Abstract
Mundo, William, Lilian Toledo-Jaldin, Alexandrea Heath-Freudenthal, Jaime Huayacho, Litzi Lazo-Vega, Alison Larrea-Alvarado, Valquiria Miranda-Garrido, Rodrigo Mizutani, Lorna G. Moore, Any Moreno-Aramayo, Richard Gomez, Patricio Gutierrez, and Colleen G. Julian. Is maternal cardiovascular performance impaired in altitude-associated fetal growth restriction? High Alt Med Biol. 23:352–360, 2022.
Introduction:
The incidence of fetal growth restriction (FGR) is elevated in high-altitude resident populations. This study aims to determine whether maternal central hemodynamics during the last trimester of pregnancy are altered in high-altitude FGR.
Methods:
In this cross-sectional study of maternal-infant pairs (FGR, n = 27; controls, n = 26) residing in La Paz, Bolivia, maternal heart rate, cardiac output (CO), stroke volume, and systemic vascular resistance (SVR) were assessed using continuous-wave Doppler ultrasound. Transabdominal Doppler ultrasound was used for uterine artery (UtA) resistance indices and fetal measures. Maternal venous soluble fms-like tyrosine kinase-1 (sFlt1) levels were measured.
Results:
FGR pregnancies had reduced CO, elevated SVR and UtA resistance, fetal brain sparing, and increased maternal sFlt1 versus controls. Maternal SVR was positively associated with UtA resistance and inversely associated with middle cerebral artery resistance and birth weight. Maternal sFlt1 was greater in FGR than controls and positively associated with UtA pulsatility index. Women with elevated sFlt1 levels also tended to have lower CO and higher SVR.
Conclusion:
Noninvasive assessment of maternal cardiovascular function may be an additional method for detecting high-risk pregnancies at high altitudes, thereby informing the need for increased surveillance and appropriate allocation of resources to minimize adverse outcomes.
Introduction
Fetal growth restriction (FGR) poses an immediate threat to newborn survival and increases disease risk across the life span (Lees et al., 2015; Korkalainen et al., 2017; Colella et al., 2018). Although FGR represents an escalating global health challenge, it disproportionally affects low- and middle-income countries (de Onis et al., 1998; Katz et al., 2013; Lausman and Kingdom, 2013; Gordijn et al., 2018). Bolivia is a low-income country with one of the highest maternal and infant mortality rates in the Western Hemisphere (World Bank, 2015a, 2015b) due, in part, to the fact that more than half of its population resides at high altitudes (≥2,500 m).
The incidence of FGR is threefold greater than in lower altitudes even under comparable health care conditions (Julian et al., 2007, 2009; Soria et al., 2013). A critical barrier to reducing perinatal mortality at high altitude is the lack of noninvasive and easy-to-use tools for early detection of high-risk pregnancies, thus allowing for increased surveillance, careful timing of delivery, and appropriate allocation of resources to minimize adverse outcomes (Lindqvist and Molin, 2005; Bujold et al., 2010; Gardosi et al., 2013).
Compared to a healthy pregnancy, maternal cardiac output (CO) and stroke volume (SV) are reduced, and systemic vascular resistance (SVR) and uterine artery (UtA) resistance indices are elevated in FGR pregnancy (Roberts et al., 2018; Perry et al., 2020; Ling et al., 2021). High maternal SVR positively associates with UtA pulsatility index (PI), a predictor of adverse perinatal outcomes independent of fetal size, and umbilical artery (UmbA) or middle cerebral artery (MCA) Doppler indices indicative of brain sparing (Monaghan et al., 2018; Perry et al., 2019; Tay et al., 2019). Although maternal CO and SV are reportedly reduced at high versus low altitudes (Kametas et al., 2004), unknown is whether maternal central hemodynamics are impaired in FGR versus healthy pregnancy at high altitudes and, if so, whether such effects are associated with increased uteroplacental and fetal vascular resistance or elevated soluble fms-like tyrosine kinase-1 (sFlt1), an indicator of endothelial dysfunction.
In this cross-sectional study, we sought to determine whether maternal cardiac performance is reduced and SVR elevated in FGR pregnancy at high altitude and, if so, to define its relationship to maternal SVR, uteroplacental vascular resistance, and fetal circulatory changes. Since one mechanism underlying altered maternal systemic hemodynamics in FGR pregnancy may be endothelial dysfunction, we measured circulating levels of sFlt1, an antiangiogenic factor elevated in vascular disorders of pregnancy (Maynard, et al., 2003a; Dávila et al., 2010; Birdir et al., 2018), and tested whether sFlt1 levels were positively associated with maternal SVR or resistance within the uteroplacental or fetal circulation. To our knowledge, this is the first integrated analysis of maternal systemic hemodynamics, uteroplacental resistance, fetal hemodynamics, birth weight, and sFlt1 in the same cohort. Terms and abbreviations used in the article are listed in Table 1.
Terms and Abbreviations
Materials and Methods
Ethical approval
Study participants were enrolled following their written informed consent to study procedures approved by the University of Colorado Multiple Institutional Review Board (No. 18-0210) and its Bolivian equivalents operated by the Caja Nacional de Salud and the Hospital Materno-Infantil.
Study population and design
Subjects were 53 mother-infant dyads (26 controls and 27 FGR cases) receiving prenatal care and scheduled to deliver at Hospital Materno-Infantil, the largest maternal-child hospital serving the high-altitude cities of La Paz and El Alto, Bolivia (3,600–4,100 m). Studies were conducted from September 2019 through May 2021. Inclusion criteria were maternal age >18 and <45 years and between 32 and 34 weeks of pregnancy at enrollment, no known risk for preeclampsia (e.g., no diabetes [type I, II or gestational], chronic hypertension, or cardiopulmonary disease), no evidence of anemia (hemoglobin <8.5 g/dl), prepregnant body mass index <30 kg/m2, nonsmoker for the duration of the current pregnancy, singleton pregnancy, residence at high altitude for at least 10 years, scheduled to deliver by Cesarean section to permit balancing cases and controls by gestational age at delivery, and maternal self-identification as being of either Andean or Mestiza ancestry.
Fetal inclusion criteria were no evidence of fetal aneuploidy, genetic disorders, or structural anomalies. Controls were defined as healthy, normotensive pregnancy with an appropriately grown fetus. While we acknowledge that there is no definitive method to differentiate between pathologically growth-restricted and constitutionally small fetuses, we distinguished probable cases of pathological growth restriction as those with an estimated fetal weight (EFW) <10th percentile according to the Hadlock C formula and an UmbA systolic and end-diastolic (S/D) velocity ratio >95th percentile or a cerebroplacental ratio (CPR; = MCA PI/UmbA PI) <1.08; CPR values below this cutoff are a sensitive index of brain sparing and adverse outcomes (Shahinaj et al., 2010; Rizzo et al., 2020).
Maternal demographics, reproductive history, and health history were obtained from medical records or questionnaires. Self-reported medical information on questionnaires was double-checked against health records. At 34 ± 2 weeks of pregnancy, maternal hemodynamics, and Doppler ultrasound studies of UtA resistance indices, fetal hemodynamics and fetal growth were assessed on the same day; at this time, study personnel also obtained maternal blood pressures by sphygmomanometer and oxygen saturation by pulse oximetry. Newborn and delivery complications, birth weight, gestational age at delivery, infant sex, length, head circumference, and Apgar scores were recorded from hospital records.
Biological sampling
Maternal peripheral blood samples were drawn by routine venipuncture from an antecubital vein into ethylenediamine tetraacetic acid-coated collection tubes at the time of hemodynamic examination; whole blood was used to measure hemoglobin concentrations in duplicate (HemoCue Hb 201+ System; HemoCue, Angelholm, Sweden), and plasma was processed for the measurement of sFlt1 by enzyme-linked immunosorbent assay (R&D Systems, Inc., Minneapolis, MN). Plasma samples were flash-frozen and stored at −80°C before analysis.
Hemodynamic studies
Maternal cardiovascular measurements were made using an ultrasound cardiac output monitor (USCOM-1A; USCOM Ltd., Sydney, Australia) as per published methods (Perry et al., 2020). Heart rate (HR) was obtained by measuring the time between each Doppler profile. CO (SV × HR), SVR (mean arterial pressure [MAP]/CO), cardiac index (CI), and SVR index (SVRI) were also calculated. SVRI was calculated as: 80 × (MAP − right atrial pressure)/CI. CI is CO adjusted for body size and is calculated by dividing CO by body surface area. Continuous-wave Doppler ultrasound estimates of CI strongly correlate with direct measurements obtained by pulmonary artery catheterization (Tan et al., 2005; Jain et al., 2008).
Transabdominal Doppler ultrasound studies of UtA, UmbA and MCA resistance indices (PI, resistance index [RI], and S/D), and fetal biometry were conducted as previously described (Julian et al., 2009) using a Medisono P25 equipped with a curvilinear transducer (3C-A) and 3D transducer (VC6-2) probes. Standard fetal biometry (biparietal diameter, head circumference, abdominal circumference, femur length) and EFW were recorded. Arterial resistance indices were examined bilaterally, and average right- and left-side measurements were reported. Fetal ultrasound measurements may be subject to intra-and interobserver variability (Sarris et al., 2012) and were therefore conducted by a single, experienced operator (L.T.J.). For each patient, maternal cardiovascular hemodynamic studies and obstetrical ultrasounds were performed on the same day.
Outcome measures
Primary outcomes were maternal hemodynamic indices, including HR (beats per min [bpm]), MAP (mmHg), CO (l/min), CI (l/min/m2), SVR (dynes/s/cm5), SVRI (dynes/s/cm5/m2), maternal UtA resistance indices (PI, RI, and S/D), fetal UmbA and MCA resistance indices, maternal plasma sFlt1 levels, and birth weight.
Statistical analysis
Continuous variables were evaluated for normality using Kolmogorov–Smirnov tests. The mean values or frequencies for variables were compared between groups using independent Student's t-tests or chi-squared statistics. Pearson correlations and linear regression models were used to test the association between continuous variables. Univariate general linear models were used to generate and compare estimated marginal means of infant birth weight, adjusted for variation in gestational age and infant sex. Data are expressed as mean ± standard deviation or the 95% confidence interval for proportions in the text, tables, and figures. A two-sided p-value <0.05 was considered evidence of association or difference in sample means or frequencies, and trends were reported when 0.05 ≤ p ≤ 0.10. Data analyses were conducted using SPSS v.26 (IBM, Chicago, IL) and graphics prepared using GraphPad Prism v. 8.3.1 (GraphPad Software, Inc., La Jolla, CA).
Data access
The data described in this article are available upon reasonable request to the corresponding author.
Results
Demographic and pregnancy characteristics
Maternal characteristics, including age, altitude of birth or childhood, years of high-altitude residence, height, pregravid body mass index, weight at the time of study, gravidity, and parity did not differ between controls and FGR cases (Table 2). Nearly all women had secondary education or greater. No women underwent fertility treatment for the current pregnancy. While no women reported smoking or consuming alcoholic beverages during their pregnancy, two fathers (one control and one FGR case) reported smoking one to five cigarettes per day. Compared to controls, FGR newborns weighed 1,032 g less (823 g less after correction for gestational age) at birth (Table 2). On average, gestational age at delivery was similar between controls and FGR, and approximately one-third of infants were born preterm in each group.
Maternal and Newborn Characteristics
For adjusted variables values are shown as the estimated marginal mean ± SEM. All other data are shown as the mean ± SD or as a proportion (%) and the 95% confidence interval in brackets. Sample sizes for each variable are in parentheses.
Andean (Aymara or Quechua) ancestry was self-reported (yes or no).
BMI, body mass index; GA, gestational age; SD, standard deviation; SEM, standard error of the mean.
Maternal cardiovascular function, UtA resistance, and fetal hemodynamics in FGR cases and healthy controls
Compared to controls, CI was significantly lower for FGR, while SVR and SVRI were elevated (30% and 20% higher, respectively) (Fig. 1). Compared to controls, FGR pregnancies had a higher maternal MAP and a lower HR (82 ± 7 mmHg vs. 90 ± 10 mmHg [p < 0.001] and 82 ± 12 bpm vs. 74 ± 12 bpm [p = 0.02], respectively). Maternal UtA resistance indices (PI, RI, and S/D ratio) were higher for FGR than controls (Fig. 2). Although UmbA resistance indices (PI, RI, and S/D ratio) were similar between controls and FGR, MCA PI, RI, and S/D were lower in FGR cases (Fig. 3). CPR was also reduced in FGR (2.01 ± 0.42 vs. 1.42 ± 0.51), with a larger proportion of FGR cases having CPR values <1.08 (27% [12, 48] vs. 81% [60, 95]; p < 0.01).
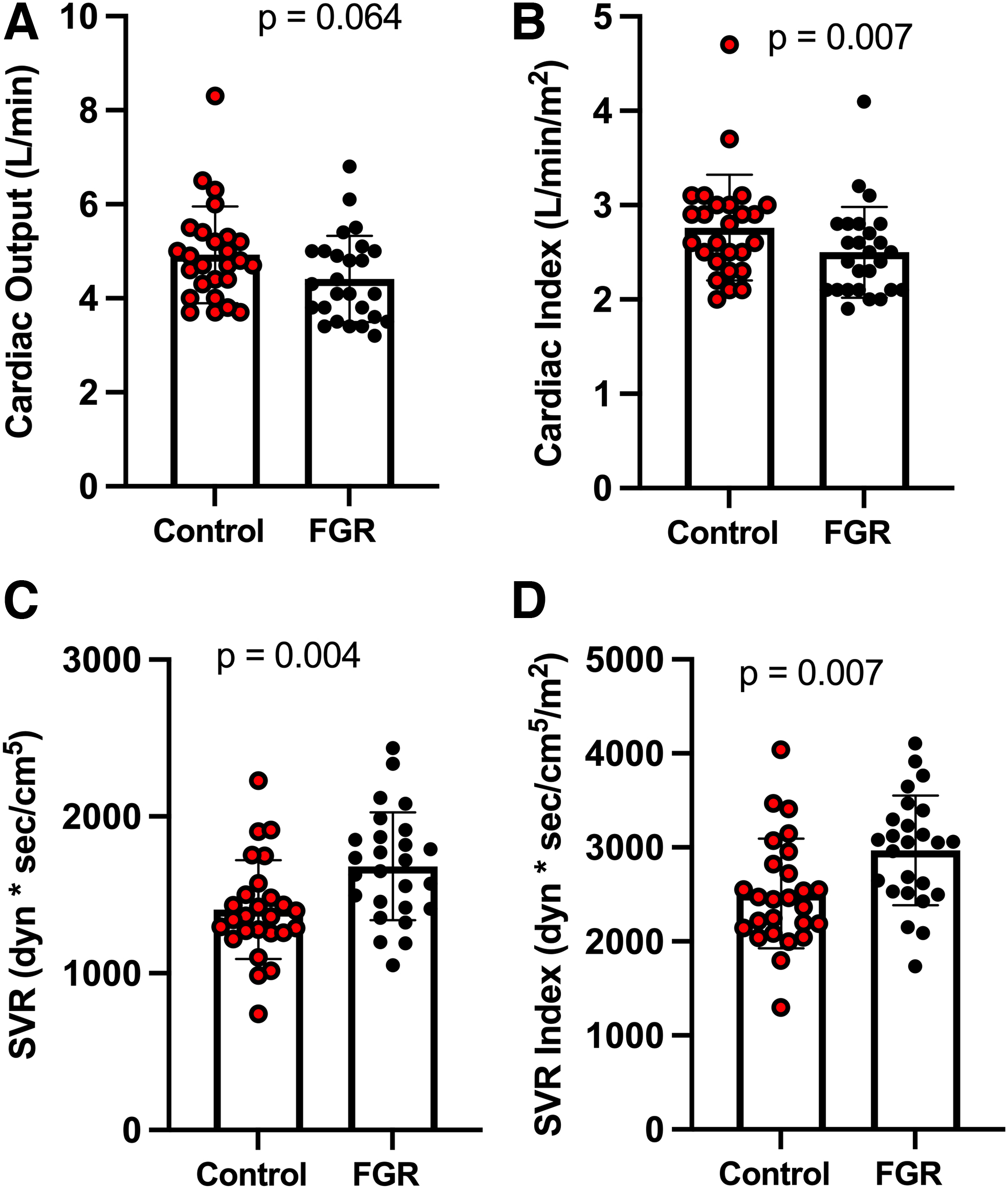
Maternal CO and SVR.
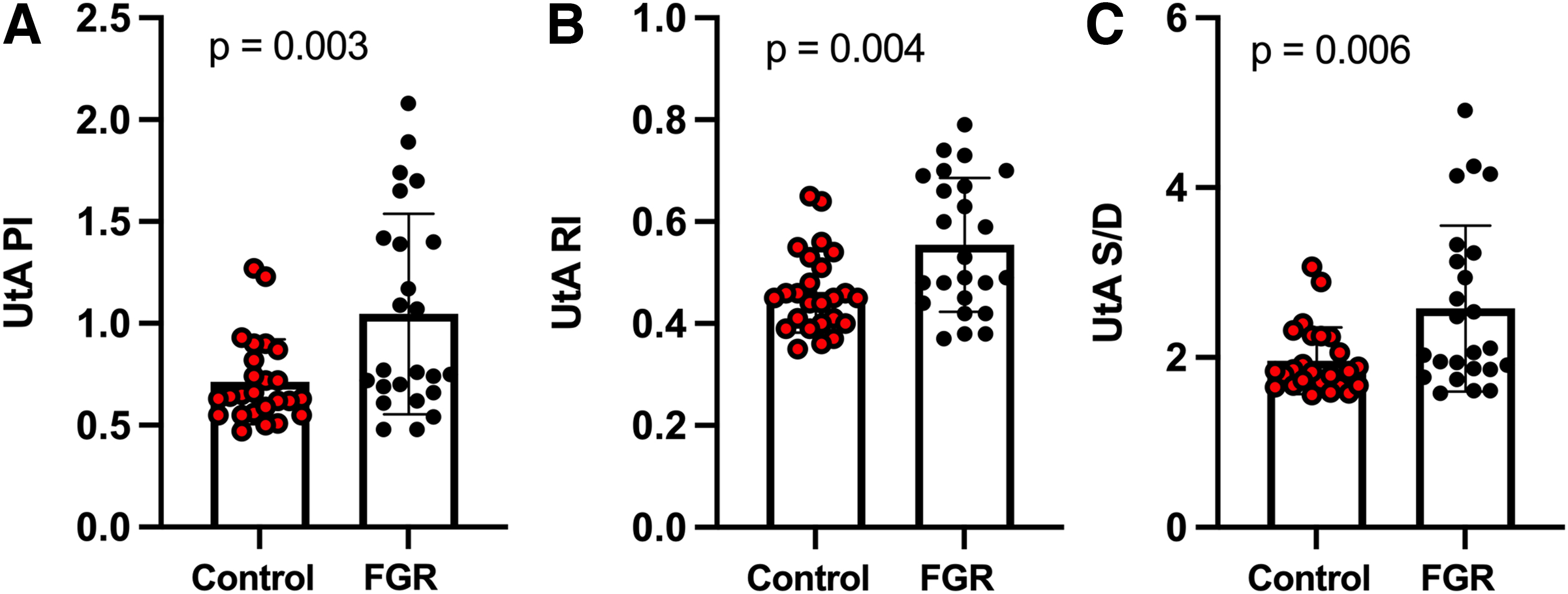
UtA resistance indices. Maternal UtA PI, RI, and S/D ratio were significantly higher for FGR (black circles) than controls (red circles), as shown in
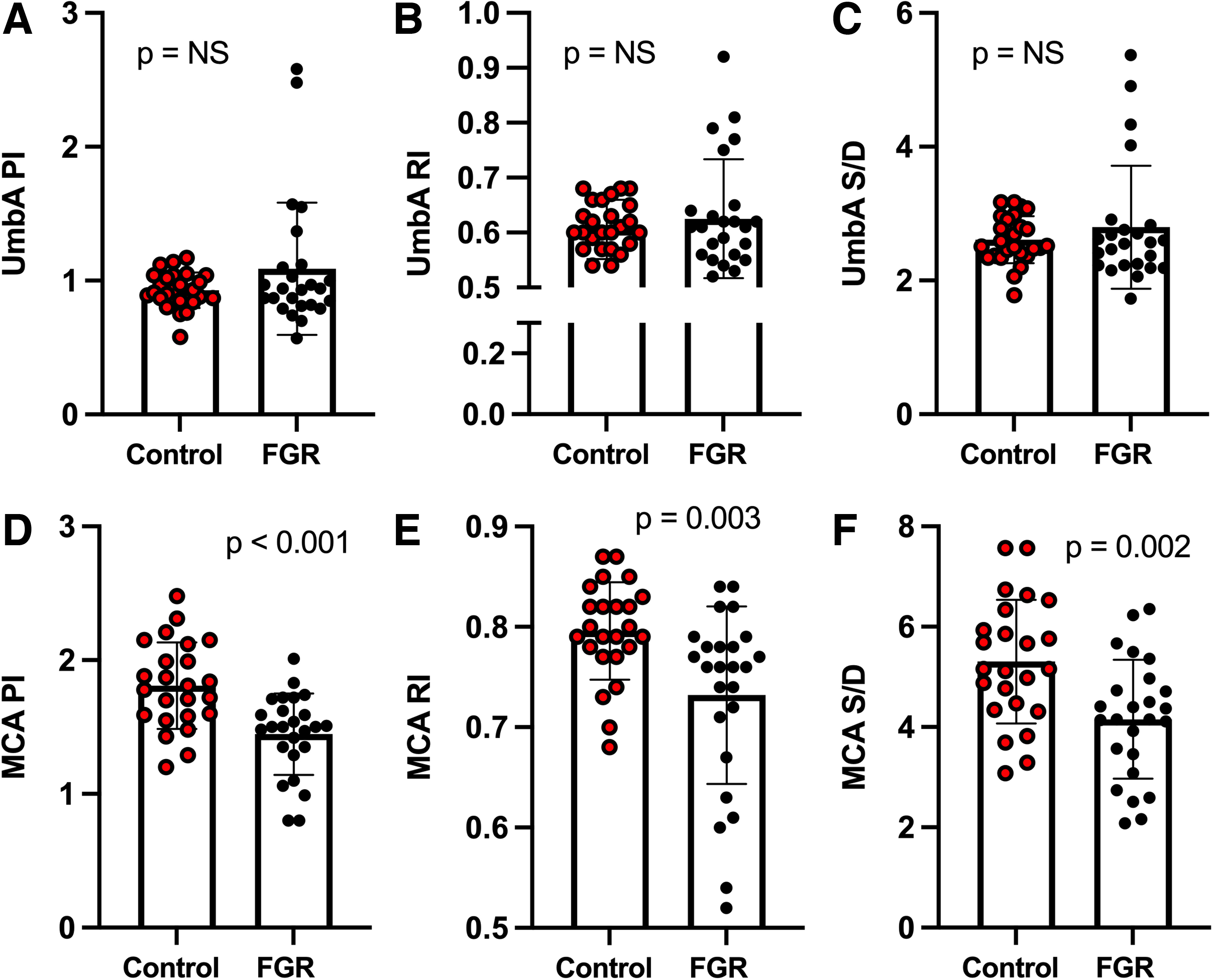
Umbilical and MCA resistance indices. There were no significant differences in UmbA resistance indices (PI, RI, and S/D ratio) between controls (red circles) and FGR (black circles)
Relationship between maternal CO, SVR, UtA resistance, and fetal hemodynamics
Higher maternal SVRI was associated with lower MCA RI and S/D and greater UtA RI (Fig. 4). Maternal CI was positively associated with CPR (r = 0.36, p = 0.01), while SVR and SVRI were inversely related to CPR (r = 0.37, p = 0.01; r = −0.41, p < 0.005). No relationship between maternal CO or SVR and UmbA resistance indices was detected. Maternal SVR was also associated with decreased birth weight even after controlling for gestational age [F (2, 45) = 15.9, p = <0.001].
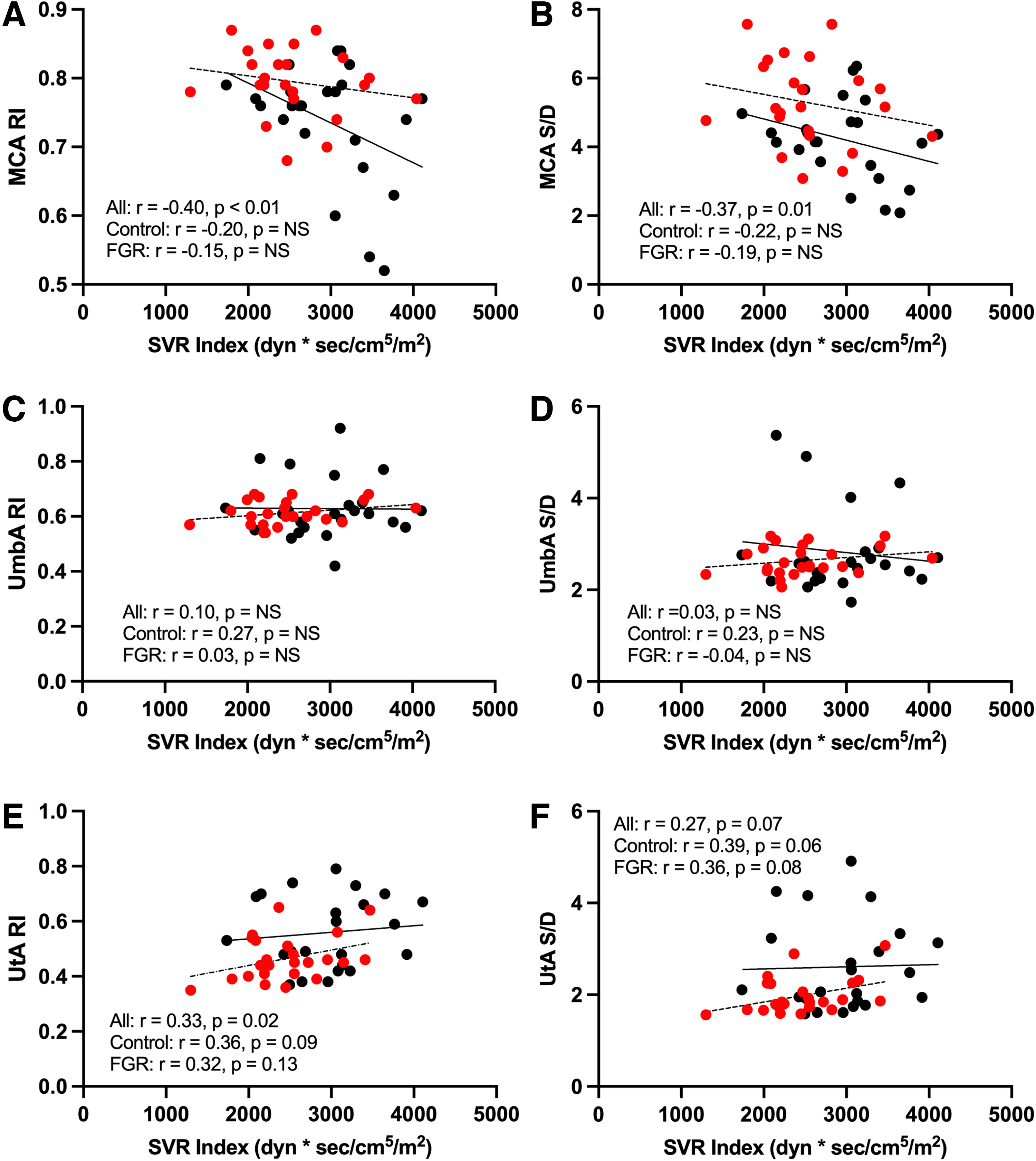
Association between SVRI and MCA, UmbA, and UtA RI and S/D. Higher maternal SVRI was associated with lower MCA RI and S/D and greater UtA RI.
Relationship between maternal sFlt1 and hemodynamic indices
Compared to controls, maternal sFlt1 values were higher in FGR (20,488 ± 10,498 pg/ml vs. 33,858 ± 19,448 pg/ml, p = 0.02). In our cohort, sFlt1 was positively associated with UtA PI (r = 0.58, p < 0.001) and negatively associated with CPR (r = −0.43, p = 0.01) and MCA PI (r = −0.44, p < 0.01) (Fig. 5). Maternal sFlt1 levels also tended to be inversely associated with CI (r = −0.27, p = 0.10) and directly associated with SVRI (r = 0.29, p = 0.08). There was no relationship between sFlt1 and UmbA PI (r = 0.23, p = NS).
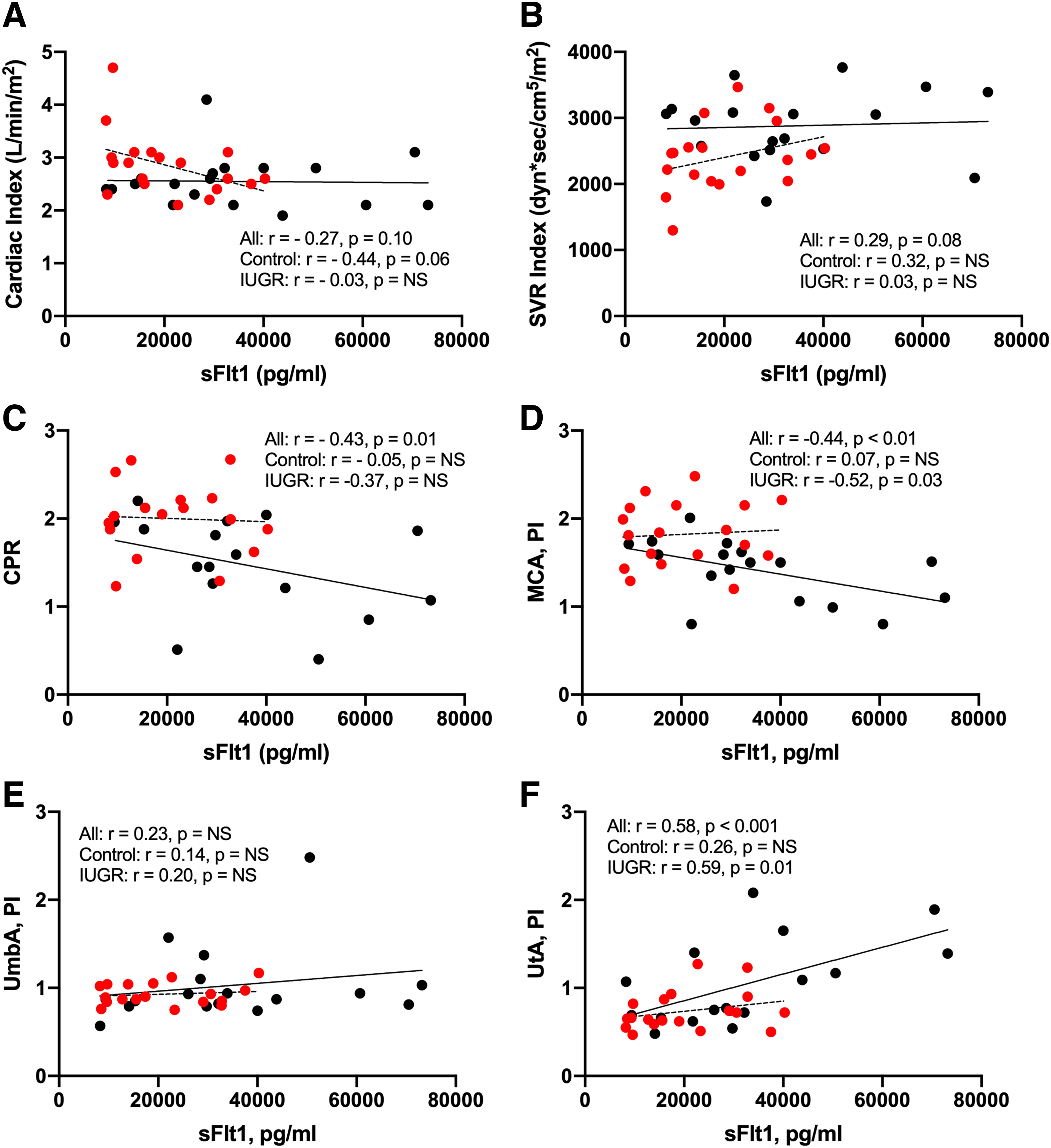
Maternal sFlt1 correlations with maternal hemodynamics. Circulating sFlt1 levels were negatively associated with CPR and MCA PI and positively associated with UtA PI.
Discussion
We found that maternal cardiovascular performance was reduced in high-altitude FGR and associated with greater maternal UtA resistance indices and fetal brain sparing. Specifically, FGR cases were characterized by a low CO-high SVR maternal phenotype that was paralleled by increased maternal UtA RI, reduced MCA RI, and lower CPR. We further demonstrated that maternal circulating sFlt1 levels were higher in FGR than controls, inversely associated with fetal MCA PI and CPR, and positively associated with maternal UtA PI. Together our data suggest a central role in reduced maternal cardiovascular performance, likely aggravated by excess circulating sFlt1 levels, contributing to the development of FGR at high altitudes. Future studies are required to determine whether such differences are apparent before FGR onset.
Our finding that maternal cardiovascular performance is reduced in high-altitude FGR agrees with observations made at low altitudes (Bamfo et al., 2008; Rang et al., 2008; Stott et al., 2017; Roberts et al., 2018; Perry et al., 2020). Our data further suggest that high altitude may raise maternal SVR in both healthy pregnancy and FGR; specifically, in our cohort, SVR values for controls and FGR cases were 25% and 29% greater, respectively, than published values for low-altitude subjects (Vinayagam et al., 2018; Perry et al., 2020). The centile position of CO and SVR in 35-week pregnancies with FGR reported at low altitude is 23% and 79% (Perry et al., 2020), compared to our cohort which was 2.8% and 98%, respectively. Altitude impact on CO and SVR among pregnant woman is further supported by studies showing that high altitude (Cerro de Pasco, Peru [4,370 m]) reduces maternal CO versus sea level (Lima, Peru [154 m]) (Kametas et al., 2004).
In healthy pregnancy, uteroplacental blood flow increases due to a rise in maternal CO, reduced SVR and uteroplacental vascular resistance, enhanced blood volume, and a consequent redistribution of blood flow to favor the uteroplacental circulation (Perry et al., 2019). High-altitude FGR has been attributed, in part, to a lesser expansion of uteroplacental blood flow and a reduced fall in uteroplacental resistance (Julian et al., 2008, 2009). In healthy, low-altitude pregnancies (35 weeks), the 50th centile of CO and SVR is 6.8 (l/min) and 1,011 (dynes × s/cm5), respectively (Vinayagam et al., 2018). In comparison, average values for CO and SVR in our entire cohort were below the 10th and above 90th centile, respectively.
We consider that the low CO and high SVR observed in high-altitude FGR may precede placental hypoperfusion and directly contribute to fetoplacental hypoxia and restricted fetal growth. Our findings support that an integrated maternal hemodynamic-placental model that considers insufficient maternal cardiovascular performance as a primary contributor to placental insufficiency may better represent the pathophysiology of high-altitude FGR as has been suggested by others for low-altitude FGR (Perry et al., 2020; Mecacci et al., 2021).
Maternal SVR begins to decline immediately following conception in a healthy pregnancy (Moore, 2003). This effect of pregnancy is due to widespread peripheral vasodilation resulting, in part, from increased nitric oxide production and reduced levels of endothelin-1, a potent vasoconstrictor, in the maternal circulation (Julian et al., 2008). Maternal vascular homeostasis during pregnancy also depends on angiogenic balance or the ratio of antiangiogenic to angiogenic factor levels. sFlt1 exerts antiangiogenic effects by sequestering vascular endothelial growth factor and placental growth factors, thereby disrupting maternal vascular homeostasis (Powe et al., 2011).
The maternal vascular endothelial dysfunction hallmark of preeclampsia has been associated with excess placental sFlt1 secretion into the maternal circulation (Maynard, et al., 2003b). Although maternal sFlt1 has been used as a biomarker to indicate placental insufficiency and a screening tool to identify FGR in high-risk groups (Herraiz et al., 2018; Visan et al., 2019; Quezada et al., 2020), a novel implication of our work is that increased maternal sFlt1 may also contribute to (or perhaps result from) the elevated SVR and reduced maternal cardiac performance.
Our study had two major strengths. First, we performed an integrated analysis of maternal systemic hemodynamics, uteroplacental resistance, fetal hemodynamics, and maternal sFlt1 levels in the same cohort. This permitted us to establish the plausibility of a reasonable pathway linking biomarkers and hemodynamic indices with fetal and maternal cardiovascular outcomes. Second, conducting this study in Bolivia was advantageous, given that more than 1.5 million people live in the La Paz-El Alto metropolitan area at an altitude that is sufficient to profoundly increase the incidence of FGR compared to sea level.
Limitations of the present study included the absence of a low-elevation Bolivian cohort to directly determine the impact of high altitude on maternal cardiovascular performance in this population. Comparisons of maternal cardiovascular performance between our cohort and previously reported values for lower altitude should therefore be interpreted cautiously. We have studies that are currently underway to address this limitation. Given prior work indicating a protective effect of Andean ancestry on fetal growth at high altitudes (Julian et al., 2007, 2009), the inclusion of a lowland ancestry population living at high altitude may also be valuable to reveal ancestry-related differences in maternal cardiovascular performance during pregnancy at high altitude.
In addition, longitudinal ultrasound screening for fetal growth or maternal hemodynamics measures were not performed, which prevented us from determining the temporal relationship between FGR onset, elevated SVR, and reduced cardiac performance. Specifically, the hemodynamic studies were performed in established FGR cases and controls. Given prior reports of diminished or static maternal CO and SV accompanied by elevated SVR before FGR onset (Stott et al., 2017), future high-altitude studies would benefit from paired, prospective measurements of maternal hemodynamics, uteroplacental resistance, and fetal biometry beginning in early pregnancy; these studies are currently underway in Bolivia and will provide further insight into whether abnormal maternal cardiac function can be used to identify pregnancies at an increased risk for FGR at high altitude.
Conclusion
Our data collectively highlight the possibility that reduced maternal cardiac performance and elevated SVR directly contribute to fetoplacental hypoxia and restricted fetal growth at high altitudes. Our findings emphasize that prospective studies are warranted to determine whether abnormal maternal central hemodynamics early in pregnancy can identify pregnancies at an increased risk of FGR at high altitudes. Early diagnosis of FGR is crucial as it is vital to establish adequate surveillance of fetal status, minimize risks of preterm birth and intrauterine hypoxia, and reduce the impact on the long-term function of developing organs. Our study underscores the unique opportunity offered by high-altitude studies to dissect the contribution of maternal vascular adaptations to pregnancy for FGR and the long-term cardiopulmonary health of both the mother and child.
Footnotes
Acknowledgments
The authors extend our sincere gratitude for the generosity of the women who participated in this research study. The authors also thank Dr. Eduardo Arcienaga, Dr. Jhonny Gonzales, Dr. Vitalio Mizutani, and Julie Houck for their assistance with study setup, subject recruitment, clinical data acquisition, data compilation, and sample collection.
Authors' Contributions
Writing—Original draft (W.M.), Conceptualization (C.G.J., L.T.J.), Writing—Review and editing (L.T.L., L.G.M., C.G.J.), Investigation (W.M., J.H., L.T.L., A.H.-F., A.M.-A., R.M., V.M.-G., A.L.-A., L.L.-V., P.G., R.G.), Formal analysis (C.G.J., W.M.).
Author Disclosure Statement
No competing financial interests exist.
Funding Information
National Institutes of Health (R21 TW010797, R01 HL138181) and the Ludeman Family Center for Women's Health Research at the University of Colorado Anschutz Medical Campus.
