Abstract
Introduction
: This article presents comprehensive safety guidelines and risk management strategies for conducting research involving bats in laboratory environments, building upon previously established field safety recommendations. With the increasing use of bats in biomedical and ecological research, proper handling, housing, biosafety, and biosecurity measures are critical for protecting researchers, the surrounding community, and animal welfare.
Methods
: This document discusses essential topics such as institutional review processes, medical surveillance, regulatory compliance, and the transportation of live bats and their biological materials. It also includes a discussion of pathogens of concern that may be found in bats or bat samples, from rabies and other lyssaviruses to filoviruses, henipaviruses, and others. It emphasizes the need for robust risk assessments tailored to various laboratory settings, highlighting procedures for handling potentially infectious tissues and fluids and managing bat housing in vivarium facilities. In addition, it addresses considerations for quarantine, environmental controls, and biosafety and biosecurity protocols that are critical for protecting researchers and the community, specifically the use of personal protective equipment and training requirements for laboratory personnel.
Discussion
: By detailing these safety practices using a holistic approach from a broad background of expertise, this article aims to equip researchers and biosafety professionals with the tools necessary to establish a safe and ethical framework for bat-related studies, facilitating responsible and effective research while minimizing zoonotic risks and supporting conservation efforts.
Introduction
As studies in medicine, biology, and beyond continue to explore new areas of science, the use of in vivo and in vitro model systems is increasing. While many model systems employ typical species such as rodents, increasingly researchers are using a wider variety of species, including bats. For example, the chiropteran resource facility in development at Colorado State University will serve as a breeding facility to raise and care for bats of various species that can be used as research models in studies on a wide range of human viruses. 1 Bats fulfill important ecological niches and are also an important economic resource. Studies with these creatures can provide important information on ecosystem health, physiology, population dynamics, and zoonotic diseases, to name a few. Safe work practice considerations for working with bats in the wild have been previously described, 2 but work with bats in captivity or with their tissues in a laboratory setting also requires appropriate experience, skills, and safety practices to protect the health and welfare of the animals, as well as the people that interact with them. As bats are known to carry a wide range of zoonotic microorganisms, many with the potential to cause lethal human disease and limited or no current therapies, working safely with bats and bat materials is of utmost importance to protect researchers and their communities.
In our previous publication, we outlined the biosafety considerations and safe work practices for field work with bats. Many of the recommendations, such as preexposure rabies vaccination and personal protective equipment (PPE), remain applicable to working with live bats and their tissues in a controlled research laboratory environment. In this article, we review housing and handling live bats in a vivarium, as well as the handling of bat tissues in a research laboratory setting. Work with bats in captivity or with their tissues and bodily fluids can be found in academic, industry, and government settings, as well as in wildlife settings such as zoos and rehabilitation establishments. Personnel handling the live bats and/or their tissues can come from a variety of backgrounds such as biomedical research and veterinary fields. Here, we recommend baseline practices and procedures to address such considerations. However, it should be noted that many of the proposed recommendations apply to settings in the United States. Other national settings may have different restrictions and requirements that are not able to be addressed in this article. For settings outside of the United States, researchers should investigate and adhere to all local, state, and Federal regulations and should also liaise with their respective institutional environmental health and safety representatives.
In addition to practices and procedures, this article discusses the transportation of materials and animals from the field to the lab, risk assessment for controlled research environments, medical surveillance, emergency response, and communication with stakeholders. In total, the goal of the recommendations provided here is to build upon the guidance issued in the first publication, and in combination with local, regional, national, and international requirements, provide information on establishing a safe working environment for researchers without compromising project goals.
Administrative Preparations
Permits
Working with captive bats and bat tissues requires extensive review and approval by permitting authorities. Permits and memorandums of understanding may be required by agencies, including city, county, state, tribal, and Federal entities, that own or manage land where bats will be captured. If applicable, personnel working with bats should also seek permission from private property owners, either informally (i.e., verbally) or through access agreements and waivers that protect property owners from liability. Wilderness permits may be required if bats are to be captured in remote areas, such as on wilderness lands managed by the United States Forest Service. 3 If crossing international borders with bats or their tissues, researchers should understand and comply with laws and regulations on either side of a border. It is also important for personnel working with bats to stay informed on changing regulations and recommendations. For example, during the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic, multiple domestic and international agencies issued recommendations limiting activities that could facilitate the spread of SARS-CoV-2 between humans and wildlife. 4
Regulations vary based on species, origin, and even tissue type, and will dictate which permits are needed. For example, specific United States Fish and Wildlife Service (USFWS) permits and specimen letters are required to capture, study, and possess threatened and endangered species, tissues, and carcasses. 5 Collection and transport of tissues and other biomaterials (e.g., DNA, RNA, blood, cell cultures, urine, feces) both internationally and domestically may require permissions issued by the Centers for Disease Control and Prevention (CDC), USFWS, International Air Transport Association, Animal and Plant Health Inspection Service (APHIS), or from international agencies (e.g., The Convention on International Trade in Endangered Species of Wild Fauna and Flora). Researchers should be prepared to describe all possible etiologic agents in the species and tissue types they are transporting, as well as any measures they have undertaken to neutralize these potential pathogens.
The use of bats in captivity also necessitates oversight by state and Federal agencies, for example, state wildlife agencies and veterinarian’s offices, United States Department of Agriculture (USDA), CDC, United States Border Patrol, and USFWS. Vaccine trials and the use of etiologic agents known to infect bats or other taxa are approved and monitored by Federal agencies, including the CDC, USDA–APHIS, and the United States Food and Drug Administration (FDA).
During the permit application review process, regulators may critique the need for a particular research project, investigate whether activities overlap with previous work, monitor skills and training of personnel, and review overall study design. The number of bats in a proposed study may be scrutinized, and applications should require power analyses to justify the chosen number. This ensures that a sufficient sample size is collected that generates meaningful results while minimizing negative impacts on wild bat populations and stress on bats in captivity. Depending on the agency, potential impacts on other natural systems and local human populations will be assessed. Agencies may also have specific reporting requirements, which researchers should review before submitting an application. For example, APHIS requires institutions to report the number of bats and which species are used (i.e., captured, sampled, housed) per calendar year. State agencies tend to require more specific information, such as numbers of females and males captured per species and the exact coordinates where bats were captured.
Not every permitting agency reviews applications for safety concerns, and thus, internal institutional committees, programs, and departments (e.g., Institutional Animal Care and Use Committee [IACUC], Institutional Biosafety Committee [IBC], Occupational Health and Safety Program, Environmental Health and Safety) complete critical reviews of proposed activities for biological and other safety concerns. These entities also perform risk assessments and assist in generating Standard Operating Procedures (SOPs) and detailed instructions related to safety that are clearly written and available to all personnel working with bats. In addition, institutional import/export offices may require Material Transfer Agreements for transport or sharing, domestically or internationally, of samples of animal origin. Similar to permitting agencies, internal oversight groups will require information specific to their review focus. For example, renewing or closing IACUC protocols requires reports stating the numbers and species of bats used, if incidental captures occurred, and whether or not any unintended adverse events (e.g., mortalities) occurred. IACUCs may also require statements on whether research goals were met.
Across external and internal entities, each level of oversight provides important opportunities to review activities for safety and scientific rigor. Ultimately, these review processes also reduce the risk of exposure to pathogens for humans and their wildlife subjects, reduce impacts on local environments and human populations, and ensure the humane and ethical use of research animals such as bats. As institutional committees often include external and community members, review processes also provide opportunities for transparency with the public about research activities involving bats.
Institutional Review and Approval Considerations
In the United States, an IACUC must approve any research or teaching activities with vertebrate animals. 6 Similar animal welfare frameworks are found in many other countries, and scientific ethics demand that animal welfare be considered regardless of legal requirements to do so. Therefore, some animal welfare review and approval process is likely necessary to work with bats in captivity for teaching or research purposes. In addition, if animal tissues or cells are being brought into a vivarium facility, the attending veterinarian and the vivarium manager should be made aware of this fact prior to the material entering the facility to help mitigate any infectious disease risks associated with those tissues. Minimally, a tissue transfer form may be required to alert the animal care program that an animal product is being received and help the institution ensure that the material is legitimately and legally sourced. For more details about performing a risk assessment for housing bats in a vivarium, see the section on captive husbandry in “Husbandry Considerations.”
Before approval of any work with captive bats, the IACUC or other animal welfare oversight entity must carefully consider the welfare of the animals throughout the process of capture, transfer, acclimation, and quarantine, and the final experimental, breeding, or teaching activities. In the United States, bats are regulated by the USDA under the Animal Welfare Act, 7 and thus, these activities must comply with guidance within the Animal Welfare Regulations. 7 The American Society of Mammologists also regularly publishes guidelines for the humane use of mammals in research, 8 and these are considered primary guidelines for wild mammals by the Public Health Service Office of Laboratory Animal Welfare 9 and AAALAC International, an independent animal welfare accrediting organization. 10 The IACUC may also want to carefully review the husbandry plans for bat species kept in captivity, to reduce the likelihood of any unexpected morbidity or mortality related to improper husbandry. Last, the IACUC must recognize the limitations of the individual knowledge of its members and seek outside experts when appropriate.6,7,9 The semiannual IACUC program review and facility inspections provide an opportunity to review capabilities of the program and facilities and help determine if research with captive, live bats is feasible. Given that bats are an incredibly large and diverse group of mammals, 11 consulting with researchers, wildlife biologists, wildlife rehabilitators, or veterinarians who have worked with the specific species of interest is highly recommended.
In addition, requirements for IBC registration, review, and approval will depend on the type of work being conducted and the institution’s policies. Use of recombinant or synthetic nucleic acids requires IBC review at institutions that receive funding from the United States National Institutes of Health for recombinant or synthetic nucleic acid work 12 or are covered by a local ordinance. Some IBCs will require registration for work with animals or animal tissues that pose zoonotic disease concerns. Even if IBC oversight is not required, it is important to ensure that the risk posed by these samples is properly addressed through a risk assessment to determine the proper biosafety practices and PPE, occupational medicine such as rabies vaccines and titers, as well as administrative controls. Therefore, registration with the IBC is strongly recommended to assist researchers in evaluating these parameters. Typically, the process of completing the IBC paperwork will prompt researchers to consider infectious diseases, PPE, disinfectants, etc. IBCs are also composed of experts in diverse disciplines; they can aid the researcher in identifying hazards that they may not have considered. At a minimum, researchers are encouraged to contact their local Biosafety Professional to review their research plan. Alternatively, many IACUCs include biosafety professionals as members of the committee or liaisons to perform a thorough review of the proposed research.
Establishing a process for cross-review by the IBC, Occupational Health and Safety (OHS), and IACUC should be prioritized for work with captive bats, if one does not already exist. Often, the specific forms used in these three different review processes collect different information that can be helpful in informing the other review groups. For example, a review of the IACUC protocol for a study of captive insectivorous bats may reveal to the IBC and OHS team that the hibernating bats will be collected from an abandoned lead mine, which could involve additional biosafety, physical safety, and chemical safety concerns that were not originally assessed for the captive bat work.
Ethics and Conservation
Bats are impacted by human activities and encroachment on their natural habitats. The International Union for Conservation of Nature (IUCN) uses its Red List of Threatened Species to communicate the conservation status of numerous animal species, including nearly 1,400 species of bats. 13 With regard to threatened bat species, over two dozen species are listed as Critically Endangered, nearly 100 as Endangered, and over 100 species as Vulnerable by the IUCN. Furthermore, IUCN has identified nine bat species which are known or presumed to be extinct. Thus, while research into pathogens for which bats routinely serve as reservoirs is vital for global public health, a balance must be struck to preserve a shrinking biodiversity. The work of the Bat Conservation Working Group within the IUCN represents one of many conservation efforts worldwide. This group puts forth yearly activities and advisories to protect bat populations globally, such as recommendations to prevent human-to-bat transmission of SARS-CoV-2, or communicating with the bat research and conservation community on safe practices for bat research. It would be unethical of the authors of this article to suggest the procedures and activities to safely perform field work with bats and bring materials back to the lab, but then fail to address conservation efforts to promote the safety of the animals themselves. Therefore, we encourage researchers and biosafety professionals responsible for assessing the risk to institutional personnel to expand that assessment to include risk to animal populations. In addition, local IACUCs promote ethical concerns of the research they review and commonly have members who specialize in ethics. Consultation with these experts can provide fresh insights on how to promote bat conservation. Alternative techniques, such as increasing tissue availability through mechanisms such as biobanking, may be effective not only at increasing conservation efforts put forth by conservation partners but also in reducing the risks undertaken by the research community when collecting samples in the field, accomplishing two parallel goals simultaneously. In the case of biobanking wild samples, researchers should be careful to ensure inclusion and credit for the communities where these samples originated to avoid bioprospecting.
Transportation of Bats
Responsible research planning should include transportation of bats and bat tissues in accordance with all transportation-related regulatory requirements. Various international and national regulations exist that apply specifically to transportation of infectious substances, including biological materials that may harbor infectious agents. In some instances, researchers will need to transport body parts or tissues to the laboratory for further analysis, but in other cases, the transportation of the live animal may be required.
According to the World Health Organization (WHO) Guidance on regulations for the Transport of Infectious Substances in its current version, 14 live animals (including genetically modified ones) which have been infected with a biological agent must be transported in accordance with the appropriate regulations in its country of origin, transit, and/or destination. Such regulations are usually associated with proper animal care, and as such, infectious substances’ transport regulations will not generally be applicable. 15 A live animal that has been intentionally infected and is known or suspected to contain an infectious substance may only be transported by air under terms and conditions approved by the appropriate national authority of the States of origin, transit, destination, and operator.14,15
In addition to consulting appropriate regulations, transport methods should be determined in coordination with veterinarians or the IACUC to consider the needs of the various species, the length of the journey, method of movement, the safety of those performing transport, and the health and welfare of the animals. One example can be found in Krutzsch and Sulkin from 1958, 16 that shares a good mode for transporting bats through collection with nets and transfer from the nets to metal rat cages, which were adapted for the transport of the animals by addition of water and temperature control. The authors direct the reader to the companion article published in this same issue of Applied Biosafety by Cardenas-Canales et al. for another case study on transporting bats from the field to the vivarium.
In Vitro Work
Risk Assessment
The risk assessment process is vital to planning and conducting safe work with bats and/or materials from animal sources (Figure 1). In our previous publication, we describe in detail the risk assessment process as applied to field research with bats. The reader is encouraged to review the article 2 for a comprehensive review of the risk assessment process. While the risk assessment process for in vivo and in vitro research is similar, there may be additional and/or unique aspects to consider.
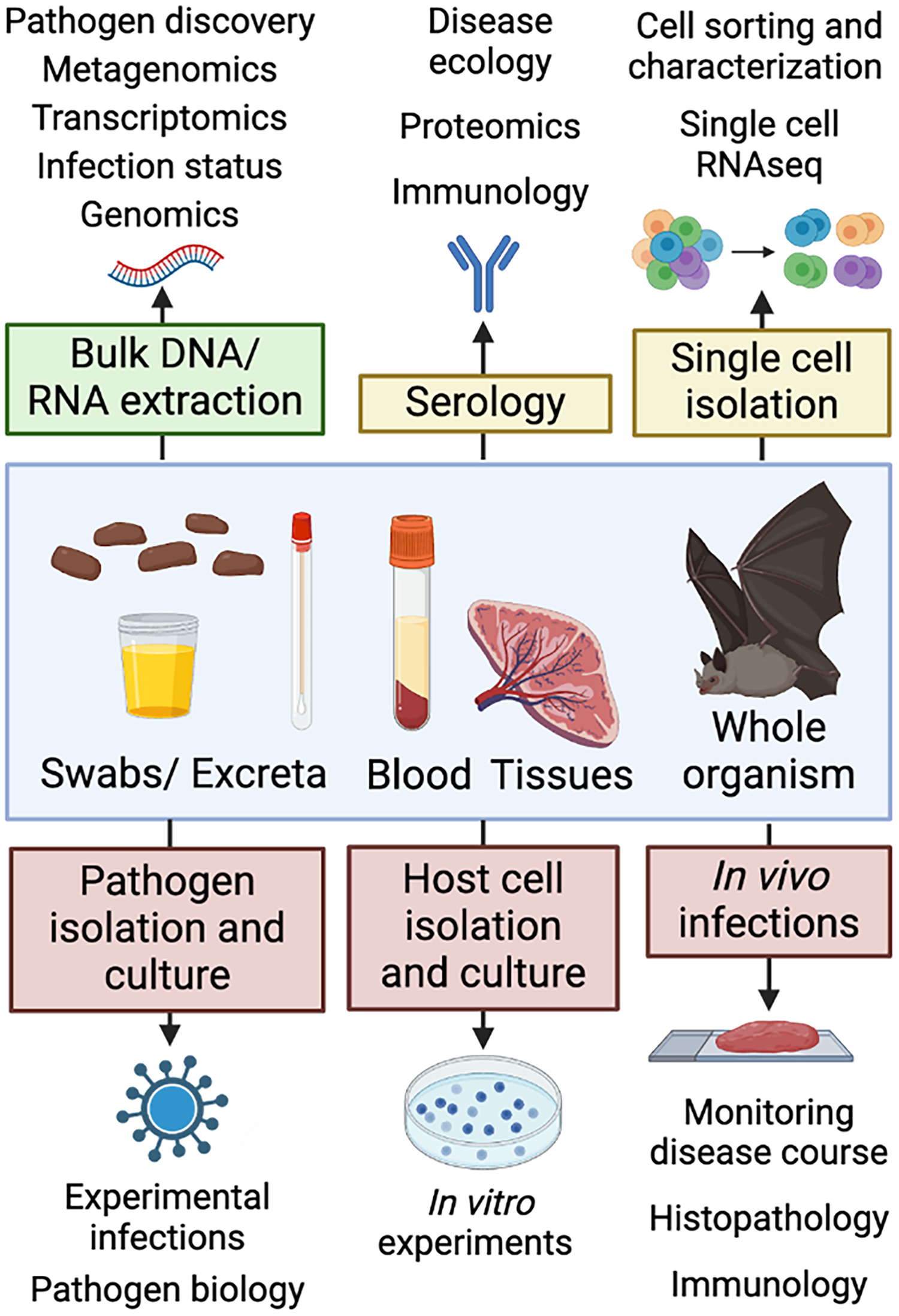
Laboratory-based studies may use a variety of samples and techniques with varying biosafety levels. Examples of studies conducted on captive bats are available in Supplementary Table S1. Some procedures that may be applied to these various samples are boxed with examples of the types of experiments and fields of inquiry that might use these techniques grouped at the end of the arrow. The color of the box indicates an approximate biosafety level. Green: Sample treatment (e.g., lysis) may allow experiments to proceed at a lower biocontainment level after risk assessment and with appropriate precautions. Yellow: Proteins cannot be denatured for serology, and cells must stay alive for single-cell isolation. These requirements often interfere with pathogen inactivation, so samples must be treated with caution and worked with at the appropriate BSL. Red: Isolation, culturing, and work with pathogens and host cells in vitro or in vivo pose the highest biosafety risk and depending on the source of the samples or agents may require BSLs as high as 3 or 4. Created in BioRender. Frank, H. (2023) BioRender.com/u21l262 . BSL, biosafety levels.
It is important to highlight that before the process starts, a thorough risk assessment must take place when working with biological samples (including the reception of animals that can be potentially contaminated with pathogens). In addition, there are important considerations that are covered in greater detail in the sections of this article and summarized in Table 1.
Considerations for the risk assessment necessary when bringing bats or bat samples back from the field to the laboratory
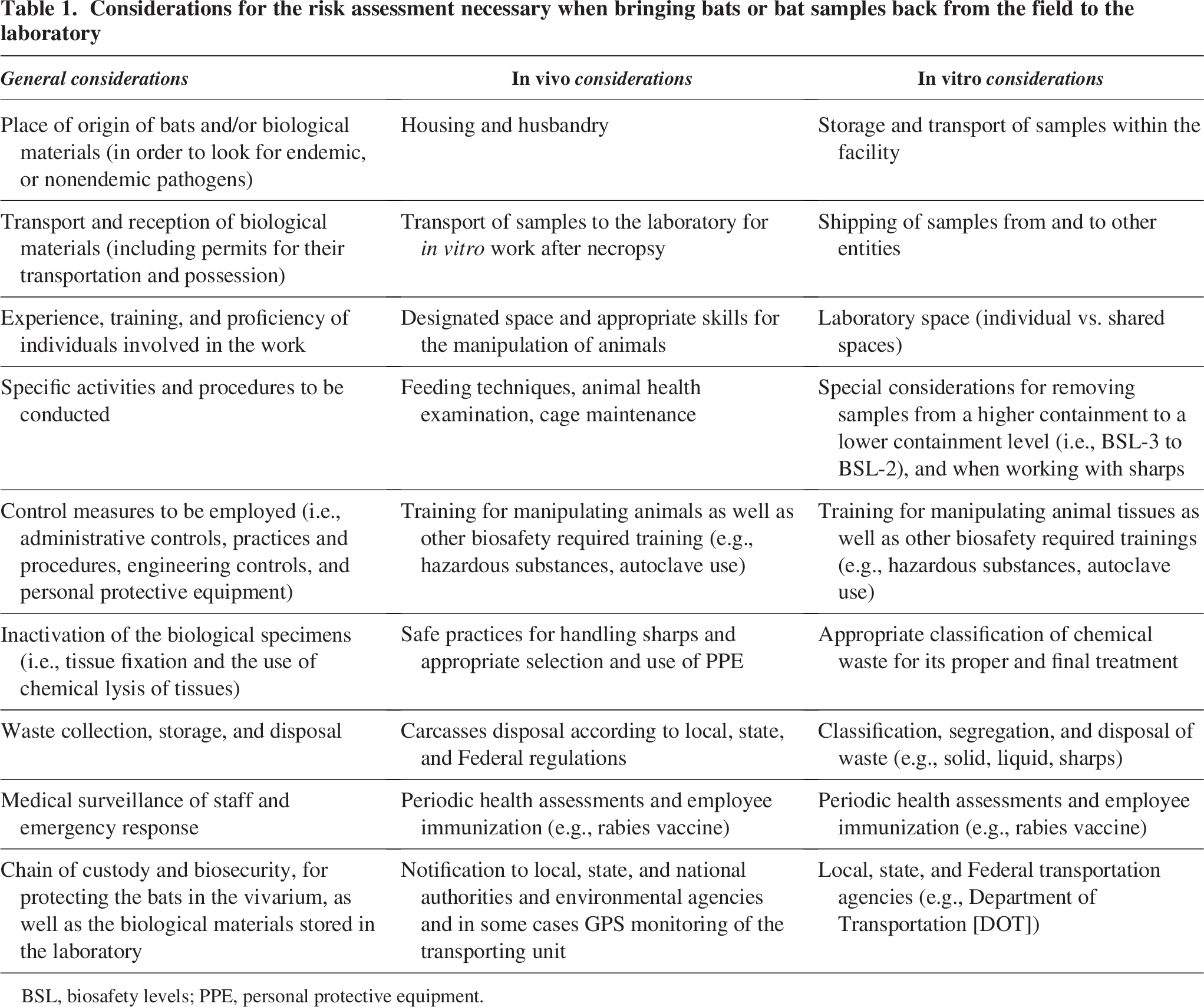
BSL, biosafety levels; PPE, personal protective equipment.
The risk assessment process depends on a team effort and requires a multidisciplinary approach. Prior to conducting the risk assessment, the reviewing team should be formed and include representatives from the research team (principal investigators and researchers), biosafety professionals (such as the Biosafety Officer), occupational health, and the vivarium and veterinary medicine staff for in vivo work. Additional members may be added based on the complexity of the project. For example, when tissue fixatives are proposed, the institution’s chemical safety officer should be involved in the review of the specific protocols for fixative chemicals. In addition, if core facility resources are to be used on unfixed tissues (e.g., fluorescent activated cell sorter), researchers must consult the facility staff to ensure the core has sufficient biosafety precautions and can be appropriately decontaminated prior to use by other research teams. All risk assessment discussions and decisions should be documented in writing and communicated to the appropriate individuals either in writing and/or as part of an in-person training session.
There are a variety of written resources that can help during the risk assessment process. These include a completed institutional form that describes the work to be performed, the locations where the work is to be performed, as well as other information as required by the biosafety team or oversight body such as an IBC. In addition, for in vivo work, a completed IACUC registration is essential. Cross-referencing both the IBC and IACUC registrations is important when conducting the risk assessment. In addition, SOPs and training materials should be developed and reviewed as part of the overall oversight and risk assessment process.
The risk assessment process is a continual activity. After conducting the initial risk assessment and prior to commencing activities, the process should be revisited any time there is a change or modification, including but not limited to changes in biological materials, SOPs, facilities, lab equipment, or staff. This should also include updating, reviewing, and approving any changes to the information provided to the IBC and IACUC. To ensure researcher safety and compliance in the dynamic research environment, IBCs and IACUCs should be available and prompt in their reviews and guidance. In addition to this risk assessment process, all the individuals involved in the activity will need to understand all the risks involved in the proposed work.
One of the most controversial and discussed issues in recent years has undoubtedly been the assignment or classification of microorganisms into risk groups and the assignment of biosafety levels (BSL) to laboratories. For many years, researchers handled biological agents following the guidelines established by different national or international agencies (e.g., CDC, WHO, WOAH [formerly Office International des Epizooties]), to carry out diagnostic, research, or reference activities according to the nature and characteristic of pathogens. However, the presence of emerging diseases in different regions of the world (i.e., COVID-19, Mpox, Ebola virus disease), the globalization phenomena, and the frequency of these epidemic outbreaks have required the readiness of institutions to be prepared and quickly respond. It is especially important to consider this during the risk assessment and evaluate both the known risk and possible anticipated risks for work with bats or their tissues.
Storage of Materials
Proper sample storage is a critical consideration both in terms of the quality of data returned by the sample and for the safety of those handling it. Typically, the nature of the sample and the downstream applications will determine storage requirements, but safety considerations should be built in at each stage of storage, including immediately following collection, during transport, and while maintaining within a lab. Given our limited understanding of bat viruses and their zoonotic potential, certain sample types should be handled with special care and personnel made aware of potential disease risk. In particular, samples containing blood, serum, guano, or organ tissues should be handled as though they are infectious. At the outset of any project involving collection of bat samples, researchers should be familiar with any relevant pathogens in the region of origin of the sample, including those that have potential to cause ecological damage (e.g., soil pathogens) or have potential to infect humans or other laboratory or domestic animals. Environmental samples or those taken from wild animals such as bats will often be considered a risk for infectious pathogens until proven otherwise or inactivated, and thus be handled at a minimum at BSL-2.
Improper storage may lead to degradation of the sample or may expose laboratory personnel to potential pathogens. For example, samples intended for RNA extraction may require specialized buffers or specific temperatures to remain viable, as RNA is highly unstable and degradation may yield spurious results during analysis. However, stabilizing buffers can also support viability for several infectious viruses.17,18 Certain viruses also remain viable under a variety of temperature conditions, including freezing, and different temperature regimes may support viral viability depending on storage duration. 19 Therefore, it is important not to assume that a sample is safe to handle without precautions if it has been frozen or preserved in buffer, unless that buffer specifically neutralizes pathogens (e.g., Zymo DNA/RNA shield).
Samples collected from bats for DNA applications are most often collected using nonlethal methods, such as small circular biopsies from the patagial or uropatagial membranes, the skin folds that connect the forelimbs and hindlimbs, or the hindlimbs, respectively. These can be stored in a variety of ways, including in ethanol, frozen, in nucleic acid stabilizing buffers, or on silica desiccant. Such samples can be easily stored long term in silica desiccant without loss of DNA yield and without the requirement of cold storage. Some fungal and bacterial pathogens can persist when stored on silica desiccant,20,21 and thus, it is important to use proper PPE, work practices, and engineering controls to mitigate the risk when processing samples.
Whole carcasses are often frozen upon collection, but frequent or multiple freeze thaws should be avoided as this can promote microbial growth, damage DNA, and compromise other tissues intended for pathology or toxicant assays. Upon collection, carcasses should be placed in at least two labeled, sealable plastic bags and frozen at a maximum of −20°C for long-term storage. Where possible, and depending on the intended use of the carcass, necropsy should be performed as close to death as possible, and each tissue of interest dissected, washed in distilled water or other appropriate buffer, cataloged, and stored frozen in well-labeled vials.
Labeled, leak-proof secondary containers should be utilized to transport samples from the vivarium to the laboratory, and from laboratory to laboratory within the institution. If samples include bone or other hard material, containers should also be puncture-resistant. If the transport route includes public roadways, the United States Department of Transportation requirements must be followed depending on the material and whether dry ice or liquid nitrogen is used in transport. However, if the collection and transportation will be done in another country, the appropriate consultations will be required with local, state, and/or national authorities.
Blood, serum, and other tissues should all be handled and stored according to current blood-borne pathogen SOPs consistent with the institution’s requirements. A variety of infectious pathogens have been isolated from bat blood, sera, or organs and are infectious to nonbat mammal species.22,23
In all cases, a sample storage database should be created and updated with the addition of each new sample and should contain basic safety information about the sample type. A well-organized storage system paired with a well-curated and frequently updated sample database is the best approach to keeping laboratory personnel and the public protected from any health threats originating from laboratory bat samples. The content of each database may vary between institutions; however, each database must contain the minimum information to allow traceability of each sample.
Use of Materials
Through the risk assessment process, the appropriate hierarchy of controls can be established. This leads to the selection of the appropriate biosafety or containment level. Ultimately, the IBC or safety committee approves the biosafety or containment level for the work. Given that bats can harbor a variety of disease agents, treating the material as potentially infectious is important. Working at BSL-2 at a minimum is good practice. If the risk assessment concludes that there are concerns that the material may contain one or more pathogens, additional controls (enhanced measures) may be needed. These enhancements may be as simple as confining the work to a small laboratory area and requiring all procedures to be conducted in a biosafety cabinet while wearing a disposable gown instead of a cotton lab coat.
When in vitro as well as in vivo work requires the use of sharps, the institution’s Biosafety Professional should be consulted. Whenever possible, sharps should be eliminated or replaced with safer versions such as self-sheathing needles/syringes. While there are a variety of commercially available sharps safety devices, some may not be compatible with the task. In that case, additional PPE such as needlestick-resistant glove liners should be utilized. Sharps injuries, as with all occupational injuries, need to be reported to the institution’s occupational health provider for immediate assessment.
Training is also a key consideration. All laboratory staff handling the bat samples must complete required animal and biosafety training, as well as demonstrate proficiency in the use of laboratory equipment and performance of the tasks. Other training, such as Ethics in Research, may also be required by the institution.
Disposal of Materials
Bat samples that are no longer required for research purposes should be disposed of in accordance with applicable local, state, and Federal biological waste disposal requirements as well as institutional policies. Appropriate waste containers should be provided for solid and sharps waste. Depending on applicable requirements, autoclaving materials prior to final disposal may be required. Freezing carcasses prior to removal from the institution by a waste vendor may be an option if odor is a concern. While small amounts of liquid associated with samples can likely be accommodated in the solid biological waste container, larger volumes of liquids should be collected in a leak-proof container for treatment with bleach or other compatible and efficacious disinfectant prior to sink disposal (where allowed). Autoclaving without chemical treatment is also an option prior to sink disposal.
In Vivo Work
In addition to bringing bat samples back to a laboratory, researchers may want to bring live, captive bats back for experiments. Depending on the location and species of bat, this may present challenges to biosafety professionals to prepare a suitable housing environment, train animal husbandry personnel, and notify occupational health and hazardous waste professionals, among others. In addition, members of the community may present strong objections to housing bats with potential pathogens. The following sections are provided to inform the biosafety community as well as institutional partners of best practices to maintain these colonies, both for the health and safety of the animals as well as the researchers and the community. While certain sections may not be commonly thought of as relating to biological safety, we encourage the community to examine how each may influence animal behavior or health and subsequently impact researchers.
Housing Bats
When studies require performing experiments with bats in the laboratory, there are several animal welfare and biosafety considerations to work through before the arrival of the bats. First, it is important to consider the natural ecology of the species of bat to recreate the natural environment, behaviors, and feeding habits as closely as possible. 24 Mimicking a bat’s natural environment can also reduce the animal’s stress level. This can make the bats easier to handle and reduce the risk of a scratch or bite, thereby reducing the likelihood of a potential zoonotic exposure. 24 The plan for sourcing food, maintaining temperature, humidity, lighting, and airflow, and the logistics for waste management and decontamination must be ready before the arrival of the bats. 8 Supplementary Table S1 provides a selection of captive studies with bats of different species and research objectives.
Housing Facility
One primary consideration is the housing facility. It is important to have a facility with adequate space to shelter the number of animals required. In the United States, the USDA Animal Welfare Regulations stipulate that bats must have space for flight. 6 This may be achieved by having cages that allow the animal room for flight, or by using the room as primary containment and allowing free flight. The housing room should always be secured by double door entry, 9 and it is recommended that the interior door be secured by a plastic mesh curtain to reduce the risk of accidental escape and potential biological release. Ensure the housing room does not have any crevices or spaces that bats can use to escape by, including mitigation strategies such as air vent covers. Mesh screens are helpful, but it should be verified that escape-proofing the room has not compromised any directional airflow to maintain negative pressure to surrounding areas if it is needed for biological containment. If housing bats in a free-flight room, methods to capture and inspect the bats at regular intervals should be designed to be safe for both the bat and the handler. Measures should be taken to exclude the bats from roosting or getting trapped in crevices in the room or around equipment. Retrieval of bats from these types of spaces can carry a risk of injury to the bat and the animal handler. Design of roosting spaces can include a way to safely enclose the bats, such that a person could walk into the room during daylight hours and simply close the daytime roost to contain them. 25 The duration of the study is also an important consideration when selecting and preparing the housing facilities, as the duration of the captive studies will demand different features and conditions to be maintained to provide adequate care to the captive bats.
As part of the risk assessment, the appropriate containment level for microorganisms should be determined as represented by an animal biosafety level (ABSL). Information on ABSL containment and microbiological practices, PPE, and other safety mechanisms, as well as assessments of certain pathogens can be found in the Biosafety in Microbiological and Biomedical Laboratories (BMBL) manual. 26 One low-risk-tolerance method is to determine the pathogen that requires the highest ABSL criteria that might possibly be found in that species and treat them as presumptive positive until quarantine and testing have ruled out that pathogen. For some pathogens, such as rabies virus (RABV), antemortem testing may not be able to adequately rule out infection. This type of risk analysis can include the prevalence of the pathogen in the area where the bats are captured, if known. In many cases, this information may not be available, and institutions must make determinations based on their risk tolerance, given the information available.
Temperature
Temperature must be adapted to the natural environment of the species and should be changed according to seasonality to maintain the physiological and biological features of the species as closely as possible, depending on the period of study. The temperature may also be used to manipulate the behavior and physiology of some bat species. For example, facultative and obligate hibernating bats may be placed into temporary torpor or hibernation simply by keeping them in cool temperatures. These temperatures can be maintained using florist coolers or environmental chambers.24,27 Individuals must have adequate fat stores to survive the planned period of hibernation, as they will not eat during torpor and should not eat during arousal periods of hibernation. 28 Conversely, if it is desired to keep hibernating species out of prolonged torpor, keeping the room temperature closer to the upper limit of their normal preferred temperature range will assist in this (but may not prevent recurrent daily torpor) (Falendysz, unpublished data). While some nonhibernating species are tolerant to high-temperature ranges and display thermoregulatory behaviors (e.g., moving and distancing from other bats), 29 some species have very narrow ranges of acceptable temperatures. For example, common vampire bats (Desmodus rotundus, a nonhibernating species) prefer 21–27°C. 30 In one report, they experienced mortality after prolonged exposure to temperatures between 2°C and 10°C and can only withstand short periods at 32–34°C. 31
Humidity
Environmental humidity should be adapted depending on the species, considering that humidity can directly affect wing membrane health32,33 and also likely affects skin microbiome. 34 Many species of bats require higher humidity. Most North American bat rehabilitation facilities recommend 60–90% humidity. 33 Cave-dependent bat species, such as little brown bats (Myotis lucifugus), develop skin disease if kept in too low humidity. 33 M. lucifugus kept at an inconsistent humidity ranging from 35% to 60% developed constrictive bands of skin on the first digit of the wing, leading to ischemic necrosis of the digit and mobility issues (Figure 2) (Falendysz, unpublished data). Likewise, the American Zoos and Aquaria Association recommends 60–90% humidity for fruit bats 34 and 40–60% for vampire bats. 30 Artibeus spp. can tolerate 40–60% humidity. Many facilities may be unable to tightly maintain humidity using their standard air handling systems. In addition, frequent air changes in a laboratory setting may work against maintaining higher humidity. Humidity chambers may be constructed as described by Lollar 33 or plastic greenhouses may be used to maintain the microenvironment at a higher humidity than the housing room. 24 Within these spaces, household warm air humidifiers or misting systems designed for amphibians and reptiles can be used to maintain humidity. Misting systems controlled by hygrometers provide consistent and tightly controlled humidity ranges. It should be noted that high humidity can cool and freeze in high-efficiency particulate air filters on the laboratory exhaust in containment facilities if the filters are not in temperature-controlled spaces (Falendysz, unpublished data). This can affect the air changes per hour and can potentially damage the filter, hence the preference for using humidity chambers for maintaining a humid microenvironment in this situation. Hibernating bats are often kept in specialized environmental chambers that simulate the low temperatures and high humidity of hibernacula.27,35
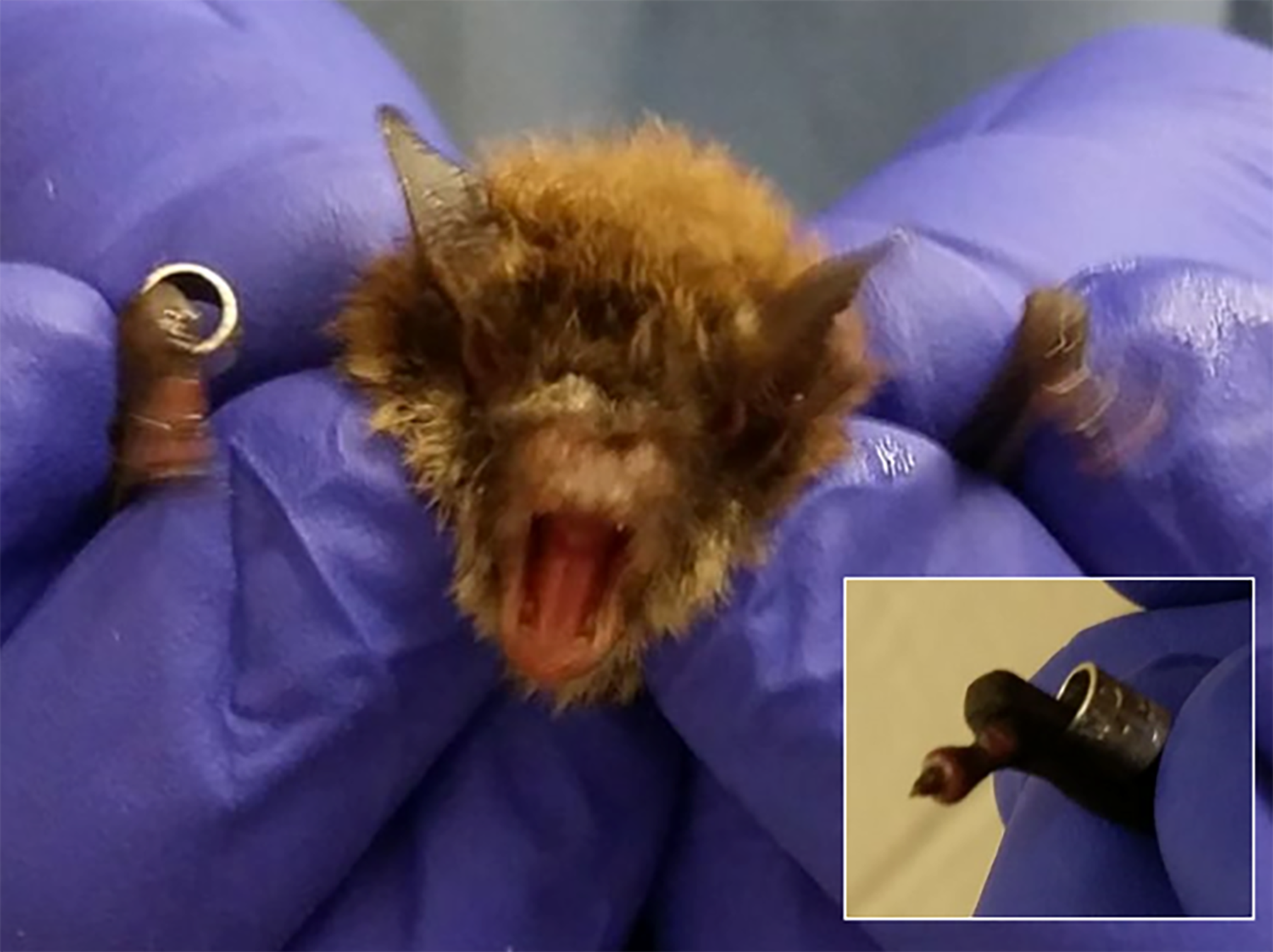
An example of an Myotis lucifugus bat that experienced ischemic necrosis of the digit (inset) and subsequent mobility issues due to inconsistent humidity conditions during housing. Photos courtesy of E. Falendysz.
Within humidity chambers used for euthermic bats, it is important to consider airflow for the frequent removal of feces and urine from the cages to reduce ammonia while maintaining sufficient humidity. 36 The dietary intake of the bats has a large influence on ammonia output. 37 Last, it is important to remember that environmental humidity above 75% leads to higher growth of bacteria and fungi, 38 and thus frequent disinfection of the environment and humidifiers is needed to prevent infections in both the bats and the animal care staff and to maintain the scientific integrity of the experiment.
Caging
If the investigators will design and custom-build cages, a thoughtful choice of caging materials can optimize safety and comfort for humans and the research subjects. Metal wire mesh has a higher potential to damage the bat’s skin and nails of the feet and the first digit of the wing, which is used for climbing. However, the advantage of these materials is that they are easy to clean and can be autoclaved or sanitized using a high-temperature cage washer. If using metal mesh or other materials for caging that have the potential to have sharp edges, care should be taken to ensure that all sharp edges are covered to protect the bats and personnel. Fabric mesh cages offer more comfort for the bats when climbing but require more manual labor to clean them. Frames for fabric mesh cages can be made from PVC pipes, which are lightweight and can withstand moderately hot temperatures in a cage washer but will melt at 180°C cage washer temperatures required by the BMBL at ABSL-1. 26 Therefore, chemical disinfection will likely be required for this type of caging. In addition to animal comfort, cages may need to be designed and adapted to the type of study that is being conducted. For example, behavioral or infection studies requiring constant direct observations of bats will necessitate cages made of materials that allow such observations.39–41 The use of clear materials such as plexiglass offers a smooth cleanable surface, and when used as at least one side of a roosting box, it allows bats to be visualized inside their roost. Again, if plexiglass is used, it should be free of sharp edges. Last, cage design should also consider the safety of the handlers to retrieve the bats for routine procedures, the accessibility to capture and manipulate the bats without potentially injuring them while being captured, and the ability to prevent escape.
Enrichment
The natural behavior of bats requires appropriate locations for resting or roosting. All species will require daytime roosting areas for sleeping. Some species will roost in open spaces, along the ceiling or walls of a cage, 42 while other species such as M. lucifugus or Eptesicus fuscus 33 have strong preferences for crevices to roost in. For the safety and well-being of the bats, it is important to give crevice-dwelling bats an appropriately tight area to roost in, or they may choose to rest in undesirable crevices, such as under floor mats or cage door frames. Metal, plastic, or wooden bat boxes may provide appropriate roosting areas. Surgical towels hung along the surfaces of a cage can provide a mobile, soft crevice, and these towels are autoclavable and do not have fabric loops that can catch the delicate nails of the bats. Wood is a readily available and cheap material that is easy to work with, but roosting areas made of wood are hard to keep clean and disinfect and therefore are not recommended.
If using metal or plastic materials for making roosts, nonstick drawer liners can be used to make the surfaces climbable for the bats, and these liners can be removed and cleaned as needed. Additional climbing and enrichment materials could include leaves, sticks, and other natural materials collected from the environment. To avoid bringing insects or microbes into the laboratory environment, these can be autoclaved and dried and then autoclaved after use.
Husbandry Considerations
Bats produce a large amount of feces for their size, and sometimes, its consistency makes cleaning cages a difficult task (e.g., vampire bat guano). 43 Large quantities of water will likely be required to clean cages, walls, and floors. Due to the potential risk of exposure to pathogens, splash protection such as safety goggles and face shields is recommended during cleaning procedures to protect mucous membranes. Respiratory protection may also be advised, due to the risk of aerosolization of pathogens during cleaning. Frugivorous bats are quite messy when eating, spreading partially eaten food and guano on nearly all surfaces of their enclosure. Walls can be covered by a plastic, washable material that will help eliminate the guano from the wall. To facilitate the cleaning procedures, the use of absorbent paper, newspaper, sawdust, or other bedding material is recommended for all bats. It is important to consider that rooms dedicated to activities such as cleaning, food storage, and food preparation should be separate from the main housing room to avoid introducing potential contamination of materials, samples, and exposure to staff and ultimately compromising the execution of the studies.
Dietary Requirements
Insectivorous bats most often eat live insects. Many insects can be bred in captivity and are available for purchase, or colonies can be maintained in the vivarium. It is generally recommended to supplement vitamins and minerals in these insects by gut-loading them with multivitamins and calcium carbonate.25,33 Consider how to contain live insects and prevent escape into other areas of the vivarium. One method is to feed only larval forms and prevent larval forms from pupating and becoming volant. For the commonly fed mealworm, Tenebrio molitor, careful monitoring and removal of pupating worms are generally sufficient to prevent flying adult beetles from escaping the room. The integration of live insects into the vivarium should be coordinated with the facility’s pest control program, to ensure that pest control activities do not kill dietary insects or contaminate them with pesticides before they are fed to bats. Insects may also be captured from the environment to feed to bats, though it is important to remember that these insects can be vectors of diseases and intermediate hosts for parasites. Also, they should not be collected from areas in which pesticides have recently been applied. 25 For frugivorous bats, the diet can be based on tropical fruits like guava, mango, orange, papaya, and sapodilla. The inclusion of fruits native to the local climate of the vivarium can also help reduce the high costs of their diet. As an additional supplement, a bird multivitamin formula such as Vitafort-A® (Parfar, Mexico) can be used. Importantly, many bat species, unlike most vertebrates, are unable to synthesize vitamin C, 42 a nutrient missing in some bird supplements, so careful attention is needed. For insectivorous and frugivorous bats, water must be always available. Obligate blood-feeding bats (D. rotundus is the most studied) can be kept in captivity successfully by feeding fresh blood.25,44 Bovine blood is generally used for feeding common vampire bats, and the sources to acquire it (e.g., abattoirs) are regularly available. However, swine or goat blood may be offered too. Blood preparation can be done by mechanical methods (i.e., blood is whisked until clots form and these are removed) or by the addition of sodium citrate that prevents coagulation. Batches can be aliquoted and kept frozen for later use. 25 Each bat can consume up to 25 mL daily,43,45,46 and feeding should be scheduled in the evening. Some vampire bats can drink the blood at room temperature or even cold. If loss of appetite is observed, prewarming the blood prior to feeding it to the bats may help. Leftover blood must be removed in the morning, to avoid bacterial growth. D. rotundus does not display a strong aversion to food sources and may be at risk of infection if blood is contaminated. Additional water is not always provided to vampire bats, as their daily water requirement can be met through their completely liquid diet. A general recommendation for all bat species is to offer their meals in several dishes or containers and evenly distribute them in the cages to avoid competition within the group. Many different feeders and dishes can be used and placed on the floor or hung along the walls, which would encourage movement as well. 25
Outdoor Housing
Some studies, especially those that do not involve a biological agent or surgical interventions, or those that aim to study environmental factors on bats, may allow for animal housing in outdoor enclosures. These types of housing set-ups can be ideal when the bat is native to the area, as they do not require as much environmental manipulation. One potential concern is that the bats could have continual exposure to other animals, insects, and pathogens, or be at risk of predation. 2 When housing bats outdoors, adequate separation of the imported animals from the native wildlife and humans should be enforced, to ensure they do not introduce human or animal pathogens to a new geographic location. Quarantine procedures may be similar to indoor housing. Depending on risk assessment, the initial quarantine period may be best conducted indoors to avoid contact with other wildlife, which risks introducing pathogens to wildlife or exposing the quarantined animals to new, locally occurring pathogens. 47
Exposure to vector-borne diseases is of particular concern in outdoor housing, or, conversely, imported animals could introduce or spread parasites, viruses, or bacteria to invertebrate vectors such as mosquitos, kissing bugs, ticks, or midges. For example, Trypanosoma cruzi, the causative agent of Chagas disease, has been well-documented worldwide in bat species. 48 Recently, DNA of T. cruzi was detected in D. rotundus in Chile, which is a first-time report with an unknown origin of infection. The role of bats in the transmission cycle of T. cruzi needs further study. Nonetheless, the overlap and contact between bat species that can serve as carriers and insect vectors highlights their importance in the maintenance or dispersion of T. cruzi to other areas. Another consideration is that outdoor-housed bats could become infected with novel pathogens through contact with local vector species. For example, Babesia canis DNA (a tick-borne parasite of domestic dogs) was found in the feces of bats from Hungary. 49 The absence of the tick vector’s DNA suggested an infection from the bats or through the consumption of other (unknown) insects carrying the pathogen. 50 Therefore, caution should be employed when considering housing bats outdoors at the expense of potential contact with free-ranging animals or insects. Routine ectoparasite treatments, as discussed in the “Quarantine” section, may be recommended for bats housed outdoors, to reduce some of the risks associated with exposure to ectoparasites and vector-borne diseases.
Pest Control
Control of vertebrate and invertebrate pests can have negative consequences for captive bats when not given adequate consideration of the risks. Invertebrates exposed to insecticides used for pest control or crop protection can be hazardous to insectivorous bats. If bats are housed outdoors, pesticides and insecticides used in the surrounding areas could become harmful if insectivorous bats consume them. Toxic pesticides such as organochlorines and polychlorinated biphenyls were found in wild bats from Germany, despite prohibitions on their use for decades prior. 51 If insect pests can enter the facility where captive bats are housed indoors, they can bring these contaminants with them. Rodenticides also pose a risk to bats, and bat mortalities from diphacinone poisoning have been reported in connection with rodent control activities in the area. 52 de La Brooke et al. 53 and Williams et al. 54 found that there was a potential for rodenticides to be ingested by insectivores through pesticide residues in insects and mollusks.
Quarantine
Bringing wild-caught bats into captivity requires careful consideration of the potential for zoonotic and other animal pathogens that they may carry. Bats are now recognized as common carriers of viruses, especially RNA viruses, which is possibly a side effect of their adaptations to metabolically demanding flight. 55 Bats can also be infected with bacteria, ectoparasites, and fungi. 56 Quarantine is an important measure to protect staff and other animals in the vivarium, and to ensure that the bats held in captivity are healthy enough to participate in their planned activities. The species of bat and their location of origin are important factors in determining the risks of various pathogens, or even nonpathogenic symbionts, that could be carried by bats. A commonly utilized quarantine period for bats is 30 days. During this time, molecular detection and serological testing for infectious agents may be warranted.
Bats from the Americas should be considered potentially infected with RABV, while nonrabies lyssaviruses are found in many other parts of the world. 57 Thirty days of quarantine is generally recommended to preclude the risk of rabies transmission among captive groups, based on the premise that infectious diseases, such as rabies, are most likely to become clinical during this period. 58 However, there is no known quarantine period that can rule out the risk of RABV in bats that originate from endemic regions. Bats infected with RABV can present incubation periods that vary greatly, even after experimental infection.41,59–61 Bats have been documented to have outbreaks of clinical rabies 4 or more months after entering captivity.59,62,63 In at least one of these occurrences, rabies was not known to occur in the local bat population. 63 Furthermore, there is no sensitive, specific antemortem test for rabies in bats, as positive serology does not always indicate active infection.63,64 Some bats will remain seronegative until after clinical signs and shedding begin, 41 and antibodies in surviving bats may wane to below detectable limits over time.59,60 Under these circumstances, careful record-keeping is encouraged to allow for tracking dates of the addition of bats and cage allocations into the groups to estimate possible exposures and isolate individuals to prevent further spread of infection. To date, there are no rabies vaccines labeled for use in bats. Nonetheless, vaccination of bats using commercial rabies vaccines for domestic animals may be utilized as a measure of protection against infection. In many countries, the use of these vaccines off-label would require the oversight of a licensed veterinarian, with the caveat that data are lacking on the effectiveness of such vaccines in different bat species. Experimental studies have reported positive results regarding the advancement of rabies vaccines for bat use. Some include the use of commercial, inactivated rabies vaccines in Tadarida brasiliensis 65 and Rousettus aegyptiacus, 66 showing that vaccination elicits a humoral immune response. Others have focused on recombinant vaccines, demonstrating the development of a humoral response and protection against rabies challenge in different species (e.g., D. rotundus,45,67 E. fuscus61,68) or the potential for reducing the risk of transmission via shedding. 41 Still, there is a need to continue conducting studies that validate the effectiveness of bat-focused rabies vaccines in various bat species for this practice to be widely implemented in captive colonies.
Flaviviruses, including Venezuelan Equine Encephalitis, are another viral group of concern in some bats from South and Central America.69,70 Bat species of the Old World have also been identified as important reservoir species for many other viral pathogens. High-consequence human pathogens such as Ebola virus, Nipah virus, Hendra virus, and coronaviruses have been described to occur naturally in species of bats in these regions.56,69 In many cases, these viruses are not pathogenic to their bat hosts, so combinations of serology and polymerase chain reaction (PCR) are recommended during quarantine for detection. Until the presence of these pathogens has been ruled out, adequate PPE and biosafety precautions are recommended, especially as many of these pathogens may be transmitted via aerosols. 26 Coronaviruses are found widely among bats, and bats have been the source of several emerging coronaviral diseases in humans. 71 Bat species vary in their ability to become infected with and shed SARS-CoV-2.72,73 Nonetheless, it is recommended that individuals wear PPE, including nonvalved N95 respirators or facemasks when handling bats, to avoid the potential of transmission of SARS-CoV-2 virus to bats.74,75
Likewise, noninfectious conditions can be identified and treated during the quarantine period. Further, this period will also provide a time for animals to adapt to the housing facility, their diet, and their allocation with other individuals in experimental groups.
Ectoparasites and endoparasites are commonly found in wildlife hosts, and bats are no exception. Mites, flies, and soft ticks have been observed in many species of bats and are likely coevolved with their host.76–78 In our experience, topical selamectin has proven a safe and effective treatment for most mites in bats when dosed at 6 mg/kg using a micropipette (Falendysz, unpublished data) and applied twice within 2 weeks (Cardenas-Canales, unpublished data). Barnard et al. recommend fibronil and other parasiticides for ectoparasites. 32 Figure 3 shows ectoparasites found on recently imported Artibeus spp. and vampire bats.

Ectoparasites can be frequently found in bats kept in captivity, mainly when experiencing overcrowding.
Alopecia (hair loss) is a common problem that can develop in many bat species while in captivity. Hair loss has been commonly observed on the chest and abdomen in Artibeus spp. While some individuals are captured presenting this condition, it has been observed to initiate in captivity. The etiology is unknown, although various fungal and bacterial infections have been ruled out. 79
Procedures with Live Bats
Blood collection is one of the most common procedures performed in bats during experiments. The technique will depend on the bat species but can be done safely with adequate training. For example, large bat species (e.g., fruit bats, flying foxes, vampire bats) can be bled from the cephalic vein. 80 In other smaller species (little brown or big brown bats), blood is obtained from the uropatagial vein.81,82 Generally, the maximum blood volume that can be collected is 0.5–1% of the body weight, which amounts to 300 µL of blood or more from larger species, whereas for small species, sometimes 50 µL is the maximum volume possible. In this case, Nobuto blood filter strips (Sterlitech, WA, USA) can be a good option for obtaining blood samples from small bat species. Given the small amounts of blood and the lower pressure in the vessels, tubes coated with anticoagulant are recommended, if not interfering with the sample type and test to be done.
Usually, two people are required to bleed bats, one handler (wearing bite-proof gloves) and another performing the venipuncture. Morgan et al. (2022) designed a device for a single individual to safely collect blood from anesthetized or nonanesthetized bats. 83 The device is a plastic restraint chamber that gives access to the bat’s wings, providing an added safety measure for the handler and increasing the efficiency of blood collection. By using the restraint devices during sampling, no needle sticks, bites, or scratches to samplers occurred, and no health issues or complications from blood collection were observed in the bats bled. Wimsatt et al. (2005) utilized a plastic tube restraint device in conjunction with anesthesia to collect small quantities of blood from free-ranging big brown bats and demonstrated no difference in short-term survival. 82 Figure 4 shows venipuncture from the uropatagial vein in a laboratory-housed big brown bat without anesthesia, using a custom-made restraint wrap (Figure 5), a thimble to protect fingers, and capillary tubes (Mircovette CB300, Sarstedt Inc., Numbrecht, Germany). Other restraint devices have been designed to obtain samples of interest such as wing biopsies 84 for white-nose syndrome detection. Blood may also be collected during the euthanasia process, if bats are decapitated under deep anesthesia and blood is collected into a small tube from the decapitation site.
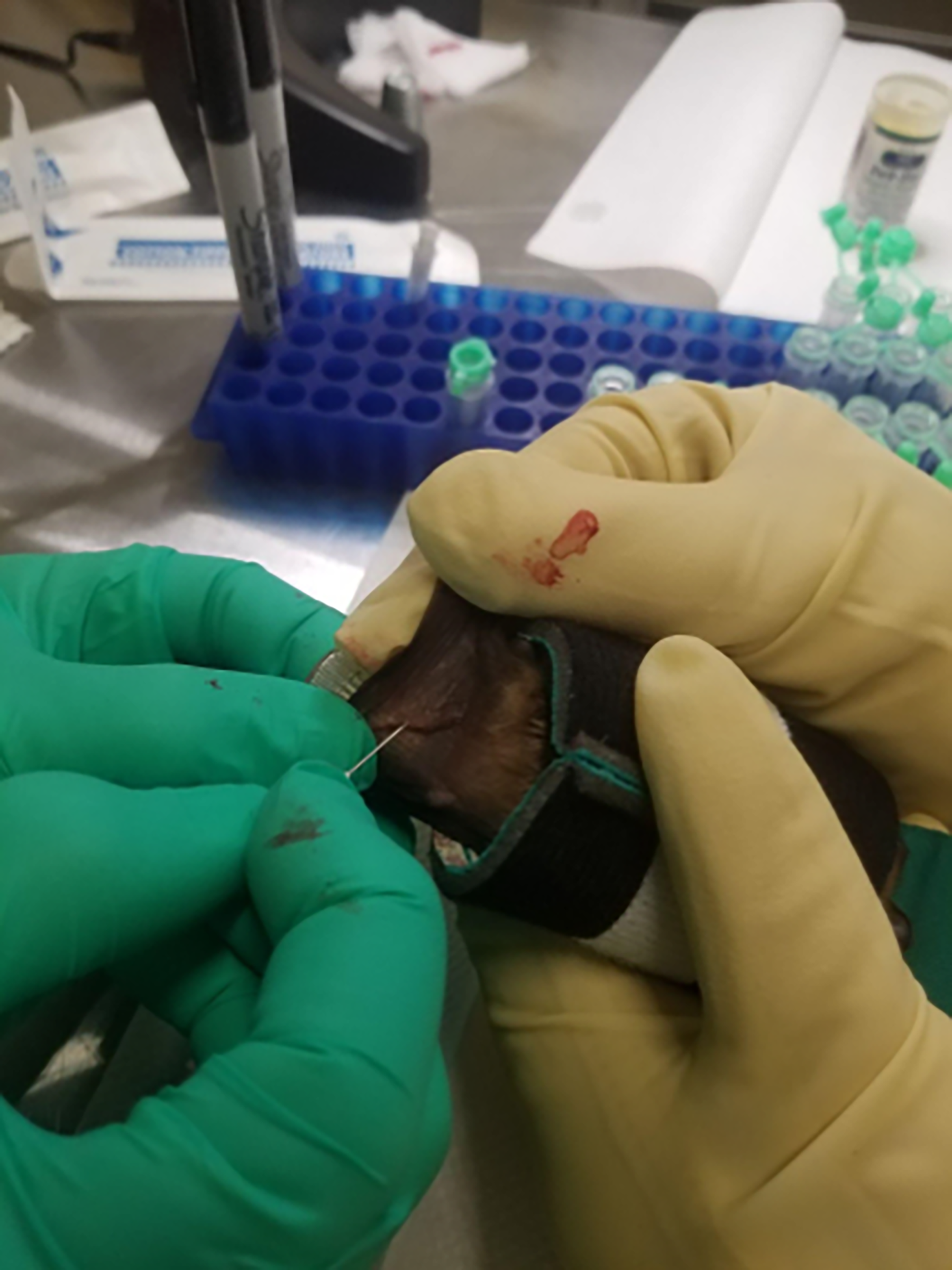
Venipuncture from the uropatagial vein without anesthesia. Photo courtesy of E. Falendysz.
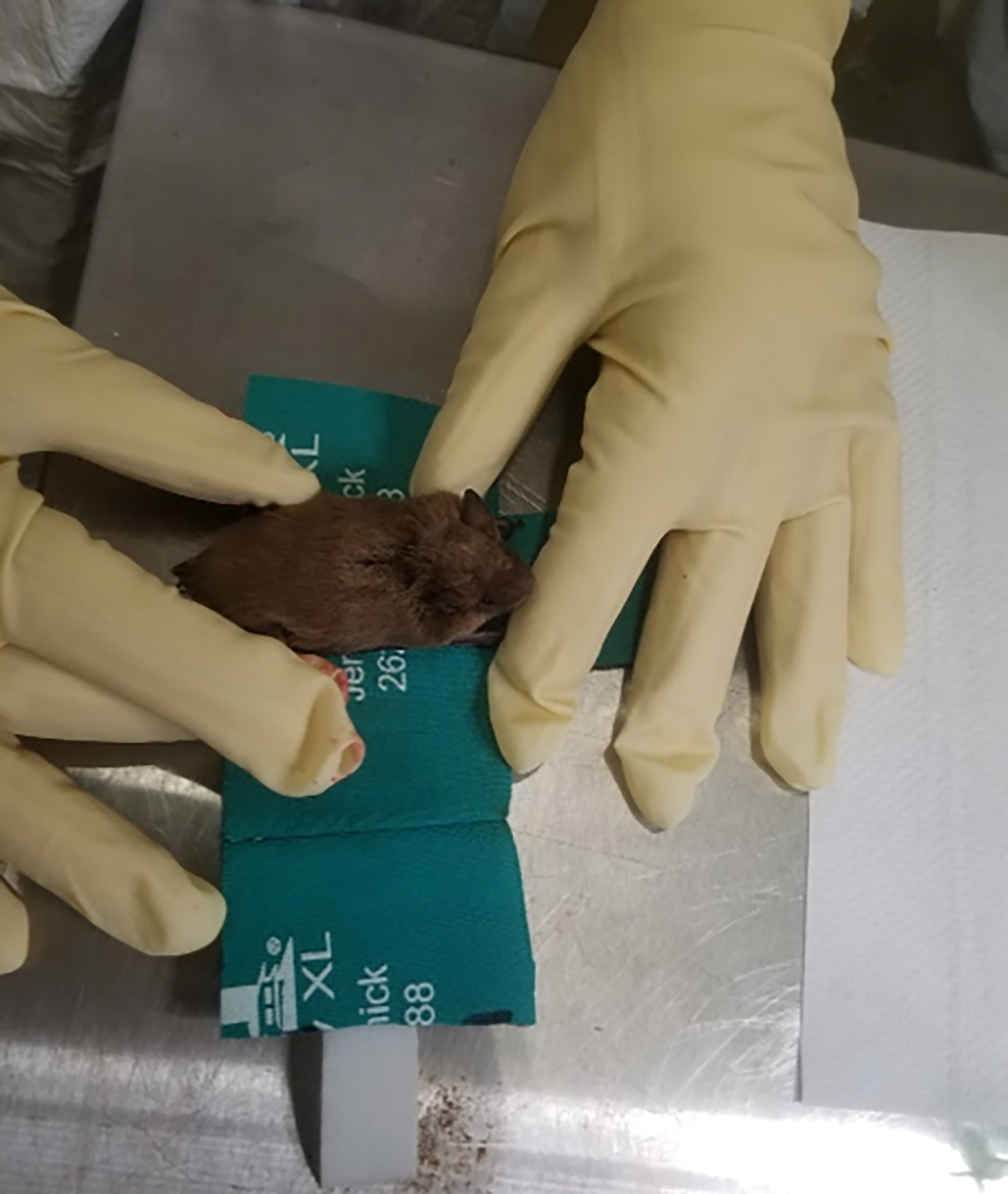
A big brown bat (Eptesicus fuscus) sits in a restraint device made from a neoprene can cooler and a Velcro® self-grip strap. The long edge is wrapped around the body and closed with the Velcro strap. The smaller flap is tucked in, over the head. The restrainer uses leather-protective gloves with latex gloves over them.
Release of Captive Bats
Given the conservation status of many bat species, there may be strong ethical reasons to release bats after a study, if they have not been infected with any known pathogens. This activity should be carefully planned in coordination with wildlife managers and wildlife veterinarians, to ensure that the animals released into appropriate geological and ecological areas are fit for release and that they do not pose a risk to free-ranging bat populations. Adequate separation of bats to be released from other animals that could share pathogens must be considered during the planning for housing the bats in captivity. Additional surveillance or testing of bats before release may be necessary to protect wild populations.
Necropsy
Necropsy of bats can be performed very similarly to other small mammals, such as rodents. One significant anatomical difference is the patagium, or wing membrane. If histological sections of the wing membrane are to be examined, the tissue can be rolled around a cylinder of dental wax during necropsy and then sliced in cross-sections after fixing in formalin or other fixative. This requires the wings to be pinned in the extended position (Figure 6), and a scalpel is used to cut the wing incrementally as it is rolled around a small cylinder of dental wax. 85 The dexterity required to tightly roll the wing membrane around the wax means that cut-proof gloves or thimbles cannot be used to protect the fingers from the scalpel, and great care must be used. Safety scalpels with retractable blades can be used to reduce the risk of a sharps injury. Necropsy is typically performed on a downdraft table or in a biosafety cabinet. A downdraft table provides some protection to the operator from biological hazards, but it is not a primary containment device as the protection is not complete like a biosafety cabinet. A risk assessment should be performed to determine if a downdraft table is appropriate for bat necropsies.

An example of pinning of wings in an extended position to allow for cut wing membrane to be rolled around a small cylinder of dental wax and then fixed in formalin or similar before cross-sections are taken. Tissues were taken during necropsy, after confirmed humane euthanasia of the animal. Photo courtesy of E. Falendysz.
Agents of Concern and Occupational Health
Disease-Causing Agents of Concern
Before working with bats, their fluids or tissues, it is important to understand that there are various disease-causing agents that may be present, including RABV. It is important to know what country and geographic region the bats and/or bat tissues are being sourced from. The potential for the bats and/or bat tissues to harbor one or more disease-causing agents is partially dependent on their point of origination. This is especially important when recommending vaccines.
In the realm of biological safety, laboratory-acquired infections (LAIs) can provide many lessons learned, particularly around the importance of properly conducting procedures, utilizing appropriate safety measures, and recognizing and reacting to possible exposures or signs and symptoms of illness. Many of these risks can and should be properly assessed prior to the initiation of the work, and close calls, near misses, potential exposures, or LAIs should be fully reviewed to determine how an assessment can be improved. Spillover from bats has led to various documented outbreaks, and as bat research expands, the potential for an exposure in the field or the lab will continue to grow. These might be known pathogens, such as those discussed in this article, or they might lead to the discovery of novel pathogens. Potential exposure to a bat during field research in 2012 led to an unknown illness in a researcher and the eventual identification of a novel paramyxovirus, named Sosuga virus. 86 This new virus is closely related to a rubella-like virus in fruit bats, with which the researcher was working.
Medical surveillance recommendations, as well as general safety, also extend to agents that are isolated and identified from bats. It is important to note that many of these agents are Risk Group 3 and 4 agents and are subject to additional regulatory oversight to possess and work with the agent. In the United States in particular, a number of these agents are regulated by the Federal Select Agent Program, 87 which has a strict set of requirements for performing work. We do not address such biosafety and biosecurity requirements for working with Risk Group 3 and 4 agents in this article.
The following agents are representative of some of the more common disease-causing agents in bats, or agents that may require special attention.
Bartonella Spp.
Many Bartonella species are zoonotic, with those responsible for human illness associated with a broad spectrum of clinical symptoms, including culture-negative endocarditis and prolonged fever of unknown origin. 88 While transmission may be through a bat bite or the bite of biting arthropods, it has been proposed that fecal droppings of blood-fed bat ectoparasites might transmit Bartonella into the human host, assisted by superficial scratching or tissue trauma of the skin. Bats have been associated with Bartonella across the globe, and exchange of Bartonella between phylogenetically distant domestic animals and wildlife, including bats, has occurred, likely due to increased contact.
Filoviruses
Ebola Virus
Researchers found a bat in Liberia that may have been infected with Ebola Zaire, as the bat harbored about one-fifth of the viral genome. 89 Bats have been suspected as being the source of Ebola infections since viral fragments have been identified in several species. Ebola is transmitted through direct contact with an infected animal (bat or nonhuman primate) or human. The virus enters through the skin or mucous membranes of the eyes, nose, or mouth. This has led to outbreaks in several African countries, with various species of the virus implicated in these outbreaks. The illness manifests with “dry” symptoms such as fever, aches and pains, and fatigue, which progresses to the “wet” symptoms of diarrhea and vomiting. These can be followed by bleeding or bruising from various organs. There are two treatments approved by the FDA involving monoclonal antibodies. There is also an FDA-approved vaccine effective against Ebola Zaire for those at high risk of infection.
Marburg Virus
The Marburg virus reservoir is the Egyptian rousette bat (Rousettus aegypticus),90,91 a cave-dwelling bat found widely across Africa. It causes an illness similar to Ebola. However, there are currently no effective vaccines for it, and there are no approved antivirals for treatment, although some, like ribavirin, have been tried.
Lloviu Virus
Few viruses have been described as lethal for bats, besides lyssaviruses; Lloviu filovirus causes a deadly disease and mass mortalities in common bent-wing bat (Miniopterus schreibersii) populations in Spain, Portugal, France, and Hungary, 92 where 5% of the Hungarian population of common bent-wing bats perished. 93 Its zoonotic potential is currently under investigation.
Henipaviruses
Hendra Virus
Hendra virus, found in Australia, is closely related to Nipah virus (see below) and is transmitted through the urine of fruit bats (flying foxes). 22 Symptoms are similar to Nipah virus, with respiratory and central nervous system involvement. Only seven cases were reported in humans between 1994 and 2013, with four out of seven cases fatal. 94 Ribavirin has been shown to be effective in lab studies, but the usefulness of this drug is not clear.
Nipah Virus
Nipah virus is a zoonotic agent found in infected fruit bats (flying foxes) or pigs in south and southeast Asia and is transmitted through direct contact or through their body fluids, including blood, urine, or saliva. 95 It can also be transmitted through food or fruit contaminated by infected bats, and human-to-human transmission has been documented. Closely related to Hendra virus (see above), it is also transmitted through fruit bats. Symptoms of Nipah virus infections range from mild to severe, with deaths reported in documented outbreaks between 1998 and 2023. Initial infections can present with fever, headache, and a respiratory illness with cough, sore throat, difficulty breathing, and vomiting followed by brain swelling (encephalitis) with sleepiness, disorientation, mental confusion occasionally progressing to coma, and death. There are no licensed treatments, although monoclonal antibody treatments have been used on a supportive basis.
Histoplasma
Histoplasmosis is a fungal infection caused by Histoplasma capsulatum, carried in bat gastrointestinal tracts where it can be shed in guano or found in soil and transmitted to humans. 96 It is the most frequent cause of fungal respiratory tract infections in humans. Fungal spores are inhaled leading most commonly to self-limited, acute, influenza-like illnesses. However, it can lead to potentially lethal infections, particularly in those with underlying health conditions. These occur more frequently in immunocompromised individuals. Treatment ranges from no care to the use of antifungal agents depending on the nature of the infection. While the highest risk for human exposure would occur when entering bat caves, it is prudent to minimize aerosolization of guano in the research setting such as when removing guano during husbandry operations.
Leptospira
Leptospirosis caused by Leptospira interrogans is a bacterial infection most often associated with exposure to infected rodents through their urine, which is then spread to soil or water. This can enter through the skin or mucous membranes (eyes, nose, or mouth), especially if the skin is broken through a cut or scratch or through drinking contaminated water. It has been detected in over 50 bat species in various tropical or subtropical areas. 97 However, the role of bats as transmitters of this infection is not confirmed, as a single case of bat to human infection was identified only by serology. Leptospirosis causes a wide range of symptoms as well as asymptomatic infections. Symptoms may include kidney damage, inflammation of the lining of the brain and spinal cord (meningitis), liver failure, respiratory illness, and even death. Treatment involves the use of appropriate antibiotics such as penicillin or doxycycline, especially in early infections.
Lyssaviruses
There are 18 major species of lyssaviruses, one of which is RABV. Bats are one of the most commonly reported rabid animals in the United States. 98 Rabies vaccine is effective against lyssaviruses belonging to phylogroup I, which includes RABV, Aravan virus, Australian bat lyssavirus, Bokeloh bat lyssavirus, European bat lyssaviruses type 1 and 2, Duvenhage lyssavirus, Gannoruwa bat lyssavirus, Khujand virus, Irkut lyssavirus, Taiwan bat lyssavirus, and Kotalahti bat lyssavirus. Many of these viruses are known to cause bat and human fatalities. However, the RABV vaccine is not efficacious against those in phylogroup II including Lagos bat virus, Shimoni bat lyssavirus, and Mokola Virus (MOKV), and those not yet been included in any phylogroup, including Ikoma lyssavirus, West Caucasian bat Virus, and Lleida bat lyssavirus. Other viruses are pending inclusion in the genus: Divaça bat lyssavirus, Phala bat lyssavirus, and Taiwan bat lyssavirus 2. 99
Among these phylogroups (and excluding RABV), only MOKV in phylogroup II is known to lead to human fatalities. However, in areas where the ability to type these viruses is not readily available, such as Africa, there may be other lyssaviruses not currently identified that are pathogenic. The most common route of infection will depend on if workers handle live animals or only infected tissues. Bites from infected animals represent the most common route of transmission, and concentrations of lyssaviruses are high in central nervous system tissue, salivary glands, saliva, and lacrimal secretions. For work with potentially infected tissues, accidental cuts or needlesticks, as well as mucous membrane exposures or contact with broken skin, represent the highest hazard.
It is also important to consider the health of the bats and prevent spread of infectious agents that can adversely impact colonies. The following are two examples:
Pseudogymnoascus destructans, agent of white-nose syndrome
Pseudogymnoascus destructans is a fungus that infects hibernating bats, causing what is known as white-nose syndrome. The fungus thrives in cold and humid conditions characteristic of caves and mines used by bats. Primary transmission is from bat to bat, although it may be carried by humans on their clothing and gear when traveling from cave to cave. The fungus currently does not appear to pose a risk to humans. 100
SARS-CoV-2, agent of COVID-19
One review of the global distribution and host range of beta-coronavirus evolutionary lineages suggests that 40+ species of temperate-zone North American bats could be immunologically naïve and susceptible to infection by SARS-CoV-2. 101 Individuals that frequently handle and come into close contact with bats can help decrease any chances of spillover by adopting many of the same controls noted for human protection such as PPE and biosafety practices while carefully evaluating how their actions might adversely affect bat populations. The CDC recommends that people with suspected or confirmed COVID-19 avoid contact with animals, including pets, livestock, and wildlife. 102 The authors have addressed SARS-CoV-2 and bats in their previous article.
Medical Surveillance and Vaccination
Due to the presence of the zoonotic agents mentioned here, as well as many more found in bats, proactive occupational health programs are essential when working with bats in the vivarium setting, and bat tissues and fluids in the research and diagnostic laboratory. It is important to realize that not all of these agents will cause the bat to appear sick, but they are capable of causing illness in humans. In addition, individuals who have an underlying medical condition such as pregnancy or immunosuppression may be at an increased risk of developing disease or experiencing adverse health events.
Pre- and Postexposure Prevention and Medical Surveillance
Laboratory work with bat tissues poses a risk to the individuals handling the tissue. In addition to the biological risks described earlier, if the tissues are preserved in formalin or similar solution, there are chemical safety risks to consider. All work with bat tissues or fluids should be registered with the institution’s IBC or similar oversight committee so that appropriate medical surveillance and vaccination information and recommendations can be provided by the occupational health team.
Individuals who may handle live bats should be included in an institution’s animal allergen surveillance program. In addition, when considering the in vivo setting, bat guano should be considered a potential allergen and source of pathogens. Respiratory protection should be used if determined necessary through the risk assessment process.
In May 2022, the CDC issued updated guidance regarding the use of rabies vaccines. 103 Given rabies is the most important public health hazard associated with bats, this vaccine should be offered to individuals at risk for RABV exposure. Table 2 presents the redefined risk categories and recommendations for vaccination and is reprinted from the Morbidity and Mortality Weekly Report (MMWR) article with permission.
Rabies preexposure prophylaxis recommendations as published in MMWR 103
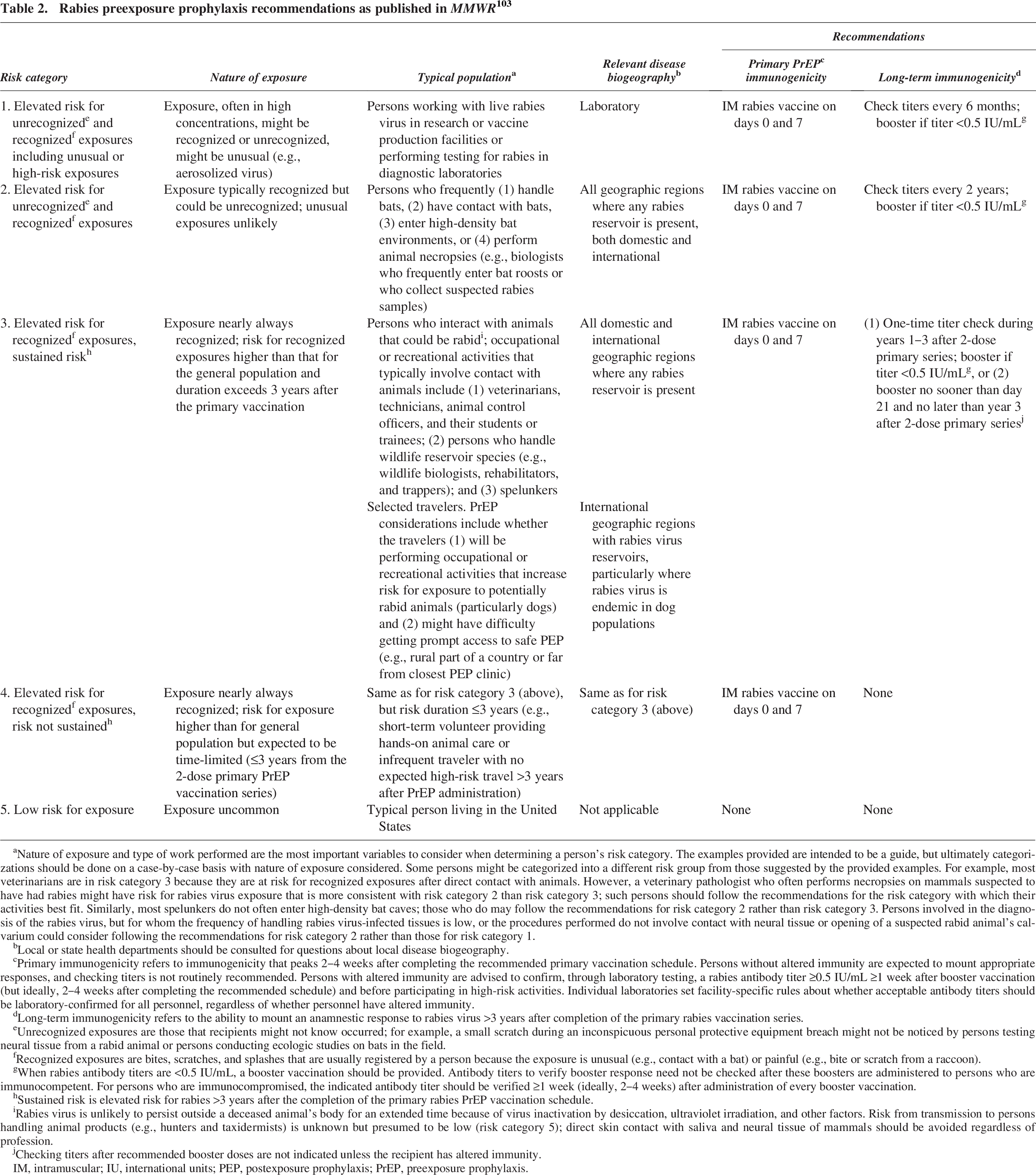
Nature of exposure and type of work performed are the most important variables to consider when determining a person’s risk category. The examples provided are intended to be a guide, but ultimately categorizations should be done on a case-by-case basis with nature of exposure considered. Some persons might be categorized into a different risk group from those suggested by the provided examples. For example, most veterinarians are in risk category 3 because they are at risk for recognized exposures after direct contact with animals. However, a veterinary pathologist who often performs necropsies on mammals suspected to have had rabies might have risk for rabies virus exposure that is more consistent with risk category 2 than risk category 3; such persons should follow the recommendations for the risk category with which their activities best fit. Similarly, most spelunkers do not often enter high-density bat caves; those who do may follow the recommendations for risk category 2 rather than risk category 3. Persons involved in the diagnosis of the rabies virus, but for whom the frequency of handling rabies virus-infected tissues is low, or the procedures performed do not involve contact with neural tissue or opening of a suspected rabid animal’s calvarium could consider following the recommendations for risk category 2 rather than those for risk category 1.
Local or state health departments should be consulted for questions about local disease biogeography.
Primary immunogenicity refers to immunogenicity that peaks 2–4 weeks after completing the recommended primary vaccination schedule. Persons without altered immunity are expected to mount appropriate responses, and checking titers is not routinely recommended. Persons with altered immunity are advised to confirm, through laboratory testing, a rabies antibody titer ≥0.5 IU/mL ≥1 week after booster vaccination (but ideally, 2–4 weeks after completing the recommended schedule) and before participating in high-risk activities. Individual laboratories set facility-specific rules about whether acceptable antibody titers should be laboratory-confirmed for all personnel, regardless of whether personnel have altered immunity.
Long-term immunogenicity refers to the ability to mount an anamnestic response to rabies virus >3 years after completion of the primary rabies vaccination series.
Unrecognized exposures are those that recipients might not know occurred; for example, a small scratch during an inconspicuous personal protective equipment breach might not be noticed by persons testing neural tissue from a rabid animal or persons conducting ecologic studies on bats in the field.
Recognized exposures are bites, scratches, and splashes that are usually registered by a person because the exposure is unusual (e.g., contact with a bat) or painful (e.g., bite or scratch from a raccoon).
When rabies antibody titers are <0.5 IU/mL, a booster vaccination should be provided. Antibody titers to verify booster response need not be checked after these boosters are administered to persons who are immunocompetent. For persons who are immunocompromised, the indicated antibody titer should be verified ≥1 week (ideally, 2–4 weeks) after administration of every booster vaccination.
Sustained risk is elevated risk for rabies >3 years after the completion of the primary rabies PrEP vaccination schedule.
Rabies virus is unlikely to persist outside a deceased animal’s body for an extended time because of virus inactivation by desiccation, ultraviolet irradiation, and other factors. Risk from transmission to persons handling animal products (e.g., hunters and taxidermists) is unknown but presumed to be low (risk category 5); direct skin contact with saliva and neural tissue of mammals should be avoided regardless of profession.
Checking titers after recommended booster doses are not indicated unless the recipient has altered immunity.
IM, intramuscular; IU, international units; PEP, postexposure prophylaxis; PrEP, preexposure prophylaxis.
Employers may want to consider rabies vaccination as a postoffer, preplacement requirement to work with bats for those employees where it is not medically contraindicated. Vaccinating at no cost to the employee within a short period of time after hire is one strategy for those who cannot provide evidence of prior vaccination. For existing employees, information on the vaccine including the May 2022 CDC guidance should be provided in addition to strongly recommending vaccination (or requiring, depending on the terms of employment) and providing it free of charge. Ongoing titers and further preexposure vaccination per CDC should be offered to those who continue to work with bats as part of an ongoing occupational health program to make sure vaccine and titer updates are provided in a timely fashion. Similarly, employees with a previous vaccination history should have their antibody titers monitored regularly per Table 2 by the employer at no cost to the employee. Those individuals not previously vaccinated and not willing to be vaccinated should be asked to sign a declination form, though the necessities and capabilities of each occupational health group may vary by institution. The decision to require a vaccination for certain activities must be made in consultation with legal and human resources personnel at each entity. In addition, all workers at risk should be trained on the need for postexposure vaccine if exposures arise, even if preexposure vaccine is up to date, again, per the CDC guidelines. These workers should know what to do and where to go to get the postexposure vaccine within the short timeframe specified by the CDC; this is important information to include in training for individuals working with or around bats or bat tissues.
Immunocompromised Status
Immunocompromised individuals have a weakened immune system with a reduced ability to fight infections and other diseases. Individuals planning to work with or around bats should consult with an occupational medicine provider and include a discussion of personal health, particularly as it relates to immune status. Medical providers should consider the implications of individual health and immune status when considering vaccine recommendations, as the vaccine schedules differ from those for nonimmunocompromised individuals, efficacy of vaccination, and availability of treatment options following potential exposures. Education and counseling regarding increased risk may need to be part of the medical conversation. In some cases, there may be no research on the risks of certain activities (e.g., a pregnant, rabies-vaccinated researcher working with infected bat tissue); individuals must therefore be allowed to opt out of activities according to their own risk tolerance without employment consequences.
Emergency Response and Planning
Advance preparation for emergency situations such as bites and scratches as well as sharps injuries from necropsies and tissue manipulation is of the utmost importance. Prior to obtaining bats, their fluids, or tissues, it is highly recommended that all possible exposure scenarios be reviewed, and plans for handling each exposure be put in place.
Written plans should be developed for postexposure assessment, treatment, and prophylaxis. Such plans should consider what occupational health resources exist within the institution and what resources may need to be procured from external resources. Signage should be posted in conspicuous areas where bats are housed and manipulated as well as areas where tissues are used. The signage should include the steps to take following an exposure as well as the relevant phone numbers and instructions for obtaining occupational health services in a prompt manner. All personnel must be reminded of the need to seek assessment promptly regardless of the severity of the exposure.
Tabletop drills are an excellent way to plan for exposure scenarios and test the institution’s response readiness. Participants should include representatives from occupational health and the individuals slated to handle the bats and/or bat tissues. This includes researchers as well as husbandry staff.
It is important for the occupational health practitioners supporting the research program to be knowledgeable about the work to be conducted with the bats or bat materials. This includes being familiar with the rabies preexposure prophylaxis recommendations (Table 2).
If the laboratory space is a communal setting with multiple laboratory groups and shared equipment, those not involved in the handling of bat materials should be advised of the work to be conducted and the potential risks. In addition, they should be offered an opportunity to discuss any personal medical concerns with an occupational health physician.
Conclusions
The information presented in this article seeks to help biologists, biosafety professionals, human and veterinary medical professionals, and others in the environmental health and safety field to broadly and holistically perform a risk assessment when reviewing potential work with bats or bat tissues. Review must consider the many aspects outlined in this and previous articles, regardless if the work is performed in the field or in the laboratory, in vitro or in vivo. Potential for RABV exposure following a cut, bite, mucous membrane, or other exposure may be a primary concern, but other zoonotic diseases should also be considered and through other routes such as aerosol exposure. Further, risks should be considered and addressed not just for those directly performing work, but those who may work in the same area, or may be working in support of the research, such as vivarium staff. Finally, risks to animal welfare should be considered to perform work in a humane manner and protect species at risk, or to protect other species housed in the vivarium. Collectively, the information discussed here can help safety professionals pursue relevant data to make informed decisions and advocate for the necessary resources, and the authors hope that while this article outlines risk considerations for work with bats, the basic concepts and considerations can be applied to work with other species that pose potential zoonotic threats to humans.
Authors’ Contributions
A.A.S., N.A.C., E.M.C.C., E.A.F., H.K.F., D.F., G.R.F., T.W.H., S.M.J., M.S.M., and C.A.S.: Contributed to conceptualization and writing of the original draft. E.G.D., L.A.O.C., G.L.P., and S.E.V.: Contributed to conceptualization, writing of the original draft, and reviewing and editing the final draft.
Footnotes
Acknowledgments
The authors thank the Eagleson Institute for their financial contribution to the Open Access publication fee. In addition, the authors thank Douglas C. Eckery, PhD (USDA APHIS Wildlife Services) for reviewing the article.
Funding Information
This work received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. The publication fee for Open Access was generously provided by the Eagleson Institute.
Authors’ Disclosure Statement
The authors declare that there are no conflicts of interest.
