Abstract
Introduction:
The use of e-cigarettes is increasing rapidly. As only scant information is available on the toxicological hazard of inhaling flavoring compounds in e-liquids, we assessed effects of the widely used flavoring ethyl maltol on bronchial epithelial cells. We investigated whether ethyl maltol and/or nicotine could induce inflammatory effects and would have the potential to become systemically available.
Methods:
Bronchial epithelial cells (Calu-3) were cultured at the air–liquid interface and exposed to e-cigarette aerosol with/without nicotine and with/without ethyl maltol using a smoking machine for one or two 30-minute periods per day for 2 consecutive days. Similar experiments were carried out using fully differentiated human primary bronchial epithelial cells (MucilAir) from different donors.
Results:
Exposure to neither of the four e-cigarette aerosols affected cell viability or membrane integrity. Both nicotine and ethyl maltol rapidly translocated to the basolateral side in both cell models. Exposure to e-liquids affected the production of MCP-1, IL-8, IP-10, IL-17A, and IL-6. For Calu-3 cells, these effects were mostly related to nicotine, and for MucilAir cells, these effects mostly related to ethyl maltol. Exposure of MucilAir cells to e-liquids with nicotine and ethyl maltol resulted in an increased mRNA expression of IL-8, and exposure of MucilAir cells to e-liquid with nicotine led to a decreased expression of MCP-1.
Conclusion:
Our data show that both cell models can be used to assess translocation of inhaled compounds using a human relevant exposure method. Inhalation of nicotine and ethyl maltol may cause local effects in the respiratory tract and could lead to systemic exposure.
Introduction
Electronic cigarettes, or e-cigarettes, are promoted by the industry as a less harmful alternative for conventional tobacco cigarettes, or are marketed as products to help users quit smoking.1–3 Although the e-cigarette vapor indeed contains fewer compounds than tobacco smoke, 4 it does contain compounds that can be harmful when inhaled. 5 For example, propylene glycol (PG) and glycerol are the main components of e-liquids and may cause irritation to the respiratory tract,6,7 which may lead to inflammation. The effects of nicotine have been assessed previously,8–10 but e-liquids also contain many flavoring compounds that could be hazardous when inhaled and make the product more attractive to use. 11
The European Tobacco Products Directive states that ingredients of e-liquids should not have carcinogenic, mutagenic, or reproduction toxic (CMR) properties and may not pose a risk to human health in heated or unheated form. 12 Especially regarding human health effects other than CMR, data on toxicological effects of flavoring compounds in e-liquids are lacking. This is a concern given the high number of flavoring compounds used in e-liquids and the widespread use of e-cigarettes or vapes among the population.13–15
In fact, for only a few of these flavoring compounds, information is available on the toxicological effect after inhalation.16,17 Considering that e-cigarette users are inhaling these flavoring compounds daily and multiple times per day, there is an urgent need to characterize the toxicological effects of e-liquid flavoring compounds.
Most, if not all, flavoring compounds used in e-liquids were found to be safe when used in food products. However, this does not imply that inhaling such flavoring compounds, when using an e-cigarette, is safe. Indeed, there are examples of flavoring compounds used in e-liquids that are safe for oral use, but hazardous when inhaled, such as cinnamaldehyde and diacetyl.18–22 Inhaling such compounds can cause an inflammatory response that may ultimately lead to chronic respiratory diseases (reviewed in Ref. 18 ). The effects of diacetyl, for example, are well known from occupational exposure where workers developed bronchiolitis obliterans. 23
Moreover, inhaled compounds may not only exert local toxic effects in the lungs and airways but can also become rapidly systemically available. It is well known that inhaling nicotine leads to a faster “nicotine-kick” than oral absorption of nicotine due to fast absorption through the airways and subsequently passing the blood–brain barrier.24,25 Similarly, other compounds, such as flavoring compounds, could enter the blood circulation and cause systemic effects in the body. This route substantially differs from the oral route, as orally ingested compounds first have to pass the acidic environment of the stomach and metabolism in the liver before they enter the blood circulation, thus resulting in a much faster increase in blood concentrations through the inhalation route.
Although compounds need to cross the endothelial cell layer as well before they enter the blood circulation, passing of the epithelial barrier is the first step toward systemic uptake of compounds. Cell models of the airway epithelium, cultured at the air–liquid interface (ALI), can be used to assess translocation across the epithelial layer and the effects of inhaled compounds at their first site of contact. In addition, ALI-cultured cell models also allow realistic exposure to tobacco product and e-cigarette aerosols. Intermittent exposure of the bronchial cells of e-cigarette users to the aerosol as it is inhaled can be simulated using a smoking machine to generate aerosols.
The aim of our research was to apply in vitro models of the airway epithelium to determine whether a flavoring compound from e-cigarettes (1) could induce toxicological effects such as cell death, cytokine responses, and altered gene expression and (2) has the potential to cross the epithelial barrier. To this purpose, we prepared experimental liquids with or without nicotine and ethyl maltol, which is one of the most commonly used flavoring compounds in e-liquids.26,27 As toxicological effect and the uptake mechanism of ethyl maltol are unknown, we have used two different cell models in our experiments.
Materials and Methods
E-liquids
We have used e-liquids prepared in-house, consisting of PG and glycerol in a 1 to 1 ratio. In addition, nicotine (10 mg/mL) and/or ethyl maltol (10 mg/mL) were added to these e-liquids. This resulted in four different e-liquids that allowed us to assess the effects of nicotine and ethyl maltol individually and in combination. E-liquids were prepared using PG (Sigma-Aldrich, Darmstadt, Germany) and glycerol (Sigma-Aldrich) 1:1 v/v to obtain the e-liquid base. For each individual liquid, 10 mL of liquid based on weight and corrected for the density was transferred in glass jars.
For the first liquid, no ingredients were added (N−/E−). For the second liquid (N+/E−), 100 μL of 1.01 g/mL of nicotine (Sigma-Aldrich) was added to obtain a concentration of 10 mg/mL nicotine. For the third liquid (N−/E+), 100 mg of ethyl maltol (Sigma-Aldrich) was added to obtain a concentration of 10 mg/mL. For the fourth liquid (N+/E+), 100 μL of nicotine and 100 mg of ethyl maltol were added to obtain a final concentration of 10 mg/mL nicotine and 10 mg/mL ethyl maltol.
The solutions were homogenized in the dark using a magnetic stir bar and magnetic stirrer until completely dissolved (10 minutes to 4 hours at room temperature). The individual glass containers were wrapped in aluminum foil to prevent photodegradation. The solutions were stored in the dark at room temperature until use.
Cell models
Initial translocation measurements and toxicological assessment were conducted using the bronchial epithelial cell line Calu-3, cultured at the ALI, which was followed by similar experiments with primary bronchial epithelial cells (PBECs): the MucilAir model from Epithelix.
Calu-3 cells were cultured as described previously.28,29 In brief, cells were seeded on 12-well inserts with 0.4 μm pore size and grown until confluent. The apical medium was removed on day 7 after seeding (cells were airlifted), subsequently cells were cultured at the ALI for 7–9 days before exposure.
MucilAir cells (Epithelix, EP01MD, donor MD0801 [referred to as donor 1] and MD0802 [referred to as donor 2]) in 24-well inserts were transferred from their nutritive transport gel upon arrival to new 24-well plates (Corning costar 24-w, clear, REF3524) and cultured according to the manufacturer's protocol in complete culture medium (Epithelix, MucilAir culture medium, EP05MM) in a 37℃ incubator (Binder, CO2 5%, air 95%). Donor 1 was a 27-year-old Caucasian male and donor 2 was a 55-year-old African female. Both donors were nonsmokers and had no pathology reported.
Exposure
ALI-cultured cells were exposed in a Vitrocell exposure module (Vitrocell, Waldkirch, Germany) that was connected to a VC-1 smoking machine (Vitrocell). This experimental setup allowed us to assess the effects of nicotine and ethyl maltol on bronchial epithelial cells.
For each condition, three inserts with cells were placed in Vitrocell exposure modules (type 6/3), for exposure to e-cigarette aerosol from a VC-1 smoking machine (see Fig. 1). A refillable e-cigarette (eLeaf model iStick Pico 21700, from Zwoofs.nl, 2019, power at 13 W and air inlet fully open) was filled with one of the e-liquids and weighed before use. For each e-liquid, a different device was used to avoid contamination of one e-liquid with another. The e-cigarette was smoked with a puff volume of 55 mL per puff, 3 s per puff inhale, 4 s per puff exhale, and 2 puffs per minute using a square-shaped curve as a standard protocol for e-cigarette use. 30
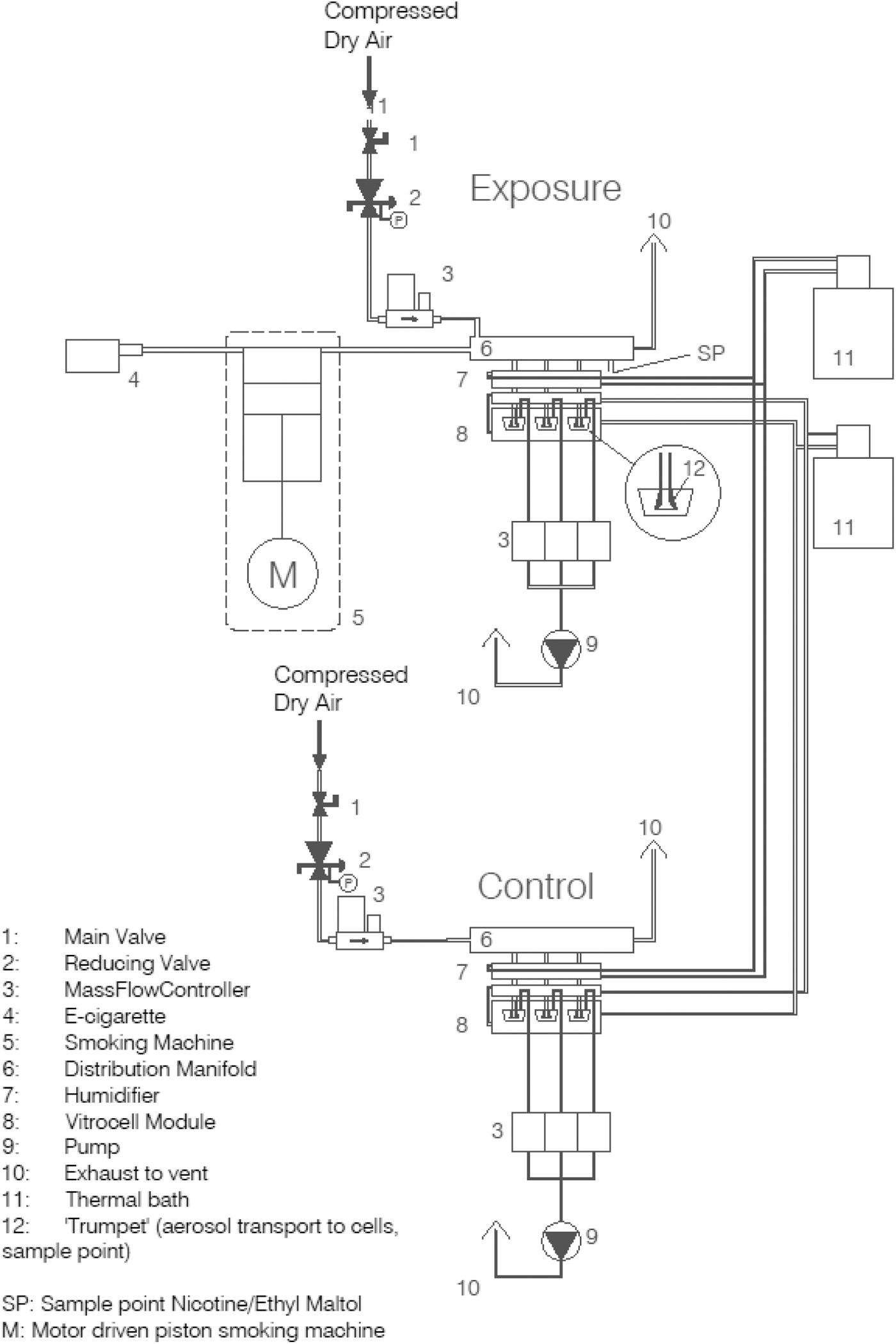
Schematic diagram of the exposure system.
This aerosol was diluted with a continuous flow of 500 mL/min (463 mL/min) and led to the distribution unit. From the distribution unit, the aerosol was directed at 5 mL/min through trumpets in the module to the cells. In this way, three inserts were exposed simultaneously in a single module. Excess aerosol (which is the total flow of 500 mL/min minus the flow needed to supply the inserts at 5 mL/min) was exhausted through the exit of the distribution unit using mass flow controllers. To reduce losses of aerosol constituents in the tubing, the smoking machine performed 10 puffs before the cells were placed in the modules.
Cells were exposed to one of the four e-liquids either once or twice per day for 30 minutes per exposure (afternoons only or mornings and afternoons) for 2 consecutive days to mimic repeated exposure over multiple days, see the exposure scheme in Figure 2. Cells exposed to clean air in a similar module at 5 mL/min for the same period and frequency served as controls. In addition, three inserts were kept in the incubator to control for effects caused by handling, the airflow during exposure, or other factors. The complete experiment was repeated once for both cell models.
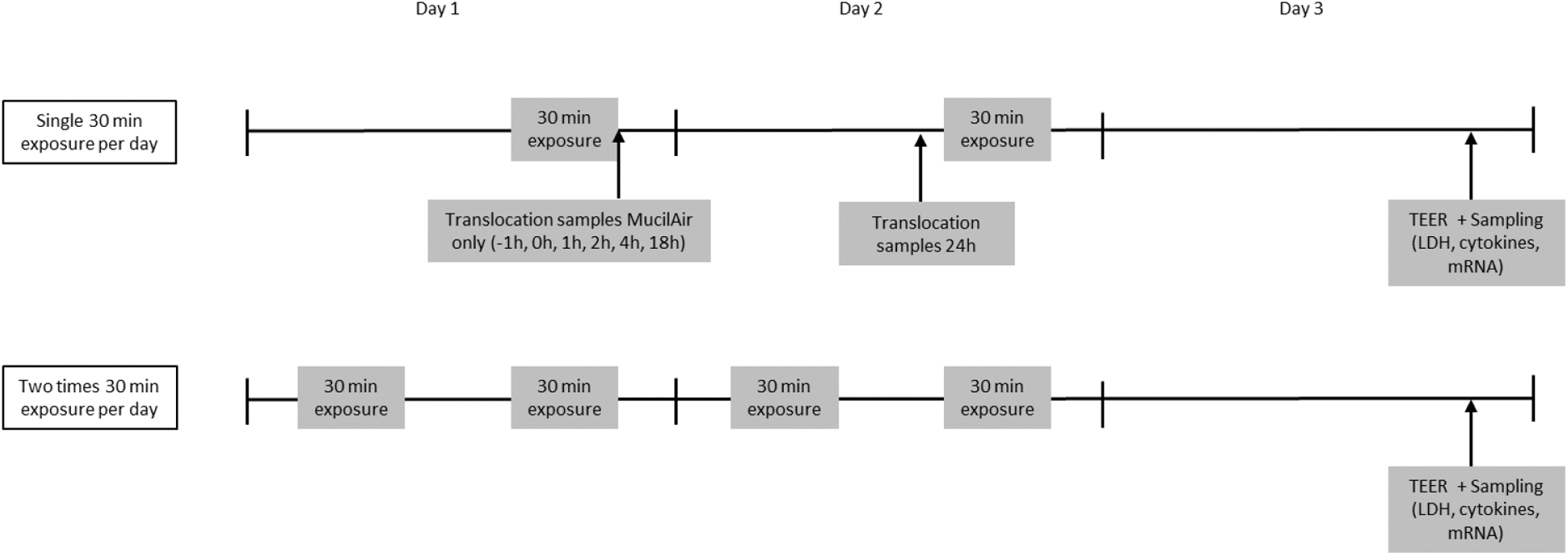
Exposure scheme for Calu-3 and MucilAir cells exposed to e-cigarette vapor once or twice per day for 2 consecutive days. Sampling for translocation was done after a single exposure, whereas samples for cell viability, cytokines, and gene expression were collected on the day after the last exposure.
Sample collection
Air samples
Air samples were taken, for concentration measurements in the air close to the cells, from the outlet of the distribution block, which was placed just above the modules and as close to the cells as possible. For Calu-3 exposures, air samples were taken in the setup after exposure of the cells. As no cells were exposed when collecting air samples, samples were also collected from the trumpets, which is more close to the cells. For the MucilAir exposure, air samples were collected during exposure.
All air samples were collected using two impingers in a row, each containing 10 mL methanol and nicotine D4 as internal standard. The internal standard was used to correct for any evaporated liquid. Samples from the distribution unit were drawn through the impingers with a flow of 5 mL/min for 10 minutes during exposure. Concentrations were measured (using LC-MS/MS Analysis section).
Cell culture samples
Basolateral medium samples of Calu-3 cells were collected after the first exposure and before the second exposure for translocation measurement (for cells exposed to a single 30 minutes period only), and on the day after the second exposure for cytokine measurements. Calu-3 cells were used for cytotoxicity analysis (MTS assay, see below) on the day after the last exposure, and MucilAir cells were lysed for mRNA isolation.
The exposure ended at t = 0 and basolateral medium was sampled from MucilAir cells after a single exposure at t = −1 hour, 0 minute, 1 hour, 2 hours, 4 hours, 18 hours, and 24 hours. For cells that were exposed twice a day, basolateral samples were collected at t = −1 hour and t = 5 hours (exposure ended at t = 0). After sampling, basolateral medium was changed completely. Samples were stored at +4°C for translocation analysis.
In addition, apical and basolateral medium samples from Calu-3 and MucilAir cells were collected after transepithelial electrical resistance (TEER) measurement (see section ‘Transepithelial electrical resistance’), split and stored at +4°C for a maximum of 7 days for the lactate dehydrogenase (LDH) assay and at −80°C for cytokine analysis. Apical medium samples of Calu-3 cells exposed once to liquids N−/E− and N+/E− were accidently not collected for cytokine analysis. For RNA isolation, 200 μL QIAzol (79306; Qiagen) was added to the cells and incubated for 15 minutes on a plate shaker (Heidolph titramax 100) at 300 rpm, room temperature. Cell lysates were then homogenized by pipetting up and down and stored in Eppendorf tubes (Eppendorf DNA Lo-Bind 1.5 mL, 525-0130) at −80°C.
Liquid chromatography-tandem mass spectrometry (LC-MS/MS) analysis
Air samples and cell culture medium samples were analyzed with LC-MS/MS. A volume of 100 μL from the glass impinger was diluted with mobile phase A (10 mM ammonium formate with 0.1% formic acid) to a total volume of 1000 μL. A volume of 100 μL of cell culture medium was transferred to an Eppendorf cup and denatured using 200 μL mobile phase B (1:1 v/v acetonitrile–methanol +0.1% formic acid). After vortexing, the samples were centrifuged at 16,000 g. Fifty microliters of the supernatant was transferred to low-volume high performance liquid chromatography (HPLC) vials and diluted using 50 μL of mobile phase A. The samples were homogenized using a vortex and stored at max. 5°C before analysis.
The LC-MS system consisted of a Shimadzu 20 series (binary pump, degasser, autosampler, and column oven; Shimadzu, Duisburg, Germany) coupled to an QTRAP6500 mass spectrometer (ABSciex, Darmstadt, Germany). The LC analysis was performed on an Acquity UPLC reversed phase column (HSS C18 1.8 μm 2.1 × 150 mm). A gradient starting with 5% mobile phase B with an increase to 100% from 2 to 4 minutes was held for an additional 2 minutes, after which it was returned to the initial conditions in 1 minute and re-equilibrated for 1 minute before the next injection cycle. An injection volume of 10 μL was used for all samples. The limit of detection for nicotine and ethyl maltol was 2 ng/mL, which corresponds to 12 ng/mL in cell culture medium.
Mass transitions for nicotine were observed as m/z 162.9 → 131.9 and a qualifier of m/z 162.9 → 116.9. For ethyl maltol, the following transitions were used m/z 140.8 → 126.0 and m/z 140.8 → 70.9.
Transepithelial electrical resistance
On the day after the last exposure, cell culture medium was added to the cells on the apical side and incubated for 5 minutes after which TEER was measured with an Evom2 Volthohmmeter equipped with a chopstick electrode (World Precision Instruments, Inc., FL, USA). An insert without cells was used to correct for background signal.
Cytotoxicity
For Calu-3 cells only, cytotoxicity was measured with the MTS assay [3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium assay] as described previously. 28 For Calu-3 cells and MucilAir cells, cytotoxicity was determined by measuring LDH with the use of a cytotoxicity kit (11644793001; Roche), according to the manufacturer's instructions and as described previously. 28
Cytokine analysis
Cytokine levels in the samples were measured with the use of LEGENDplex™ HU Essential Immune Response Panel (13-plex) (740930, Lot No. B317382 and Lot No. B335764; Biolegend). After rehydrating the standard with 250 μL mQ for 10 minutes 300 rpm, RT, an 1:3 serial dilution was made with assay buffer. Beads were vortexed for 1 minute after which a mixture was prepared with assay buffer, bead solution, and detection antibodies 1:1:1. Of this mixture, 15 μL was pipetted in each well of a 96-well plate (Falcon, V-bottom, pp, REF353263) after which 5 μL of standard, sample, or assay buffer as control was added to the wells.
This was incubated for 2 hours on a plate shaker (Heidolph titramax 100) at 600 rpm, RT, while the plate was protected from light with the use of aluminum foil for all the coming steps of the assay. Next, 5 μL SA-PE was added to all wells and incubated for 30 minutes on the plate shaker at 600 rpm, RT. Wash buffer was prepared by 20 times dilution in mQ and 150 μL was added to all wells. The plate was centrifuged for 5 minutes at 1000 g, setting 7 for acceleration, setting 2 for break (Eppendorf centrifuge 5810R), after which the plate was decanted once to remove the supernatant, pressed onto absorbent paper and turned upside up carefully.
Each pellet was resuspended in 80 μL wash buffer and the fluorescence was measured on the FACS Canto II. Data were then processed with the online LEGENDplex software (BioLegend, Qognit), gating (selection of successful measurements) was reviewed for inconsistencies in the measurements.
RNA isolation and quantitative real-time polymerase chain reaction (qPCR)
MucilAir cells were resuspended by vigorous pipetting and transferred to vials on ice. Vials were then transferred and stored at −80°C until further processing. Whole RNA extraction was performed following the manufacturer's protocol using the Rneasy Lipid Tissue mini kit (Qiagen) with a Dnase digestion step (Qiagen). The RNA concentration was determined using the Qubit3 (Invitrogen, Carlsbad, CA, USA), and RNA quality was analyzed using the 2100 Bioanalyzer (Agilent Technologies, Amstelveen, the Netherlands).
Total RNA (400 ng) was reverse transcribed into cDNA using iScript™ cDNA synthesis kit (Bio-Rad, the Netherlands), including a no reverse transcription control and a no template control. This cDNA was diluted in milliQ (1:50) and stored at −20°C until use.
Real-time quantitative PCR amplification was performed by mixing 4.4 μL of 1:50 diluted cDNA, 5 μL 2xSensiMix™ SYBR® & Fluorescein Kit (Bioline, the Netherlands), and 0.6 μL primers specific for genes of interest in white LightCycler480 384 multiwell plates (Roche, Switzerland) and subsequently running the thermal cycling protocol: 10 minutes at 95°C, 55 cycles of 10 seconds at 95°C, 20 seconds at 60°C on the LightCycler480 machine (Roche).
Qualitative analysis of the melt curves was conducted using LightCycler480 software (Roche) and quantitative gene expression analysis was performed in LinRegPCR software 2014.x (the Netherlands). The geometric mean of a combination of three reference genes (β2-microglobulin [B2M], ribosomal protein L13A [RPL13A] and Cyclophillin A [cyclo]) was calculated in GeNorm software 3.4 (Primerdesign, USA) and used for normalization of the gene expression levels of interest. Samples with no amplification, no plateau phase, too low Cq value, or outside 5% of group mean were automatically excluded by LinRegPCR analysis, as well as qualitative analysis of melt curves and peaks in LightCycler480 software resulted in exclusion of outliers. Primers are listed in Table 1.
Primer Sequences Used to Measure Gene Expression
Data analysis and statistics
For TEER, LDH, and MTS measurements, background values (responses of cell culture medium only) were subtracted from all measurements. For these parameters and for cytokine responses, averages of the three inserts per condition were calculated (each e-liquid, each exposure duration, and, for MucilAir cells, for each donor). For LDH analysis, averages were calculated after determining the relative response compared with LDHmax. The cytokines that had >30% of the values below the detection limit or where the average of air control sample was <20 ng/mL were excluded from further analysis. For LDH, MTS, TEER, cytokine responses and gene expression data significant differences with corresponding air controls were calculated using a two-sided t-test. All plots were prepared in Microsoft Excel.
Results
Air concentrations
For the Calu-3 exposures, air concentrations of ethyl maltol and nicotine were, respectively, 236 ng/mL (± 96 ng/mL) and 287 ng/mL (± 62 ng/mL) as measured in the distribution units (see Supplementary Table S2.1). As samples were collected after exposure, this also allowed sampling from the trumpets. Air concentration measurements in the distribution unit and trumpets showed that ∼35% of ethyl maltol and nicotine in the distribution unit reached the cells, meaning that 65% was lost in the humification tubing (7 in Fig. 1) and the trumpets of the Vitrocell module (8 in Fig. 1). This relatively large loss is mainly due to the additional tubing and humidification before the aerosol reaches the cells.
During the exposure of MucilAir cells, concentrations were measured from the distribution unit only as sampling from the trumpets is not possible during exposure, meaning that losses in the humidification step could not be accounted for. During exposure of cells from the donor 1, the average ethyl maltol concentration was 297 ng/mL (± 103 ng/mL) and the average nicotine concentration was 197 ng/mL (± 75 ng/mL). During exposure of cells from the donor 2, concentrations of ethyl maltol and nicotine were 246 ng/mL (± 78 ng/mL) and 137 ng/mL (± 41 ng/mL), respectively. More detailed information on the concentrations and the individual measurements is given in Supplementary Table S2.2.
The quantity of e-liquid evaporated is the nominal concentration. The average e-liquid use was 13.0 mg per puff (±0.6 mg per puff) and 11.8 mg per puff (±1.2 mg per puff) during Calu-3 and MucilAir exposures, respectively (see Supplementary Table S1). Approximately 80% of ethyl maltol and nicotine in the e-cigarette vapor was measured in the distribution unit.
Barrier integrity and cytotoxicity
TEER was measured pre-exposure (to check for pre-exposure differences in the culture, data not shown) and postexposure (Supplementary Table S4).
Exposure to e-cigarette aerosol gave a small but significant reduction in TEER of Calu-3 cells for the e-liquid with nicotine and ethyl maltol. As the reduction was small, it indicates that the barrier was still largely intact, and the cells were suitable to assess translocation. Similarly, LDH levels of Calu-3 cells were not increased by any of the exposures (basolateral all <10% of LDHmax (Supplementary Table S5). This effect on LDH was, however, not supported by the cell metabolism as measured with the MTS assay, as air exposure showed no effect. Significant reductions in cell metabolism were found for e-liquids with nicotine or with ethyl maltol (not for the e-liquid with both nicotine and ethyl maltol).
The results of TEER measurements postexposure of MucilAir cells can be found in Supplementary Table S4. In general, the donors responded with similar effects on TEER upon exposure but TEER levels were slightly higher for donor 1. Exposure of these cells for two 30-minute periods to e-liquid containing ethyl maltol/no nicotine per day resulted in a decrease in TEER. For donor 2, two 30-minute exposures to both liquids containing ethyl maltol (with or without nicotine) also resulted in a significantly lower TEER. Single 30-minute exposures did not give an effect on TEER, indicating that this exposure was suitable to assess translocation of nicotine and ethyl maltol.
Translocation of nicotine and ethyl maltol
Translocation of nicotine and ethyl maltol across the Calu-3 epithelial barrier was determined once, on the day after the single 30-minute exposure, which is indicative of the total translocation over time as the medium was not refreshed in that period. Both ethyl maltol and nicotine were able to cross the cell barrier. The use of a continuous flow system for exposure means that the exact quantity of nicotine and ethyl maltol reaching the cells is unknown, therefore, translocation is not expressed as a percentage of the total exposure. Results are shown in Figure 3.
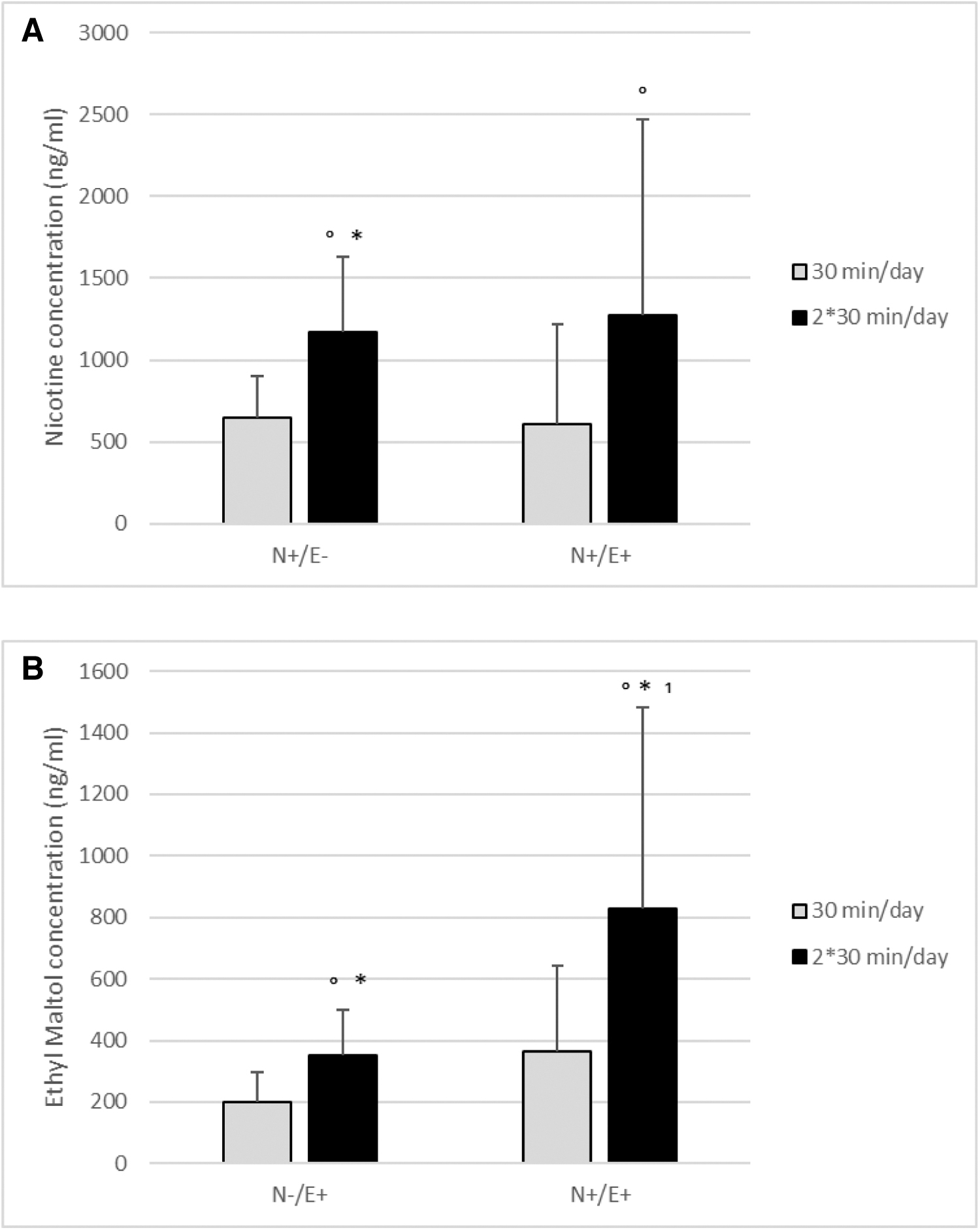
As expected, the exposure duration was associated with the translocation, for example, 60 minutes of exposure resulted in a significantly higher concentration of nicotine and ethyl maltol in basolateral medium compared with 30 minutes of exposure. The relatively large standard deviations reflect the variation between experiments. Still, when comparing concentrations within experiments, the concentrations of ethyl maltol were consistently higher when the cells were also exposed to nicotine (significant for 2 × 30 minutes exposure per day). This suggests that translocation of ethyl maltol is enhanced by the presence of nicotine.
Translocation of nicotine and ethyl maltol was followed over time for MucilAir cells after exposure for 30 minutes to e-cigarette aerosol. Results are shown in Figure 4A and B and more detailed data are given in Supplementary Table S3.
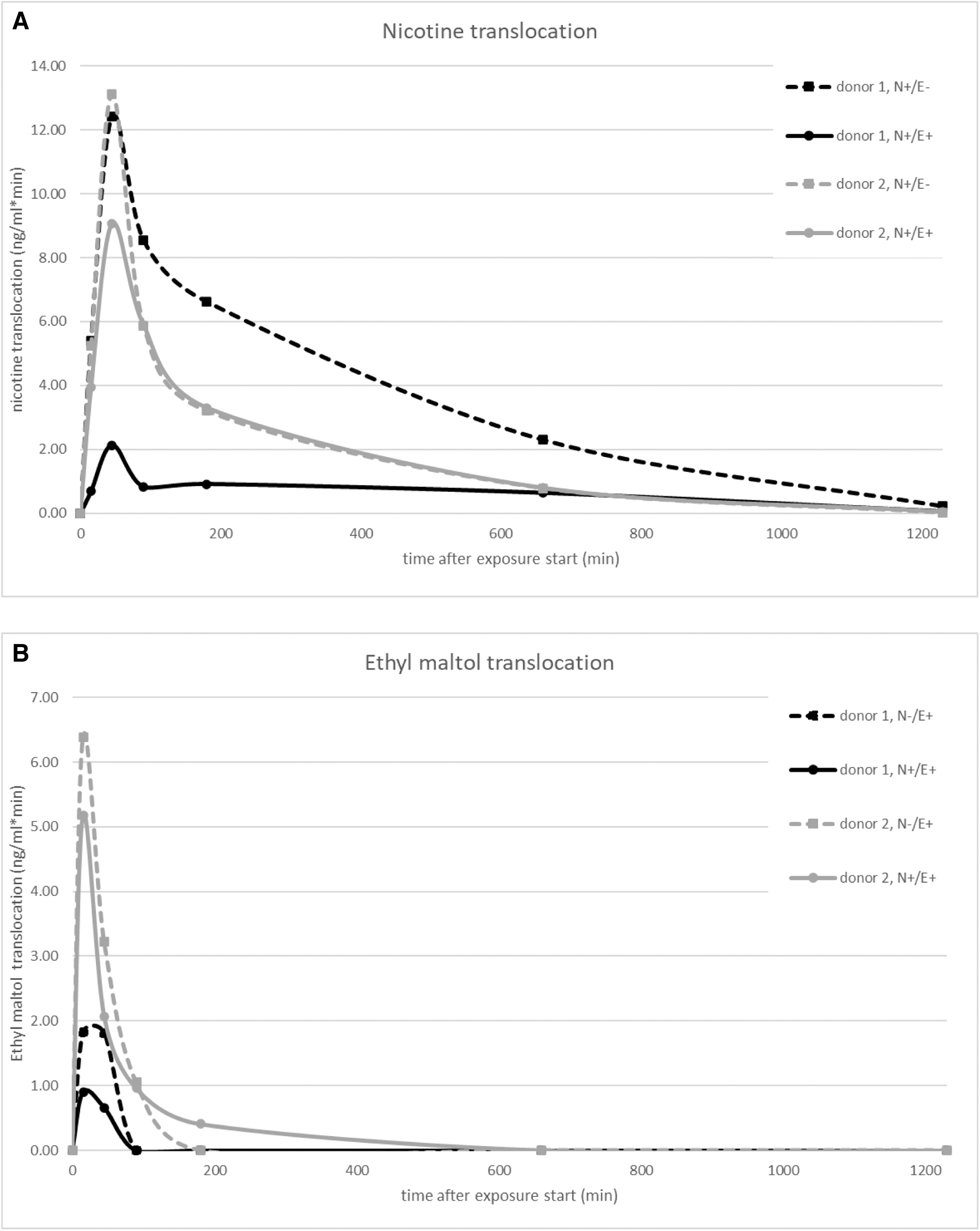
As expected, nicotine was found to cross the cell barrier quickly; the majority was translocated within an hour after the initiation of exposure for all donors and e-liquids. Within donors, there was a difference between translocation of nicotine from the liquid with nicotine only and the liquid containing both ethyl maltol and nicotine; nicotine translocation was faster for N+/E− than for N+/E+ for both donors. There was also a difference between donors, donor 2 was found to have a higher total translocation of nicotine from liquid N+/E+ than donor 1.
Translocation of ethyl maltol across the epithelial layer was even faster than for nicotine. Similarly to nicotine, the total translocation of ethyl maltol was higher for donor 2 than for donor 1.
Inflammatory response
Only cytokine responses showing significant effects by any of the exposures are included. The inflammatory response was measured by determining the production of a selected set of 13 cytokines and chemokines. Responses on the apical side as well as the basolateral side of cell cultures were measured. Exposure of Calu-3 cells resulted in changes in cytokine production on the apical side only (Fig. 5A). Significant changes in cytokine response of MucilAir cells were seen on the apical and basolateral side (Fig. 5B). Supplementary Table S6 contains absolute cytokine responses.
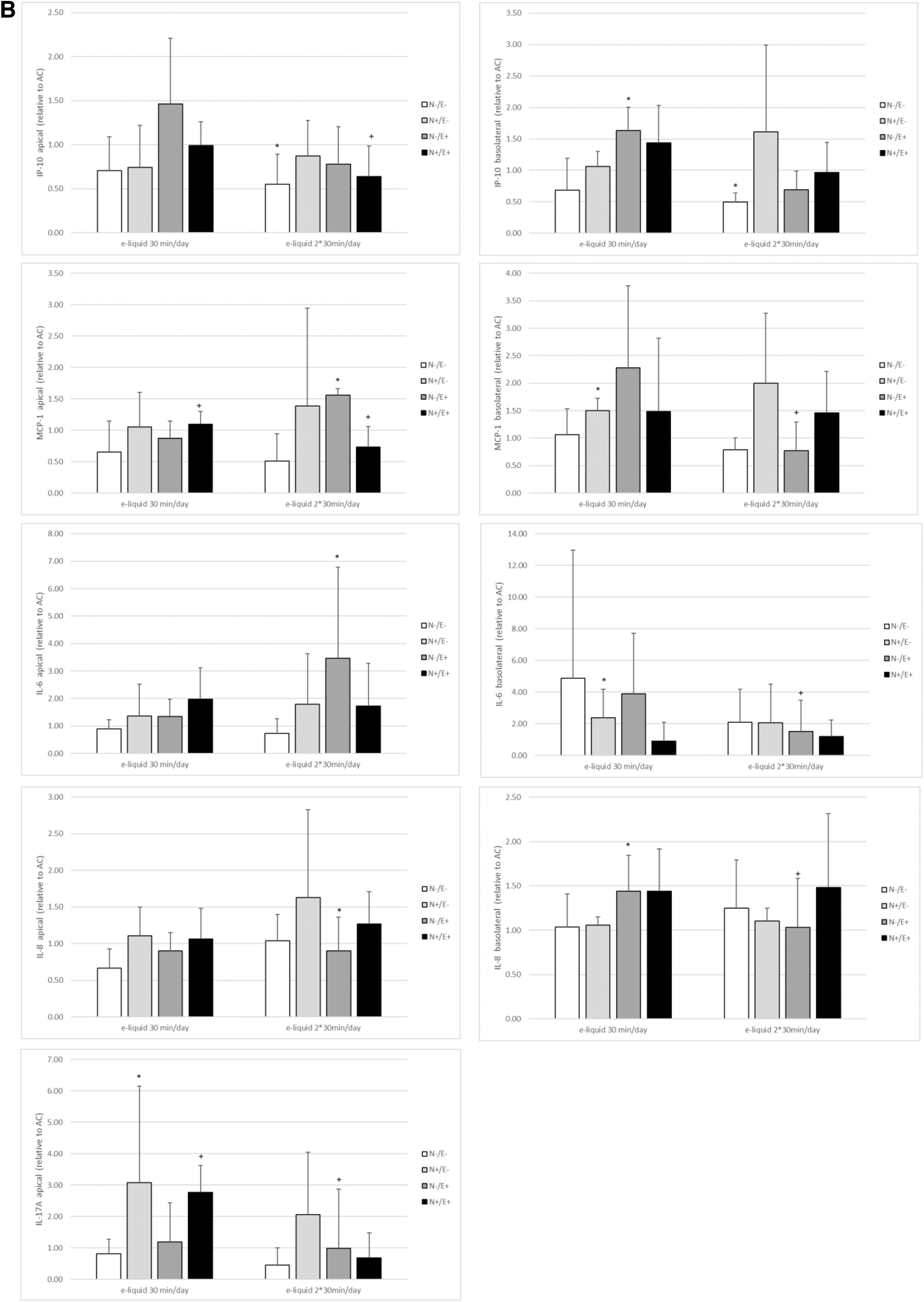
Our data show that some e-liquids induced effects on MCP-1, IP-10, and IL-8, and for MucilAir cells also on IL-6 and IL-17A. The statistically different effects cannot be linked to the presence of nicotine or ethyl maltol in the e-liquids. However, for Calu-3 cells, significant differences were found for nicotine containing e-liquids (liquid N+/E−), whereas MucilAir cells seem to respond mostly to ethyl maltol containing e-liquids (N−/E+ and N+/E+). The carrier only (N−/E−) induced no significant effects on cytokine levels.
IL-8 secretion of Calu-3 cells was decreased after exposure for 2 × 30mins per day, whereas it was increased after exposure for 30 mins per day. IL-8 secretion of MucilAir cells was only increased on the basolateral side. On the apical side of MucilAir cells, the responses were limited. MCP-1 response was slightly decreased in Calu-3 cells exposed to ethyl maltol containing liquids (N−/E+ and N+/E+), but was increased in MucilAIr cells exposed to the liquid with nicotine only (N+/E−).
Gene expression analysis
The expression of IL-8, ICAM-1, MCP-1, SOD, NRF2, CHRNA, and HMOX was determined on the day after the last exposure of MucilAir cells. The expression of MCP-1 and IL-8 was significantly affected (Fig. 6; Supplementary Table S7). Other genes did not show exposure-related changes in gene expression (data not shown). The IL-8 gene expression was increased for ethyl maltol containing liquids (N−/E+ and N+/E+), 24 hours after the last exposure. MCP-1 expression was decreased in nicotine containing e-liquids (N+/E−).
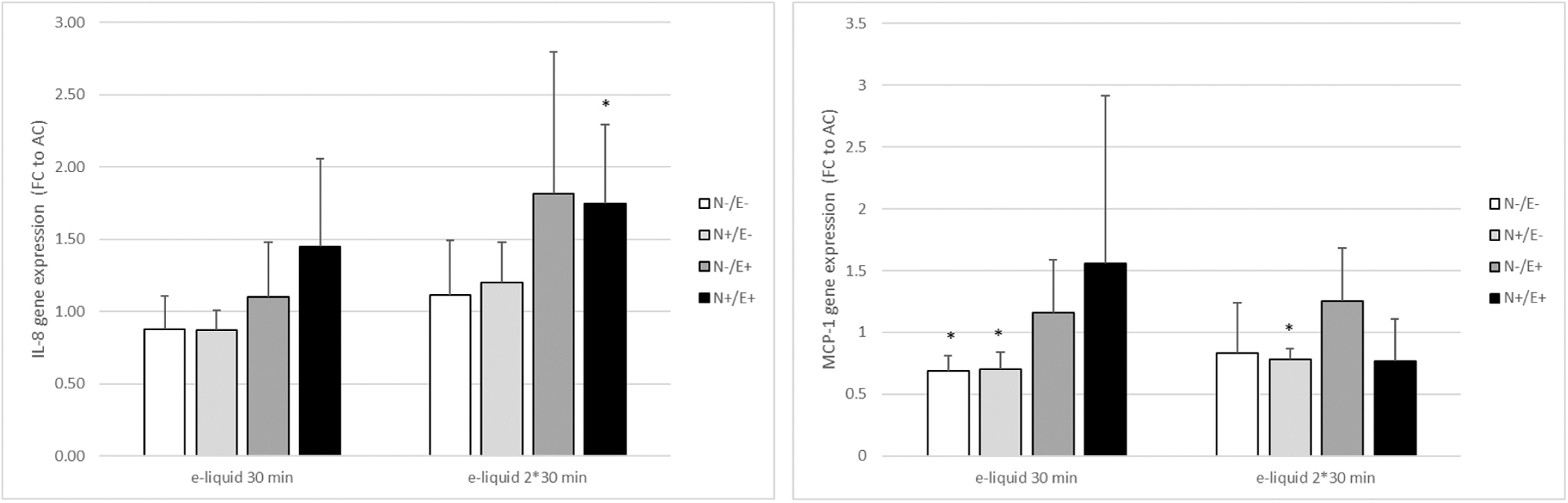
Fold changes in expression of IL-8 and MCP-1 24 hours after the last exposure of MucilAir cells exposed once or twice for 30 minutes per day to an e-liquid aerosol, on 2 consecutive days, relative to air controls. N−/E−: no ethyl maltol, no nicotine; N+/E−: nicotine only; N−/E+: ethyl maltol only; N+/E+: both ethyl maltol and nicotine. *Significant effect compared with air control of the same exposure duration (p < 0.05).
Discussion
The aim of the research described in this article was to apply in vitro models of the airway epithelium to determine whether a flavoring compound from e-cigarettes could induce inflammatory effects and would have the potential to become systemically available. ALI-cultured bronchial epithelial cells, Calu-3 or MucilAir cells, were exposed to e-cigarette vapor containing nicotine and/or ethyl maltol. Our results showed that both cell models responded to this exposure with an inflammatory response, and translocation of nicotine and ethyl maltol could be detected.
Calu-3 versus MucilAir
Our results show a difference between Calu-3 cells and MucilAir cells with respect to their cytokine response. Cytokine levels were only significantly different on the apical side for Calu-3 cells, whereas exposure of MucilAir cells resulted in significant differences on both the apical and basolateral side of the culture. Also the effect and the e-liquid inducing this effect were not consistently the same, meaning that Calu-3 and MucilAIr cells respond differently to exposure to the e-liquids. This is not surprising as the cell models are largely different.
Whereas MucilAir cells are differentiated PBECs, with cilia and a mucus layer, ALI-cultured Calu-3 cells only form cilia after a long ALI-culturing period and do not produce a mucus layer. When cultured at the ALI, Calu-3 cells differentiate to cells that are comparable with the bronchial epithelium. 31 The cell models may differ in receptor expression, but as the mechanism for translocation of ethyl maltol is unknown, it is not possible to explain the observed differences based on the mechanism of translocation.
Our results show that there is likely a difference in transport mechanism between Calu-3 and MucilAir cells for nicotine and ethyl maltol, since in the MucilAir model, ethyl maltol translocation was less in the presence of nicotine, whereas in the Calu-3 model, the opposite was found. MucilAir cells share more characteristics with human bronchial epithelium in vivo than Calu-3 cells, nevertheless information on transport mechanisms is needed to decide which model best predicts human systemic exposure and toxicological effects.
The results of our experiments show quite large variation in translocation, not only between donors, as seen for MucilAir cells, but also between replicates, as illustrated for Calu-3 cells. The variation between experiments cannot be explained by the differences in air concentration, as this was approximately similar between the two replicates. We also found differences in MucilAir cells. As these cells originate from different individuals, a possible explanation could be a difference in uptake of nicotine and ethyl maltol between individuals. Such differences may be important for systemic availability of inhaled nicotine and ethyl maltol and their subsequent toxicological or addictive effects. This is especially important as the blood concentration curve determines systemic exposure and possible systemic effects.
Previous study on the use of ALI-cultured cell models has been conducted using droplet exposure (or quasi-ALI exposure) not air exposure. Calu-3 and MucilAir cells have mainly been used to assess drug transport,.32,33 The application of these cell models to assess transport of chemicals or e-liquid constituents across the epithelial barrier is relatively new and has not been done for airborne compounds before. 34 Previously, we have assessed nicotine translocation in a different cell model (A549), 35 which may be considered a less suitable model to study translocation as it does not form tight junctions, has no mucus layer and no cilia.
Others have also assessed the translocation of glucose in combination with PG and glycerol in PBECs, and found a reduced uptake of glucose in the presence of PG and glycerol. 36
Toxicological response
Exposure of Calu-3 cells to e-cigarette vapor resulted in a response of MCP-1, IL-8, and IP-10, which was consistent with the results found for donor 2 of MucilAir cells, where IL-6 also showed a response. The liquid containing only nicotine (N+/E−) was found to induce most effects on the cytokine response in Calu-3 cells, whereas for MucilAIr cells, the effects on cytokines were mainly found for ethyl maltol containing liquids (N−/E+ and N+/E+). IP-10 response was variable but significant decreases were found for both cell models. The effects could, however, not be directly attributed to either nicotine or ethyl maltol exposure.
Whereas cytokine levels reflect the cytokine production over the past 24 hours, gene expression reflects a momentary response. This may explain differences between cytokine secretion and gene expression response. The expression of IL-8 and MCP-1 was affected in response to e-cigarette vapor exposure, however, we did not find effects on ICAM-1, SOD, NRF2, CHRNA, or HMOX expression although changes were reported previously.37–41 This may be explained by the time of measurement, as the responses of gene expression occur relatively quick after exposure and we measured expression 24 hours after the last exposure.
Previous research showed an inflammatory response after exposure to e-cigarette vapor. 18 Muthumalage et al. found an increase of IL-8 and MCP-1 in lung epithelial cells (BEAS2B or 16-HBE) in response to exposure to e-cigarette aerosol. 42 They also found effects on other cytokines, which we did not find in our experiment, probably due to differences in cell models and/or e-liquid composition. Changes in the expression of inflammatory genes were determined by Pinkston et al., 43 who found an increased IL-6 expression in a coculture model with macrophages.
This increase was most pronounced for macrophages, and to a lesser extent for epithelial cells. The effect of e-cigarette aerosol extracts was also found to have an effect on inflammatory response, 44 where IP-10, IL-6, and IL-8 overlapped with our findings. Other researchers have found similar effects of e-cigarette aerosol, either on monocultures or on cocultures.45–48
Effects of e-cigarette aerosol on mitochondrial function have been reported previously. 49 Although we have not measured mitochondrial function, such findings may explain the effect we have found in the MTS assay, which measures metabolic activity, on Calu-3 cells for the liquids with nicotine only or ethyl maltol only (N+/E− and N−/E+). Surprisingly, we did not observe this for the liquid containing both nicotine and ethyl maltol (N+/E+). Measurement of other parameters for mitochondrial function may help to explain this effect.
The importance of concentration measurements
Concentration measurements for in vitro experiments and more specifically for exposure of ALI-cultured cells are not common practice, which is surprising as it is a standard for in vivo inhalation studies and important to relate in vitro to in vivo effects. 50 Deposition measurements in static exposure systems can be included by measurement of the deposited dose. 51 In our continuous exposure setup, we have included concentration measurements of nicotine and ethyl maltol in air. During exposure, we sampled at a location close to the cells, and after exposure, at the site of the cells during exposure.
The results of these measurements were found to be variable, which is not surprising considering the way of aerosol generation and the specific compounds used. Nicotine is a compound that binds with high affinity to various materials, which greatly affects the amount of nicotine reaching the cells. Nevertheless, the long tubing and piston pump in the smoking machine, together with the tubing toward the distribution manifold, were not accountable for the largest loss of nicotine and ethyl maltol. It appeared that the humidification step, between the distribution manifold and the Vitrocell module, was accountable for the majority of the loss of nicotine and ethyl maltol.
This humidification is required to avoid any cell damage caused by low humidity. These measurements of concentration, at different locations of the aerosol generation setup, show the importance of concentration measurements in ALI-exposed cells. In our experiments, even though a substantial amount of nicotine and ethyl maltol was lost in the tubing, concentration measurements allowed us to quantify the exposure. However, concentration measurements are not common practice yet. Therefore, as others have already proposed,52,53 we argue for inclusion of concentration measurements at the site of the cells in cell exposure experiments, since only this gives information on exposure and allows assessment of dose-related effects, which is important for use of in vitro data in risk assessment. 51
Strengths and limitations
Our data show that both nicotine and ethyl maltol can cross this epithelial barrier. For nicotine, this was already known from human studies, 54 but we are the first to show translocation of ethyl maltol across the epithelial barrier. This is important as also in human airways, inhaled compounds need to cross the epithelial barrier to become systemically available. Further research is needed on the systemic effects of ethyl maltol, as the inhalation route avoids first pass metabolism in the liver and may, therefore, induce different systemic effects than orally consumed ethyl maltol.
We also provide data on the toxicological hazards of inhaling ethyl maltol on bronchial epithelial cells. Only little inhalation toxicity data were available on this compound, whereas this flavoring compound is commonly used in e-liquids, and thus many e-cigarette users will be exposed through inhalation. From a public health perspective, it is important to have information on its toxicity. As no information is available on the uptake mechanism of ethyl maltol, we have used two different cell models. With their different characteristics, they cover different aspects of transport and toxicity.
The effects seen in vitro show that the cells respond differently to exposure to e-cigarette aerosol with nicotine and ethyl maltol. Especially for primary cells, such as MucilAir, the donor variation may lead to high variation in results. Although the results of some measurements indeed show relatively high variation for some parameters, we have seen effects on cytokine production and gene expression. These potential hazards for human exposure through inhalation indicate a need for further research, specifically for ethyl maltol.
In the future, a mechanistic approach in assessment of toxicological effects, such as the adverse outcome pathway approach, would allow more specific identification of the human health risks that could occur. Ideally, a change in cytokine response could, with such an approach, then be related to the chance of development of health effects.
From a practical perspective, our results also show the importance of concentration measurement close to the site of the cells as the quantity that is added to the system, the nominal concentration, which in this case is the e-liquid evaporated, can differ substantially from what actually reaches the cells. This is even more important for (complex) mixtures of compounds. Using only the nominal concentration will result in a shift of the dose–response curve: effects are thought to occur at higher concentrations than they actually do. From a risk assessment perspective, this is especially important as compounds may be hazardous at lower concentrations than assumed.
Implications of this research
Our results show that Calu-3 cells and MucilAir cells can be used for assessment of translocation and toxicological effects of e-cigarette aerosol using a human relevant exposure method. Information on translocation, and subsequent blood concentration curves over time are important for assessment of systemic toxicological effects. This is especially important for mixtures of compounds, in this case as e-cigarette aerosol, as compounds may interact, 36 and the form in which they reach the cells may influence the effects. Such possible interactions are especially important to assess for e-cigarettes and tobacco products as these form a complex mixture of compounds with relatively large changes due to aerosol aging.
Conclusions
Based on our experimental study, we can conclude that both nicotine and ethyl maltol can cross the bronchial epithelial barrier, indicating that both compounds may become systemically available. Furthermore, nicotine and ethyl maltol induced inflammatory effects on bronchial epithelial cells. Further research should, therefore, focus on local and systemic effects of inhaled e-cigarette aerosols, using human relevant exposure and toxicity parameters.
Footnotes
Acknowledgments
The authors thank Wouter Visser for his help with the choice of e-cigarette and e-liquids, Michele Davigo for his contribution to the gene expression measurements, and Rob Vandebriel and Peter Keizers for critically reviewing the article.
Authors' Contributions
Y.C.M.S. contributed to conceptualization, data curation, formal analysis, methodology, project administration, supervision, visualization, and writing (original draft, review, and editing). E.G. and G.D. were involved in cell culture and toxicological read-outs—investigation, data curation, and methodology. E.D. and P.F. carried out continuous exposure of cell models and air sampling—investigation, data curation, and methodology. D.L. took charge of chemical analysis—investigation, data curation, and methodology.
H.M.H. took charge of RNA isolations—investigation, data curation, and methodology. L.M. carried out gene expression measurements—investigation, data curation, and methodology. A.R. was in charge of data curation (gene expression data), formal analysis (gene expression data), and writing (review). R.T. was in charge of conceptualization (supporting), funding acquisition, project administration, supervision, and writing (review).
Author Disclosure Statement
No competing financial interests exist.
Funding Information
This research was funded by the Dutch Ministry of Health, Welfare and Sports (project number 5.7.1).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
