Abstract
Background:
The efficacy of cannabis treatment is determined by the active pharmaceutical ingredients (APIs) of the ingested composition. Despite smoking predominancy in cannabis treatment, very little is known regarding its yield and provision rate of cannabis APIs.
Material and Methods:
Ten experiments were performed, studying changes in APIs content during smoking, using a designated smoking machine. APIs content was evaluated via analysis of a cigarette's residuals and of the smoke composition; cannabinoid and terpene content were assessed.
Results:
Results demonstrated increased cannabinoid content in the cigarette sections closer to the mouth, as compared with those closer to the lit end. Similarly, cannabinoid content in the inhaled smoke increases as smoking progresses. Similar results are found for sesquiterpenes. Monoterpenes, having lower boiling points reach the smoke before the sesquiterpenes and cannabinoids do.
Conclusion:
A mechanism is proposed, including: (i) decarboxylation and evaporation of APIs adjacent to the lit end, (ii) transition of API vapors away from the hot zone, (iii) condensation of APIs in cigarette's sections closer to the mouth, and (iv) re-evaporation of APIs as the hot zone approaches, thereby reaching the smoke. Differences in the boiling points between the various APIs result in varying composition along the cigarette and in the inhaled smoke. The main implications are: (i) APIs delivery through smoking cannot be uniform, (ii) APIs amount per puff increases as smoking progresses, and (iii) terpenes are inhaled before the cannabinoids are. Thus, in addition to its known health-threatening hazards, smoking entails nonuniform provision of APIs, even within the same cigarette.
Introduction
Cannabis sativa L. is one of the oldest domesticated plants known to humans.1,2 It has long been recognized and valued as an important source of food, fiber, and medicine.3,4 Cannabis is currently used to treat a variety of conditions and symptoms, including, pain, fibromyalgia, insomnia, and post-traumatic stress disorder.5–8 Its active pharmaceutical ingredients (APIs) include cannabinoids and terpenes. More than 100 cannabinoids were identified so far,9–11 the most common ones being Δ9 tetrahydrocannabinol (THC), cannabidiol (CBD), and cannabigerol (CBG). Cannabinoids appear in the cannabis plant in their acid form, and decarboxylate to the neutral form on temperature elevation, as in smoking. The number of terpenes in the cannabis plant exceed 200, out of which about 20 are the most prevalent. These include myrcene, limonene, pinene, linalool, terpinolene, β-caryophyllene, and humulene. 12
Most cannabis terpenes belong to one of the following groups, arranged here according to increasing boiling point: monoterpenes, monoterpenoids, sesquiterpenes, and sesquiterpenoids.
While the majority of the preclinical and clinical studies have focused on the effects of extracts,13–17 a large fraction of the medical cannabis users consume their drug through smoking, 18 (in Israel, more than 80% of them 19 ). This preference is plausibly derived from nonmedical recreational cannabis use and from the rapid effect obtained on inhalation (shorter onset time). 20
Given the known health-threatening effects, most studies of cannabis smoking have focused on the hazards of smoking, manifested as harmful, mostly carcinogenic, compounds in the cannabis smoke.21–28
Several cannabis smoking studies have investigated the delivery yield of cannabinoid provision, defined as the fraction of cannabinoids in the cigarette smoke, out of total cannabinoids in the cigarette.29–31 Yields were relatively low. Sheehan et al. 29 have shown THC mean delivery yields of 12.6% (ranging from 7.2% to 28.0%), similar to those reported earlier by Van der Kooy et al. 30 (11–26%). These relatively low yields are consistent with the estimations that up to 50% of the cannabinoids are lost in side-stream smoke, up to 30% are destroyed by pyrolysis, and 10% are trapped in the butt.29,32,33
Additional studies have shown that the yield notably depends on the smoking parameters, that is, puff frequency, puff length, and puff volume. 30 Higher yields were found for higher frequency (15 sec as compared with 30 and 60 sec), longer suction (4 sec as compared with 2 and 3 sec) and higher puff volume (50 mL as compared with 25, 35, and 45 mL). Yield dependency on puff parameters was also supported by Fehr and Kalant. 34
Also tested was the effect of mixing cannabis with tobacco, a common practice among cannabis users. 31 Generally, the yield of THC was higher in such mixes compared with those of pure cannabis cigarettes. 31 The authors have related the improved yield to a more efficient burning due to different combustion temperatures and also suggested differences in vapor pressure equilibrium, without elaborating on this point. 31
The nature and efficacy of cannabis medical treatment is determined by the effects of each one of the consumed APIs and by those of some interactions between the APIs.35–37 Yet, very little is known about smoking delivery yield and rate of provision of APIs other than THC. Sheehan et al. 29 have reported total terpene content in the smoke, ranging between 50 and 4300 μg/g of cannabis smoked. Fischedick et al. 24 reported the concentration in the smoke of some specific terpenes, and compared the effects on CB1 activation of pure THC to those of cannabis smokes. No significant differences were observed.
Moreover, based on our experience, cannabis cigarettes are mostly consumed in several portions (rather than consuming the entire cigarette at once). As such, plausible differences in the delivery yield of the various APIs—derived from differences in their partial vapor pressures—might result in different compositions of the different inhaled fractions.
Indeed, we have recently demonstrated a significant difference in the evaporation rate of various cannabis APIs, inhaled through a vaporizer. Specifically, monoterpenes and monoterpenoids were mostly evaporated and the sesquiterpenes and sesquiterpenoids were markedly evaporated, before there was any significant evaporation of THC. 38
To evaluate the actually inhaled multi-API component compositions during cannabis smoking, the current study focuses on (i) reproducibility of the results of smoking the cigarette between portions; (ii) distinguishing between the yields of delivery of the various APIs and (iii) mechanistic explanation to the results.
Materials and Methods
Plant material
Inflorescences of three commercial cannabis chemovars were obtained from Bazelet Pharma (Or Akiva, Israel), an authorized medical cannabis manufacturer in Israel: (i) a THC-rich chemovar with total THC (T-THC) and total CBD concentrations of 14.3% and <0.1%, respectively; (ii) a THC-rich chemovar with T-THC and total CBD concentrations of 23.5% and <0.1%, respectively; and (iii) a balanced chemovar with T-THC and total CBD concentrations of 9% and 10.6%, respectively. The inflorescences were stored in a freezer for 48 h and then ground to particles of about 2 mm.
Unsmoked cigarettes were used as a control (Experiments 1A, 1B, 1C, and 1D). In each experiment, controls and smoked cigarettes shared the same cannabis inflorescence and had similar weight.
Blank material
A fraction of the ground THC-rich inflorescence was extracted twice with 95% ethanol at solvent to plant material weight ratio of 10:1. After the removal of the ethanol, the ground extracted material contained <1% THC. This pre-extracted ground material is referred to in the following as blank material.
Preparation of cannabis cigarettes
Commercially available cannabis cigarette's conic tubes (109 mm length, 40 mm filter) were used (Jware). For the various experiments, the cigarettes were prepared to contain 125, 250, or 500 mg of ground material each. The exact weight for each cigarette was determined using an analytical balance.
Smoking machine
A suction bottle was filled with 30 mL of 95% analytical grade ethanol. Then, using a rubber Guko gasket, a 10-mL serological pipette was inserted into the suction bottle until it approached the bottom of the ethanol solution. The cigarette was inserted into the tip of the serological pipette. The suction bottle was connected to a peristaltic pump (crpump, BT300LC) (Supplementary Fig. S1), precalibrated to 35 mL in 5.6 sec. The intervals between the puffs were set to 15 sec. These parameters were selected based on the conclusions of a previous study, 30 to obtain optimal yield. The smoke, trapped in the ethanol was sent for analysis in high-performance liquid chromatography (HPLC) and gas chromatography (GC), to measure the concentration of cannabinoids and terpenes, respectively.
To verify that no loss of cannabis smoke is made through the peristaltic pump, a second suction bottle filled with 30 mL ethanol was connected to the pipe exiting the peristaltic pump. The smoke trapped in this second suction bottle was also sent for analysis. Results demonstrated that above 98% of the cannabinoids are captured in the first tube.
Analysis of the inflorescence samples
Each sample (either nonsmoked inflorescence or smoking residues) was weighed and mixed with ethanol at a ratio of 45 mL ethanol per 500 mg plant material in a 50-mL tube. Then, the mixture was sonicated for 30 min, at 40°C in a properly closed tube. After removal of the plant material, 500 μL of the ethanol solution was diluted with 25 mL methanol. The diluted solution was filtered through a 0.22 μ filter and then analyzed.
Analysis of the smoke samples
The smoke, trapped in the 30 mL ethanol (see Smoking machine section above) was sent for analysis. No additional dilution was performed.
HPLC and GC analysis
High-performance liquid chromatography
The analysis of cannabinoids was carried out on HPLC Waters PDA 2996 (Waters Corporation, Milford, MA), equipped with a pump, autosampler, column-oven, and a photodiode array detector (PDA). The analytical balance was Mettler Toledo MS205DU (Mettler Toledo, Columbus, OH). The HPLC column used was Phenomenex Luna Omega C18 column (Phenomenex, Torrance, CA). The mobile phase was buffer (ammonium acetate):acetonitrile, at 1:1 ratio, at a constant flow of 0.1 mL/min. Detection used wavelength of 220 nm, and injected volume of 10 μL.
The method is fully validated for 12 cannabinoids in line with the requirements of the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) guidelines, 39 Israeli Medical Cannabis Association (IMCA), European Pharmacopoeia (EP), 40 and United States Pharmacopeia (USP). 41 The nominal working concertation is 100 μg/mL and the method range is 0.1–120.0% of that nominal working concentration, proved by linearity, precision, and accuracy studies.
The limit of detection of the method is 0.1 μg/mL and limit of quantitation −0.2 μg/mL. Uncertainties were within 5% of the reported value. Total cannabinoid content was calculated as if all of the cannabinoids were in their decarboxylated form.
Gas chromatography
Terpene analysis was carried out on Agilent Technologies GC system model 6890 N (Agilent Technologies, Santa Clara, CA) equipped with Flame Ionization Detector. Identification was based on the retention times of the Certified Reference Material (CRM) standards and was verified by GC mass spectrometry at AminoCann (Aminolab, Ness Ziona, Israel). A CTC autosampler (Pal RTC; CTC Analytics, Zwingen, Switzerland) was used. The column used was Phenomenex ZB-624plus (Phenomenex) with helium as carrier at 1.2 mL/min constant flow. The method is fully validated for 25 terpenes likely to be present in cannabis. The method is fully validated according to the requirements of the ICH 39 guidelines, IMCA, EP, 40 and USP. 41 The range of the method was between 200 and 4000 μg/mL, proved by linearity, precision, and accuracy studies. The limit of reporting was 200 ppm. Uncertainties were within 5% of the reported value.
Standards
Cannabinoid standards
Cannabinol (CBN), cannabichromenic acid (CBCA), cannabichromene (CBC), cannabigerolic acid (CBGA), CBG, cannabidiolic acid (CBDA), CBD, Δ9-tetrahydrocannabinoic acid (THCA), and Δ9-THC were purchased from Cerilliant (Cerilliant Corporation, Round Rock, TX). Cannabidivarinic acid (CBDVA), cannabidivarin (CBDV), tetrahydrocannabivarin (THCV), and Δ8-THC were purchased from Restek (Bellefonte, PA). All cannabinoid standards are CRM standards, at 1000 μg/mL in methanol.
Terpene standards
Terpene standards were purchased from Restek, Merck (Rosh-Ha'ayin, Israel), and PhytoLab (Vestenbergsgreuth, Germany) (see Supplementary Data for further details). All terpene standards are CRM standards, at 2500 μg/mL in isopropanol. Ethanol for standard solutions and sample preparation is HPLC grade (J.T. Baker, Phillipsburg, NJ).
Statistical analysis
Statistical analysis was conducted using Statistical SPSS 20.0 software (IBM Corp., Armonk, NY). One-way ANOVA or paired/independent t-test was performed to quantify the API measurements within the various cigarette sections or between these and a control. Pairwise comparisons using Bonferroni adjustment for multiple comparisons were used in all post hoc analyses.
Experimental procedure
Experiment 1: Changes in the cigarette composition during smoking
Experiment 1A
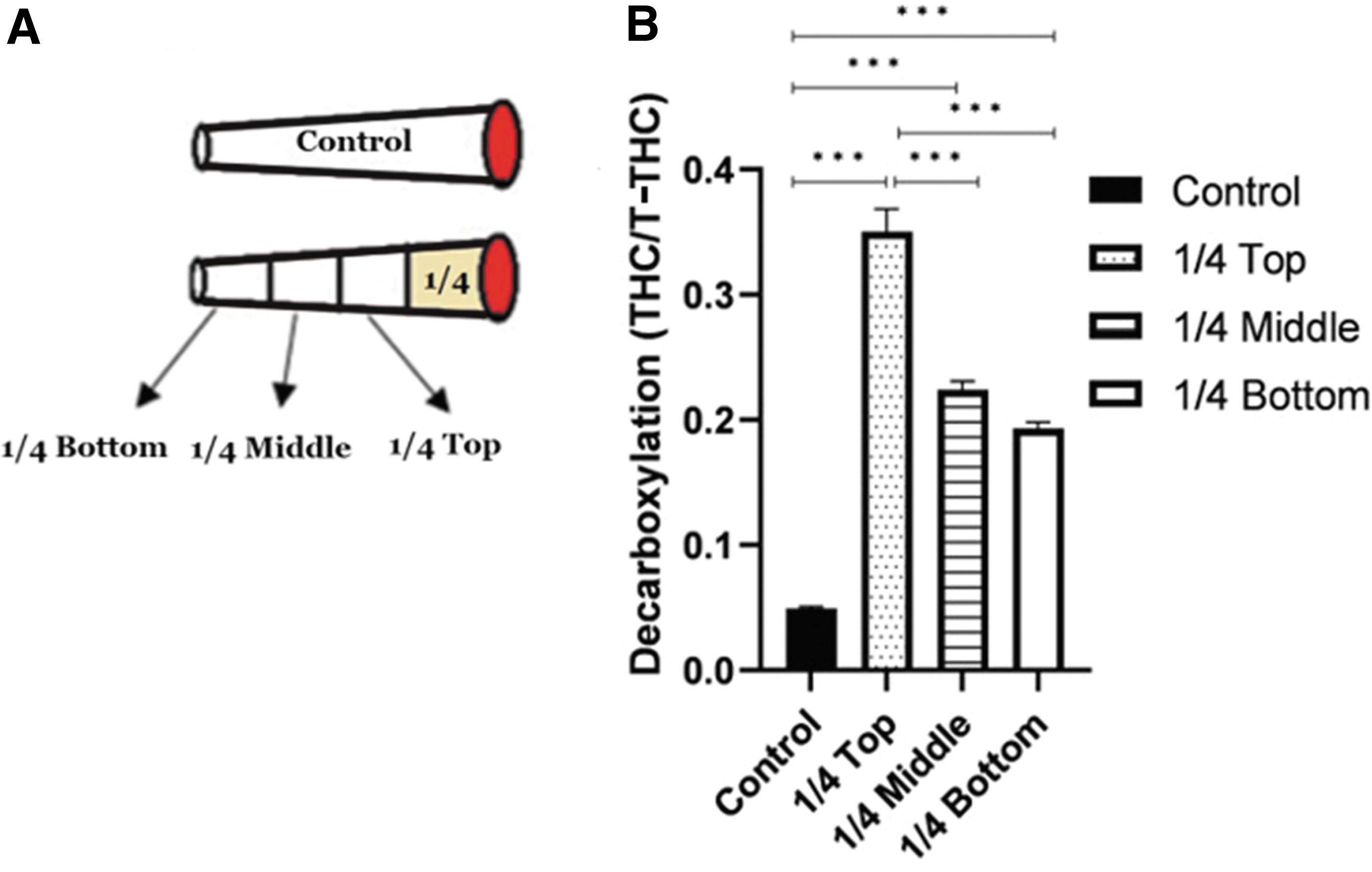
Seven tubes were filled with 500 mg of THC-rich inflorescence, containing 14.3% THC and <0.1% CBD. A quarter of each cigarette was smoked on the smoking machine and then the cigarette was extinguished. The residue, the nonsmoked part of the cigarette, was divided into three equal sections designated top, middle, and bottom, wherein the top was the one closest to the burning part and the bottom—the furthest (Fig. 1A). THCA and THC concentrations were determined in each one of the quarters (n=7), and compared to those in the control (unsmoked cigarette containing 500 mg of the same cannabis inflorescence, n=3).
Experiment 1B
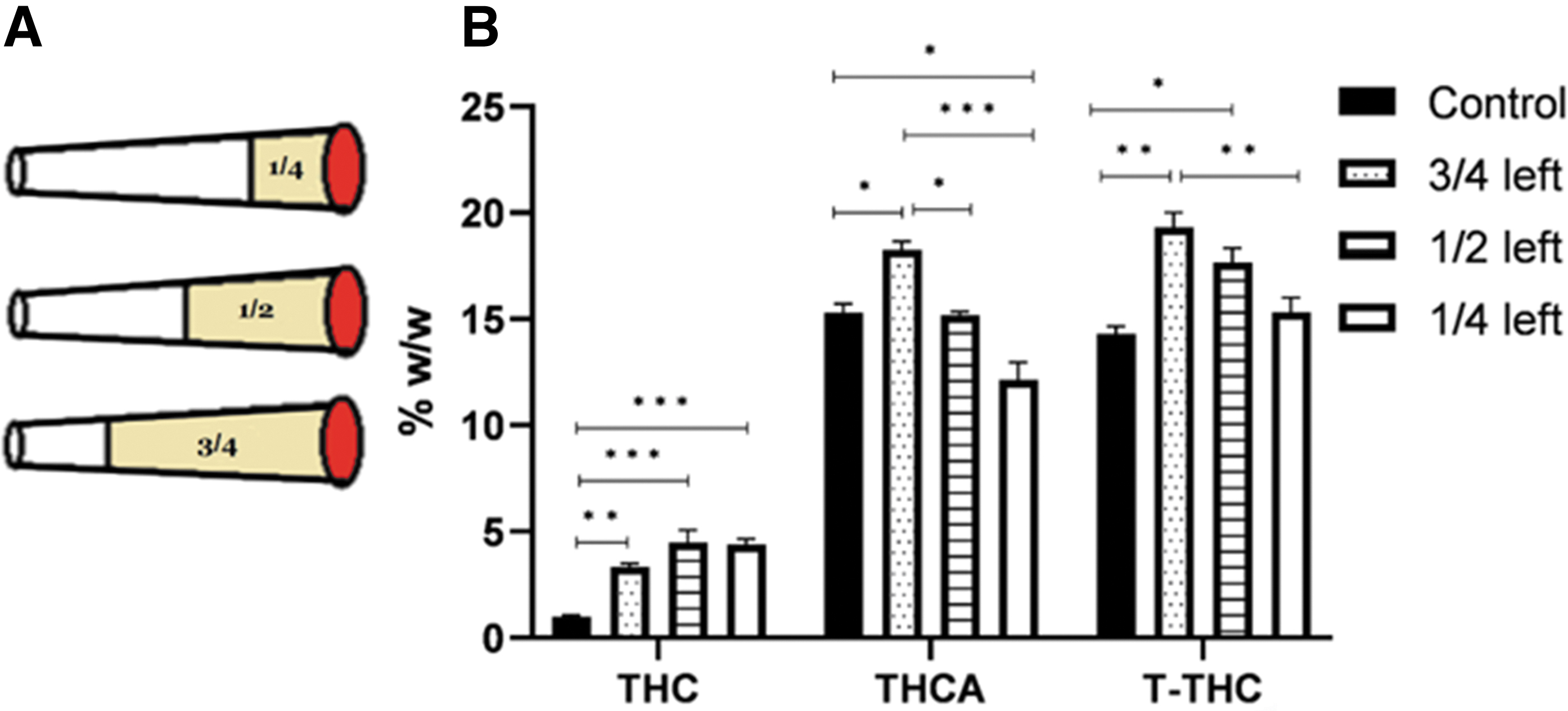
Nine tubes were filled with 500 mg each of THC-rich inflorescence (14.3% THC, <0.1% CBD). These cigarettes were divided into three groups, varying in the fraction of the cigarette smoked, that is, one-quarter, one half, or three-quarters smoked (n=3 in each) (Fig. 2A). THC and T-THC concentrations were analyzed in the residues, and compared to those in the control (n=3).
Experiment 1C
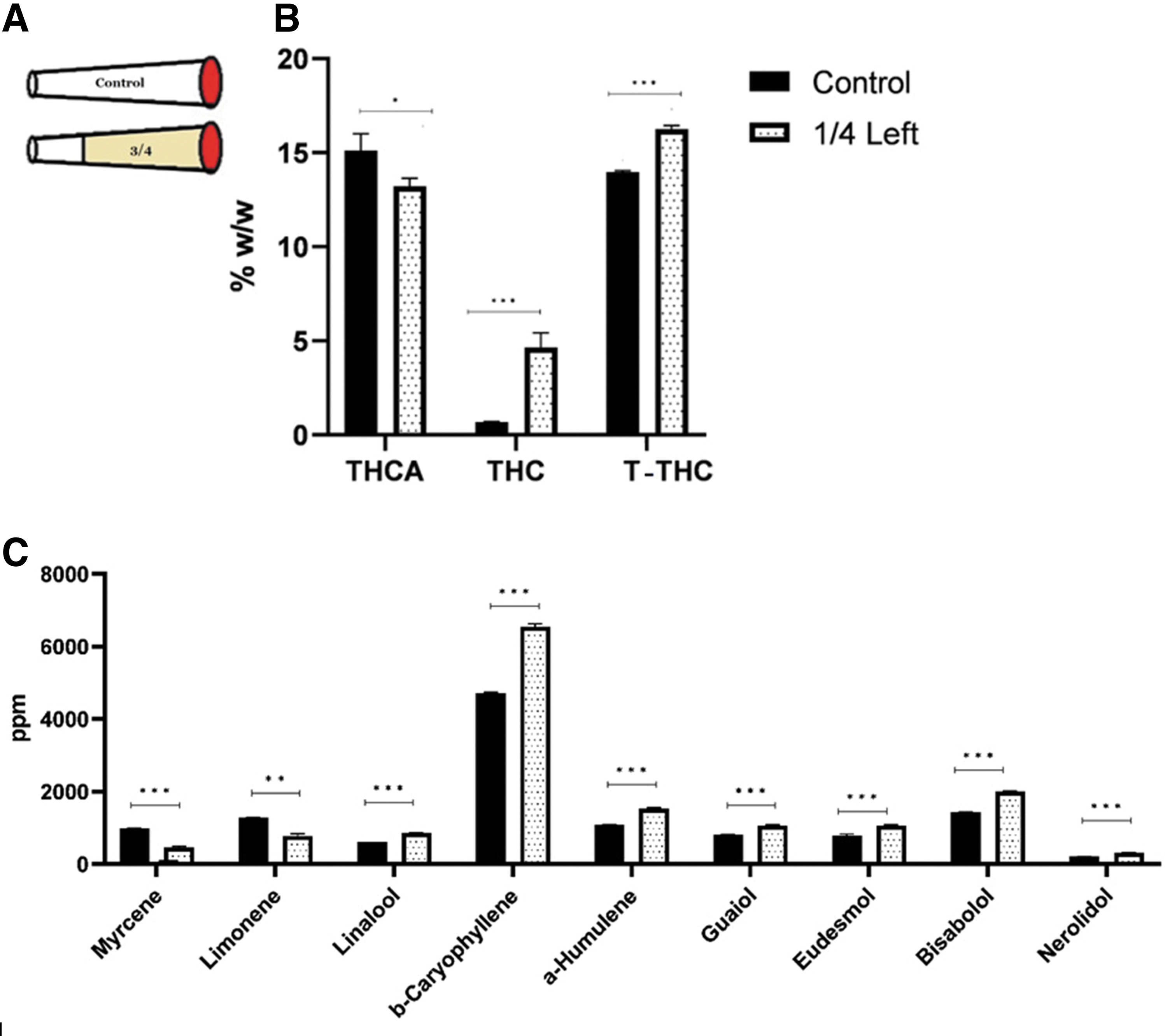
Six tubes were filled with 500 mg each of THC-rich inflorescence (14.3% THC, <0.1% CBD). Three-quarters of each cigarette were smoked on the smoking machine (Fig. 3A) and the residues were analyzed for THC, THCA and terpene concentrations, and compared to those in the control (n=3).
Experiment 1D
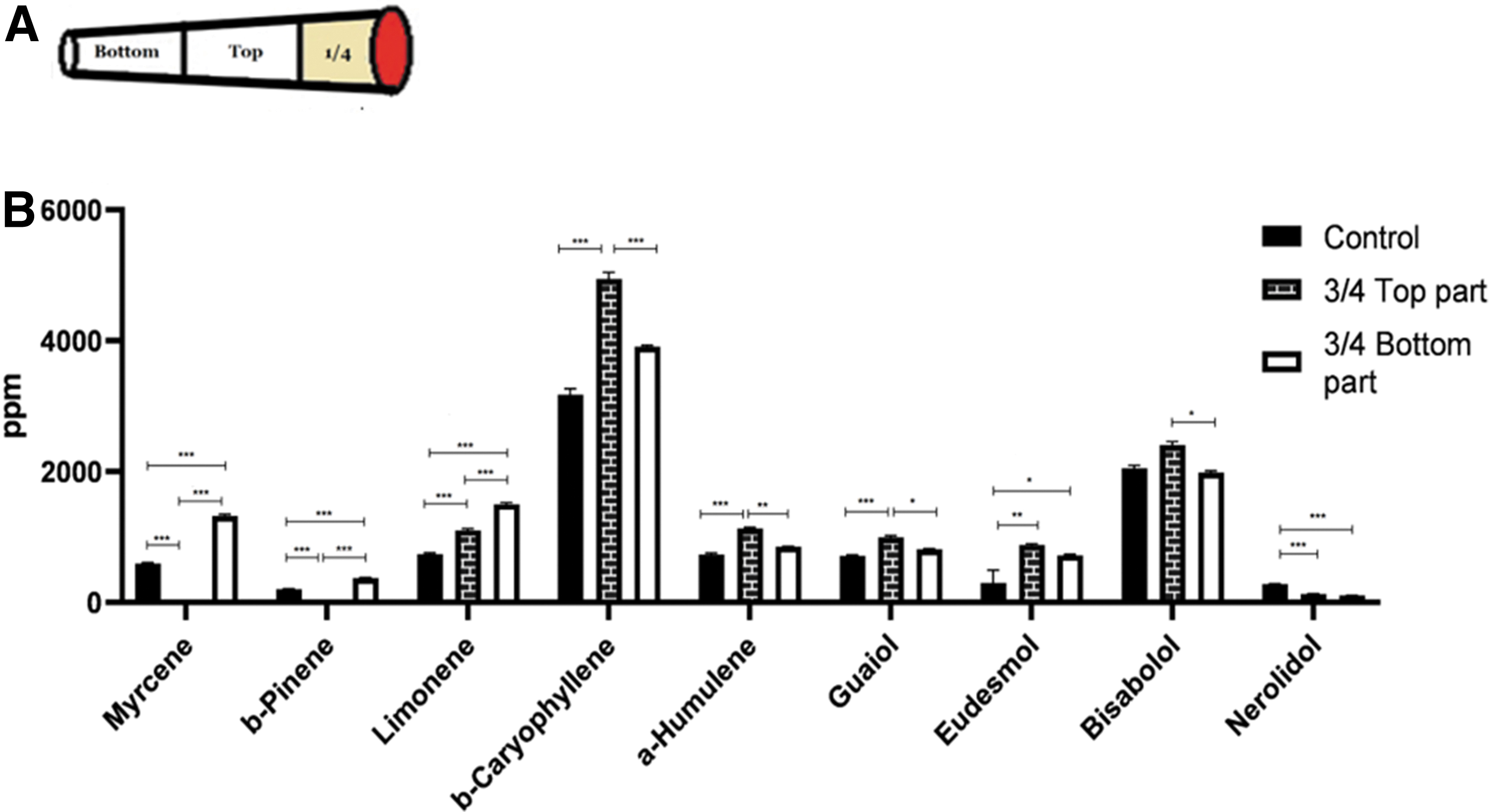
Three tubes were filled with 500 mg each of THC-rich inflorescence (14.3% THC, <0.1% CBD). One-quarter of each cigarette was smoked on the smoking machine. Each residue was divided into two halves, the top one, which was closer to the burning end, and a bottom one (Fig. 4A). Those residue halves were analyzed for their terpene concentration and compared with controls (n=3).
Experiment 2: Changes in the smoke composition during smoking
Experiment 2A
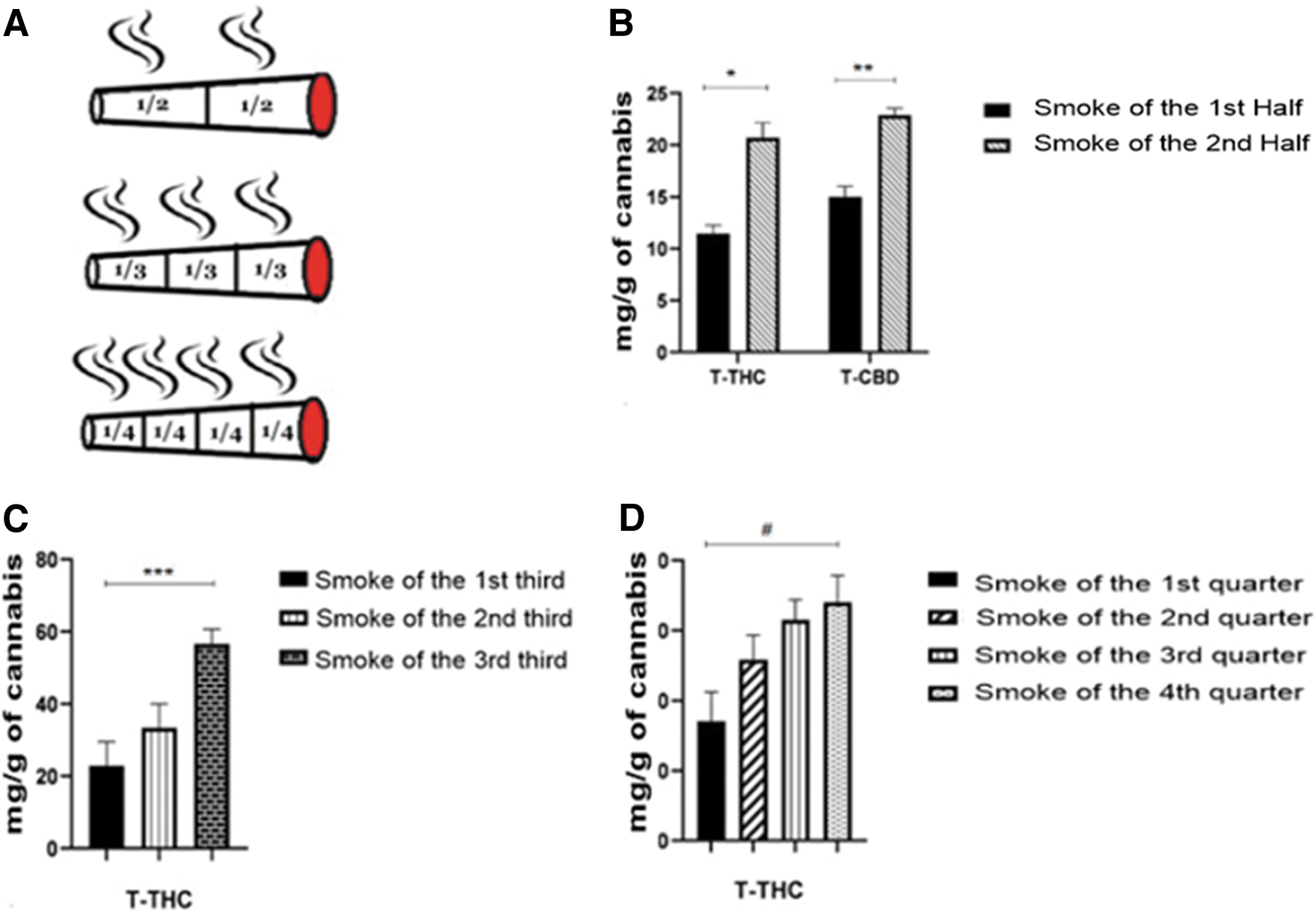
Six tubes were filled with 500 mg each of balanced inflorescence (9% THC and 10.6% CBD). One half of each cigarette was smoked using the smoking machine and then extinguished. The smoke was collected. Then, the second half was lit and smoked and its smoke was collected (Fig. 5A). The smokes of the first halves and those of the second halves were analyzed for their cannabinoid and terpene content.
Three tubes were filled with THC-rich chemovar (23.5% THC and <0.1% CBD) and were smoked in three parts, using the procedure detailed above (Fig. 5A).
Three additional tubes were filled with THC-rich chemovar (23.5% THC and <0.1% CBD) and (Fig. 5A) were smoked in four parts, using the procedure detailed above.
Experiment 2B
Three tubes were filled with 125 mg each of THC-rich inflorescence (14.3% THC, <0.1% CBD), smoked on the smoking machine and the smoke was collected and analyzed (these will serve as the control cigarettes. Scon=for smoke of the control cigarette). Three additional tubes were each filled with 375 mg blank (pre-extracted) material and then 125 mg of the same THC-rich inflorescence was added on top of the blank material (in the part to be first lit). These cigarettes were smoked on the smoking machine, a quarter after quarter (being extinguished after each quarter and lit again for the next one). Smokes of each quarter were collected separately and analyzed (SC, SB1, SB2, and SB3, for smoke of the cannabis part, smoke of the top blank material, smoke of the middle blank material, and smoke of the bottom blank material, respectively) (Fig. 7A).
Experiment 2C
Three tubes were filled with 250 mg balanced inflorescence (9% THC and 10.6% CBD) each, smoked on the smoking machine, and the smoke was collected and analyzed (Scon). Three tubes were filled with 250 mg balanced inflorescence in the bottom part, followed by 250 mg blank material (pre-extracted) in their top part (the part to be first lit). These cigarettes were smoked on the smoking machine, one half after the other (being extinguished after each half and lit again for the next one). Smokes of each half were collected separately and analyzed (SBT and SCB for smoke of the blank material at top, and for smoke of the cannabis material at bottom, respectively). Three additional tubes were filled with 250 mg blank material in the bottom part followed by 250 mg balanced inflorescence in their top part (the part to be first lit). These cigarettes were smoked on the smoking machine, one half after the other (being extinguished after each half and lit again for the next one). Smokes of each half were collected separately and analyzed (SCT and SBB, for smoke of the cannabis material at top, and for smoke of the blank material at bottom, respectively) (Fig. 8A).
Terms
Control—ground inflorescence before smoking
Residue—the nonsmoked fraction of a cigarette left after smoking
Blank material—pre-extracted inflorescence
SC—smoke of ground cannabis inflorescence
SB—smoke of blank material
Top (first half)—the part which is close to the first-to-lit end
Bottom (second half)—the part which is close to the filter
T-THC—total calculated THC content (THC + THCA×0.88)
T-CBD—total calculated CBD content (CBD + CBDA×0.88)
Results
Decarboxylation during smoking (experiment 1A)
First, we have studied the degree of decarboxylation and its dependence on the distance from the burning end. Decarboxylation of the residues' quarters (top, middle, and bottom, see Experimental Procedure section) was compared and referenced with the decarboxylation of a control, a non-smoked cigarette (Fig. 1. Decarboxylation degree is presented in the form of THC fraction out of calculated T-THC). Decarboxylation of each one of the residue's quarters was greater than that in the control and decarboxylation of the top quarter was greater than those of the other quarters (one-way ANOVA, Supplementary Table S1).
Cannabinoids left in the nonsmoked fractions of a cigarette (experiment 1B)
Figure 2B presents the concentrations of THC, THCA and T-THC in the residues after smoking one quarter, one half or three quarters of a cigarette as compared to those in a control cigarette (see details in the Method section).
In accordance with the prior experiment, THC contents of all residues were higher than those in the control. T-THC contents of the 3/4 and 1/2 residues (obtained following smoking one-quarter and one-half of a cigarette, respectively) were significantly higher than those in the control. T-THC content of the 3/4 residue was significantly higher than T-THC content of the 1/4 residue (one-way ANOVA, Supplementary Table S1).
Cannabinoid and terpene content in the residue after smoking three quarters of the cigarette (experiment 1C)
Figure 3 compares the concentrations of THC, THCA, T-THC and terpenes in the residues following smoking three quarters of a cigarette to those in a control. Both THC and T-THC concentrations in the residues were higher than those in the control. As expected, THCA levels in the residues were lower than those in the control (Fig. 3B) (independent t-test, Supplementary Table S2).
Higher concentrations in the residue as compared with control were also found for linalool, caryophyllene, humulene, guaiol, eudesmol, bisabolol, and nerolidol. However, in cases of myrcene and limonene, the concentrations in the residues were lower than those in the control (Fig. 3C) (independent t-test, Supplementary Table S2).
Terpenes left in different parts of the cigarette after smoking one-quarter of it (experiment 1D)
Figure 4B presents terpene concentrations in the top and bottom halves of a residue after smoking one quarter of a cigarette, as compared with those of a control. The terpene concentrations in the bottom parts were higher than those in the top parts and those in the control for myrcene, β-pinene, and limonene. An opposite result is found for β-caryophyllene, humulene, guaiol, eudesmol and bisabolol, where the concentrations in the top parts were higher than those in the bottom parts (one-way ANOVA, Supplementary Table S1).
Comparison of the content of cannabinoids and terpenes in the smoke of different parts of a cigarette (experiment 2A)
Comparing the cannabinoid content in the smoke of the first and second halves of a cigarette (Fig. 5B), demonstrates that both T-THC and T-CBD contents of the smokes of the second halves are higher than those of the first halves (independent t-test, see Supplementary Table S2 for details).
Similar results were obtained when smoking the cigarettes in a larger number of fractions. Thus, analyzing the smoke of the three-thirds of a cigarette demonstrates that T-THC contents of the smoke increased in consecutive fractions, that is, 1st third <2nd third <3rd third (Fig. 5C). Significant difference was obtained between the 1st and 3rd thirds (one-way ANOVA). Similarly, dividing the cigarette into four parts revealed that T-THC content of the smoke increases in the order, first quarter<second quarter<third quarter <fourth quarter (Fig. 5D). A trend was obtained between the first and fourth quarters (one-way ANOVA, Supplementary Table S1).
Calculating the average delivery yields, that is, the fraction of the cannabinoids in the smoke, out of the total cannabinoids in the cigarette (as done in Refs.29–31 ), demonstrated yields of 18% (for both CBD and THC) when smoking the cigarettes in two halves, and yields of 16% and 12% when smoking the cigarettes in three-thirds and four-quarters, respectively.
Figure 6B presents the terpene data of smoking cigarettes containing the balanced inflorescence (9% THC and 10.6% CBD) in two halves, using the above protocol. In accordance with the cannabinoid data, higher contents in the second half are found for all terpenes, expect for myrcene (paired t-test; Supplementary Table S2).
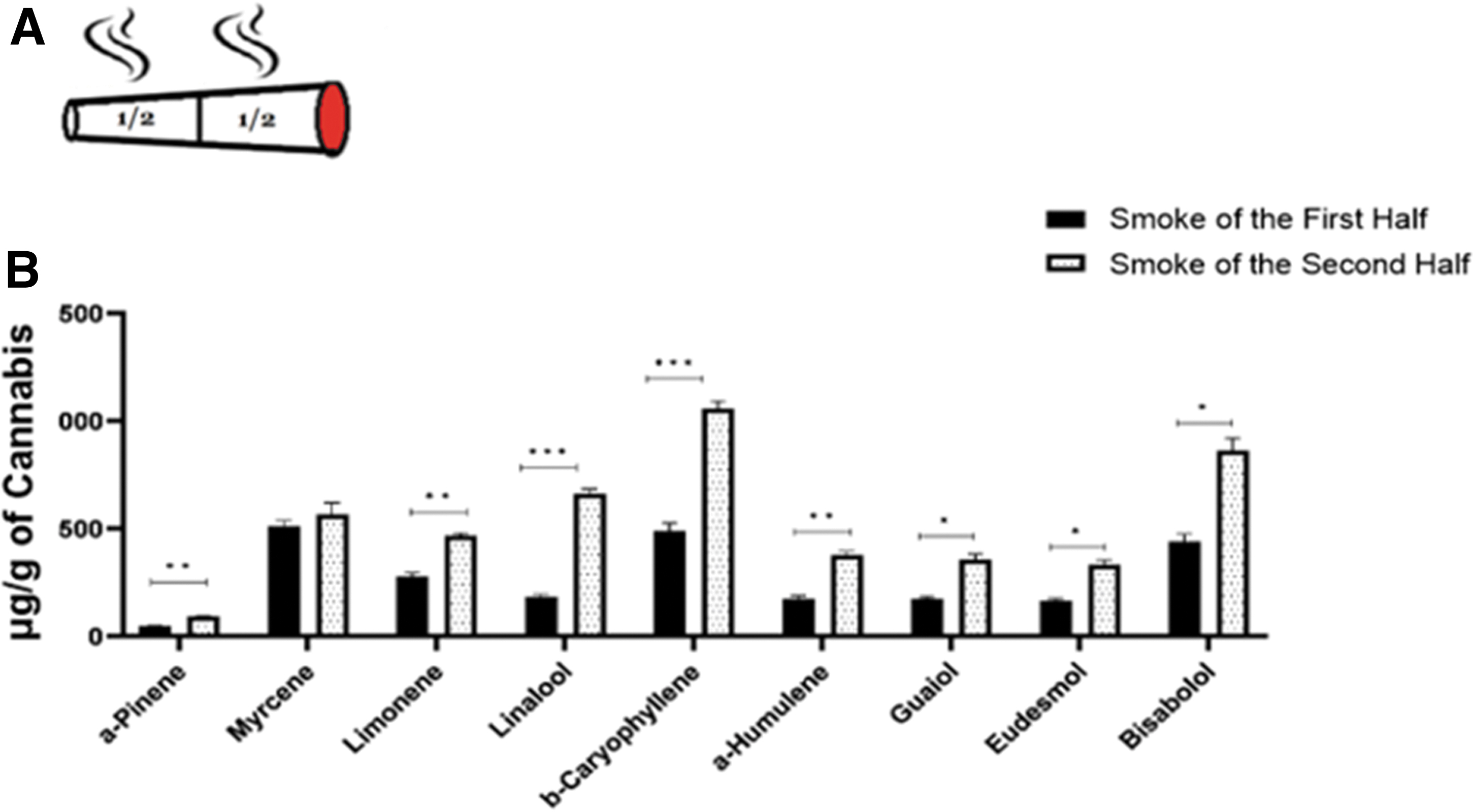
Terpene content in the smoke of two halves of the cigarette. Mean+SEM, *p<0.05, **p<0.01, ***p<0.001.
Cannabinoid content of smoke generated by smoking different sections of a cigarette containing cannabis and blank material (experiment 2B)
Figure 7B compares the total THC content in the smoke of a 125 mg inflorescence in a control cigarette (Scon) to that of a 125 mg of the same inflorescence at the top of a cigarette containing blank material bottom (SC). The content in the latter is only a small fraction of that in the former. Additional fractions of the cannabinoids are found in the smoke of the blank quarters although originally containing no cannabinoids (SB1, SB2, and SB3 for smoke of the top blank material, middle blank material, and bottom blank material quarters respectively). T-THC level in Scon was significantly greater than that in each one of the other cigarette sections; one-way ANOVA; Supplementary Table S1. The cumulative amounts of T-THC in the SC and the various blank material sections of the cigarette, resembled that in Scon (Fig. 7C).
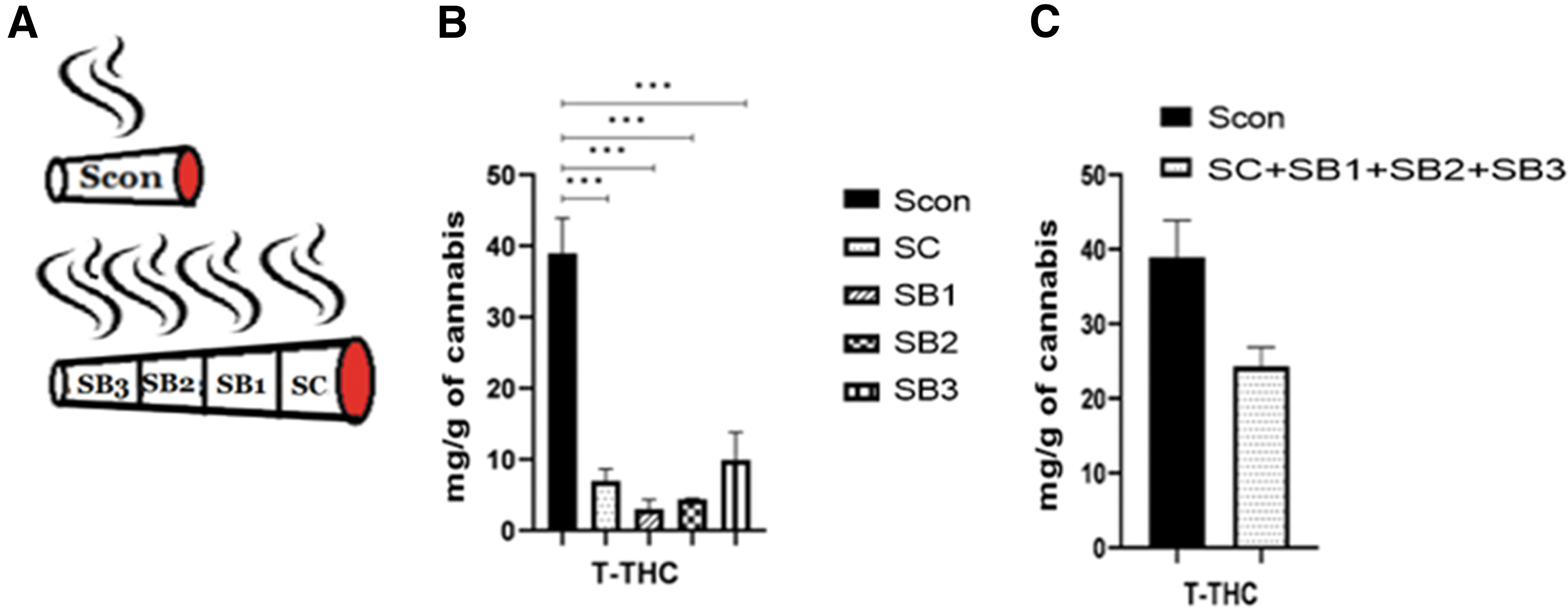
The results demonstrate a yield of 28% in the smoke of the control (Scon) cigarettes and of 17% in the combined sections of the cigarettes composed of inflorescence and blank material (SC+SB1+SB2+SB3).
Cannabinoid and terpene content of smoke generated by smoking different sections of a cigarette containing cannabis and blank material (experiment 2C)
Figure 8 presents the cannabinoid and terpene contents in the smoke of a cigarette composed of half cannabis inflorescence and half blank material. Consider first the smokes generated in smoking cigarettes composed of a top blank material half and a bottom balanced inflorescence half—SBT and SCB, respectively. SBT contained little to no cannabinoids, as expected from smoking of pre-extracted material. SCB had cannabinoids content similar to that of cigarettes containing 250 mg balanced inflorescence (Scon), again as expected. The picture changes in the case of the smokes of cigarettes composed of a top inflorescence half and a bottom blank material half—SCT and SBB, respectively. The cannabinoid content of SCT was only about one half that of Scon and SCB. At the same time, the smoke of the pre-extracted blank material SBB had cannabinoid content similar to that of SCT. This observation is true for both THC and CBD (Fig. 8B) (one-way ANOVA, Supplementary Table S1).
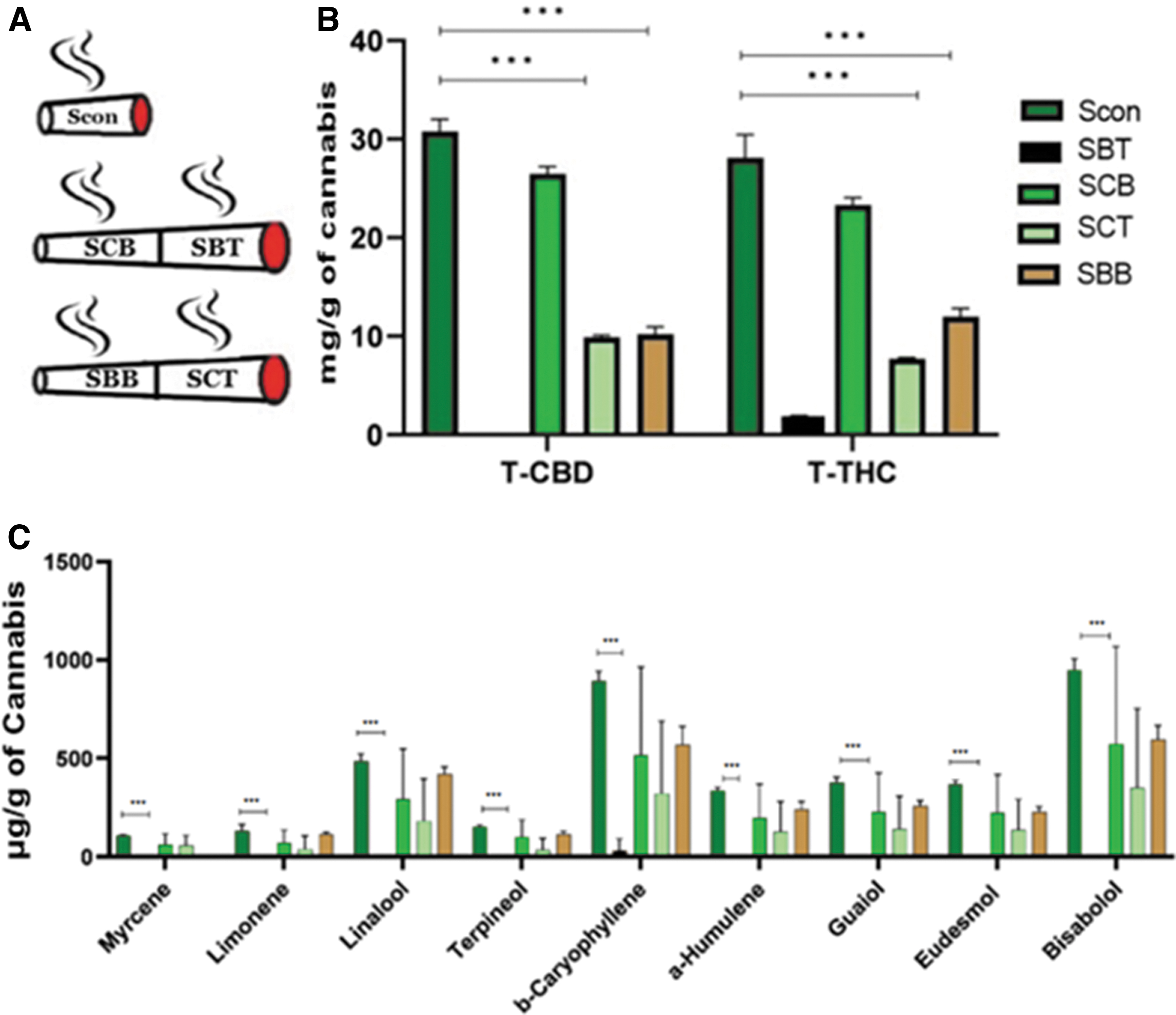
The results demonstrated a yield of 29% for CBD and 31% for THC in the control (Scon) cigarettes, of 26% for CBD and 27% for THC in the combined sections of the cigarettes composed of a top blank material half and a bottom balanced inflorescence half (SCB+SBT), and of 19% for CBD and 22% for THC in the combined sections of the cigarettes composed of a top balanced inflorescence half and a bottom blank material half (SBT+SCB).
Similar results were found for the terpene data. That is, the content of terpenes is low in SBT, but that in SCB is similar to that of the control. Yet, the content of SCT is relatively small and a major content is found in SBB (Fig. 8C) (one-way ANOVA, Supplementary Table S1).
Discussion
The results in this study show modifications, during the smoking progress, in the cannabinoid and terpene content of the smoke, as well as in the composition of the various fractions of the cigarette residues. A mechanism is proposed below for explaining these observations, based on the temperature gradient along the cigarette and on the physical properties of these APIs.
A temperature gradient exists along a burning cigarette. According to Ermala and Holsti, 42 at the burning tip, the temperature ranges between 470°C and 812°C and the neighboring section is at >300°C. The following section is between 100°C and 300°C and the rest is under 100°C. This “heat front” is moving along the cigarette with the progress of smoking. During inhalation, air is drawn in through the glowing end, increases its temperature, 43 heats up, and moves toward the mouth end of the cigarette. At each point along the cigarette, there is fixed ground cannabis matter at a given temperature and, during inhalation, there is also some moving hot air. The chances are that the moving air is hotter than the fixed cannabis.
This temperature gradient is demonstrated by the heat-driven decarboxylation process (as shown in Figure 1).44,45 After smoking a quarter of a cigarette, the fraction of decarboxylated THC out of the T-THC increased in the section that was closest to the burning end from 0.05 before smoking to 0.47 after it. The farther the section is from the burning end, the lower is the extent of decarboxylation, but in all cases, some decarboxylation does take place. This result demonstrates that the whole cigarette warms up to some extent on smoking a quarter of it, probably due to the movement of the hot air.
Cannabinoids are labile organic molecules that fully oxidize to CO2 and water next to the glowing tip, or partially oxidize at the neighboring hot region of >300°C. These reactions are fast. If there was no movement of cannabinoids away from these elevated temperature zones, not much API would have been left for smoking.
But THC and CBD do reach the smoke (as shown in Figures 6–8), which indicates that cannabinoids do move away from the hottest zones (THCA and CBDA decarboxylate early in the process and are therefore hardly found in the smoke). 46 At a given distance from the burning end, the temperature is low enough to slow degradation, but is still high enough to evaporate the cannabinoids. THC and CBD boiling points are about 425°C, but these cannabinoids do evaporate at lower temperatures, at a rate affected by their partial vapor pressure, which is about 6.7 and 40 Pa at 155°C and 190°C, respectively. 38
Thus, during inhalation, the cannabinoid vapors are drawn toward the mouth in the stream of hot air and move away from the degradation zone. This explains the way cannabinoids escape degradation and reach the smoke, but is not sufficient to explain the changes in smoke composition as smoking progresses. There is still another step.
As the vapor-carrying air moves, it reaches cooler regions and cools down. As a result, the contained cannabinoid vapors exceed saturation, leading to condensation, similar to dew formation in cooling air. A fraction of the cannabinoid vapors is therefore converted into small cannabinoid droplets, which are carried forward with the moving air. A fraction of those droplets finds its way to the inhaled smoke, but other droplets are blocked and trapped in the relatively small openings between ground particles and/or settle on those particles in sections where air flow rate decreases. As a result, cigarette sections that are remote from the hot tip get enriched with cannabinoids.
This enrichment due to movement of cannabinoid vapors, is demonstrated in the results of Figure 2, where T-THC concentrations in the residues after smoking one-quarter or one-half of the cigarettes are higher than those in the control. The same is true for the residue of smoking three-quarters of the cigarette (Fig. 3).
In agreement with the suggested mechanism, a fraction of the condensed droplets does reach the smoke, as demonstrated in the results of smoking a cannabis cigarette in two halves (Fig. 5). The smoke of the first half, the one, that is farthest from the mouth end, does contain cannabinoids (which travel through the second half). At the same time, the cannabinoid content of the smoke of the second half is significantly greater. That is since cannabinoids from the first half that did settle in the second half, re-evaporate when the elevated temperatures approach their location and thereby get into the smoke. This is further demonstrated when smoking the cigarettes in three or four segments (Fig. 5), as cannabinoids from the first-to-lit segments settle and then re-evaporate at consequent segments.
Another demonstration is found in the results summarized in Figure 7. The smoke of smoking 125 mg inflorescence at the top of a 500 mg cigarette, contains much less cannabinoids compared with smoking a 125 mg inflorescence cigarette. Much of the rest of the cannabinoids is found on smoking the other three-quarters. It is important to keep in mind that those three-quarters were of blank material, which originally contained no cannabinoids. The cannabinoids found in their smoke are therefore cannabinoids of the inflorescence top part, which got trapped in the blank material fraction.
Similar observations are found in the results summarized in Figure 8. In one set of the experiments, the smoke of a first half filled with blank material (SBT), had essentially no cannabinoids and that of the second half, containing 250 mg inflorescence (SCB), contains a similar amount of cannabinoids as that of a cigarette containing only 250 mg inflorescence. Yet, in the other set, a large fraction of the cannabinoids of the first inflorescence half (SCT) got trapped in the second half, which originally contained no cannabinoids (SBB), and was found in the smoke of smoking that second half.
To summarize the discussion up to this point, the following steps are found in the mechanism of smoking, as illustrated in Supplementary Figure S2: (i) When the temperature in a cigarette region approaches 100°C, cannabinoids decarboxylate. (ii) As it gets to about 200°C (similar to that of common vaporizers) cannabinoids evaporate and are carried, in the stream of the air, away from the glowing end. (iii) Reaching cooler regions, the vapors cool down and condense to small droplets, some of which continue the move with the air and reach the smoke. (iv) The rest of the droplets are trapped and settle in the sections of the cigarette that are closer to the mouth. (v) The settled cannabinoids re-evaporate as the hot zone approaches, move with the air, and thereby reach the smoke.
Our delivery yields, the fraction of the cannabinoids that eventually reaches the smoke—are similar to those reported by Van der Kooy et al. 30 While this is not the focus of the study here, it is important to keep in mind, as already noted, that moving away from the elevated temperature increases the delivery yield by reducing the extent of degradation. Both loss to degradation and the move away from the degradation zone are accelerated by temperature elevation, but their dependencies on temperature may differ.
There are also additional affecting parameters, such as those of smoking. In fact, the analysis in this study explains the results of Van der Kooy et al. 30 Each one of shorter time between puffs, a larger volume inhaled, and a longer duration of inhalation, improve the yield of THC delivery since all of them improve its transfer away from the elevated temperature, where degradation takes place. The results presented herein further demonstrate that the delivery yield decreases as the cigarette is consumed in multiple fractions (Figs. 5, 7, and 8). Repeated lightening of the cigarette leads to increased degradation of the cannabinoids.
Another support to the discussed mechanism is found in the content of CBN, an oxidation product of THC. 47 According to Van der Kooy et al., 31 there is a correlation between increased THC delivery yield in the smoke and reduced proportion of CBN. Additionally, CBN proportion in smoking is greater than that in vaping. 46 In both cases, higher yields and lower temperatures lead to reduced THC oxidation.
A similar analysis applies to CBD, which has a similar decarboxylation temperature and a similar vapor pressure dependence on temperature, as shown in Figures 5 and 8. The same is probably true also for other cannabinoids (see the results for CBG in Van Der Kooy et al. 31 and Pomahacovaet al. 46 ). Differences between cannabinoids may result from difference in their rate of oxidation. Smoking cannabinoids that are more stable to oxidation would result in a somewhat greater delivery yield and less partially oxidized reaction products (high-temperature full oxidation to CO2 and water would still be about the same).
Similar considerations apply to the case of the terpenes, with a couple of important differences: (i) terpenes do not decarboxylate (but do oxidize); (ii) terpenes are more volatile than cannabinoids, and (iii) terpenes differ from each other in their boiling points and vapor pressures more than major cannabinoids do.
The terpenes of higher boiling points (typically sesquiterpenes and sesquiterpenoids) are affected similarly to the cannabinoids—evaporate, move away from the hottest zone, settle, and re-evaporate. This is demonstrated in the above-summarized results. Thus, as per Figure 3, in the residue after smoking three-quarters of the cigarette, the concentrations of sesquiterpenes and sesquiterpenoids are higher than those in the control, as is the case for the cannabinoids. As for the cannabinoids, the contents of sesquiterpenes are higher in the smoke of the second half of a cigarette (Fig. 6). They also appear in the smoke of the second half blank material (SBB) when smoked after smoking a first inflorescence half (SCT) (Fig. 8). In all those cases, the sesquiterpenes evaporated, moved away into a cooler zone, settled there, and then re-evaporated to reach the smoke.
The lower-boiling terpenes, that is, the monoterpenes, also show these steps, however, the section of the cigarette that is hot enough for evaporating them is larger (extending relatively farther from the glowing end). At the same time, as smoking progresses, the cooler part gets shorter, so that there is no condensation or just very little condensation of these terpenes. As a result, monoterpenes move away more rapidly from the hot zone and reach the smoke first. Thus, Figure 3 shows that in the residue after smoking three-quarters of the cigarette, monoterpenes content is lower compared with the control. The effect of the lower boiling point of monoterpenes is also shown in Figure 4, wherein monoterpenes evaporate and move all the way to the far end (bottom part), while sesquiterpenes and sesquiterpenoids settle in the closer section (top part), the temperature of which is sufficient to keep in vapor form only the low-boiling monoterpenes.
Conclusions
A mechanism was conceived for the transfer of cannabinoids and terpenes from the cannabis inflorescence into the smoke and was supported by dedicated experiments. The mechanism involves the steps of (i) partial degradation of the cannabinoids, (ii) decarboxylation of the rest, (iii) evaporation of the decarboxylated cannabinoids and move of vapors away from the hottest zone, (iv) condensation of vapor to droplets that partially settle in regions that are more remote from the hot part, and (v) re-evaporation as the hot zone approaches, to reach the smoke.
A similar mechanism applies to the sesquiterpenes and sesquiterpenoids, the boiling points of which approach those of the cannabinoids, except that terpenes do not decarboxylate. Same considerations apply to the monoterpenes, but since they boil at a lower temperature, they evaporate earlier than the cannabinoids and the sesquiterpenoids and therefore condense less.
This mechanism explains the observed changes in the cigarette composition and in the smoke composition as smoking progresses. It also explains the effects of smoking parameters on the delivery yield and the correlation between yield and CBN formation.
An important outcome is that delivery of the pharmaceutically active ingredients (cannabinoids and terpenes) cannot be uniform. Their amount per puff increases as smoking progresses. Thus, for example, if one half of the cigarette is used in the morning and the other half for the night, the dose in the latter might approach twice that in the former.
Due to this mechanism, in smoking, terpenes are inhaled before the cannabinoids are. The same is true for using vaporizers. 38 Additionally, terpenes' delivery yield is greater, due to reduced degradation on faster move away from the heat. These are particularly true for the monoterpenes, the boiling points of which are relatively low. Thus, during smoking there are changes in both the amount of APIs and their composition.
This finding further increases the difference between cigarettes and oils (extracts), where monoterpenes are lost during processing. 48
Footnotes
Acknowledgment
The authors thank Inna Perutski for HPLC and GC analyses.
Data Availability Statement
The data presented in this study are available on request from the corresponding authors.
Authors' Contributions
Writing—original draft preparation: A.M.E., D.H.-S., and N.R.; writing—review and editing: A.M.E., D.H.-S, and N.R.; Statistical analysis: D.H.-S.; and Experimental procedure: D.S and D.H.-S. All authors have equally contributed to the work. All authors have read and agreed to the published version of the article.
Author Disclosure Statement
A.M.E., D.H.-S., D.S, D.B.-Z., and N.R. are employers of the Bazelet group, a medical cannabis manufacturer in Israel.
Funding Information
No funding was received for this article.
Abbreviations Used
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
