Abstract
Introduction:
As opioid-related drug overdoses remain a public health crisis, there is a critical need for innovative approaches to developing safer analgesics with improved safety profiles. BDH-001 is a fixed-dose combination of low-dose buprenorphine (BUP) and cannabidiol (CBD) being developed as a safer analgesic than currently available opioids. The purpose of this study was to examine the analgesic and opioid-sparing effects of BDH-001 and to complete an in vivo safety assessment in rats.
Methods:
Analgesic effect of BDH-001 was assessed using the chronic constriction injury model of chronic neuropathic pain with pain threshold assessed via Von Frey testing. Drug-drug interaction effects on pharmacokinetic (PK) parameters were assessed in a single dose PK study in rodents. The effects on respiratory depression were also assessed and confirmed in two separate rodent studies performing blood gas analysis and measuring O2 saturation.
Results:
BDH-001 (combination of subanalgesic BUP dose and CBD) resulted in statistically significant increases in pain threshold compared to saline (p < 0.001), CBD alone (p < 0.01), and BUP alone (p < 0.05). The half-life of BUP was significantly shorter in the presence of CBD compared to BUP alone (p = 0.008), with no significant changes in any other BUP pharmacokinetic parameter assessed. CBD was found to attenuate BUP-induced respiratory depression in rats when assessing blood gases (p < 0.05) and O2 saturation (p < 0.05) over several time bins.
Conclusions:
Data obtained in the present study indicate the addition of CBD to BUP was opioid-sparing and attenuated BUP- but not morphine-induced respiratory depression. There was no evidence these findings were the result of a PK interaction. Results support the hypothesis that BDH-001, a fixed-dose combination of BUP and CBD, may provide effective analgesia with a more favorable safety profile.
Introduction
Drug overdose remains a U.S. public health crisis, with an estimated 100,000 deaths in 2021. Deaths from drug overdoses were more than six times higher in 2021 compared with 1999 and more than 75% of overdose deaths in 2021 involved an opioid.1,2 There have been three “waves” of the US opioid overdose epidemic: wave 1 in the 1990s with increased opioid prescribing, wave 2 showed opioid prescriptions decreased and users transitioned to heroin in 2010, and wave 3 in 2013 with major increases in overdose deaths involving illicit fentanyl. 3 The current, or fourth wave is characterized by continued use of illicit fentanyl, and additionally, deaths due to co-involvement with stimulants have skyrocketed. 4 While opioid prescribing has decreased, pain management and opioid prescribing practices continue to play a role in overdose deaths. Following the 2016 CDC guidelines for opioid prescribing for chronic pain, forced tapering of prescription opioids has been associated with a 69% increase in overdose risk and a 130% increase in risk for a mental health crisis. 5 Among patients prescribed opioids, opioid misuse, taking opioids for longer or in higher doses or for reasons other than prescribed is common and may lead to the development of opioid use disorder (OUD). 6 In 2023, an estimated 5.7 million Americans aged 12 or older had OUD in the past year, with only 18% receiving OUD medications. 7
Though high opioid doses are a major risk factor for OUD and overdose, 8 opioids remain FDA-approved for acute, chronic, and breakthrough pain. Chronic opioid therapy is associated with diversion and abuse, physical dependence and withdrawal, and death from respiratory depression. In addition, opioid treatment can cause nausea and vomiting, constipation, sedation, and dizziness.9,10 A variety of drug classes have been investigated to counter opioid-induced respiratory depression, but none have been successful. 10 Thus, there is a critical need for safer analgesics with reduced risk of abuse, dependence, and respiratory depression.
BDH-001 is a fixed-dose combination (FDC) of low-dose buprenorphine (BUP) and cannabidiol (CBD) being developed as a potentially safer analgesic. CBD is a phytocannabinoid derived from the Cannabis sativa plant that is non-psychoactive with minimal abuse liability. 11 A CBD oral solution is FDA-approved to treat seizures associated with Lennox-Gastaut syndrome, Dravet syndrome, and tuberous sclerosis complex. Preclinical and clinical studies suggest that CBD also has anti-anxiety, anti-inflammatory, and analgesic effects,12–16 and can mitigate opioid withdrawal symptoms.17,18 BUP is an FDA-approved mu opioid partial agonist and kappa antagonist with sublingual, buccal, and depot injectable formulations for OUD and transdermal and buccal formulations for chronic pain. Although safer than full agonists, BUP use is still associated with dose-related negative effects, including diversion and abuse, physical dependence and withdrawal, constipation and gastrointestinal adverse events, and overdose, especially when combined with sedatives.19,20 Preclinical studies assessing the potential opioid-sparing effects of cannabis-related compounds with full agonist opioids in different models of pain found varying results including no effect, additive effects, and one report showing synergy with morphine on acute inflammatory pain, but no studies have examined the effects of CBD and a partial opioid agonist on chronic pain.21–27
The purpose of this study was to examine the analgesic and opioid-sparing effects of BDH-001 and to complete an in vivo safety assessment in rats. Results indicate BDH-001 displays a greater than additive analgesic effect when combining low-dose BUP and CBD and improves safety through the attenuation of BUP-induced respiratory depression.
Materials and Methods
Study drugs
In the chronic constriction injury (CCI) study, saline was administered orally (PO, 10 mL/kg) or intravenously (IV, 1 mL/kg). Buprenorphine stock (10 mg/mL) was diluted with saline to 0.04 mg/mL (Group 2) and further diluted to 0.01 mg/mL (Groups 3 and 5) and administered via IV. Cannabidiol (0.7 mg/mL) was prepared with 50 mg of CBD dissolved in 2.5 mL DMSO to obtain a solution of 20 mg/mL and further diluted with saline to obtain a solution of 0.7 mg/mL (Groups 2 and 5) and administered via oral gavage.
In the single dose pharmacokinetic (PK) study, Buprenex Hydrochloride (0.3 mg/mL) was diluted with sterile saline and administered via IV. Cannabidiol was prepared and administered as described above. For the combination administration, CBD gavage was administered 1 h prior to BUP.
In the confirmatory respiratory depression study, BUP was diluted to 0.04 mg/kg with sterile saline (dose volume of 1 mL/kg) and administered via IV. Morphine was diluted to 10 mg/kg with sterile saline (dose volume of 1 mL/kg) and administered subcutaneously. Cannabidiol was prepared and administered as described above. Cannabidiol/Vehicle 1 was administered 1 h before opioid/Vehicle 2.
As this is a pilot study, study drug routes of administration were based on previous studies to support study design and interpretation. 28
Animals
Healthy rodents were used and drug dose volume was administered per body weight. Rodents were acclimated to laboratory conditions before study initiation. In the CCI study (MD Biosciences), 40 male Sprague-Dawley rats (Envigo RMS Israel Ltd, 120–150 g) were housed within a limited access facility with 3–5 rats/cage, temperature 62.6–73.4°F, relative humidity (RH) 30–70%, a 12-h light-dark cycle and 15–30 air changes/h. In the PK study (BATTS Laboratory), 25 male and 25 female Sprague-Dawley Rats (Charles River, 8–10 weeks), with vascular catheterization of the jugular vein and carotid artery were housed 1 rat/cage, temperature 68–73°F, RH 34–44%, and a 12-h light-dark cycle. In the confirmatory respiratory depression study (Melior Discovery), 52 male Sprague-Dawley rats (Charles River, 200–225 g) were housed 1–2 rats/cage in a ventilated rack system on a 12-h light-dark cycle. In all studies, animals were provided standard rodent chow and water ad libitum. All animal protocols were conducted according to the USDA Animal Welfare Act, the PHS Policy on Humane Care and Use of Laboratory Animals, and the US Interagency Research Animal Committee Principles for the Utilization and Care of Research Animals. Each study protocol was approved by the facility Institutional Animal Care and Use Committee.
CCI study and analysis
The CCI model was established by surgical ligation of the sciatic nerve in rat. 29 The left sciatic nerve was exposed above the femoral joint and three loose knots were applied. Treatment groups and dosing regimen detailed in Table 1. Rats were placed in an enclosure and positioned on a metal mesh surface but allowed to move freely. Von Frey tests began after cessation of exploratory behavior and were conducted at baseline (day −1) and on day 14 (at 5 min before IV dosing and 60 min post-IV dosing). Von Frey monofilaments were applied at right angles on the plantar surface of the foot to induce the paw withdrawal reflex, with a maximum force of 60 g. Decreases in force needed to induce withdrawal are indicative of allodynia, as the force applied is a non-painful stimulus under normal conditions. Data are presented as means ± SEM (Fig. 1). All groups were compared to Group 1 or to each other using one-way ANOVA followed by Dunnet’s test or Tukey post-test, respectively (GraphPad). A two-way mixed ANOVA was also completed to assess the effects of time and treatment group comparing day 14 pre- vs. post-dose. Potential for synergistic interaction of BUP + CBD was assessed using a Combination Index (CI) value to determine if the observed effects of the combination is greater than the expected additive effects, with CI <1 consistent with a synergistic interaction and this interaction was further assessed in a 2 × 2 factorial ANOVA examining day 14 post-dose paw threshold for CBD and subanalgesic BUP.30,31
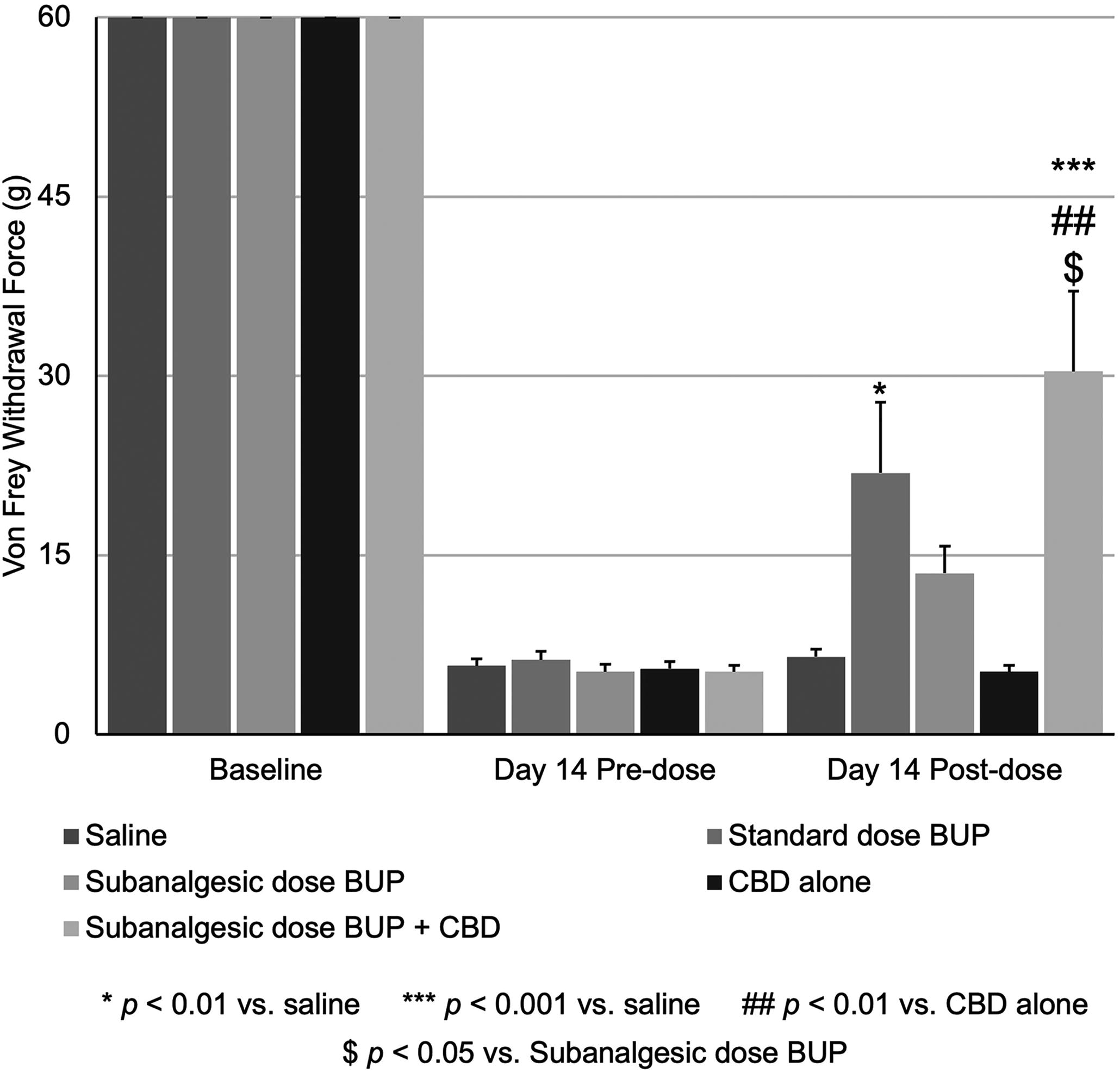
Pain threshold before and after standard and subanalgesic BUP and CBD. Male Sprague-Dawley rats underwent surgical ligation of the sciatic nerve using the CCI model of chronic neuropathic pain, treatment groups and dosing regimen described in Table 1. On study day 14, pre-dosing, all animals were tested and experienced high sensitivity to Von Frey testing. Following pre-dose Von Frey testing, animals were administered an IV injection of either saline or BUP and were retested 1 h post-dose. Data are presented as means ± SEM, all groups were compared to Group 1 or to each other using one-way ANOVA followed by Dunnet’s test or Tukey post-test, respectively (GraphPad).
Treatment Groups in Proof-of-Concept CCI Study
IV, intravenous; PO, oral; QD, once a day.
Single dose PK study and analysis
Treatment groups and dosing regimen are detailed in Table 2. For safety purposes and to minimize distress, animals were anesthetized inside an induction chamber with isoflurane (2% ISO and 98% oxygen) for approximately 1 min. Due to this, pO2 and sO2 values have been excluded from the blood gas analysis. Outside the induction chamber, a nose cone was applied for approximately 45 sec while 100–150 µL blood was collected 5 min prior to CBD dosing, and 1, 3, 5, and 24 h post-BUP dose from the arterial catheter for analysis via ABAXIS iSTAT Affinity Version 5 Veterinary Blood Gas Analyzer. For PK measurements, 100–150 µl blood was collected from the venous catheter at 5 min prior to CBD dosing and 5, 30 min, 1, 2, 3, 24 and 48 h post-BUP dose for analysis via high-performance liquid chromatography with in-line tandem mass spectrometry (LC-MS, Agilent Instrument LC1200 series MS G6410). Whole blood was subjected to centrifugation at 500 x g for 6 min at room temperature, plasma was separated and stored at −80°C. Non-compartmental analysis was used to estimate PK parameters (Microsoft Excel v. 2010) and data were analyzed in IBM SPSS Statistics (ver28.0). PK parameter means were compared using an independent samples t-test. For blood gas analysis, a repeated measures ANOVA with a Tukey HSD post-hoc test was used to compare groups for each parameter and confirmed via ANCOVA with baseline as a covariate to account for physiological variations.
Treatment Groups in Single Dose Pharmacokinetic Study
Confirmatory respiratory depression study and analysis
To account for the inability to measure pO2 on blood gas analysis, we assessed pulse oximetry in awake rats. Blood oxygen saturations were measured with the MouseOx pulse oximeter system (Starr Life Sciences). Prior to testing, animals were acclimated to probes and were briefly anesthetized with isoflurane, and their necks were shaved (probe location). Testing occurred over 4 consecutive days with a maximum of 4 cohorts of 4 rats (n = 16) per day. An average oxygen saturation value was measured every 5 min for 120 min. After time bins were assigned, O2 saturation, heart rate, and respiratory rate within each 5-min time bin were individually sorted (highest to lowest). In those sorted time bins, the 7 middle values were selected and averaged. If there were less than 7 values, then all collected values were averaged. On the day of the test, each animal was habituated to the device for 30 min, followed by a 30 min baseline recording period before dosing. Animals were then dosed with vehicle 1/CBD, and one h later were dosed with opioid/vehicle 2 followed by a 90-min recording period. Data were analyzed in Prism (v9.4.1) by two-way ANOVA followed by Fisher’s LSD post-hoc test to determine differences between treatment and vehicle control or between CBD/opioid and vehicle/opioid.
In all statistical analyses, a p-value of less than 0.05 was used to indicate statistical significance.
Results
Preclinical proof-of-concept study
Results of the CCI study are shown in Figure 1, and treatment groups are listed in Table 1. At baseline, pain threshold (Von Frey testing) was equivalent for all groups. At day 14 pre-dosing, all groups had developed hyperalgesia on Von Frey testing. Using a one-way ANOVA, an analgesic dose of BUP (0.04 mg/kg) significantly increased pain threshold compared to saline (Fig. 1, standard dose BUP versus saline, p < 0.01), while the subanalgesic dose of BUP (0.01 mg/kg) was not statistically significantly different from saline. BDH-001 (combination of subanalgesic BUP dose and CBD) resulted in statistically significant increases in pain threshold compared to saline (p < 0.001), CBD alone (p < 0.01), and subanalgesic BUP alone (p < 0.05). Further, the mean pain threshold for the subanalgesic BUP dose plus CBD combination, 30.38 g, was higher than the sum of the pain thresholds for the subanalgesic BUP alone (13.50 g) and CBD alone (5.25 g) demonstrating that the addition of CBD to the subanalgesic dose of BUP produced greater than additive analgesic effects, suggesting a synergistic interaction. In addition, using a two-way mixed ANOVA, we assessed the effects of time and treatment group, comparing day 14 pre- vs. post-dose. There is a main effect for time (p < 0.001) and treatment group (p < 0.001) and a significant interaction between time and treatment group (p < 0.001) such that the post-dose pain thresholds are significantly different compared to pre-dose thresholds across groups. Using Dunnett’s t two-sided posthoc test, the subanalgesic BUP plus CBD combination was significantly different compared to saline (p = 0.002), CBD alone (p < 0.001), and subanalgesic BUP alone (p = 0.027). To further assess the effects of the combination, we calculated a CI score using the Response Additivity effect-based approach, 31 which assesses if the observed combination effect (EAB) is greater than the expected additive effect given by the sum of the individual effects (EA + EB), CI = (EA + EB)/(EAB). The CI for BUP+CBD was 0.617, consistent with a potential synergistic interaction (CI <1). Further, the interaction between CBD and subanalgesic BUP was significant (p = 0.017) in a 2 × 2 factorial ANOVA, further supporting synergy. 31
Single-dose PK study and in vivo safety assessment
A single-dose PK and safety assessment was completed to examine drug-drug interactions and respiratory depression in rats. Treatment groups are described in Table 2. Results of PK analysis on plasma samples assayed are shown in Figure 2. Due to limitations in our method, data obtained in groups 1 (0.01 mg/kg BUP), 3 (7 mg/kg CBD), and 4 (0.01 mg/kg BUP and 7 mg/kg CBD) were outside the limits of quantitation for the method and excluded from analysis (data not shown). Graphs displaying concentration-time curves (mean ± SEM) for BUP in group 2 (0.04 mg/kg BUP) and group 5 (0.04 mg/kg BUP + 7 mg/kg CBD) are shown in Figure 2.
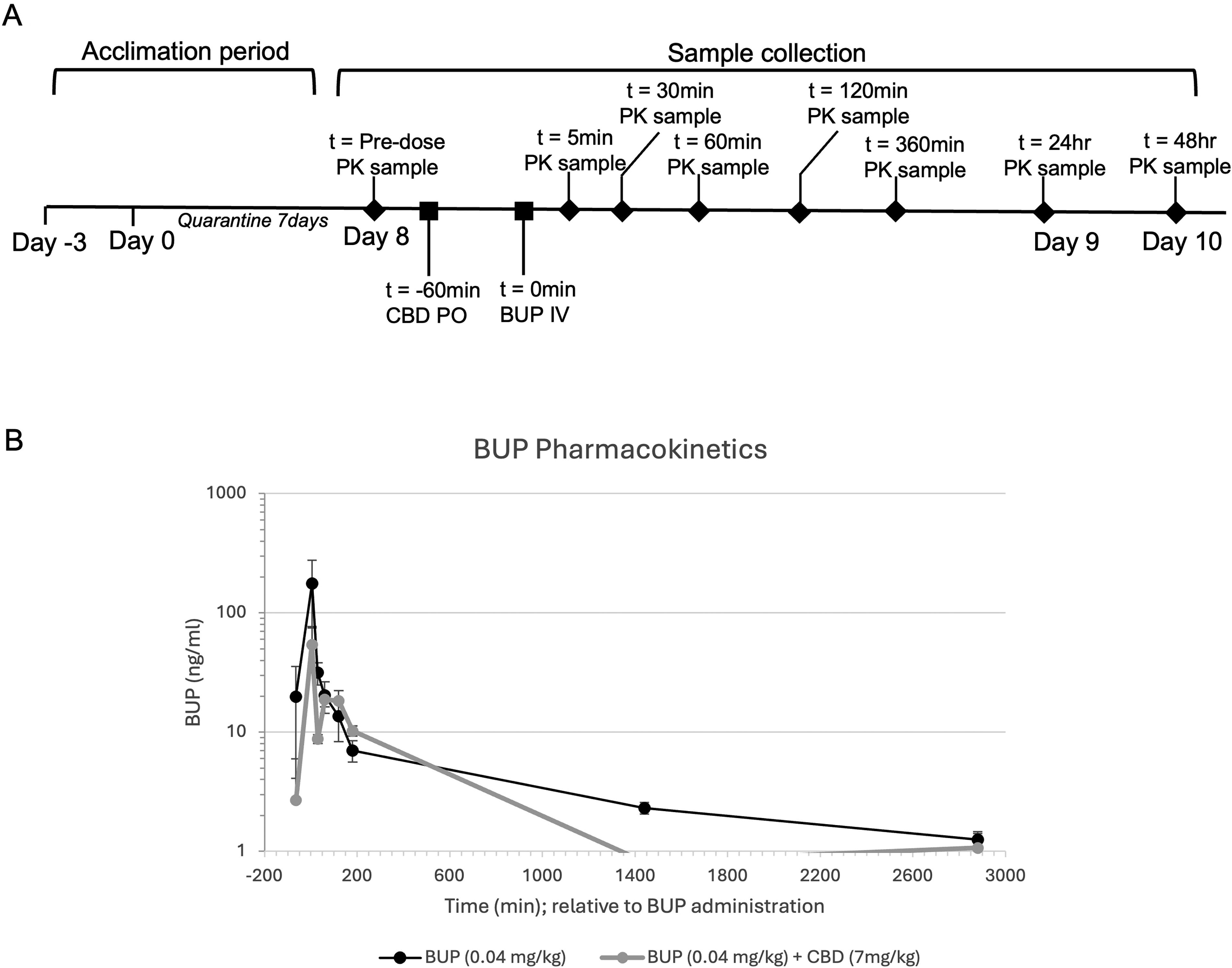
Concentration time curves for Buprenorphine in single dose PK study. Male and female Sprague-Dawley rats were administered CBD (7 mg/kg) or saline via oral gavage followed 60 min later by an IV bolus of BUP (0.01 mg/kg or 0.04 mg/kg) or saline, treatment groups listed in Table 2. Blood was collected at time points −5 min, 5 min, 30 min, 1 h, 2 h, 3 h, 24 h, and 48 h and assayed via HPLC with in-line tandem mass spectrometry (MS) method.
Pharmacokinetic parameters
Non-compartmental analysis was used to estimate PK parameters (Table 3). The half-life of BUP in the presence of CBD (group 5) was significantly shorter than for BUP alone (group 2), p = 0.008. All other pharmacokinetic parameters, including AUC, Vz, and Cl for BUP, were not significantly different in the presence of CBD.
Pharmacokinetic Parameters of Buprenorphine
p < 0.05 when compared to Group 2 BUP alone.
AUC, Area under the curve; Vz_obs, Volume of distribution during the terminal phase; Cl_obs, total Clearance.
In vivo assessment of respiratory depression
Averages of blood gas parameters (mean ± SEM) are shown in Figure 3B–F for all groups. Group means were compared via a repeated measures ANOVA with a Tukey HSD post-hoc test and confirmed by ANCOVA with baseline values as a covariate to account for physiological variations in baseline activity. For pH, there were no statistically significant differences (p > 0.05). For pCO2, tCO2, HCO3, and Base Excess (ecf) there were statistically significant differences between group means (p < 0.05), and pCO2, tCO2, HCO3, and Base Excess were significantly lower for BUP+CBD groups compared to BUP alone (p < 0.05), suggesting that CBD was able to attenuate BUP-induced respiratory depression.
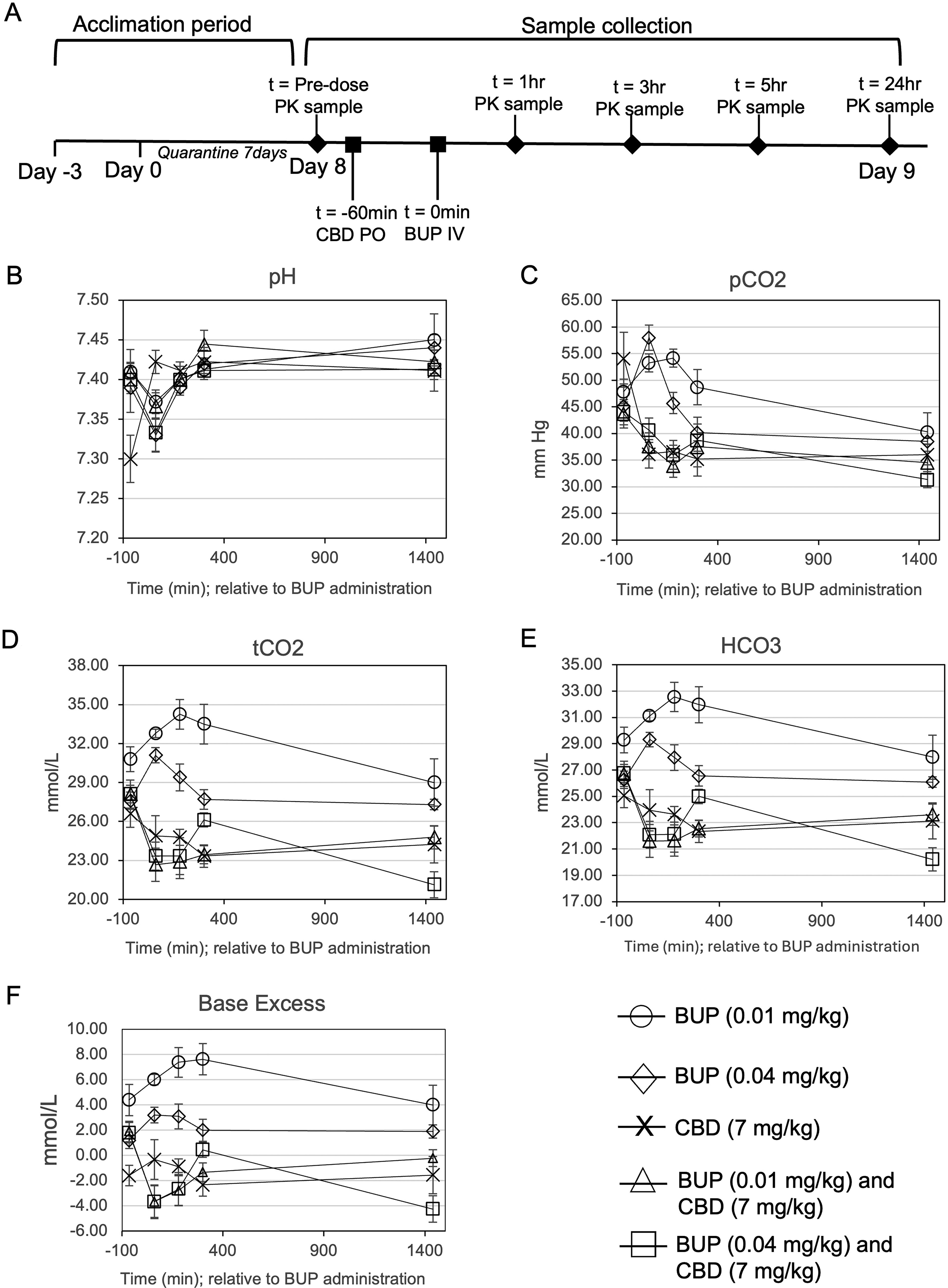
Blood gas analysis in in single dose PK study. Male and female Sprague-Dawley rats were administered CBD (7 mg/kg) or saline via oral gavage followed 60 min later by an IV bolus of BUP (0.01 mg/kg or 0.04 mg/kg) or saline, treatment groups listed in Table 2. Blood was collected (100–150 µl) at each time point (−5 min, 60 min, 3 h, 5 h, and 24 h) from the arterial catheter for Blood/Gas analysis via ABAXIS iSTAT Affinity Version 5 Veterinary Blood Gas Analyzer.
Confirmatory respiratory depression assessment
Figure 4 depicts the mean ± SEM oxygen saturation via pulse oximetry over time for each of the treatment groups (Table 4). Neither opioid produced consistent changes in respiratory rate or heart rate over time, and consistent effects of CBD on either morphine- or BUP-induced breath rate and heart rate were not observed (data not shown). All opioid groups had significantly reduced O2 saturation by 5–10 min post-opioid as compared to vehicle/vehicle. Morphine produced respiratory depression with a nadir oxygen saturation around 60%, while BUP produced lesser degrees of respiratory depression (trough at 84% O2 saturation). CBD attenuated BUP-induced reduction in O2 saturation, and CBD/BUP was statistically different from vehicle/BUP over several time bins (see Fig. 4). The addition of CBD to morphine did not reverse morphine-induced respiratory depression.
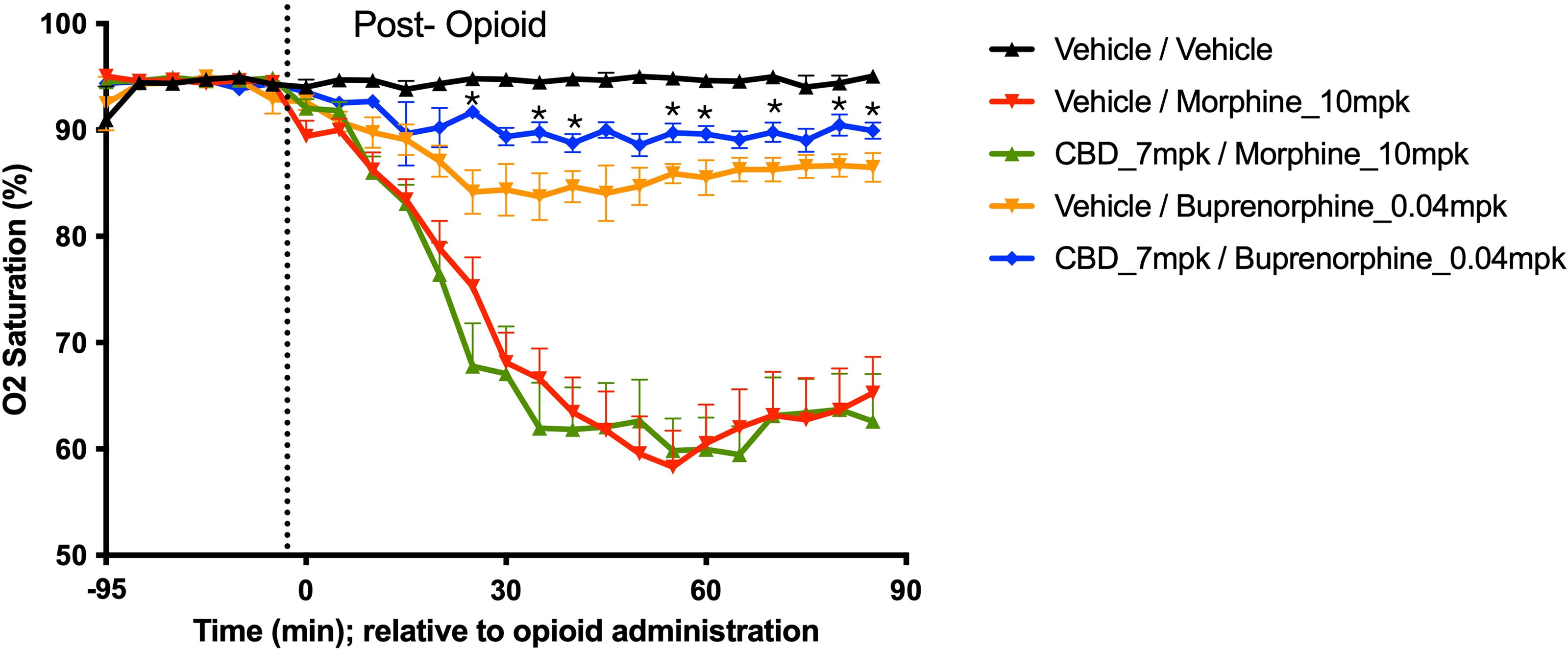
Effects of CBD on BUP-induced respiratory depression. Blood oxygen saturations were assessed in awake male Sprague-Dawley rats with the MouseOx pulse oximeter system, treatment groups detailed in Table 4. On the day of the test, each animal was habituated to the device for 30 min, followed by a 30 min baseline recording period before dosing. Animals were then dosed with vehicle 1 or CBD and 1 h later were dosed with opioid or vehicle 2 followed by a 90-min post-opioid or vehicle 2 recording period. Data shown are mean ± SEM oxygen saturation over time for each of the treatment groups and were analyzed in Prism (v9.4.1) by two-way ANOVA followed by Fisher’s LSD post-hoc test to determine differences between treatment and vehicle control, or between CBD / opioid and vehicle / opioid. * indicates p-value <0.05 when comparing CBD/BUP vs vehicle/BUP.
Confirmatory Respiratory Depression Study Design
PO, oral; IV, intravenous; SC, subcutaneous.
Discussion
In the present study, results indicate that the combination of CBD and a subanalgesic dose of BUP demonstrated enhanced analgesia using the CCI model of chronic neuropathic pain, suggesting a synergistic interaction. The addition of CBD to a subanalgesic dose of BUP resulted in a statistically significant increase in pain threshold compared to the disease-only saline group. In addition, there was a greater than additive increase in analgesia in the CBD plus subanalgesic BUP group, resulting in a CI = 0.617, suggesting synergistic analgesia between CBD and BUP. Increased analgesia was not explained by a BUP + CBD PK interaction, as the addition of CBD did not alter PK parameters for BUP with the exception of a shorter BUP half-life with CBD, which would decrease, not increase, BUP effects. Further, CBD attenuated BUP-induced respiratory depression when assessed via blood gas and pulse oximetry. Together, these data support the hypothesis that BDH-001, a fixed-dose combination of low-dose BUP and CBD, may be a safer analgesic than currently available opioids.
Previous studies have examined the potential of cannabis and cannabis-related compounds on opioid analgesia but have either focused on acute pain21–27 or the effects of cannabinoid agonists, such as THC.32,33 Using CBD would reduce the negative effects associated with cannabis, including elevated heart rate and blood pressure, paranoia, anxiety, depression, depersonalization, and hallucinations. In addition, recent efforts have shown that CBD was able to mitigate withdrawal symptoms in heroin-dependent mice 17 and reduce clinical symptoms commonly observed in OUD patients undergoing withdrawal. 18 Further, studies show that combining BUP and CBD reduces cue-induced craving and attentional bias to drug-related cues34,35 suggests that BDH-001 may offer analgesia without increased abuse liability.
Limitations of this study include testing only two opioids and a limited number of study drug doses. The confirmatory respiration study found that adding CBD to BUP but not morphine mitigated opioid-induced respiratory depression. Whether this difference suggests the effect of CBD is specific to BUP or not requires additional testing. It is possible that different doses of CBD may be required to alter the analgesic or respiratory effects of full versus partial opioid agonists. The combination of BUP + CBD exhibited a greater than additive effect on analgesia, but we were unable to perform a formal test of synergy (e.g., isobologram analysis) as this requires more than one dose level of CBD. When assessing the interaction of BUP + CBD using an effects-based approach, we find the interaction to be significant in a 2 × 2 factorial ANOVA, and the CI = 0.617, which is consistent with a synergistic interaction (CI <1).30,31 Additional studies examining different dose ratios will provide further data to confirm synergy and will inform clinical trial dose selection.
Conclusions
Preclinical data display enhanced analgesia with lower, safer doses of buprenorphine, suggesting that BDH-001 has the potential to be a safer alternative than currently available opioids. Formulation development of BDH-001 is currently underway, and clinical trials are planned to determine if BDH-001 can replicate these findings in humans.
Footnotes
Authors’ Contributions
M.S.B., D.Z.D., and K.G.H.: Conceptualization, methodology and validation. M.S.B.: Supervision, funding acquisition, formal analysis and data curation. M.S.B.: Writing—original draft. M.S.B., D.Z.D., and K.G.H.: Writing—review and editing, Visualization. M.S.B. and D.Z.D.: Project administration and resources.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
Research reported in this publication was supported by the National Institute on Drug Abuse of the National Institutes of Health under Award Numbers R43DA050397 and R44DA050397. Animal studies were performed at MD Biosciences, BATTS Laboratories and Melior Discovery.
