Abstract
Introduction:
The explosive growth of the cannabis industry in the United States over the past decade has spurred a multitude of products derived from phytocannabinoids produced by Cannabis sativa L. Decades of cannabis prohibition coupled with the more recent 2018 Farm Bill have lead to several unanticipated consequences and the widespread availability of synthetic cannabinoids derived from hemp CBD, including Δ8-THC, Δ10-THC and HHC.
Methods:
Herein, we review the available literature of the complexity of the chemistry of its current manufacture, namely, the acid-catalyzed ring closure of cannabidiol (ACRCC), the myriad of issues involving the unsolved technical problems with quality control of ACRCC-Δ8-THC and the multitude of isomerized byproducts, and the lack of consistent regulation regarding consumer safety and labeling.
Results:
We provide what we believe is the first comprehensive listing of all the documented ACRCC-Δ8-THC byproducts. Perhaps, most importantly, we highlight the growing concern that, other than Δ8-THC itself, the compounds in ACRCC-Δ8-THC product mixtures have not been subjected to any human toxicological evaluation. This is especially troubling as ACRCC-Δ8-THC products relate to vaping, and their contribution to a growing and lethal epidemic of electronic cigarette, or vaping, product use–associated lung injury (EVALI).
Conclusions:
Quality control is totally inadequate in the newly emerging Δ8-THC industry. American consumers are ingesting products that are mislabeled with many compounds that have never received any toxicological testing. EVALI cases continue to be reported with a fatality rate approaching 2% (in California).
Introduction
The explosive growth of the cannabis industry in the United States over the past decade has spurred a multitude of new and innovative products derived from the naturally occurring phytocannabinoids produced by Cannabis sativa L. Entrepreneurs have incorporated CBD
Chemical Structures
The 2018 Farm Bill
2
expressly removed industrial hemp from the definition of marihuana, defined as cannabis with <0.3% by weight Δ9-tetrahydrocannabinol (Δ9-THC)
The focus of this article is to highlight the myriad of non-natural THC isomers formed in the ACRCC conversion reaction, including Δ8-tetrahydrocannabinol, (Δ8-THC)
The Legal Landscape
Despite the 2018 legislation exempting the hemp plant and its derivatives from the regulatory prohibition of the CSA, all other naturally occurring cannabinoids produced from C. sativa, including the psychoactive cannabinoid, Δ9-THC, are still listed as Schedule 1 substances. According to the CSA, 5 a Schedule 1 substance is a drug that has
no currently accepted medical use
a high potential for abuse or addiction
a lack of accepted safety for use under medical supervision.
By default, Schedule 1 substances like cannabis (marijuana) remain illegal under federal statutes and in the 32 states in which recreational cannabis has not been legalized; it is in these states where the emerging market for Δ8-THC, which has psychoactivity similar to Δ9-THC, is most lucrative. Ironically, despite the Schedule 1 designation for the phytocannabinoid Δ9-THC (and its isomeric derivatives), the U.S. Food and Drug Administration (FDA) has approved a number of pharmaceutically produced Δ9-THC drugs such as Marinol® and Syndros®, prescribed for the treatment of severe nausea and vomiting, 6 and Epidiolex,® a plant-derived CBD for the treatment of intractable forms of epilepsy. 7
There is no known biosynthetic pathway that synthesizes Δ8-THC in C. sativa; however, to a small extent, the Δ9 isomer can isomerize to the more thermodynamically stable Δ8 isomer.
8
Through the actions of the plant enzyme tetrahydrocannabinolic acid (THCA) synthase, the plant produces the Δ9 isomer exclusively. Within the glandular trichomes of C. sativa, Δ9-THC is produced as its acidic precursor, namely Δ9-tetrahydrocannabinolic acid (Δ9-THCA)
Through an unforeseen loophole in the 2018 Farm Bill, ACRCC-Δ8-THC has been construed as legal because it may be produced in a chemical process directly from CBD isolated from industrial hemp. Unfortunately, the Bill's language directly contradicts the CSA, where all tetrahydrocannabinol isomers (code 7370) specifically list Δ8-THC along with its isomeric cousin Δ9-THC as a Schedule 1 substance.
12
Further ambiguity regarding the legality of ACRCC-Δ8-THC comes from a recent September 2021 letter to the Alabama Board of Pharmacy in which the Drug Enforcement Administration (DEA) wrote as follows:
cannabinoids extracted from the cannabis plant that have a Δ9-THC concentration of not more than 0.3 percent on a dry weight basis meet the definition of “hemp” and thus are not controlled under the CSA.
13
Since being introduced to consumer markets, ACRCC-Δ8-THC products have become a substantial revenue generator in many states where cannabis-derived products containing Δ9-THC are not legal, either medicinally or recreationally. These ACRCC-Δ8-THC products, often promoted to consumers as a less potent form of Δ9-THC, are currently being produced and sold without any regulatory control or quality assurance oversight. ACRCC-Δ8-THC products are not legally subjected to the same type of third-party testing at certified testing laboratories as required under state cannabis regulations. 14
Throughout the era of federal cannabis prohibition, illicit products have been, and will continue to be, made available to consumers to meet market demand. The principles of contraband economics dictate that if there's a market for an illegal product, producers will provide that product particularly when substantial profit can be made.
The Chemistry of THC Isomers
Δ8-THC is an isomer closely resembling Δ9-THC as seen from their three-dimensional structures (Figs. 1 and 2).15,16 The sole distinction is the position of the double bond in the methyl-cyclohexene ring between carbons 9 and 10 in Δ9-THC and 8 and 9 in Δ8-THC. However, even with this subtle conformational shift, a change in their corresponding pharmacological properties is observed. This includes their respective psychoactivity where Δ8-THC is reported to be less potent than Δ9-THC isomer in receptor binding studies.17,18
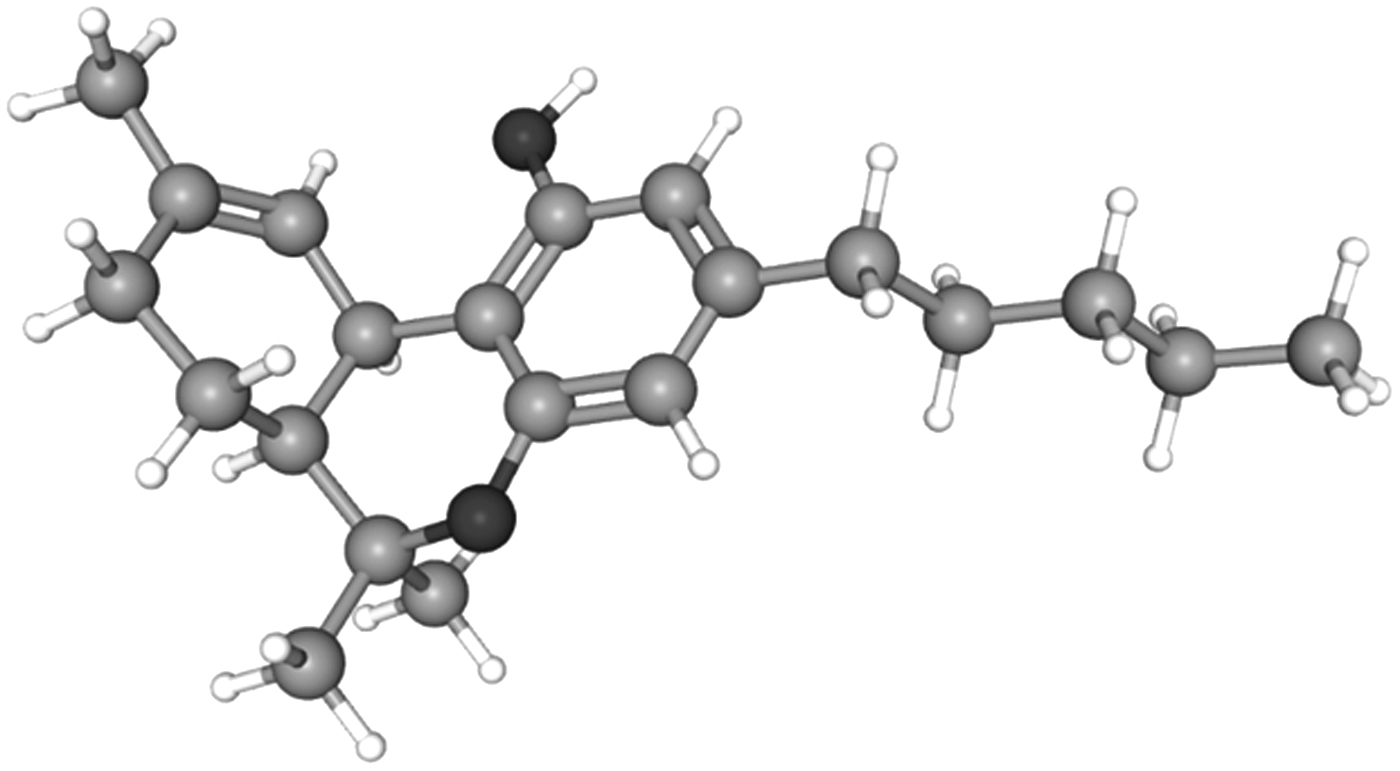
3D structure of Δ9-THC. 3D, three dimensional. Figure adapted with permission from PubChem. 15
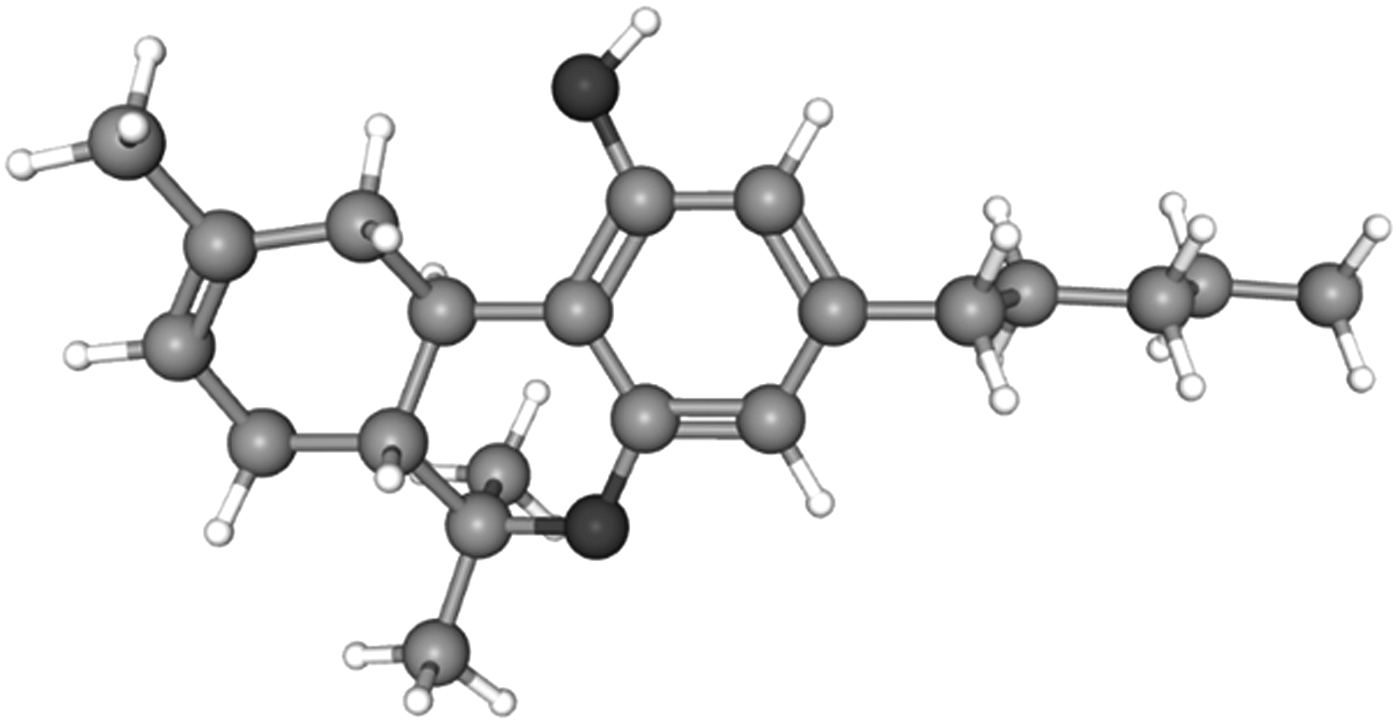
3D structure of Δ8-THC. Figure adapted with permission from PubChem. 16
Structurally speaking, CBD and all the THC isomers, including Δ7, Δ8, Δ9, and Δ10, have the identical molecular formula, C21H30O2. It is the subtle variation of how the atoms are three dimensionally arranged that confers major pharmacological differences between isomers. This is classically represented by the tremendous pharmacological differences between CBD and Δ9-THC. The differences in molecular topologies (shape) and spatial flexibility center around the fact that CBD is a constitutional isomer of THC with free rotation of the terpene ring along the axis of the phenyl ring of CBD.
Another way of looking at these differences is that CBD is a resorcinol with two meta-phenolic groups, while THC possesses one phenol, which provides for a significantly different and more varied pharmacological action. It is this nuanced difference that affects the isomer's respective receptor binding affinity to cannabinoid receptors. In this case, with both cannabinoid-1 receptor (CB1R) and cannabinoid-2 receptor (CB2R), Δ8-THC acts pharmacologically similar to Δ9-THC, as a partial agonist, with receptor binding at CB1R > CB2R. Also similar to Δ9-THC, the Δ8-isomer is metabolized in its first pass through the liver by the P450 cytochrome system to 11-hydroxy-Δ8-tetrahydrocannabinol (11-OH-Δ8-THC)
When organic compounds are used as reactants in chemical reactions, conducted both at small, research-scale and at larger production-scale facilities, the process chemistry is often complicated. In the case of ACRCC-Δ8-THC, depending on the reaction conditions, numerous additional THC isomers are formed with unknown pharmacological and safety profiles in humans. The crux of the problem is that ACRCC-Δ8-THC products are currently being manufactured and sold without any appreciable consideration for customer safety. The concern stems from the complexity of chemical processes involved in the manufacture of ACRCC-Δ8-THC. The issue in the crosshairs of the safety debate is that ACRCC-Δ8-THC is a “designer drug” synthesized from hemp-derived CBD and not extracted from naturally grown C. sativa material.
Historically, ACRCC-Δ8-THC products are not the first cannabinoid designer drugs. Professor James W. Huffman, a medicinal chemist at Clemson University, never anticipated that his library of laboratory synthesized CB1R agonists, such as JWH-018 and JWH-073, would end up on the street as psychoactive synthetic cannabinoids of abuse, namely K2 and Spice. 20 These compounds were designed solely for research purposes to study the pharmacological effects of potent CB1R full agonists, some exceeding 100 times greater binding affinity than Δ9-THC. These synthetic cannabinoids carry serious adverse side effects that often require emergent medical attention, including severe cardiovascular, neurological, gastrointestinal, renal, metabolic, and psychiatric sequelae. 21
The chemical conversion process of CBD to Δ8-THC was first described in the early 1940s by Professor Roger Adams and published by Drs. Yechiel Gaoni and Raphael Mechoulam in 1966. 22 The technical information describing this acid-catalyzed cyclization reaction can be easily accessed on various internet sites in step-by-step detail. 23 However, minor changes in reaction conditions, including reaction temperature, type of acid, solvent, exposure to atmosphere, the presence of water or alcohol, and duration of the reaction, can significantly affect the yield and mix of reaction byproducts. 24 After the passage of the 2018 Farm Bill, professional and inexperienced, amateur chemists began looking at the synthetic possibilities of the surplus of CBD; thus, the race to market new psychoactive products containing laboratory manufactured cannabinoids began.
ACRCC-Δ8-THC production requires the use of flammable reaction solvents (e.g., benzene), highly corrosive acidic reagents such as boron trifluoride, sulfuric acid, and hydrochloric acid, and heat to drive the cyclization reaction to completion. Typically, under most reaction conditions, CBD is converted to Δ8-THC, as well as several other non-natural isomers of THC, which could include Δ7-tetrahydrocannabinol
Furthermore, it has been demonstrated that even under physiological conditions, CBD may be converted to both Δ8-THC and Δ9-THC through this same ACRCC reaction in simulated gastric fluid 26 and in Wistar rats. 27 This may potentially explain reports of many patients reporting somnolence and psychoactivity after oral CBD ingestion. In fairness, the physiological conversion of CBD into THC isomers and derivatives is still being debated.28–30
Interestingly, studies have indicated the presence of other oxygenated cannabinoids in CBD vape products.
31
When the conversion reaction is performed using alcohol solvents, such as methanol or ethanol, a mixture of the corresponding 9- and 10-methoxy-hexahydrocannabinols
In another recent report, a full stoichiometric equivalent of an extremely reactive acidic reagent, phosphorus oxychloride (POCl3),
33
produced ACRCC.
34
It was found that, in addition to ACRCC-Δ8-THC and Δ4(8)-iso-tetrahydrocannabinol
The Quality Control Dilemma
Similar to other aspects of the cannabis industry, serious product quality control issues plague this new ACRCC-Δ8-THC marketplace.
35
The most important consideration of these products is the current absence of accepted product specifications or proper analytical standards and techniques for testing commercially produced ACRCC-Δ8-THC products. As this article was completed, a new certified analytical standard was made commercially available for the identification and quantification of Δ8-iso-tetrahydrocannabinol
Traditionally, high-performance liquid chromatography (HPLC) has been the analytical instrumentation utilized by most laboratories to analyze and quantify cannabinoid mixtures. 37 However, the mixtures of THC isomers formed in the ACRCC reaction present unique challenges in their identification, isolation, quantification, and purification, since some isomers co-elute with the other known isomers of THC. In practical terms, because of their similarity in polarity, the ACRCC-Δ8-THC byproducts may appear as a single unresolved peak in the HPLC chromatogram (Personal communication; Sams R, February 14, 2022).
Simply put, with current methodology, HPLC is unable to separate, identify, and quantify these myriad byproducts present in ACRCC-Δ8-THC products. Consequently, proper analysis and quantification of the full spectrum of these substances require different and more experienced data analysis, including the utilization of more sophisticated analytical techniques such as gas chromatography/mass spectrometry (GCMS), which have been demonstrated to be fit-for-purpose for the determination of Δ8-THC. 38
A recent publication illustrated the necessity for a more sophisticated analytical methodology. Researchers performed detailed chemical analysis of fluid from several commercially available vaporizers (vape pens) containing ACRCC-Δ8-THC using nuclear magnetic resonance, GCMS, and ion-coupled plasma/mass spectrometry.
39
The authors found that none of the products tested had accurate labeling and significant discrepancies were discovered in actual Δ8-THC content. Many of the products tested contained known ACRCC-Δ8-THC byproducts such as Δ4(8)-iso-tetrahydrocannabinol
Several of the products contained unlabeled cutting agents used in vape pen fluid formulations such as medium-chain triglycerides (MCT), triethyl citrate, and parts per billion levels of heavy metals such as chromium, nickel, copper, zinc, lead, mercury, and others, which likely leached from the vape pen hardware.
Perhaps most troubling was the finding of olivetol (3,5-dihydroxy-pentylbenzene)
The detection of olivetol further supports the formation of abnormally ortho-substituted THC regioisomers such as ortho-Δ9-tetrahydrocannabinol
The presence of olivetol in the ACRCC-Δ8-THC products further implies that the recombination of the olivetol and the limonene cation in the reaction leads to the formation of the terpene p-cymene (4-isopropyl toluene)
Vaping and ACRCC-Δ8-THC Pathology
A sizable amount of the ACRCC-Δ8-THC is sold for the vape market with no safety data for inhalation exposures. The FDA considers both polyethylene glycol (PG) and vegetable glycerin (VG) as “Generally Recognized as Safe’’ (GRAS). However, the GRAS designation applies only to dermal application or oral ingestion and does not address or imply the safety of inhalation exposure to these products. Goods meant to be eaten or swallowed are not necessarily meant to be inhaled deeply into the vast microenvironment of the pulmonary alveoli system. PG/VG-containing e-liquids, when heated, generate pulmonary irritants as well as known and suspected carcinogenic carbonyl compounds such as formaldehyde, acetaldehyde, and acrolein.46,47
Since the advent of the commercial availability of ACRCC-Δ8-THC in 2019, there has been a marked increase in reports of electronic cigarette, or vaping, product use–associated lung injury (EVALI). 48 Patients with EVALI present with a constellation of respiratory, gastrointestinal, and constitutional symptoms, including shortness of breath, cough, chest pain, diarrhea, abdominal pain, fever, and fatigue. To meet the Centers for Disease Control (CDC) criteria for a “confirmed” EVALI case, patients must have vaped within 90 days before symptom onset, have bilateral infiltrates on chest imaging, have a negative evaluation for infection, and have no other plausible alternative diagnosis. 49
In the current marketplace, ACRCC-Δ8-THC mixtures are solubilized with a multitude of diluents, including limonene, which individually have been suspected as being problematic for inhalation. Vape pen base fluids such as PG and VG, which when heated can generate pulmonary irritants including carcinogenic carbonyl compounds (formaldehyde, acetaldehyde, and acrolein) in addition to various metals contained within the heating coils and cartridge casings in vaping devices, leach into the inhaled vapor as well. 50 The CDC has postulated that the additive vitamin E acetate (VEA), a tocopherol, is a causative factor for EVALI, 51 as is diacetyl, a common buttery flavoring agent, has well-documented pulmonary toxicity leading to bronchiolitis obliterans, sometimes called “popcorn lung.”52,53
The role of VEA as a causative factor for EVALI has been postulated to be associated with its long, 16-carbon aliphatic tail that is thought to penetrate into the surfactant layer within the alveoli. Increasing amounts of tocopherols affect the transition of surfactant from a gel to a liquid, which affects its ability to maintain the necessary surface tension within the alveoli, and therefore has been postulated as a mechanism for EVALI lung injury. 54
Despite the aliphatic tail of ACRCC-Δ8-THC and its byproducts being only five carbons in length, we postulate that this may still be long enough to affect the phase state of the phosphatidylcholines acting as a surfactant in the alveoli, although likely not to the same degree as VEA. Regrettably, there are no toxicological data on ACRCC-Δ8-THC and its byproducts, yet their contributive role in EVALI is circumstantial. Definitive pulmonary vaping studies need to be done to definitively tie ACRCC-Δ8-THC products to the EVALI epidemic. To ignore the coincidence of the sudden appearance of ACRCC-Δ8-THC, vaping with the emergence of EVALI seems shortsighted.
A newly published national study reveals the breadth of the EVALI epidemic and its connection to cannabis vaping products, including ACRCC-Δ8-THC: as of January 2020, a total of 2558 nonfatal hospitalized patients and 60 patients with fatal cases of EVALI have been reported to the CDC. 55 This group observed a direct relationship between the frequency and duration of vaping with patient morbidity and mortality. Of note, secondary to the COVID-19 pandemic, the CDC stopped tracking EVALI cases in February 2020. In addition, because of the novelty of EVALI, the potential pulmonary morbidity seen with ACRCC-Δ8-THC products substances may be significant, and clinically is oftentimes untested, overlooked, and goes undiagnosed; therefore, the true breadth of the EVALI epidemic remains unknown.
Recently, 13 adolescents were admitted to a large university-teaching hospital for clinical signs consistent with EVALI, 30% of whom necessitated intensive care unit, and one required intubation with prolonged mechanical ventilation. Ninety-two percent of those patients admitted tested positive for Δ9-THC. 56 What number of those patients were also vaping ACRCC-Δ8-THC? We will never know as few hospital laboratories test for Δ8-THC exposure, but considering the large and growing market for ACRCC-Δ8-THC vape products, we know this number is not likely zero. In a study of computed tomography-diagnosed EVALI, the researchers looked at 160 cases in 14 states, and 133 cases (83%) were found in states where recreational cannabis was not legal. 57 It is in these states where ACRCC-Δ8-THC vape products are most prevalent.
Consumer Protection Concerns
The purification and isolation of purified ACRCC-Δ8-THC reaction products are problematic because the byproducts formed are chemically similar to both Δ8 and Δ9-THC. The process chemistry expertise needed to develop a suitable purification and analytical method to establish product specifications is not something that most manufacturers currently possess. Any perturbation in the reaction conditions such as reaction temperature, reaction time, concentration, and type of acid catalyst used changes the distribution of the various components in the final end-product mixture.
Consequently, finished consumer goods are being produced using ACRCC-Δ8-THC, which contain various amounts of the isomeric and degradation byproducts in substantial amounts (more than 30%) and being sold to consumers labeled as solely containing Δ8-THC (Personal communication; Sams R, February 14, 2022). As emphasized earlier, critical to the quality control and safety issue of these products is the lack of standardization of quality assurance or analytical methods being performed by accredited third-party testing laboratories.
A principal issue with the ACRCC-Δ8-THC products is the lack of relevant human toxicological data to the numerous isomeric byproducts and degradants found in commercial products today. Although extrapolation of animal data may be misleading, it is an important first step in understanding potential human toxicity. The toxicology data for Δ8-THC date from 1978 with a relative flurry of studies done in the early 1970's. Perhaps the most interesting finding was the lethal toxicity seen with both Δ8-THC and Δ9-THC in rats, dogs, and monkeys, between 225 and 3600 mg/kg. 58 Available data suggest that the bioactivity of Δ8-THC is similar to that of Δ9-THC, including euphoria, paranoia, dry mouth, reddened eyes, dizziness, blurred vision, relaxation, and small increases in heart rate. 59
Adverse Effects of ACRCC-Δ8-THC Products
Ultimately, consumer safety should drive the need for appropriate quality control and regulation of all products whether they are derived from cannabis or industrial hemp. Notably, health concerns regarding the use of ACRCC-Δ8-THC products have not been infrequent. From December 2020 through July 2021, the FDA received adverse event reports from both consumers and law enforcement agencies describing 22 patients who consumed ACRCC-Δ8-THC products; of these, 14 presented to a hospital or emergency room for treatment following its ingestion. 60 Adverse events included vomiting, hallucinations, trouble standing, respiratory distress, and loss of consciousness.
In addition, poison control centers across the United States reported 660 exposure cases of ACRCC-Δ8-THC products between January 1, 2021, and July 31, 2021. Of these cases, 41% involved unintentional exposure to Δ8-THC with 77% of those affected being pediatric patients <18 years of age. Eighteen percent of these patients required hospitalization, including several children who required intensive care unit admission following exposure to these products, and one required emergency intubation and ventilatory support. 61
On September 14, 2021, the FDA stated that there could be serious health risks to humans who use ACRCC-Δ8-THC products and CDC issued a health advisory to health care professionals and the public of the
increased availability of cannabis products containing delta-8 tetrahydrocannabinol (THC) and the potential for adverse events due to insufficient labeling of products containing THC and cannabidiol (CBD). 62
Although the CDC health advisory was welcomed, there is concern regarding the FDA's lack of enforcement to labeling requirements of ACRCC-Δ8-THC products from its manufacturers. If the FDA can require both tobacco and alcohol manufacturers to accurately label their products, warning consumers of health risks they accept by consuming their products, surely the FDA should mandate that all product manufacturers do the same. At the very least, consumers should be aware of the potential health risks associated with the use of all ACRCC-Δ8-THC-containing products.
Currently, a plethora of ACRCC-Δ8-THC products are being produced without any regulatory oversight, process standardization, product specification, or standardized third-party testing requirement. Without industry-wide regulation and governmental oversight, these companies may be, or may not be, following strict pharmaceutical standards for quality control in this manufacturing process.
Our apprehension centers around the possibility that ACRCC-Δ8-THC products may potentially parallel the classic example of a designer drug disaster: the story of MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine)
The case of the “frozen addicts” represents the potential for serious long-term and unintended health consequences when the steps of a chemical reaction are not strictly followed, resulting in the formation of unforeseen (and unquantifiable) byproducts. The MPTP story clearly demonstrates that even very minor differences in chemical structure may produce radically different pharmacological and toxicological effects in humans. The lesson from MPTP highlights the importance of caution when considering the use of a synthetically produced drug and the potential catastrophic consequences that may occur when drugs are manufactured using nonpharmaceutical quality control standards. Currently, the nonpharmaceutical production and widespread consumer use of cannabinoids like ACRCC-Δ8-THC exist and should prompt concern among regulators, medical professionals, and consumers alike.
Fortunately, there has been no report in the literature of “frozen vapers” from consumption of products containing ACRCC-Δ8-THC, but the emergence of EVALI aligns exactly with the appearance of ACRCC-Δ8-THC. The lesson of the MPTP “frozen addicts” should be that we do not know what could seriously hurt us. There is compelling data to show that these products being sold contain various reaction derivatives formed in the production process, resulting in inaccurate product quality testing and labeling. Introduction of these products into the cannabis consumer marketplace has been done without any thoughtful and empirically driven discussion regarding the issues and possible dangers of these designer cannabinoids. We have no understanding of their potential clinical impact for both the casual and chronic consumer.
We assert that the public deserves a consistent and accurate cannabinoid testing program for all cannabis-based products being sold, within both the recreational and medical marketplaces. These products need to be produced reproducibly with a focus on quality assurance much like that required of over the counter and pharmaceutical ingredients. In addition, these products need to be consistently tested by certified laboratories using standardized methods and accurately labeled, just like other consumer products.
Cannabis consumers are witnessing a large-scale human experiment with the introduction of these synthetically produced ACRCC-Δ8-THC products. Ultimately, the issues surrounding these products are simply another chapter in the continuing saga of cannabis prohibition, unregulated capitalism, and the political and racially motivated intentions of the CSA.
Instead, we envision a futuristic and enlightened Congress and FDA to implement a well-regulated cannabis industry (both medical and recreational) in which consumer safety and product reliability are paramount over profits.
Footnotes
Acknowledgment
The authors would like to express their sincere appreciation and gratitude to Richard A. Sams, PhD, Scientific Director at KCA Laboratories in Nicholasville, KY, whose contributions, support, and editorial direction made this article possible.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
No funding was received for this article.
