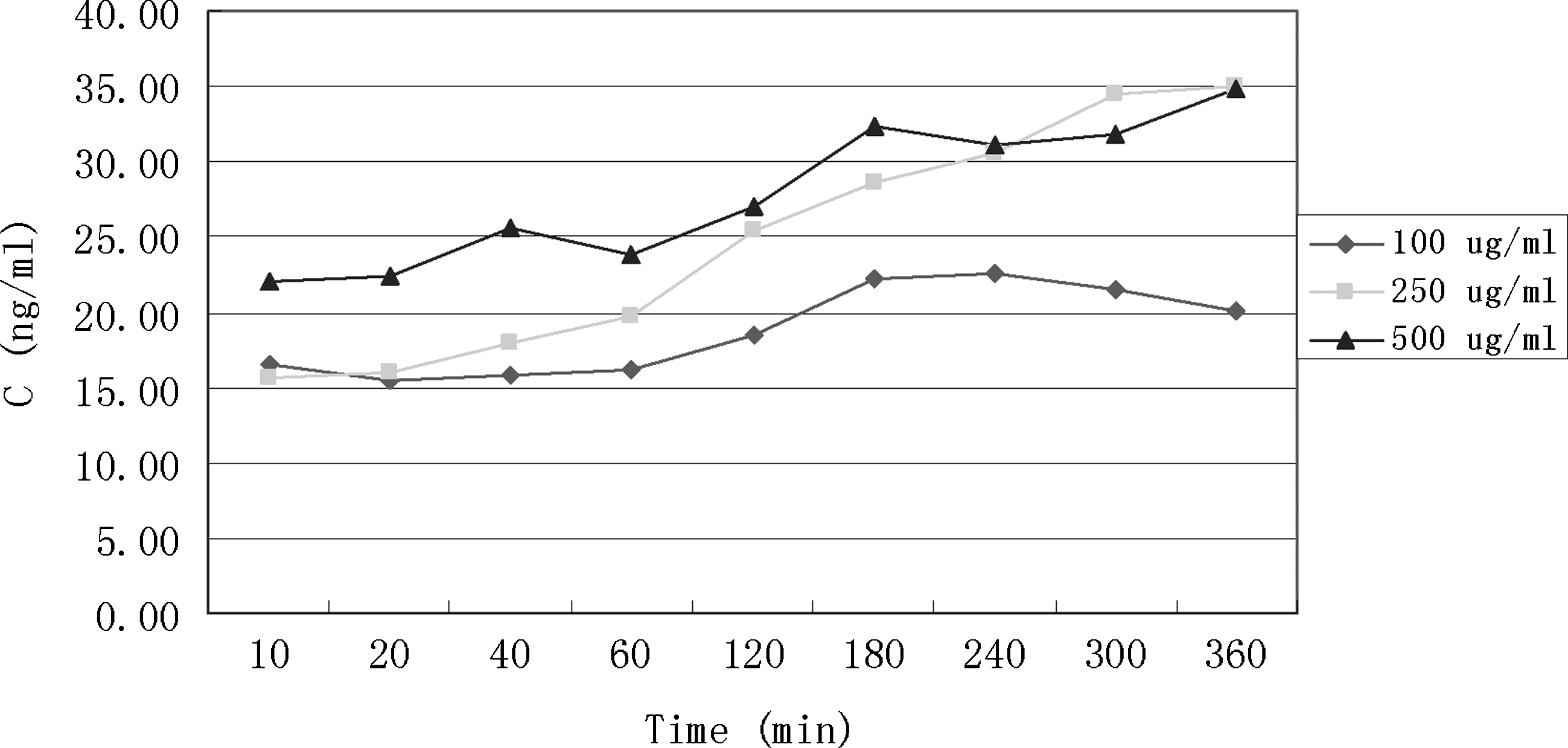
Human FCVB released levofloxacin in a dose-dependent manner. When dosages of levofloxacin in the FCVB increased, then external content of levofloxacin rose. The 500 μg/mL group released less levofloxacin than the 250 μg/mL group at 300 and 360 min. According to total tendency, the 500 μg/mL group released more levofloxacin than the other 2 dosage groups.
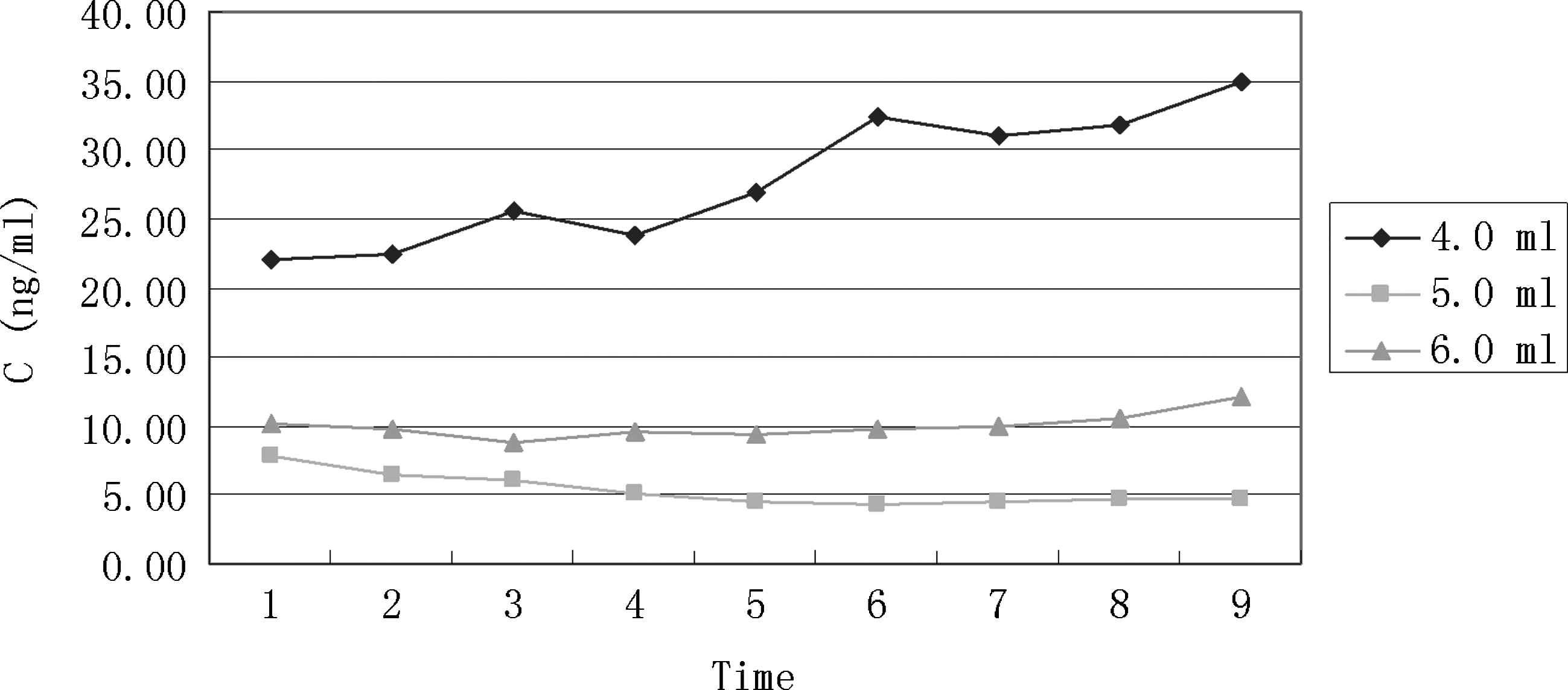
Human FCVB released levofloxacin not in a volume-dependent manner with 4.0, 5.0, and 6.0 mL volume. The 4.0 mL volume group released more levofloxacin than the other 2 volume groups.
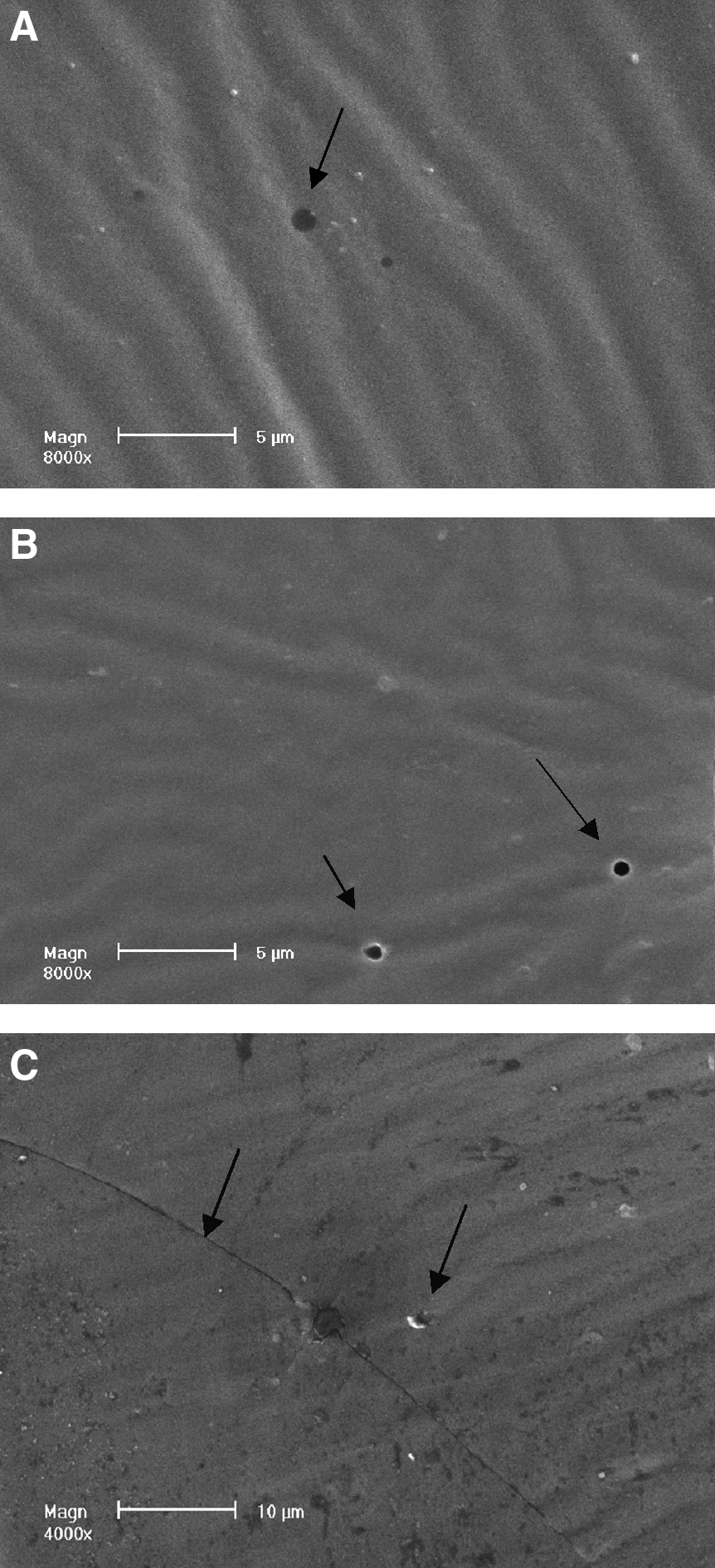
Scanning electron microscopic images of the capsule of the human FCVB. Three hundred nanometer mili apertures in the capsule were observed at the beginning and end of the release study
SD, standard deviation; RSD, relative standard deviation; RE, relative error.
The HPLC-MS/MS parameters were optimized to produce the maximum response for levofloxacin in the positive ion mode. Figure 2 shows the production mass spectra of [M+H]+ of levofloxacin. After electrospray ionization, positive ion fragments of m/z 361.7 were detected in the SRM mode with a triple quadrupole tandem mass spectrometer, and the fragment ions of m/z 318.0 were chosen as the production for monitoring levofloxacin.
Representative HPLC-MS/MS chromatograms of levofloxacin are shown in Fig. 3. The retention time of levofloxacin was 2.3 min, with no interference observed from H2O, thus demonstrating that the method chosen had good selectivity and acceptability.
The accuracy (relative error [RE] %) and precision (relative standard deviation [RSD] %) results for the QC samples are summarized in Table 1. The results of RE ≤±15% and RSD ≤±15% showed acceptable accuracy and precision for the proposed method.
The data for levofloxacin release from the FCVB were fitted to different kinetic models to explain the release mechanism and pattern using the zero-order, first-order, and Higuchi equations (Table 2). Results show that the Higuchi equation best fits the release characteristics.
Levofloxacin was released from the human FCVB in a time-dependent manner in 100, 250, and 500 μg/mL groups at 10, 20, 40, 60, 120, 180, 240, 300, and 360 min (Figs. 4 and 5). The concentration (Q) had good linear relationships with time (t1/2), especially in the 250-μg/mL levofloxacin group (Higuchi equation: Q=1.3381t1/2+10.2818, r=0.9954). The concentrations, at different time points, showed no significant differences (F=0.72, P>0.05, Fig. 4). According to the linear tendency, the FCVB continued to be released.
Levofloxacin was released from the human FCVB in a dose-dependent manner with varied dosages of 100, 250, and 500 μg/mL. When the dosages of levofloxacin in the FCVB increased, the outside content of levofloxacin rose. However, at 300 and 360 min, the 500 μg/mL group released less levofloxacin than the 250 μg/mL group. According to the total tendency, the 500 μg/mL group released more levofloxacin than the other 2 dosage groups (Fig. 5).
Levofloxacin was not released from the human FCVB in a volume-dependent manner under varied volumes of 4.0, 5.0, and 6.0 mL. The 4.0 mL volume group released more levofloxacin than the other 2 volume groups, as shown in Fig. 6.
The scanning electron microscope revealed apertures in the capsule of the human FCVB. Three hundred nanometer mili apertures in the capsule were observed before the release study and at the end of the observation time (Fig. 7). Traces after the expansion of silicone rubber were also observed in the 6.0 mL volume of levofloxacin groups.
Discussion
Endophathalmitis is defined as any inflammation of the internal ocular spaces, and it usually results in poor visual prognoses for the majority of patients. 4 DDS is the ideal method for in vitreous body drug administration. 15 This study demonstrates that the human FCVB can sustainably, mechanically release levofloxacin in vitro, and it may provide a new, combined research and therapeutic strategy as a vitreous substitute and DDS for the treatment of bacterial endophthalmitis.
The use of levofloxacin as a potential agent for the treatment of endopthalmitis has been previously described by Jackson et al., 4 Herbert et al., 22 Yildirim et al., 12 Ferrer et al., 8 and Gurler et al. 23 Jackson et al., reviewing 267 reported cases, demonstrated that the most common Gram-positive organisms are Staphylococcus aureus and Streptococcus pneumoniae. Herbert et al. reported that levofloxacin possesses a wide spectrum in vitro against both Gram-positive and Gram-negative pathogens, and suggested that the MIC90 for S. aureus is 0.25–0.50 μg/mL (MIC90 is the minimum inhibitory concentration required to inhibit the growth of 90% of organisms). Yildirim et al. demonstrated that intravitreal levofloxacin appeared to be effective in S. epidermidis endophthalmitis in rabbits, and the culture results of the treatment groups were sterile. Gurler et al. established the level of toxicity of intravitreal levofloxacin; doses of 50, 100, 250, and 500 μg in 0.1 mL injected into the mid-vitreous of rabbits were not toxic to the retina.
In this study, the human FCVB is demonstrated to have a sustained release of levofloxacin. Three hundred nanometer mili apertures in the capsule give the FCVB the capability of a DDS. Since the molecular mass of levofloxacin is 415.85 Da, the drug molecules will diffuse freely through the apertures. The osmotic pressure, so long as the FCVB with levofloxacin is immersed in water, forces the levofloxacin molecules to move through the apertures to the outer circumference. In this study, levofloxacin is demonstrated to continue to be released. On the other hand, the number of apertures also restricts the total flow rate and limits the rapid loss of the levofloxacin; sustained drug release is thereby achieved. Particularly in vivo, since the released levofloxacin will be taken away by blood or metabolized quickly, the osmotic pressure will maintain the strain. In addition, the literature shows that FCVB causes no retinal toxicity on rabbit eyes during a 3-month observation, 17 and that FCVB is a safe intraocular tamponade during a 3-month implantation time in human eyes. 20 Gurler et al. 23 and Ohkubo et al. 24 report that intravitreal levofloxacin has no retinal toxicity in rabbit eyes with dosage of 500 μg/mL or less, whereas the oscillatory potential transiently deteriorated with 1,000 and 2,000 μg/mL of levofloxacin. We chose 500 μg/mL as the highest concentration of levofloxacin inner FCVB. In this case, the released levofloxacin is much less than 500 μg/mL. Even if FCVB leakage or rupture occurs, leaked levofloxacin will remain nontoxic to the retina.
When the dosages of levofloxacin in the FCVB were increased, then the external content of levofloxacin rose. However, when the volume of levofloxacin in the FCVB increased, then the external content of levofloxacin decreased. The noninflating volume of the FCVB is 3.8±0.05 mL. At this volume, the capsule closely mimics the shape of the natural vitreous body, and there is an anterior fovea for the lens; the internal pressure of the capsule is 12 mmHg. For easy calculation and control, we injected 4.0 mL levofloxacin inside as the basic volume. At 4.0 mL volume, the anterior fovea of the lens bulges forward slightly, and the internal pressure was 23 mmHg. At volumes of 5 and 6 mL, the capsule is expanded, the thickness of the capsule is reduced, and the internal pressure is increased to 55 and 68 mmHg, respectively. However, the expansion of the capsules is not uniform. In particular, at 6 mL volume, this spherical-shape capsule expanded into an oval shape. With uneven inflation, the apertures in the capsule expanded unevenly and the amount of levofloxacin released was reduced.
Intraocular DDS is becoming increasingly important, and the need for intraocular DDS has resulted in a variety of controlled-release systems such as implantable devices and injectable particles.15,16 Current DDS, such as the dexamethasone intravitreal implant for macular edema and the intravitreal fluocinolone acetonide implant for uveitis,25,26 confront several major disadvantages. For example, they are opaque, may interfere with vision, or result in a nonuniform drug distribution.15,16 Drugs are centralized and should pass from the vitreous body to the retina. In contrast, the FCVB has good transparency, induces very little refractive shift, and permits the levofloxacin in solution to evenly disperse in the capsule and permeate externally in a uniform manner. With the FCVB implanted, levofloxacin touches the retina directly. Another study on rabbit FCVB demonstrated that, in a S. aureus endophthalmitis rabbit model, the FCVB with 625 μg/mL levofloxacin tamponaded, and the culture results of aqueous humor were sterile (Supplementary Fig. S1; Supplementary Data are available online at www.liebertonline.com/jop). Therefore, the human FCVB may provide a novel DDS for bacterial endophthalmitis.
Currently, a clinical trial to ascertain the biocompatibility and effectiveness of human FCVB as a silicone oil substitute in human eyes has been completed at Zhongshan Ophthalmic Center in China. The clinical trials were approved by the Sun Yat-sen University Medical Ethics Committee (Zhongshan Ophthalmic Center Medical Ethics [2009] No. 07), and have been successfully registered with ClinicalTrias.gov (ClinicalTrials.gov ID: NCT00910702), and in the Chinese Clinical Trial Register (ChiCTR-TNC-00000396). Results show that the FCVB was a flexible, effective, and safe vitreous substitute over a 3-month implantation period. 20 Thus, the FCVB may provide a combined vehicle for a vitreous substitute and endophthalmitis DDS.
Our future research will focus on the effect of the human FCVB, combined with levofloxacin, on human bacterial endophthalmitis. In addition, research on the investigation of other drugs is in progress, including antibiotics, antiproliferation agents, and vascular endothelial growth factor antagonists.
In conclusion, the human FCVB can, in vitro, mechanically and sustainably release levofloxacin in a time-dependent and dose-dependent manner, but not in a volume-dependent manner. This study provides a novel combined research and therapeutic strategy as a vitreous substitute and DDS for the treatment of bacterial endophthalmitis.
Footnotes
Acknowledgments
This study was supported by the National High-tech R&D Program of China (863 Program, 2009AA2Z404) and the National Major Projects for Science and Technology Development (2009ZX09304-003).
Author Disclosure Statement
No competing financial interests exist.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
