Abstract
Citherlet, Tom, Antoine Raberin, Giorgio Manferdelli, Nicolas Bourdillon, and Grégoire P Millet. Impact of the menstrual cycle on the cardiovascular and ventilatory responses during exercise in normoxia and hypoxia. High Alt Med Biol. 26:55–62, 2025.
Introduction:
Ovarian hormones influence several physiological functions in women. This study investigated how the hormonal variations across the menstrual cycle (MC) impact cardiovascular and ventilatory responses during rest and moderate exercise in normobaric hypoxia.
Methods:
Thirteen eumenorrheic women were tested during the early follicular (Fol1), late follicular (Fol2), and mid-luteal (Lut3) phases with measurement of hormonal levels. Heart rate (HR) variability, blood pressure, and baroreflex sensitivity (BRS) were evaluated at rest in normoxia. Ventilation (VE), peripheral oxygen saturation, and HR were monitored at rest and during moderate-intensity cycling exercise in hypoxia (FiO2 = 14%).
Results:
Despite expected hormone level variations, no significant changes were observed across the MC in HR variability (root mean square of successive differences; 64 (95% confidence interval [47, 81]) at Fol1, 54 [42, 66] at Fol2, 60 [44, 77] ms at Lut3), blood pressure (mean blood pressure; 85 [79, 90]), 87 [81, 93]), 84 [77, 92] mmHg), BRS (26 [17, 36], 28 [20, 35], 23 [17, 29] ms/mmHg), VE (rest: 8.9 [7.9, 9.8], 9.5 [9.0, 9.9], 9.0 [8.1, 9.9]; exercise: 53 [41, 66], 51.1 [36.4, 65.7], 54.4 [34.0, 74.8] l/min), peripheral oxygen saturation (rest: 89.8 [87.4, 92.1], 91.9 [88.7, 95.0], 90.2 [87.8, 92.6]; exercise: 80.5 [77.4, 83.5], 84.4 [80.4, 88.3], 81.9 [78.3, 85.4] %) HR (rest: 69.7 [60.2, 79.1], 70.8 [63.2, 78.3], 70.5 [64.0, 77.0]; exercise: 148 [136, 160], 146 [132, 161], 146 [132, 160] bpm), and cycling efficiency (0.17 [0.16, 0.18], 0.17 [0.13, 0.21], 0.16 [0.15, 0.18] %) (all p > 0.05).
Discussion:
From a practical point of view, there is no strong evidence of any usefulness of monitoring hormonal variations and the MC phases for women exercising in hypoxia.
Introduction
The underrepresentation of women in sports medicine research highlights an urgent need for targeted research on women’s unique physiology (Costello et al., 2014). One of the main characteristics of female physiology is the variation in ovarian hormone levels across the menstrual cycle (MC). The early follicular phase (Fol1) is characterized by low levels of estradiol (E2) and progesterone (P4), with E2 levels rising substantially during the late follicular phase (Fol2) while P4 levels remain low. Subsequently, during the mid-luteal phase (Lut3), a peak in both E2 and P4 levels occurs, followed by a progressive reduction as the cycle reaches its end (Elliott-Sale et al., 2021).
These physiological fluctuations in ovarian hormones impact several systems, particularly the cardiovascular and respiratory systems, and therefore could have numerous implications for female health and athletic performance (D’Souza et al., 2023). Given the central role of the cardiovascular and respiratory systems in adapting to acute reductions in inspiratory and arterial oxygen partial pressure as observed in high altitude, exploring the potential repercussions of ovarian hormone fluctuations on cardiovascular and ventilatory responses during both rest and exercise in hypoxic conditions is of interest. Acute adaptations to hypoxia include increased heart rate (HR) and ventilation (V̇E) (Burtscher et al., 2022). These rapid adaptations are critical for maintaining sufficient oxygen delivery to vital organs. Several specific responses in women to altitude have been highlighted in recent literature reviews (Burtscher et al., 2023; Horakova et al., 2023; Raberin et al., 2023).
The autonomic cardiovascular control can be assessed noninvasively with HR variability (HRV) and baroreflex sensitivity (BRS), critical markers of cardiovascular health (Task Force, 1996).
HRV is the analysis of the fluctuations in time intervals between consecutive heartbeats. It reflects the regulatory capacity of the autonomic nervous system over cardiac activity by assessing the balance between sympathetic and parasympathetic modulations to the heart (Task Force, 1996). Fluctuations in HRV can occur during different menstrual phases due to E2 and P4 level variations which are thought to influence the autonomic nervous system’s control. In particular, HRV was shown to decrease from the follicular to the luteal phase (Schmalenberger et al., 2019).
Baroreflex is a crucial regulatory system for the short term and chronic control of blood pressure (BP), ultimately ensuring adequate tissue perfusion throughout the body (La Rovere et al., 2008). BRS is quantified by measuring the changes in beat-to-beat HR in response to changes in beat-to-beat BP. Ovarian hormones, in particular E2, were shown to modulate BRS, yet the research findings on the effect of the MC on BRS remain unclear (Wenner and Stachenfeld, 2012).
In terms of the respiratory system, V̇E and peripheral oxygen saturation (SpO2) are critical markers, also potentially influenced by ovarian hormones. P4 has been shown to stimulate V̇E and E2 to potentiate its effect (Behan and Wenninger, 2008). It may therefore be expected that the responses to hypoxia and exercise are enhanced during Lut3, when the levels of E2 and P4 are elevated.
A large cohort study of 3,994 sea-level residents examined the effect of the MC phases during a hypoxic exercise test (Richalet et al., 2012). Unfortunately, no values of V̇E during exercise nor values of HR, SpO2, V̇E, hypoxic ventilatory response, or HRV measured at rest were reported in this study, but the authors clearly showed that during exercise both the SpO2 and hypoxic ventilatory response were maximal during the early luteal/mid-luteal phase, in line with a previous report (Schoene et al., 1981). Conversely, exercise V̇E was not higher in normoxia or acute hypoxia in the mid-luteal phase, when compared with the follicular phase in eight female sea-level residents (Beidleman et al., 1999). Moreover, resting SpO2, V̇E, or hypoxic ventilatory response were similar between 14 women in their follicular phase and 8 other women in their luteal phase, both in normoxia and at 4300 m of altitude (Muza et al., 2001). No difference between the MC phases was also reported on factors that may influence the cardiovascular responses and consequently the exercise capacity in altitude; for instance, the sympatho-adrenal response to exercise (i.e., plasma epinephrine and norepinephrine) (Mazzeo et al., 2000) as well as the change in HR or mean and diastolic BP (Mazzeo et al., 1998) were similar between the follicular and the luteal phases at sea level and at 4300 m.
Overall, a recent introductory paper of a series of reviews by the International Climbing and Mountaineering Federation (UIAA, Bern, Switzerland) Medical Commission (Horakova et al., 2023) highlighted the paucity of literature on women at high altitude.
Due to the large intra-individual variability in MC phases duration and in ovarian hormone concentrations (Elliott-Sale et al., 2021), one limitation of such retrospective analyses of large cohorts is that the MC phase determination being self-declared is likely inaccurate since no ovarian hormones were measured. The comparison of different groups is also not optimal due to the inter-individual variability. To our knowledge, there is no previous study using an intra-individual randomized protocol to test the ventilatory and cardiovascular responses to hypoxia in the same female participants both at rest and exercise at three different time points of their MC.
Therefore, the present study aimed to determine whether the MC influences the cardiovascular and ventilatory responses at rest and during exercise in normobaric hypoxia in healthy eumenorrheic women. Based on the hypothesis that hormonal variations during the MC can influence these outcomes, we expected that Lut3 could result in modified cardiovascular and ventilatory responses to hypoxia in comparison with Fol1.
Methods
Participants
Thirteen eumenorrheic women (EW) participated in this study. Inclusion criteria were 20–40 years old, with regular menstruation (cycles 21–35 days, no reported menstrual irregularities, and no hormonal treatments 3 months before participation). Participants were required to be healthy, non-smoking, not pregnant, not have an overnight stay above 3000 m in the last 3 months, and to not be obese (body mass index <30 kg/m2). They were required to have no cardiovascular, respiratory, or central nervous system disorders, not be on β-blocker therapy, not have documented low Fe status, and not engage in competitive swimming or breath-hold diving.
Study design
Participants underwent blood sampling, cardiovascular function assessment, and exercise testing in normobaric hypoxia at three different MC phases (Fol1, Fol2, and Lut3) demonstrating unique hormonal states. Historical cycle data were used to forecast subsequent menstrual lengths, resulting in the measurement of Fol1 at 15 ± 11%, Fol2 at 44 ± 9%, and Lut3 at 76 ± 9% of the MC duration (Fig. 1). The participants’ MC duration was on average 27 ± 3 days. This study protocol received approval from the Research Ethics Committee of Canton Vaud (2022–00178), complied with the Declaration of Helsinki, and all participants provided written informed consent before participating.

Study design.
Blood sampling
A 6 ml venous blood sample was collected from the antecubital vein of the participants and immediately centrifuged at 3000 G for 7.5 minutes. The serum was placed into microtubes and stored at −80°C. Measurements of E2 and P4 in the serum were conducted to ensure the menstrual phase identification. Serum markers for iron storage, including ferritin, Fe2+, and C-reactive protein (CRP), were also assessed to eliminate confounding factors. E2 and P4 levels were quantified using a competitive enzyme-linked immunosorbent kit (E2 ELISA kit, MyBioSource®, San Diego, CA, USA and P4 ELISA kit, Abnova®, Taipei City, Taiwan, respectively). CRP and ferritin levels were measured using sandwich enzyme-linked immunosorbent kits (CRP ELISA kit, Abnova®, Taipei City, Taiwan and Ferritin ELISA kit, Elabscience®, Houston, Texas, USA, respectively). A colorimetric assay was used for the quantification of ferrous ion concentrations (Ferrous iron colorimetric assay kit, Elabscience®, Houston, Texas, USA).
Heart rate variability, blood pressure, and baroreflex sensitivity
HRV, BP, and spontaneous cardiovagal BRS were evaluated during a 6-min rest period with participants lying down in a quiet room following a 10-minute rest in the same position. They were evaluated with continuous BP measurements on the index and middle fingers using a photoplethysmography device combined to a double cuff (NIBP100D, Biopac Systems Inc., Goleta, CA, USA). The device was automatically calibrated before each test by measuring BP on the left arm of the participant.
BRS and average BP were quantified through custom MATLAB (R2023b, The MathWork Inc, Natick, MA, USA) routines. Mean blood pressure was calculated as follow:
HRV was derived from continuous BP measurements, as pulse rate variability has been shown to provide reliable results comparable with traditional HRV assessments under resting conditions (Schäfer and Vagedes, 2013). A custom MATLAB routine was used to identify BP peaks and calculate HRV values. The parameters monitored included mean HR, root mean square of successive differences (RMSSD), power spectrum density in the low-frequency (LF), and high-frequency (HF) bands.
Acute normobaric hypoxia
Tolerance to hypoxia was assessed during 5 minutes of rest, followed by 5 minutes of moderate-intensity cycling (1.5 W/kg) in a hypoxic chamber set with a FiO2 of 14% which simulates an altitude of ∼3500 m (LowOxygen Systems, Berlin, Germany). SpO2 and HR were monitored via an earlobe pulse oximeter (WristOx 3150, Nonin Medical, Plymouth, MN, USA), while V̇E was measured using a portable metabolic cart (MetaMax 3B, Cortex Biophysik GmbH, Leipzig, Germany). Data from SpO2 and HR were averaged over the last 10 seconds of rest and exercise, and V̇E values were averaged from the last 10 respiratory cycles. Cycling gross efficiency was calculated by dividing work by energy expenditure, expressed as a percentage. Energy expenditure was derived from the formula ([3.869 x V̇O2) + (1.195 x V̇CO2) x (4.186/60) x 1000]), where V̇O2 is oxygen consumption and V̇CO2 is carbon dioxide production (Moseley and Jeukendrup, 2001).
Statistical analysis
A statistical power analysis was conducted using G*Power 3.1 to determine the appropriate sample size for this study. A large effect size (Cohen’s d = 1.16) was previously reported by the study of Takano (1984), which showed a significant increase of 0.127 in mean hypoxic sensitivity from the follicular phase to the luteal phase in a longitudinal design. Therefore, for repeated measures one-way analysis of variance with three time points, a large effect (Cohen’s F = 0.04) was expected.
The power analysis was performed with a significance level (alpha) of 0.05 and a desired power (1-beta) of 0.80. This calculation indicated that a sample size of 12 participants would be necessary to ensure adequate power to detect statistically significant differences.
The comparison between the three phases was performed using a general linear model with repeated measures and corrected for multiple comparisons using Bonferroni correction. If Mauchly’s test indicated a violation of the assumption of sphericity, further corrections were applied to adjust the degrees of freedom. Shapiro–Wilk test, skewness, and kurtosis were used to check data distribution, and no additional corrections were needed. All statistical analyses were performed using SPSS, version 26 (IBM Corp., Armonk, NY, USA). The significance level for all tests was set at p < 0.05.
Results
The participant characteristics are summarized in Table 1.
Participant Characteristics
Values are mean [95% confidence intervals].
Fig. 2 describes the hormonal levels across the three MC phases. E2 and P4 levels were 5.9 (4.6, 7.2) pg/ml and 0.2 (0.1, 0.4) ng/ml at Fol1, 50.8 (1.6, 99.0) pg/ml and 1.0 (−0.1, 2.1) ng/ml at Fol2, and 16.9 (6.9, 27.0) pg/ml and 13.4 [8.8, 18.0] ng/ml at Lut3. Notably, P4 exhibited a significant increase at Lut3 compared with both Fol1 and Fol2 (p < 0.001).
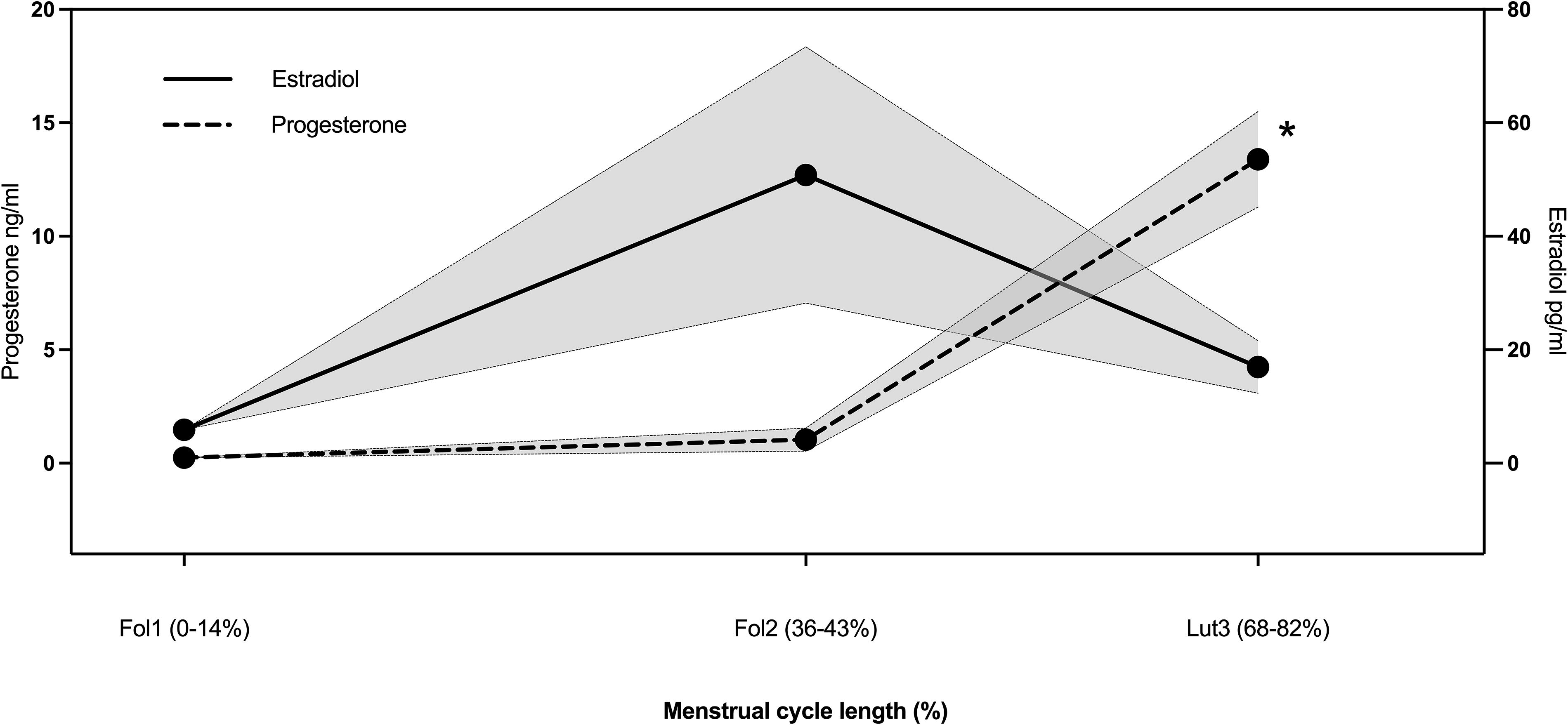
Changes in ovarian hormones at early follicular (Fol1), late follicular (Fol2), and mid-luteal (Lut3) phases of the menstrual cycle. *Significantly different from Fol1 and Fol2. Each symbol represents mean±SEM. SEM, standard error of the mean.
There was no significant difference in HRV, BRS, or BP across the three phases of the MC (Table 2).
Cardiovascular Responses in Normoxia
HF, high frequency; HR, heart rate; LF, low frequency; RMSSD, root mean square of successive differences between heartbeats. Values are mean [95% confidence intervals].
No significant difference in cardiovascular or ventilatory responses at rest and during moderate exercise in hypoxia was found across the MC (Table 3) except for SpO2 at exercise (p = 0.025). However, the Bonferroni correction revealed no differences in the paired comparison.
Cardiovascular and Ventilatory Responses to Hypoxia
HR, heart rate; SpO2, peripheral oxygen saturation; V̇E, ventilation. Values are mean [95% confidence intervals]. P-value below 0.05 is in bold.
Discussion
This study aimed to evaluate the effects of the MC on autonomic control in normoxia, as well as cardiovascular and ventilatory responses at rest and during exercise in hypoxia. Although hormonal levels did vary as expected during the MC, we did not observe any significant effect of these variations on the autonomic, cardiovascular, and ventilatory responses.
HRV at rest in normoxia did not differ across the MC, which aligns with previous studies (Kondo et al., 1989; Leicht et al., 2003). However, an extensive meta-analysis by Schmalenberger et al. (2019) highlighted medium to large variations in HRV between cycle phases. Most notably, they reported a significant decrease in HRV from the follicular to the luteal phase. A more fine-grained analysis revealed significant HRV decreases from the menstrual to the premenstrual and from the mid-to-late follicular to the premenstrual phase. It has been suggested that E2 and P4 affect HRV through both genomic and nongenomic pathways (Salerni et al., 2015). E2 seems to affect parasympathetic activity, whereas P4 modulates sympathetic activity (Leicht et al., 2003). Such a relationship provides further support for the reported cardioprotective effects of E2 (Leicht et al., 2003). The discrepancy between our results and these findings might be due to differing methods of defining cycle phases, participants characteristics (e.g., age, resting HR, athletic state, diet, and psychological traits like neuroticism and premenstrual symptomatology [Schmalenberger et al., 2019]) and HRV measurement (e.g., body position as assessing cardiovascular parameters in the supine position, tend to capture maximal parasympathetic activity and minimal sympathetic activity). Therefore, although our study did not find significant differences in HRV, it is worth noting that evidence suggests potential variations may exist.
BP at rest in normoxia did not change across the MC while previous research has shown inconsistent results. Some studies have reported higher BP levels during the luteal phase or at menstruation onset, while others have found decreased levels in the luteal phase, or no change throughout the cycle (Thakrar, 2024). Research indicates that E2 promotes vasodilation through both endothelium-dependent and endothelium-independent actions (Sarrel, 1999). E2 could thus lower BP and reduce the risk of hypertension in young women compared with men (Charkoudian et al., 2017). P4, on the other hand, opposes the effect of E2 on vascular tone and is vasoconstrictive (Sarrel, 1999). The divergent effects of E2 and P4 may explain the inconsistent findings observed between our study and existing literature, as well as within the literature itself. Further research is warranted to clarify these relationships.
BRS at rest in normoxia did not change across the MC. The impact of E2 on BRS has been nevertheless evidenced by the parallel pattern of BRS and E2 fluctuations, its elimination post-ovariectomy in rats, and the known effects of E2 in enhancing BRS via brainstem activity (Brooks et al., 2012). The absence of difference reported in the present study as in previous ones (Brunt et al., 2013; Fu and Ogoh, 2019; Vollebregt et al., 2006) may be attributed to the counteractive effects of P4. Although E2 has demonstrated its impact on BRS through various models, P4 has been shown to blunt BRS. Overall, conclusive evidence regarding the MC’s influence on BRS is still lacking.
V̇E in hypoxia at rest did not vary throughout the cycle phases aligning with some studies (Regensteiner et al., 1989; Takase et al., 2002), while increased V̇E during the luteal phase has also been reported (Takano, 1984). Similarly, during exercise, V̇E remained unchanged that corroborates previous findings (Beidleman et al., 1999; Dombovy et al., 1987; MacNutt et al., 2012; Regensteiner et al., 1989; Takase et al., 2002), whereas increased V̇E during the luteal phase has also been reported (Brutsaert et al., 2002).
Similar findings are observed in normoxia, with higher V̇E noted during exercise in the luteal phase in some studies but not in others (Beidleman et al., 1999). A higher V̇E during the luteal phase has been attributed to P4 stimulating V̇E and E2 potentiating this effect by increasing the number and affinity of P4 receptors (Brodeur et al., 1986).
Similarly, the hypoxic ventilatory response has been shown inconsistently to be higher during the luteal phase (Gargaglioni et al., 2019), with a suggested mechanism being increased excitability of the medullary respiratory center by P4 (Raberin et al., 2023).
Overall, it appears that the ovarian hormones influence is subtle—if not negligible—resulting in their effect on V̇E not being systematically detected, as in the present study.
The levels of SpO2 in hypoxia did not vary across the MC at rest and, while a difference was found at exercise, there was no difference in the paired comparison.
During the luteal phase, an increase in V̇E (Behan and Wenninger, 2008) and pulmonary diffusion capacity (Smith et al., 2015) could have contributed to higher SpO2 levels. Conversely, the potential minor decrease in oxygen affinity due to elevated body temperature, following the hemoglobin–oxygen association curve (Woyke et al., 2022) could have counteracted this effect. However, despite these physiological considerations, neither V̇E nor SpO2 exhibited significant fluctuations across the MC. In comparison, some found it unchanged during exercise in hypoxia (Dombovy et al., 1987; MacNutt et al., 2012), and others higher at Lut3 (Beidleman et al., 1999; Richalet et al., 2020). These physiological considerations appear to have a subtle influence on SpO2 levels, resulting in limited observable changes.
No HR difference across the cycle in hypoxia, both at rest and exercise was observed, which is in line with the literature (Beidleman et al., 1999; Brutsaert et al., 2002; Dombovy et al., 1987; Takase et al., 2002). During the luteal phase, decreased HRV (Schmalenberger et al., 2019), increased stress (Jain et al., 2023), or the potential impact of elevated body temperature on the sinoatrial node (de Jonge, 2003) could have contributed to an increased HR. Mazzeo et al. (1998) reported that for a given urinary norepinephrine excretion rate, HR was lower in the follicular compared with the luteal phase, but outside of it, no differences between phase assignments were observed. Overall, these factors seem to exert minor influence as many studies, including the present one, report no significant change in HR during rest or exercise across the MC (de Jonge, 2003).
Cycling efficiency in hypoxia did not vary throughout the MC, which is consistent with the absence of difference in the cardio-respiratory responses. These results align with some studies (Dombovy et al., 1987) but not with another (Hessemer and Bruck, 1985) that found it to be lower during the luteal phase. While some studies suggest some changes, the evidence is not conclusive.
Limitations
Several limitations need to be acknowledged. The alignment of the test with the peak hormonal phases is challenging due to individual variations in cycle length and fast hormone fluctuations (Hirschberg, 2022). Even though the hormonal concentrations were measured, it is unknown whether the timing could have been more precise and would have affected the results. One limitation is the low sample size (n = 13) of our study. However, despite that an a priori power calculation (see statistical analysis) resulted in a minimal sample size of 12 participants, one cannot rule out that some small—and probably non physiologically significant—differences may have been observed between the MC phases with a larger group of EW. Lastly, emerging evidences show that normobaric hypoxia is not a substitute of hypobaric hypoxia (Millet and Debevec, 2020). Although this point remains highly debated (Richalet, 2020) and beyond the strict differences in barometric pressure, due to the other environnemental factors (e.g., solar radiation, temperature, wind, and humidity), the present results in normobaric hypoxia may not necessarily be extrapolated to hypobaric hypoxia conditions and how female participants would respond and perform in real-world high altitude.
Conclusion
Overall, the present study reported that the MC does not exert a significant effect on cardiovascular function at rest in normoxia. In addition, we found that cardiorespiratory responses to hypoxia, both at rest and during exercise, remained consistent across different menstrual phases. This is in line with emerging evidence that the differences in most of the various physiological outcomes between MC phases, if present, are small (D’Souza et al., 2023; Elliott-Sale et al., 2020).
These findings suggest that the MC phase may not require specific consideration when evaluating cardiovascular and ventilatory responses in healthy women, particularly in the context of acute exposure to hypoxia.
Moreover, the absence of significant differences offers reassurance to women that their cardiovascular and respiratory functions are likely to remain stable throughout the MC and thus are unlikely to impact their overall functioning and submaximal performance.
Footnotes
Acknowledgment
The authors would like to thank the foundation SpArk for funding this study.
Authors’ Contributions
Contribution to conception or design: T.C., A.R., G.M., and G.P.M. Data collection: T.C. All authors contributed to data interpretation, drafting or critically revising the article, and have given final approval of the version to be published.
Data Availability
Data supporting this study’s findings can be obtained from the corresponding author upon reasonable request.
Author Disclosure Statement
The authors declare no competing interests.
