Abstract
Grimm, Mirjam, Lucie Ziegler, Annina Seglias, Maamed Mademilov, Kamila Magdieva, Gulzada Mirzalieva, Aijan Taalaibekova, Simone Suter, Simon R. Schneider, Fiona Zoller, Vera Bissig, Lukas Reinhard, Meret Bauer, Julian Müller, Tanja L. Ulrich, Arcangelo F. Carta, Patrick R. Bader, Konstantinos Bitos, Aurelia E. Reiser, Benoit Champigneulle, Damira Ashyralieva, Philipp M. Scheiwiller, Silvia Ulrich, Talant M. Sooronbaev, Michael Furian, and Konrad E. Bloch. SARS-CoV-2 Transmission during High-Altitude Field Studies. High Alt Med Biol. 25:197–204, 2024.
Background:
Throughout the severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2) pandemic, virus transmission during clinical research was of concern. Therefore, during high-altitude field studies performed in 2021, we took specific COVID-19 precautions and investigated the occurrence of SARS-CoV-2 infection.
Methods:
From May to September 2021, we performed studies in patients with chronic obstructive pulmonary disease (COPD) and in healthy school-age children in Kyrgyzstan in high-altitude facilities at 3,100 m and 3,250 m and at 760 m. The various implemented COVID-19 safety measures included systematic SARS-CoV-2 rapid antigen testing (RAT). Main outcomes were SARS-CoV-2-RAT-positive rate among participants and staff at initial presentation (prevalence) and SARS-CoV-2-RAT-positive conversion during and within 10 days after studies (incidence).
Results:
Among 338 participants and staff, SARS-CoV-2-RAT-positive prevalence was 15 (4.4%). During mean ± SD duration of individual study participation of 3.1 ± 1.0 day and within 10 days, RAT-positive conversion occurred in 1/237(0.4%) participants. Among staff working in studies for 31.5 ± 29.3 days, SARS-CoV-2-RAT-positive conversion was 11/101(10.9%). In all 338 individuals involved in the studies over the course of 15.6 weeks, the median SARS-CoV-2-RAT-positive incidence was 0.00%/week (quartiles 0.00; 0.64). Over the same period, the median background incidence among the total Kyrgyz population of 6,636 million was 0.06%/week (0.03; 0.11), p = 0.013 (Wilcoxon rank sum test).
Conclusions:
Taking precautions by implementing specific safety measures, SARS-CoV-2 transmission during clinical studies was very rare, and the SARS-CoV-2 incidence among participants and staff was lower than that in the general population during the same period. The results are reassuring and may help in decision-making on the conduct of clinical research in similar settings.
Introduction
During the severe acute respiratory syndrome coronavirus type 2 (SARS-CoV-2) pandemic, many clinical research projects were interrupted or considerably delayed because of the risk of viral transmission among study participants or staff and due to limited human and logistical resources (Audisio et al., 2022; Lasch et al., 2022). As in most countries of the world, the health care system in the Central Asian Republic of Kyrgyzstan was confronted with major challenges owing to the coronavirus disease 2019 (COVID-19) pandemic (Bumburidi et al., 2023; Dzushupov et al., 2021). In view of the fast spread of COVID-19 and the unknown effects of a high-altitude environment on SARS-CoV2 transmission (Pun et al., 2020), our international research consortium, the Swiss-Kyrgyz High-Altitude Medicine and Research Initiative, canceled all clinical studies on health effects of high-altitude exposure scheduled for 2020. After the decline of the COVID-19 incidence in the Kyrgyz Republic in spring 2021 high-altitude field studies were resumed.
The studies carried out in the summer 2021 focused on altitude-related adverse health effects in children and in patients with COPD, the latter being considered particularly vulnerable to COVID-19 (Leung et al., 2020). These research expeditions were conducted both in remote, isolated high-altitude mountainous regions under challenging conditions (e.g., lacking cell phone connection, deficient of convenient means of transportation requiring several hours of driving in difficult terrain to nearest settlements), as well as in the capital of Bishkek where communication and travel were easy and exposure to a large number of persons was common. We were thus concerned that circumstances that could not be avoided during the studies, such as staying in common eating and sleeping facilities or bus travel, would increase the risk of SARS-CoV-2 transmission. The decision to relaunch clinical research required a thorough analysis of epidemiological, logistical, and ethical aspects weighing potential harm to participants against desirable advances in research (Vissers et al., 2021). The considerations also comprised a strategy to mitigate the risks. Unfortunately, no robust evidence was available to reliably assess the specific risks and the feasibility and effectiveness of various measures for preventing SARS-CoV-2 transmission in the setting of the planned studies. Therefore, we had to design a safety concept based on existing general recommendations of the Kyrgyz Republic and the World Health Organization (WHO, 2022a, WHO, 2022b) and adapted it to the specific settings of our studies. The main pillars of the safety concept are outlined in Table 1 and further detailed in the Supplementary Data.
Main Points of the COVID-19 Safety Concept Implemented during the 3 Studies a
The full COVID-19 safety concept is available as an online supplement.
The aim of the current investigation was to analyze the prevalence of SARS-CoV-2 infection at the initial presentation of participants and staff, as well as the incidence of SARS-CoV-2 infection among these individuals during 3 high-altitude field studies carried out in the summer of 2021 in the Kyrgyz Republic. Data were compared to the SARS-CoV-2 background infection rate reported by the World Health Organization for the general population of the Kyrgyz Republic for the same time period (WHO, 2021) to evaluate whether being involved in the studies was associated with an increased risk of acquiring a SARS-CoV2 infection compared to staying in the usual environment (at home).
Methods
Study design and setting
We analyzed data from participants and staff of high-altitude field studies carried out by the Swiss-Kyrgyz High Altitude Medicine and Research Initiative from May 27 to September 12, 2021, in the Kyrgyz Republic at three sites. Data were collected as part of these studies and according to the specifically implemented safety concept (Table 1 and Supplementary Data).
Study 1 was a diagnostic accuracy study evaluating the performance of structured self-monitoring in detecting signs of impending altitude-related adverse health effects (ARAHE) in lowlanders with chronic obstructive pulmonary disease (COPD). The participants stayed for 1 day and night at the National Center of Cardiology and Internal Medicine (NCCIM), Bishkek, and for 2 days and nights at a high-altitude clinic in Too Ashu, 3,100 m, ClinicalTrials.gov NCT03957759 (Furian et al., 2022).
Study 2 was a randomized controlled trial evaluating acetazolamide for late prevention of ARAHE in patients with COPD in whom structured self-monitoring indicated impeding ARAHE during a stay for 2 days at the Too Ashu clinic, 3,100 m. ClinicalTrials.gov NCT04913389.
Study 3 was a case–control study comparing the prevalence of sleep apnea among school-age children living at high and low altitudes (Tien Shan mountains, 2,500–3,500 m, and Bishkek, 760 m). The study was carried out in the Aksay health post, 3,200 m, Naryn Oblast, and at the National Center of Cardiology and Internal Medicine, Bishkek, 760 m (Grimm et al., 2023).
Participants
All participants and study staff members of the three studies were included in the analysis after approval of the protocol by the Ethics Committee of the NCCIM, Bishkek, Kyrgyz Republic, and the Cantonal Ethics Committee Zurich, Zurich, Switzerland, and after obtaining written informed consent of participants. All 237 study participants were Kyrgyz residents. Among 101 staff, 82 were Kyrgyz health care workers (physicians, nurses, medical students) and logistic staff (drivers, personnel responsible for maintenance of infrastructure, cleaning and kitchen personnel), and 19 staff were Swiss health care workers.
Interventions
In studies 1 and 2, groups of 5 to 10 patients with COPD had baseline examinations in Bishkek (760 m) and subsequently traveled to the Too Ashu high-altitude clinic at 3,100 m by bus within 6 hours and stayed there for 2 days. During altitude travel, participants monitored their symptoms and pulse oximetry according to a predefined protocol. In study 2, patients with symptoms and/or signs of impending ARAHE were randomized to receive either acetazolamide capsules (375 mg/d) or placebo for the duration of their further stay at 3,100 m. In study 3, groups of 2–9 school-age children living in the Aksay region (3,200 m) and a control group of similar age living in Bishkek were examined clinically and underwent respiratory sleep studies (Grimm et al., 2023).
Participants and staff of the 3 studies followed instructions delineated in the specifically designed COVID-19 safety concept (Table 1, Supplementary Data).
Assessments
Questionnaire and clinical data
Date, time, location, and results of SARS-CoV-2 rapid antigen testing (RAT) and travel and clinical data collected in studies 1–3 were recorded.
SARS-CoV-2-testing
All participants and staff had to undergo systematic point-of-care RAT according to predefined rules (Table 1 and Supplementary Data). Thus, all participants and staff were tested before admission to the study site; staff were additionally tested every 7 days during involvement in study activities; kitchen staff were tested every 4 days. Admission to studies as participant or staff was permitted only if he/she was asymptomatic and a RAT, performed outside the study building in a designated area, was negative (Rhee et al., 2023). RAT was performed on nasopharyngeal swabs and processed according to manufacturer instructions of the Quidel Corp. SOFIA SARS Antigen Fluorescence Immunoassay. It was the first validated point-of-care kit available at the time of the studies (CDC, 2020; Dinnes et al., 2021; Porte et al., 2021). It provides results within 15 minutes after insertion of the sample into the device. The negative percentage agreement with a reverse transcription polymerase chain reaction (RT-PCR) comparator assay was 99.6% (95% CI = 98.5 to 99.9) in 581 individuals with symptoms of respiratory infection and a RT-PCR positivity rate of 18.8% (Quidel, 2023).
Outcomes
The number and characteristics of study participants and staff, the locations, and results of RAT were recorded. In RAT-positive persons, the severity of illness was recorded in terms of requiring hospital admission for treatment and whether any sequelae persisted for more than 1 month or not.
Data analysis and statistics
Descriptive statistics are presented as counts and proportions, medians (quartiles), or mean ± SD. For the purpose of this study, the SARS-CoV-2 prevalence was defined as proportion of persons involved in studies (participants and staff) with a positive RAT at initial presentation of the study. The SARS-CoV-2 incidence was defined as the proportion of persons who converted to RAT positive during the study or within 10 days thereafter divided by exposure time, i.e., the time of study participation (patients with COPD, school-age children) or working period in the study (staff). The 10-day observation covered a range up to the 95th percentile of the distribution of the SARS-CoV-2 incubation period (Lauer et al., 2020). Weekly incidence rates for successive weeks were computed over the entire study period and expressed in the percentage of participants and staff present during the corresponding week. Weekly incidence rates were also computed for the entire Kyrgyz population based on data retrieved from official resources (National Statistical Committee of the Kyrgyz Republic, 2021; WHO, 2021). Between-group comparison of incidence rates was performed by the Wilcoxon rank-sum test. A probability of p <0.05 was considered significant.
Results
In all three studies, a total of 338 persons, 237 participants and 101 staff, were involved during a cumulative exposure time to the studies of 549.7 person × weeks. Table 2 presents the characteristics of the population and results of RAT. At initial presentation to the study, 15 individuals (4.4%) were RAT positive. According to the COVID-19 safety concept (Table 1 and Supplementary Data), these individuals were not admitted to the study site for at least the next 2 weeks and subsequently were considered for study participation only if they were asymptomatic and RAT negative. Individuals who were asymptomatic and RAT negative at initial presentation were admitted and remained in the study for a mean ± SD individual duration of 11.4 ± 20.7 days. During this period and within 10 days, RAT-positive conversion occurred in 11/338 individuals (3.3%). Correspondingly, the median of weekly SARS-CoV-2 incidence rates over the course of the 109 days (15.6 weeks) of the three studies was 0.00 (quartiles 0.00; 0.64). As four staff members became SARS-CoV-2 RAT positive within two consecutive days in studies 1 and 2 (Fig. 1), studies were halted for 5 days (July 12 to 17, 2021). All current participants were discharged and no new individuals were admitted during this period. Affected staff was isolated, and close contacts were quarantined for at least 8 days. The majority of SARS-CoV2 RAT-positive cases diagnosed in the three studies were asymptomatic; some were febrile, and had cough, headache, or general malaise for a few days. None of the COVID-19 cases had to be hospitalized. Fever was treated symptomatically; one staff member used an inhaled corticosteroid for cough. Staff was readmitted to the studies after a minimal isolation period of 8 days if asymptomatic and RAT negative. One patient with COPD reported that he had tested positive for SARS-CoV-2 and developed minimal illness 10 days after the end of his study participation. No other study participant acquired SARS-CoV-2 infection. All infected individuals recovered without sequelae within a few days.
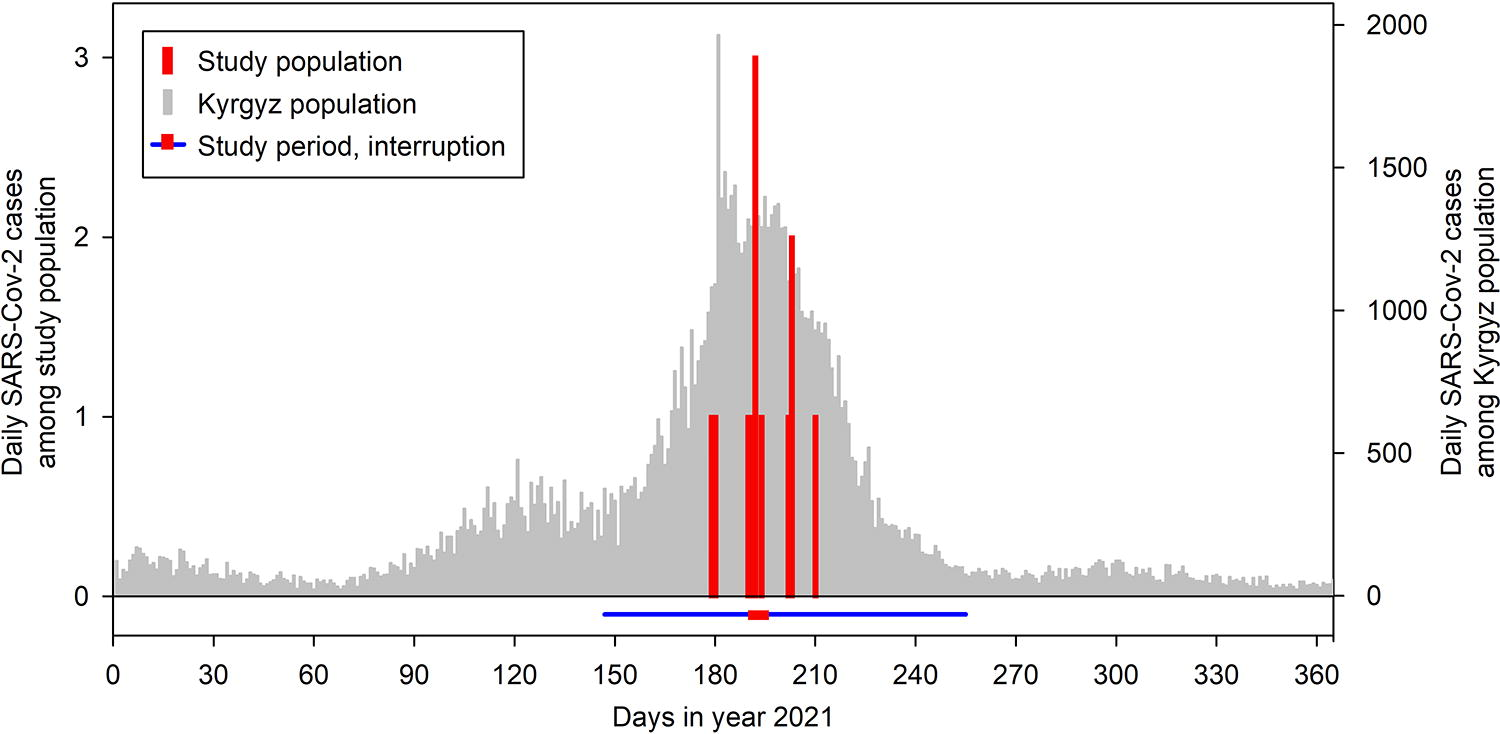
Daily SARS-CoV-2 cases diagnosed by rapid antigen testing among the 338 individuals involved in the 3 studies (participants and staff, red columns, left y-axis). The study period was from May 27 to September 12, 2023 (blue line); studies were interrupted for 5 days, from July 12–17, 2023 (red bar overlying the blue line). Gray columns (right y-axis) represent daily SARS-CoV-2 cases reported for the Kyrgyz population over the course of the year 2021.
Population Characteristics and Results of SARS-CoV-2 Rapid Antigen Testing of Participants and Study Staff in All 3 Studies
Mean ± SD, n (percent); RAT, rapid antigen testing.
Over the course of the three studies, i.e., from May 27 to September 12, 2021, at total of 42,959 cases of SARS-CoV-2 infection, corresponding to 3,967 cases (quartiles 2,106;7,462) per week, were reported for the Kyrgyz Republic with a population of 6,636,800 individuals (National Statistical Committee of the Kyrgyz Republic, 2021; WHO, 2021). Based on these data, the median SARS-CoV-2 incidence of the Kyrgyz population over the period of the studies was 0.06%/week (quartiles 0.04; 0.11), p = 0.013 versus the median incidence among participants and staff involved in the three studies. The figure illustrates that the study period fell into the time of the peak of the third COVID-19 wave in the Kyrgyz Republic.
To further investigate the occurrence of SARS-CoV-2 infection, separate analyses were performed for the study participants with COPD, the school-age children, and the staff that attended all three studies (Table 3). A total of 173 individuals participated in studies 1 and 2 in patients with COPD who were older (mean age 59.0 years) than the staff (49.8 years) and the children (11.2 years). Thirteen of 173 patients with COPD (7.5%) and 1 of 64 children (1.6%) were RAT positive at initial presentation compared with 1 of 101 staff (1.0%).
Population Characteristics and Results of SARS-CoV-2 Rapid Antigen Testing of Participants and Study Staff Listed according to Studies
One patient with COPD was diagnosed 10 days after completion of study participation.
p = 0.242 Wilcoxon rank order test staff vs. participants in studies 1, 2, and 3.
RAT, rapid antigen testing.
Over the course of the studies, 10/101 (9.9%) staff converted to RAT positive compared with 1/237 (0.4%) participants. The mean duration of stay in the three studies was 31.5 ± 29.3 days for staff and 2.8 ± 1.0 days for participants. The median incidence of RAT-positive conversion among participants and staff was similar (0.00%/week [quartiles 0.00; 0.00] and 0.00%/week [0.00; 6.48], p = 0.242, Table 3). One patient with COPD reported to have tested SARS-CoV-2 positive 10 days after the end of his study participation. It was not possible to identify the source of his infection and whether it occurred during his study participation or in the 10 days he spent at home thereafter. None of the school-age children converted to RAT positive during or within 10 days of study participation.
Discussion
We studied SARS-CoV-2 infection among participants and staff of three clinical studies carried out in remote mountain areas and in the capital of Kyrgyzstan during the emergence of the third COVID-19 wave in that country. The main finding was that despite a prevalence of 4.4% of SARS-CoV-2 RAT positivity among individuals presenting for study involvement as participants or staff, the median conversion rate (incidence) among those admitted to the studies was very low (median 0.00%/week). This was even lower than the background incidence reported for the entire population of the Kyrgyz Republic over the same period (0.06%/week). Our data suggest that the safety measures (Table 1 and Supplementary Data) implemented during the study to prevent or reduce SARS-CoV-2 transmission were effective and at least counterbalanced the increased risk of viral spread related to study participation. The 15 individuals who tested SARS-CoV-2 positive at initial presentation to the study (prevalence of 4.4%) presumably reflected the epidemiological situation prevailing in Kyrgyzstan at that time, consistent with the high caseload in the Kyrgyz community (Fig. 1) (WHO, 2021). As the vast majority of the 15 RAT-positive individuals were asymptomatic at presentation, our practice of performing systematic RAT in each individual before admission to study locations has prevented exposure of participants and staff to SARS-CoV-2 and very likely contributed to the low viral spread within the study. A similar positive effect of admission testing during significant community transmission has been reported for hospital settings (Pak et al., 2023; Rhee et al., 2023; Talbot et al., 2023).
In only one participant, a patient with COPD, conversion to SARS-CoV-2 RAT positive was associated with exposure to the study. However, the conversion was diagnosed only 10 days after study discharge. Taking the mean incubation time of SARS-CoV-2 of 5.0 days (Lauer et al., 2020; Wu et al., 2022) into consideration it is much more likely that the patient acquired the infection after returning to his community rather than during the study.
Although the absolute proportion of SARS-CoV-2 RAT positive conversion was higher among staff (9.9%) than among study participants (0.4%), the median incidence was similarly low in both groups (0.00%/week) due to the considerably longer exposure of staff to the study environment (Table 3). We assume that strict adherence to the COVID-19 safety measures by the staff effectively prevented excessive viral transmission even though the staff was quite active and mobile within and between study locations and exposed to a greater number of contacts over longer time periods compared with study participants.
Mo and coworkers (Mo et al., 2021) reported SARS-CoV-2 transmission in 10.5% of patients and 11.0% of staff in four teaching hospitals in the UK assuming incubation periods of 4–7 days over the course of 10 months in 2020. Our data compare favorably with these and other related observations (Chou et al., 2020), possibly due to a more controlled setting in our studies and as we did not admit acutely ill patients. Moreover, the risks of health-care associated SARS-CoV-2 transmission such as the number and duration of contacts and proximity between persons, oxygen administration, and exposure to secretions could be largely avoided in our studies while various measures that reduce transmission such as hand washing, use of gloves, and disinfection were implemented (Chou et al., 2020). Thus, the goal of our safety measures (Table 1), i.e., to protect vulnerable study participants and staff from contracting SARS-CoV-2, was achieved. This is remarkable as during the study period, the incidence of SARS-CoV-2 cases in general (Fig. 1) (WHO, 2021) and that of the delta variant (Dzhumakanova, 2022) associated with a high transmissibility and disease severity, in particular, was rising in the Kyrgyz population, while immunity, as evaluated by SARS-CoV-2 antibody seroprevalence in a representative sample of the Kyrgyz population, was only 48.7% (Popova et al., 2022).
The COVID-19 safety concept implemented in our studies included all WHO recommendations for avoidance of SARS-CoV2 spread, in particular, hygiene measures (hand washing, disinfection, wearing a mask) and distancing (reducing the number of individuals within rooms, time-based separation) (WHO, 2022b). In addition, several organizational measures were taken to reduce exposure, i.e., the time each participant stayed in the studies and the number of assessments were reduced as feasible without compromising the main study goals. Most importantly, by systematic admission testing of all newcomers for SARS-CoV-2 by RAT and preventing access of RAT-positive and/or symptomatic persons to study locations, exposure to SARS-CoV-2 was considerably reduced (in 15 instances, Table 2) (Pak et al., 2023; Talbot et al., 2023). Because of the delay of 2–4 days between infection and/or PCR-based detection of SARS-CoV-2 in nasopharyngeal swabs and emergence of a positive RAT (Mallett et al., 2020; Quidel, 2023), and accounting for a minor false negative rate of RAT of 0.4% (upper 95% confidence limit 1.5%) (Quidel, 2023), a small risk of SARS-CoV-2 introduction to the study cohort remained. This and the known imperfect efficacy of other safety measures explain why SARS-CoV-2 transmission took place within the study in 10 staff members and possibly one patient (Table 3). Performing RAT at regular intervals in staff and a timely response to the emergence of RAT-positive cases by interrupting the study for 5 days (Fig. 1) may have helped to prevent further viral spread.
Our study has several limitations. Although it was a prerequisite for Swiss staff to present official evidence of either prior vaccination or SARS-CoV-2 infection and a negative RT-PCR test for SARS-CoV-2 at the time of immigration into the Kyrgyz Republic in the summer of 2021, Kyrgyz staff and participants did unfortunately not have access to vaccination and their individual status of previous exposure to SARS-CoV-2 could not be objectively verified. The comparison of the incidence of SARS-CoV-2 infection among individuals involved in the current studies and the Kyrgyz population may be biased to some extent by underreporting of cases in the community and by the reduced accuracy of the available RAT (CDC, 2020; Dinnes et al., 2021; Porte et al., 2021) compared to RT-PCR testing which was not feasible in our studies. However, this did not affect the main conclusions of a very low transmission rate and absence of severe COVID-19 cases among individuals involved in the studies.
Conclusions
Our data collected during the third wave of the SARS-CoV-2 pandemic in the Kyrgyz Republic in participants and staff of clinical studies carried out under challenging conditions in remote high-altitude locations and in a university hospital demonstrate a very low virus transmission rate among persons involved in the studies. Our safety concept based on WHO recommendations (WHO, 2022c) and complementary measures specifically designed for the study settings most likely contributed to the prevention of viral transmission offsetting some unavoidable risks related to study participation. The feasibility and effectiveness of the described preventive measures are reassuring for investigators responsible for the conduct of clinical high-altitude investigations, as well as for individuals (patients, staff) considering participation in such studies. We propose that the concept may serve as a template that can be further elaborated and validated for application in future clinical trials in environments at high risk of infection transmission or during pandemic times.
Footnotes
Authors’ Contributions
M.G., K.E.B.: Substantial contributions to the conception and design of the work, acquisition, analysis, and interpretation of data for the work, drafting it and final approval of the version to be published, and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. L.Z., A.S., M.M., K.M., G.M., A.T., S.S., S.R.S., F.Z., V.B., L.R., M.B., J.M., T.L.U., A.F.C., P.R.B., K.B., A.E.R., B.C., D.A., P.M.S., S.U., T.M.S., and M.F.: Substantial contributions to the acquisition, analysis, and interpretation of data for the work and reviewing it critically for important intellectual content and final approval of the version to be published and agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Author Disclosure Statement
The authors have declared no conflict of interest.
Funding Information
This work was supported, in part, by grants from the Swiss National Science Foundation (grant number 32003B_192048), the Bockhoff Foundation, and the Swiss Lung Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
