Abstract
Abstract
Purpose:
Effects of a long-acting ophthalmic formulation of carteolol containing alginic acid on the corneal epithelial barrier function and its water retentive effect were investigated.
Methods:
Using 10 healthy adult subjects, 2% Mikelan Ophthalmic Solution® (MK) was instilled in the eye once daily for 7 days (MK group) and then after a washout period of at least 28 days, 2% Mikelan LA Ophthalmic Solution® (MKLA) was instilled in the eye once daily for 7 days (LA group). As an index of the corneal epithelial barrier function, the fluorescein uptake was measured using Kowa FL-500. A Schirmer test was conducted to evaluate the tear dynamics. In another 10 subjects, 0.5% Timoptol® (TM) was instilled in the eye unilaterally twice daily for 7 days (TM group), and the tests were conducted in the same manner.
Results:
Concerning the fluorescein uptake before and after initiation of instillation, the levels before and at 7 days after initiation of instillation were 20.7 and 26.5 ng/mL, respectively, in the LA group and 20.6 and 26.4 ng/mL, respectively, in the MK group, showing no significant difference between levels before and after initiation of instillation in either group. In the TM group, the levels were 21.4 and 65.5 ng/mL, respectively, showing a significant increase after initiation of instillation. In the Schirmer test, the values before and after initiation of instillation were 16.8 and 20.7 mm, respectively, in the LA group and 13.7 and 12.7 mm, respectively, in the MK group, showing a trend toward increase in the LA group.
Conclusions:
The findings suggest that the long-acting ophthalmic formulation of carteolol containing alginic acid does not affect the corneal epithelial barrier function and that it may possess a water retentive action.
Introduction
This time, a long-acting ophthalmic formulation of carteolol containing alginic acid, that is, 2% Mikelan LA Ophthalmic Solution® (hereinafter, referred to as 2% MKLA), prepared by adding alginic acid to an ophthalmic formulation of carteolol resulting in improved retention of the drug on the ocular surface, increased amount of carteolol transferred into the eye and prolonged duration of the action 6 became available also in Japan. While 2% MKLA possesses an intraocular pressure-lowering action comparable to that of an existing ophthalmic formulation of carteolol hydrochloride 2% Mikelan Ophthalmic Solution® (hereinafter, referred to as 2% MK), it has a marked characteristic that the frequency of instillation of 2% MKLA is once daily, less than that of 2% MK (twice daily). Although it is expected that this may lead to improved instillation adherence of patients, there is also concern about effects on the ocular surface due to prolonged duration of contact of the drug to the cornea. However, since the alginic acid added to 2% MKLA may possess water retentive action, 6 2% MKLA may act protectively on the ocular surface. Therefore, this time, we investigated the effects of 2% MKLA on the ocular surface, and the results are reported.
Subjects and Methods
Effects of 2% MKLA on the ocular surface in healthy adults
Using 10 healthy adult subjects with written consent who did not meet the following exclusion criteria (mean age: 27.9±5.5 years), the effects of 2% MKLA and 2% MK on the ocular surface were investigated by a crossover study employing the protective effects on the ocular surface as the primary endpoint and the equivalence of the intraocular pressure-lowering effect between 2% MKLA and 2% MK as the secondary endpoint.
Exclusion criteria
(1) Subjects using contact lenses (2) Subjects with−4 diopter or severer astigmatic eye (3) Subjects with underlying diseases that may cause corneal disorder (4) Subjects using any ophthalmic preparation that may induce corneal disorder (5) Subjects with severe dry eye (6) Subjects with insufficiently controlled heart failure, sinus bradycardia, atrioventricular block (second or third degree) or cardiogenic shock, subjects with bronchial asthma, bronchospasm or such a history, or subjects with severe chronic obstructive pulmonary disease
First, before initiation of drug instillation, refraction test using an Auto Ref/Keratometer ARK-530A (NIDEK®), measurement of the intraocular pressure using a noncontact tonometer RKT-7700 (NIDEK®), slit-lamp microscopic examination, measurement of the tear film break up time (hereinafter, referred to as BUT) 7 and quantification of the corneal epithelial barrier function by modified Schirmer test-1 (hereinafter, referred to as Schirmer test) 8 and fluorophotometry were conducted. Concerning the BUT, fluorescein sodium solution was instilled in the eye, and the subjects were instructed to blink several times to diffuse the solution in the tear homogeneously and then the eyelids were kept opened and the time required to develop a dry spot on the cornea was recorded. The measurement was conducted thrice, and the mean value was employed. In the Schirmer test, 0.4% oxybuprocain (Benoxil®) was instilled in the eye and 5 min later, a folded (with the width of 5 mm) part of a Schirmer Strip® (Showa Yakuhin Kako Co., Ltd.) was put on the palpebral conjunctiva at the position of ∼1/3 of the lower eyelid and then another 5 min later, the length of the strip wet with tear was measured. For evaluation of the corneal epithelial barrier function, a slit-lamp-type fluorophotometer for anterior ocular segment (Kowa FL-500®) was used. According to the method of Yokoi and Kinoshita 9 the background fluorescence intensity at the central part of the cornea was measured as the background value first and then, 0.5% fluorescein sodium solution (dissolved in BSS PLUS®) at a volume of 3 μL was instilled in the lower palpebral conjunctival sac in a noncontact manner. Ten minutes later, the test eye was washed with 20 mL of BSS PLUS while turning the eyelids so that fluorescein did not remain in the conjunctival sac. Measurement of the fluorescein uptake was conducted at the central part of the cornea in the same manner as measurement of the background value at 30 min after instillation of fluorescein (20 min after eye washing), and the background value was subtracted from the mean value and then the fluorescein uptake was calculated using the built-in calibration curve in FL-500.
After execution of the tests just mentioned, the subjects received instillation of 2% MK (5 subjects) or 2% MKLA (5 subjects) in 1 eye and instillation of physiological saline in the other eye as the control at the prescribed frequency, respectively, over a period of 7 days. Measurement of the intraocular pressure, slit-lamp microscopic examination, and quantification of the corneal epithelial barrier function by fluorophotometry were conducted on days 1, 3, and 7 after initiation of instillation and on day 3 after completion of instillation. Refraction test, measurement of the BUT, and a Schirmer test were also conducted on day 7 after initiation of instillation and on day 3 after completion of instillation only. After drug instillation for 7 days, a washout period of at least 28 days 10 was prepared; then, 2% MKLA was instilled to 5 subjects who had been treated with 2% MK, 2% MK was instilled to 5 subjects who had been treated with 2% MKLA at the prescribed frequency over a period of 7 days, and the tests were conducted in the same manner. In addition to the measurer N.T., another person J.I. was prepared as a controller who recorded the details about the ophthalmic preparations transferred to the subjects.
Using another 10 subjects (mean age: 26.4±1.1 years), 0.5% timolol maleate ophthalmic solution [0.5% Timoptol® (TM)] was instilled in 1 eye twice daily, and physiological saline ophthalmic preparation was instilled in the other eye twice daily. Measurement of the intraocular pressure and quantification of the corneal epithelial barrier function were conducted in the same manner before initiation of instillation, on days 1, 3, and 7 after initiation of instillation and on day 3 after completion of instillation.
In all the investigations, analyses were conducted using paired t-test (intergroup comparison was conducted using unpaired t-test), and the significance level was set at P<0.05. Table 1 shows preservative toxicities of 2%MKLA, 2%MK, and 0.5% TM, respectively, in the 3 drugs that employed 0.05% benzalkonium chloride (BAC). 11
The present study was approved by the institutional review board of St. Marianna University School of Medicine (Approval No. 1677).
Results
Among the 3 groups of 2% MKLA group, 2% MK group, and 0.5% TM group, no significant difference was observed in the intraocular pressure, refractive value, or fluorescein uptake before initiation of drug instillation.
Concerning the time courses of the intraocular pressure in respective groups, the values in the drug-instilled eye before initiation of instillation and on day 7 after initiation of instillation were 16.3±3.7 and 15.1±3.2 mmHg, respectively, in the 2% MKLA group, 15.7±3.5 and 13.8±3.5 mmHg, respectively, in the 2% MK group, and 14.2±1.8 and 12.6±1.8 mmHg, respectively, in the 0.5% TM group, showing significantly decreased intraocular pressure after initiation of drug instillation in respective groups (P<0.05, paired t-test).
Figure 1 shows the time courses of the fluorescein uptake in groups in which 2% MKLA was instilled for 7 days and then, 2% MK was instilled for 7 days after a washout period and in which 2% MK was instilled for 7 days and then 2% MKLA was instilled for 7 days after a washout period. No significant difference was observed at any measurement time point in comparison with the value before initiation of instillation in either group.

Time courses of the fluorescein uptake before and after initiation of instillation of 2% MKLA and 2% MK.  Instillation of 2% MK→ washout → instillation of 2% MKLA group (n=5).
Instillation of 2% MK→ washout → instillation of 2% MKLA group (n=5).  Instillation of 2% MKLA→washout→instillation of 2% MK group (n=5). In both groups, the fluorescein uptake showed time courses with no significant difference in comparison with the value before initiation of instillation of 2% MKLA or 2% MK. MK, Mikelan Ophthalmic Solution®; MKLA, Mikelan LA Ophthalmic Solution®.
Instillation of 2% MKLA→washout→instillation of 2% MK group (n=5). In both groups, the fluorescein uptake showed time courses with no significant difference in comparison with the value before initiation of instillation of 2% MKLA or 2% MK. MK, Mikelan Ophthalmic Solution®; MKLA, Mikelan LA Ophthalmic Solution®.
Figure 2 shows the time courses of the fluorescein uptake in the drug-instilled eye and noninstilled eye in the 0.5% TM group. In the 0.5% TM-instilled eye, the fluorescein uptake significantly increased from 21.4±12.3 ng/mL before initiation of instillation to 61.6±17.5 ng/mL from day 1 after initiation of instillation (P<0.05, paired t-test).

Time courses of the fluorescein uptake in the drug-instilled eye and noninstilled eye in the 0.5% Timoptol® group before and after initiation of instillation.  Fluorescein uptake in the 0.5% TM-instilled eye (n=10).
Fluorescein uptake in the 0.5% TM-instilled eye (n=10).  Fluorescein uptake in the physiological saline-instilled eye (n=10). *p<0.05, paired t-test. In the 0.5% TM-instilled eye, the fluorescein uptake showed a significant increase from day 1 after initiation of instillation in comparison with the level before initiation of instillation.
Fluorescein uptake in the physiological saline-instilled eye (n=10). *p<0.05, paired t-test. In the 0.5% TM-instilled eye, the fluorescein uptake showed a significant increase from day 1 after initiation of instillation in comparison with the level before initiation of instillation.
Compared with the fluorescein uptake in the drug-instilled eye in the 2% MKLA group, 2% MK group, 0.5% TM group, and 0.5% TM group, the fluorescein uptake showed significantly higher values in comparison with the 2% MKLA group and 2% MK group at all the measurement time points from day 1 to day 7 after initiation of instillation (P<0.01, unpaired t-test).
Figure 3 shows the results of measurement of BUT before and after initiation of drug instillation. Concerning the BUT values before and after initiation of instillation of 2% MKLA and 2% MK, the values in the 2% MKLA group were 12.0±4.1 s before initiation of instillation and 13.8±5.5 s on day 7 after initiation of instillation, showing a prolongation after initiation of instillation while there was no significant difference (P=0.29, paired t-test). The values in the 2% MK group were 10.2±3.4 s before initiation of instillation and 10.8±5.7 s on day 7 after initiation of instillation, showing similar values between before and after initiation of instillation (P=0.42, paired t-test).
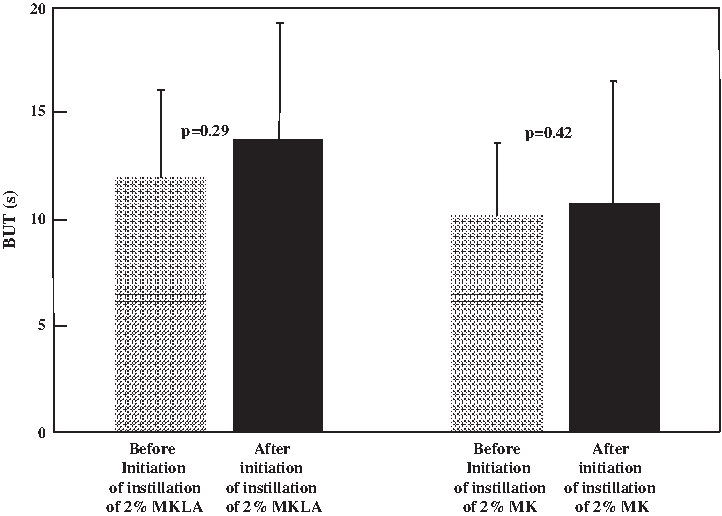
Time courses of the break-up time (BUT) in the 2% MKLA group and the 2% MK group. In the 2% MKLA instillation group, the BUT showed a trend toward prolongation on day 7 after initiation of instillation, although there was no significant difference (p=0.29, paired t-test).
Figure 4 shows the results of measurement by modified Schirmer test-1 before and after initiation of drug instillation. Concerning the results of measurement by modified Schirmer test-1 before and after initiation of instillation of 2% MKLA and 2% MK, while the values in the 2% MKLA group were 16.8±10.9 mm before initiation of instillation and 19.8±9.2 mm on day 7 after initiation of instillation, showing an increase after initiation of instillation although there was no significant difference (P=0.32, paired t-test), those in the 2% MK group were 15.2±5.7 mm before initiation of instillation and 12.8±5.6 mm on day 7 after initiation of instillation, showing a decrease after initiation of instillation.
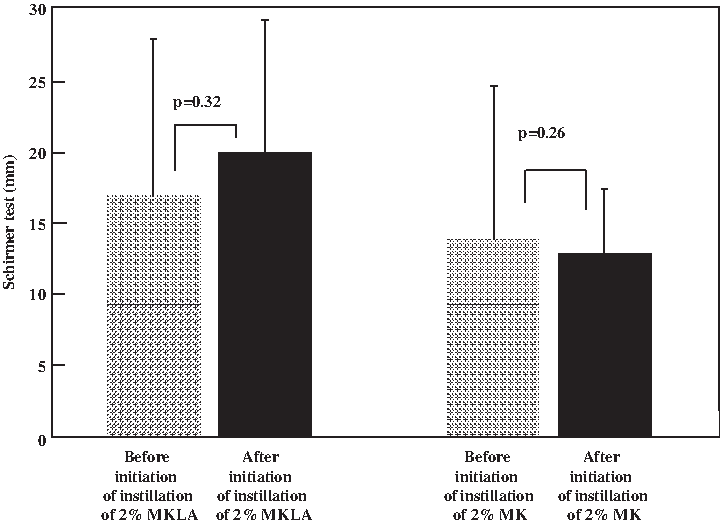
Time courses of the amount of tear in the 2% MKLA group and the 2% MK group. In the 2% MKLA instillation group, the amount of tear showed a trend toward increase on day 7 after initiation of instillation, although there was no significant difference (p=0.32, paired t-test).
Discussion
In the present study, 2% MKLA, 2% MK, and 0.5% TM were instilled to healthy adults, and the effects on the ocular surface before and after initiation of instillation of respective drugs were compared. As a result, while the corneal epithelial barrier function was decreased after initiation of instillation of 0.5% TM in comparison with before initiation of instillation, no decrease in the corneal epithelial barrier function was observed after initiation of instillation of 2% MKLA or 2% MK in comparison with before initiation of instillation.In addition, in the results of measurement of the BUT and modified Schirmer test-1 before and after initiation of instillation of 2% MKLA and 2% MK, while little change was observed between before and after initiation of instillation of 2% MK, trends toward prolonged BUT and increased amount of tear in the modified Schirmer test-1 were observed after initiation of instillation of 2% MKLA, although there was no significant difference. These results are discussed next.
As factors that may affect the corneal epithelial barrier function, the effects of preservatives, particularly those of BAC, have been reported11,12 in addition to effects of the principle ingredient of the ophthalmic preparation. Since 0.05% BAC was employed as the preservative contained in all the 3 formulations of 2% MKLA, 2% MK, and 0.5% TM used in the present study, 11 it is considered that the effects due to the concentration of BAC were equal among the 3 groups. Therefore, the time courses of the corneal epithelial barrier function were compared between the 2% MK group and the 0.5% TM group that employed the same instillation frequency first. As a result, while the corneal epithelial barrier function was decreased from an early stage after initiation of instillation of 0.5% TM, no effect on the corneal epithelial barrier function was observed during the period of 7 days after initiation of instillation of 2% MK. This result suggests that the effects of carteolol hydrochloride on the corneal epithelial barrier function are less than those of timolol maleate. Inoue et al. 5 reported that among patients using anti-glaucoma ophthalmic preparations in the clinical setting, the group using timolol showed a significantly higher incidence of superficial punctate keratopathy (hereinafter, referred to as SPK) in comparison with the group using carteolol, and this result is consistent with our present result. In addition, Kawai et al. 13 reported that carteolol possesses actions to suppress the production of TNF-α and IL-6 and that anti-inflammatory effects can be expected by these actions. This mechanism may possibly be involved in the maintenance of the corneal epithelial barrier function and decreased incidence of SPK after instillation of carteolol.
Next, carteolol preparations with different instillation frequency and base were compared. Since the fluorescein uptake showed time courses with no marked change before and after initiation of instillation in both periods treated with 2% MKLA and 2% MK, it was suggested that carteolol shows less effects on the corneal epithelial barrier function even when the instillation frequency or base is different.
Modified Schirmer test-1, which is an index of the amount of tear, and the BUT, which is an index of the oily layer of the tear film, were also investigated. Since carteolol-induced decrease in the amount of tear 1 and its effect on the corneal perception due to local anesthetic effect are intrinsically less than those of timolol, 3 the decrease in the amount of tear after instillation of carteolol was expected to be small, and what further change in the tear would occur by addition of alginic acid to carteolol was investigated. As a result, in the group in which 2% MKLA was instilled for 7 days, the BUT showed a trend toward prolongation, and the amount of tear section showed a trend toward increase in modified Schirmer test-1 also. Since these results were obtained in a very short instillation period of 7 days, it is considered that further investigation with a longer period is required in the future, however, the possibility that alginic acid contained in 2% MKLA possesses an ability to retain the tear to some degree was suggested.
As just described, the effects of 2% MKLA on the ocular surface were investigated. It was clarified that 2% MKLA shows an intraocular pressure-lowing effect without an effect on the ocular surface in healthy adults and the possibility that alginic acid contained in 2% MKLA acts in a complementary manner for this result was suggested. It was considered that the effects of 2% MKLA in patients with already-decreased corneal epithelial barrier function or patients with dry eye and the effects of 2% MKLA in corneal disorder models should also be investigated in the clinical setting in the future.
Footnotes
Author Disclosure Statement
No competing financial interests exist.
