Abstract
Introduction:
The Vitrocell® Mammalian 48 exposure module offers a high throughput platform for air-liquid interface (ALI) exposures that mimic human exposure conditions for assessing the potential cytotoxicity of a test article. The aim of this study was to evaluate the in vitro cytotoxic response of whole smoke in Chinese hamster ovary (CHO) cells, using the Neutral Red Uptake Assay across two modules. The secondary aim was to further characterize the system utilizing three dosimetry techniques.
Methods:
Whole smoke generated from 3R4F Kentucky Reference cigarettes was examined over a diluting airflow range 10 to 1 L/min. Cigarettes were smoked according to both standard and intense International Organisation for Standardisation (ISO) smoking regimens. Aerosol photometers (using area under curve, AUC), fluorescence of anhydrous dimethyl sulfoxide (DMSO), and nicotine quantification. Photometer AUC, particulate deposition (measured by fluorescence), and nicotine determination were used to characterize the modules.
Results and Discussion:
Dosimetry measures (AUC, fluorescence, nicotine) demonstrated consistent aerosol delivery across rows within and between the Vitrocell® Mammalian 48 exposure modules. Cytotoxicity was achieved using the intense ISO smoking regimen with IC50 calculated as 5.6 ± 2.3 and 3.8 ± 1.6 L/min, respectively for both modules, with no statistical difference observed between the modules (p > 0.05). We have also demonstrated that the cytotoxic response with ISO smoking regimens is consistent with previously published data using Vitrocell® 12/4 modules.
Conclusion:
The data in this study support the validation and characterization of the Vitrocell® Mammalian 48 module for whole smoke exposure at the ALI for cytotoxicity assessment with the Neutral Red Uptake assay.
Introduction
Cigarette smoke is a complex aerosol consisting of both gaseous and particulate phases. The latest research indicates more than 7000 unique chemical components can be found in cigarette smoke 1 with at least 98 compounds considered hazardous to human health. 2 Even though smoking rates have decreased in more developed countries, smoking is among the leading causes of premature death 3 and diseases caused by smoking include are not limited to cardiovascular disease, chronic obstructive pulmonary disease, stroke, and cancer. 4
Regulatory agencies have started to implement guidance for the submissions of nonclinical data to assess the potential harm to human health of tobacco and next-generation products (NGPs).5–8 In recent years, Electronic Nicotine Delivery Systems (ENDS) with multiple flavors and nicotine levels as well as heated tobacco products (HTPs) have become increasingly popular.9–11 As a result of this new and evolving class of products, there are less extensive toxicological data sets available. Therefore, new technologies and equipment are required to increase experimental throughput and the understanding of biological consequences to make appropriate assessments of potential toxicological effects.
Traditional testing methods, such as those published by regulatory agencies,6,12 have recommended testing for genotoxicity and cytotoxicity using the gaseous and particulate fractions either separately or in combination thereof. These assays have been used historically for product assessment.13–17 However, these assays may not be able to fully evaluate the complex aerosol that is generated from tobacco or NGPs. For the past decade, there has been an increased move to test systems that can be exposed to the whole aerosol at the air–liquid interface (ALI).18–24
Exposures at the ALI have the added advantage that both phases of the aerosol interact with the biological assay while maintaining and allowing any chemical interactions between both phases and transient chemical reactions to interface with the test system, which would be missed in standard in vitro testing practices. In addition, exposure at the apical side of the ALI cultures is able to create an environment more comparable with that of “real life” exposure of tobacco products and NGPs since the exposure uses the whole smoke/aerosol.
There are some limitations with the testing of the particulate and vapor fractions as the interactions that occur between the two phases are not assessed and the loss of volatile compounds particularly from the gaseous phase may occur before application in the test system. As a benefit, however, it has been shown that there are no effects of long-term storage on particulate matter genotoxicity and cytotoxicity responses. 25 Trapping procedures may not represent the complete chemical composition of the aerosol due to the selective capture of aerosol constituents.
For these reasons there has been movement into whole aerosol exposures at the ALI. However, exposures at the ALI introduce a level of complexity in terms of aerosol generation, handling, and delivery that is not observed in the standard aerosol collection and exposure methods. There are a wide variety of smoke generating machines available by various companies, including Vitrocell® Systems GmbH (Waldkirch, Germany; e.g., Vitrocell VC10®),26–28 Borgwaldt KC (e.g., Borgwaldt RM20S, LM4E),29–31 Burghart Messtechnik GmbH (SAEVIS smoking machine),23,24 and some in-house made examples.32,33
These smoking engines are then attached to the cell exposure modules through a dilution system, examples of which are available from Vitrocell Systems GmbH,20–22,34 Cultex® modules, 18 and bespoke examples.35,36 Even though there are many system configurations available, the concept is consistent in delivering fresh whole aerosol to the cell cultures at the ALI. What is most important is any system being used is understood through characterization, qualification, and validation.
The primary aim of this study was to evaluate the cytotoxic response of whole smoke generated from 3R4F Kentucky Reference cigarettes utilizing the Vitrocell Mammalian 48 module system with a Vitrocell VC10 smoking machine and compare with previously generated data. The secondary aim was to further characterize the system by utilizing three dosimetry techniques; aerosol photometers (using area under curve, AUC), fluorescence of anhydrous dimethyl sulfoxide (DMSO) as previously described 28 and analytical determination of nicotine in DMSO. The Neutral Red Uptake (NRU) assay is based around guidance from the Interagency Coordinating Committee on the Validation of Alternative Methods. 37
The NRU assay is a cell survival/viability assay based on the ability of viable cells to incorporate and bind neutral red, a supravital dye. Neutral red is a weak cationic dye that readily penetrates cell membranes by nonionic diffusion and accumulates intracellularly in lysosomes.38,39 Thus, the NRU assay examines the cellular membrane integrity as well as the cellular energy status since Neutral red is taken up into lysosomes by an energy-dependent process. Changes brought about by the action of xenobiotics result in a decreased uptake and binding of neutral red. It is thus possible to distinguish between viable and damaged or dead cells, which is the basis of this assay.
The NRU assay and other cytotoxicity endpoints are part of a battery of assays used to assess the toxicological profile of a chemical, including chemical mixtures, and has been recommended as part of Cooperation Centre for Scientific Research Relative to Tobacco (CORESTA) and Health Canada. 40 In addition, this document also recommends the use of the Bacterial Reverse Mutation Test41,42 commonly known as the Ames Assay and a mammalian mutagenicity assay either the in vitro micronucleus (IVMN) assay 43 or the Mouse Lymphoma Assay (MLA). 44
The aforementioned assays, including the NRU assay, have been used as biological endpoints to assess either whole smoke from cigarettes or whole aerosol from ENDS and other HTPs.19–22,34,45–48 Thorne et al. demonstrated that an ALI approach is feasible for the MLA. 49 These assays can help contribute to an in vitro toxicological assessment of the aerosol test article in its natural state as opposed to fractionation or constituent testing. The ability of having these in vitro assays conducted at the ALI also aligns with the testing requirements of ICH 50 and UKCOM. 51
The aim of this study was a qualification of the Vitrocell Mammalian 48 exposure module using the NRU assay and substantiate the results against previously published data. A successful validation will allow further alignment with toxicological testing requirements, increasing the throughput and allowing a holistic assessment of the whole smoke/aerosol.
Materials and Methods
Chemicals and reagents
Chemicals and reagents were obtained from the following suppliers: saline (CAS No. 7647-14-5) from Bunzl Healthcare (London, United Kingdom), neutral red (aqueous 1%; CAS No. 553-24-2) from Pyramid Innovation (Polegate, United Kingdom), ethanol (CAS No. 64-17-5) from Hayman Ltd. (Witham, United Kingdom), acetic acid (CAS No. 64-19-7) from Merck Millipore (Darmstadt, Germany), phosphate buffered saline (PBS; CAS No. 20-74-10) from Severn Biotech Ltd. (Kidderminster, United Kingdom) and trypsin-EDTA (CAS No. 9002-07-7) from Sigma-Aldrich Co. Ltd. (Gillingham, United Kingdom). Tissue culture media (McCoy's 5A) was obtained from Invitrogen Life Technologies (Paisley, United Kingdom).
Product used
3R4F Kentucky Reference cigarettes were obtained from the University of Kentucky. Before smoking, cigarettes were conditioned for at least 48 hours and no more than 10 days at 22°C ± 1°C and 60% ± 3% relative humidity. 52
Cell culture preparation
CHO-WBL cells supplied by the European Collection of Cell Cultures were maintained in tissue culture flasks in McCoy's 5A medium (supplied containing HEPES buffer) supplemented with 10% (v/v) heat inactivated fetal bovine serum and 0.52% penicillin/streptomycin (McCoy's 5A complete) at 37°C ± 1°C in an atmosphere of 5% ± 1% (v/v) CO2 in the air. Cultures were subcultured regularly to prevent overgrowth. Cells were not used beyond passage 15 after removal from liquid nitrogen. The measured cell cycle time of the cells was ∼13 hours.
Near confluent cultures were trypsinized and resuspended in McCoy's 5A complete. The cell number in the suspension prepared was determined using a Coulter counter. All Transwell™ used (including blanks) were presoaked for 1 hour at 37°C ± 1°C in McCoy's 5A complete to allow for equilibration of the membrane before cell seeding. For 24 mm Transwell (high-throughput modules) a concentration of 1 × 105 cells/mL were seeded into the required number of Transwell in 2.0 mL of McCoy's 5A complete media.
In addition, 2.0 mL of McCoy's 5A complete was placed in the well underneath the Transwell. For 12 mm Transwell (12/4 standard modules) a concentration of 7.5 × 104 cells/mL was dispensed into the required number of 12 mm Transwell in 0.5 mL McCoy's 5A complete. In addition, 1.5 mL of McCoy's 5A complete was placed in the well underneath the Transwell. All Transwell were incubated at 37°C ± 1 C for 18–24 hours in a humidified atmosphere of 5% (v/v) CO2 in the air to achieve approximately half confluent monolayers. Blank Transwell had McCoy's 5A complete added, (i.e., media with no cells present, 2.0 mL in Transwell and 2.0 mL beneath Transwell for 24 mm, and 0.5 mL in Transwell and 1.5 mL beneath Transwell for 12 mm).
Vitrocell VC10 smoking robot and Mammalian 48 high-throughput module
Cigarette smoke was generated using a Vitrocell VC10, serial number VC10/210311 (Vitrocell Systems GmbH). Different concentrations of whole smoke were achieved by altering the diluting airflow using mass flow controllers (Analyt-MTC GmbH, Mülheim, Germany). For all experiments, concentrations were expressed in terms of airflow in liters per minute (L/min). The vacuum rate was fixed at 5 mL/min. Operation and confirmation of functionality, including installation, operational, and performance qualification, were performed as previously described. 21
Cigarettes were smoked according to the International Organisation for Standardisation (ISO) intense smoking regimen42,53 (55 mL puff, 2 seconds duration, 30 seconds frequency, with 100% vent blocking). Cigarettes were also smoked according to the International Organization for Standardization 3308, 54 namely 35 mL puff volume, 2 seconds puff duration, 60 seconds frequency with a bell-shaped puff profile and no vent blocking to compare with previously published data. 21
This study utilized the Vitrocell Mammalian 48 exposure module climatic chamber. The Vitrocell Mammalian 48 module utilizes the same technology as previously described;19,21,22,26 however, this equipment allows higher throughput with up to seven doses and one air exposed control, all with up to six replicates per dose (Fig. 1). Additional individual dosimetry modules were located on each of the seven dose rows to allow quantitative assessment of the delivered dose. Exposures conducted in the Vitrocell 12/4 exposure modules that were used to compare with the results in the HTP modules were conducted as previously described. 21

Vitrocell® Mammalian 48 exposure module. Image supplied by Vitrocell Systems GmbH (Waldkirch, Germany).
Photometers
Photometers were purchased from Vitrocell Systems GmBH. For 3R4F combustible cigarettes, seven photometers were harmonized to whole smoke using the ISO intense regimen and a diluting airflow of 1.5 L/min so that the voltage for each photometer was ∼4.0 V. Harmonization was performed as previously described. 28 Each photometer was connected to the VC Photometer Control Box, which was then connected to a computer. Photometer data were output as both comma separated value (CSV) and virtual compact disk (VCD) file formats.
DMSO fluorescence
3R4F pad-collected total particulate matter (TPM) in DMSO at 24 mg/mL was serially diluted (twofold dilutions) with DMSO (0.0024–5 mg/mL). The Ex355/Em485 readings were taken from these dilutions to establish a standard curve of relative fluorescent units (RFU) versus TPM concentration (mg/mL). Immediately postexposure, all DMSO samples were retained and triplicate 100 μL aliquots from each well were plated into 96-well plates.
Analysis was performed using a VERSAmax™ Microplate reader (Molecular Devices, San Jose, CA). Values were represented in RFU485, the mean values for each smoke exposure were subtracted from the mean RFU for the air control to give relative fluorescence per smoke dose. An equivalent TPM concentration was extrapolated from the standard curve.
Nicotine
Samples in DMSO were separated using a binary gradient and a Phenomenex Kinetex 2.6 μm EVO C18 High-performance liquid chromatography (HPLC) column. Detection and analysis of nicotine was performed using a triple quadrupole tandem mass spectrometer utilizing electrospray ionization (ESI) and multiple reaction monitoring (MRM). The linear range of the method was ∼5–500 ng/mL with an limit of quantification (LOQ) defined as the nominal concentration of the lowest level calibration standard and an limit of detection (LOD) defined as 1.5 ng/mL.
Controls
For all experiments, incubator controls (IC), ALI controls, and positive controls were included. IC consisted of Chinese hamster ovary (CHO) cells cultured in Transwell in McCoy's 5A complete at 37°C ± 1°C and incubated in an atmosphere of 5% ± 1% (v/v) CO2 in air, for the same “exposure” time as the cultures at the ALI. ALI controls consisted of CHO cells cultured in Transwell in McCoy's 5A complete, exposed to air at the ALI at a flow rate of 0.2 L/min.
Data from cells subject to mainstream aerosol exposures were compared with the ALI controls to provide a relative assessment of survival/toxicity. Positive controls comprised CHO cells cultured in Transwell and treated with 2000 μg/mL sodium dodecyl sulfate (SDS) in McCoy's 5A complete at 37°C ± 1°C and incubated in an atmosphere of 5% ± 1% (v/v) CO2 in air, for the same “exposure” time as the cultures at the ALI.
Whole smoke exposures
3R4F Kentucky Reference Cigarettes were evaluated for cytotoxicity in six separate experiments using the ISO intense smoking regimen 53 on two Mammalian 48 systems (serial Nos. VC48 260417 and VC48 081217) with a total number of three cigarettes (33 puffs in total). Dilution airflow rates of 10, 8, 7, 6, 5, 4, and 2 L/min were evaluated, using five replicate Transwell inserts per concentration and control. In addition, three experiments were conducted with the ISO intense smoking regimen 53 using the standard Vitrocell 12/4 modules with a total number of three cigarettes (33 puffs in total).
Dilution airflow rates of 10, 8, 4, and 1 L/min were evaluated using three replicate Transwell inserts per concentration and control. Finally, three experiments were evaluated for cytotoxicity using the ISO smoking regimen (ISO 3308) conducted using two Mammalian 48 systems with a total number of eight cigarettes (64 puffs in total). Dilution airflow rates of 10, 8, 7, 6, 5, 4, and 2 L/min were evaluated using five replicate Transwell inserts per concentration and control.
Before the exposure, media was carefully aspirated out of the apical side of each Transwell. Transwell for ALI controls and mainstream aerosol dosing were transferred to exposure chambers, with each well previously filled manually with 7 mL McCoy's 5A complete. IC and blank Transwell had 2 mL fresh McCoy's 5A complete added apically to the Transwell, and 2 mL was placed in the well underneath the Transwell.
Positive control Transwell had 2 mL fresh McCoy's 5A complete containing a final concentration of 2000 μg/mL SDS added apically to the Transwell, and 2 mL fresh McCoy's 5A complete containing a final concentration of 2000 μg/mL SDS was placed in the well underneath the Transwell. For 12 mm Transwell (12/4 standard modules) ALI controls and mainstream aerosol dosing were transferred to exposure chambers, with each well previously filled manually with 3.5 mL McCoy's 5A complete.
IC and positive control Transwell were transferred to fresh plates. IC and blank Transwell had 0.5 mL fresh McCoy's 5A complete added apically to the Transwell, and 1.5 mL was placed in the well underneath the Transwell. Positive control Transwell had 0.5 mL fresh McCoy's 5A complete containing a final concentration of 2000 μg/mL SDS added apically to the Transwell, and 1.5 mL fresh McCoy's 5A complete containing a final concentration of 2000 μg/mL SDS was placed in the well underneath the Transwell. Control (incubator and positive) and blank Transwell were incubated at 37°C ± 1°C in a humidified atmosphere of 5% (v/v) CO2 in air for the length of the exposure.
In each whole smoke exposure experiment, the seven individual externally mounted dosimetry modules (one for each diluting airflow), and the third well in each airflow contained a stainless steel Transwell insert containing 3 mL of DMSO. One “in-line” photometer was placed between the externally mounted dosimetry module and the dilution system. For 12 mm Transwell (12/4 standard modules) 0.9 mL of DMSO was aliquoted into a stainless steel Transwell in the first position of the modules.
The whole smoke was exhausted from two ports of the VC10 and directed to the first and fifth rows in dilution system, respectively. The smoke was then serially diluted through the addition of diluting air through the air inlets to achieve the desired concentration for the diluted smoke doses (Fig. 2).
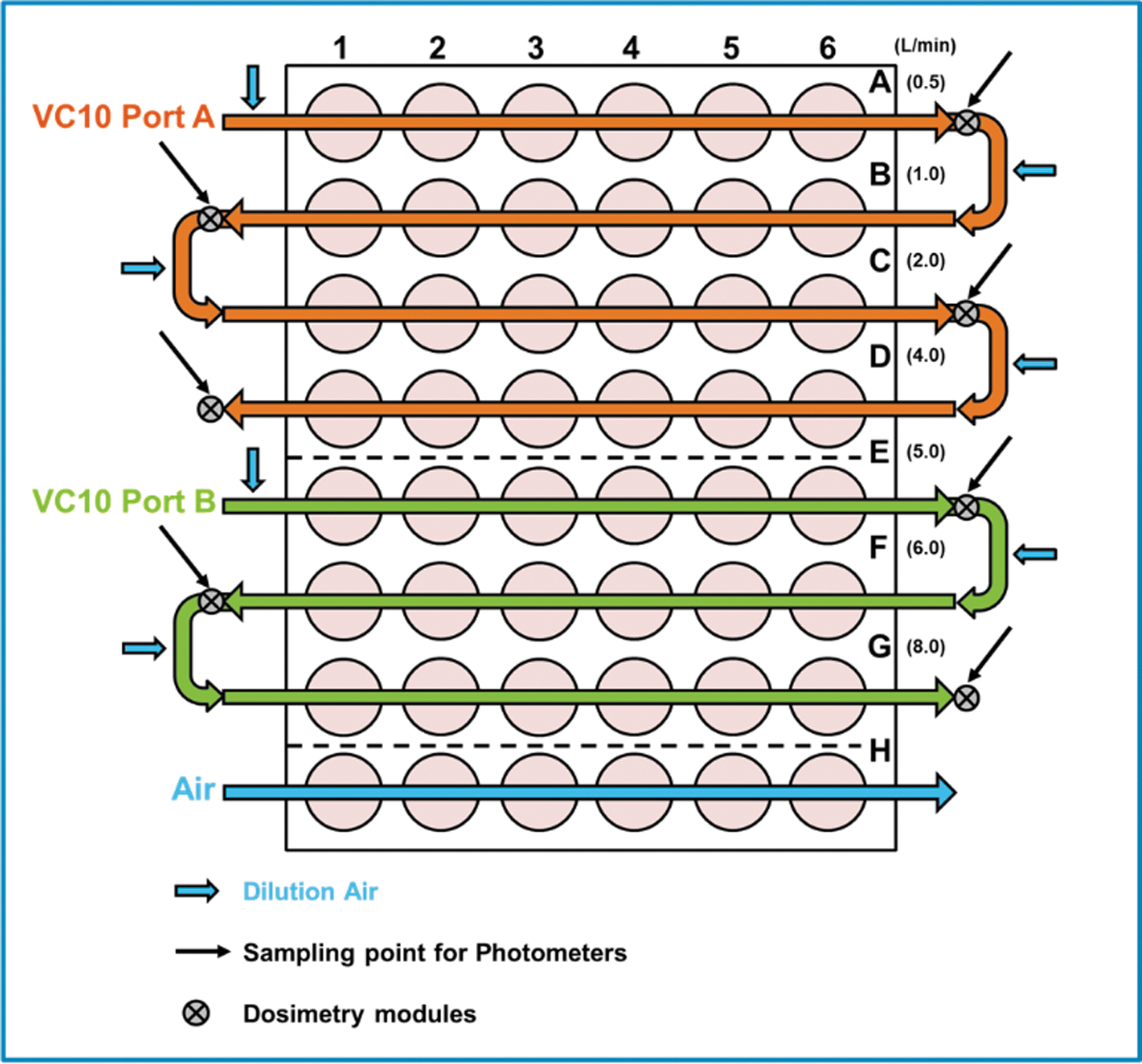
Schematic representation of airflows; diluting airflows from rows A to D for 2–6 L/min, and rows E to G for 7–10 L/min.
Neutral red uptake assay
Following the microscopic evaluation after the 24-hour postexposure period, media was aspirated from the Transwell and 1.75 mL (0.35 mL 12/4 modules) of neutral red solution (50 μg/mL) was added to each Transwell and 2.0 mL (0.5 mL 12/4 modules) of neutral red solution (50 μg/mL) was placed in the well underneath the Transwell. Neutral red solution was also added to blank Transwell (no cells) that had been equilibrated as described previously. All Transwell were incubated at 37°C ± 1°C for 3 hours ±10 minutes in a humidified atmosphere of 5% ± 1% (v/v) CO2 in the air. After incubation, cells were examined microscopically for signs of cytotoxicity.
Immediately after the visual assessment, the neutral red solution was removed, and the cells were washed three times through a series of three large dishes containing PBS. The PBS rinse was decanted from the Transwell after each wash. After the third wash, the Transwell were tilted at an angle onto absorbent tissue (e.g., Kimwipe™) to drain the residual PBS rinse and then the bottom of each Transwell was blotted dry.
After washing, 2.2 mL (0.5 mL 12/4 modules) neutral red destain solution (ethanol:acetic acid:distilled water 50:1:49 [v/v/v]) was added to each Transwell. Plates were shaken on a plate shaker for ∼40 minutes until it was considered that all the neutral red had been extracted from the cells. Neutral red destain solution was removed from the Transwell and placed in the well below. Transwell were then discarded. One hundred microliters aliquots of neutral red destain solution was placed into each of 3 wells on a flat bottomed 96-well plate for each Transwell.
Analysis was performed using a SpectraMax™ plate reader at a wavelength setting of 540 nm. Neutral red absorbance was expressed in terms of optical density (OD540).
Data analysis
A calculation of cell viability expressed as NRU was made for each condition by using the mean neutral red absorbance of the replicate Transwell, corrected by the mean blank Transwell value and the plate blank (destain only). The mean OD540 value for each whole smoke exposure dilution was compared with the mean OD540 value for the concurrent ALI control. Relative cell viability was expressed as a percentage of the concurrent ALI control. Percentage survival and standard deviation were calculated for each mainstream smoke exposure dilution. The statistical method used to calculate IC50 values was a Sigmoidal model with the top parameter fixed at 100 as detailed hereunder:
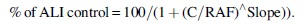
The top parameter (fixed at 100 above) is the maximum percentage of ALI survival that the model can attain. Parameter C is the Reciprocal Air Flow for which the percentage of ALI survival is equal to half the top parameter ( = 50% with top = 100). With top parameter = 100, IC50 = C. The slope parameter reflects the steepness of the descent of the model.
Photometer data were output as both CSV and VCD file formats. Voltage for each airflow was recorded every second and from this an area under the curve value was calculated for each experiment. For DMSO captured material an equivalent TPM concentration was calculated using the Ex355 and Em485 readings against a standard curve of known 3R4F ISO intense TPM concentrations (0.0024–5 mg/mL). Nicotine values were reported in ng/mL.
Results
NRU assay
NRU data using the ISO intense regimen 53 and ISO regimen 54 are presented in Table 1; data are presented as the mean from six individual experiments on the two Mammalian 48 high-throughput modules (named HTP-1 and HTP-2) for ISO intense regimen and three individual experiments per ISO regimen. For HTP-1 and HTP-2 Mammalian 48 systems, whole smoke induced cytotoxicity was observed in all experiments with marked decreases >50% relative to the concurrent ALI control.
Mean % Survival with Standard Deviation (n = 6 Experiments) from Chinese Hamster Ovary Cells Exposed to Whole Smoke from 3R4F Kentucky Reference Cigarettes Using ISO Intense Smoking Regimen (ISO:20778 2018) and ISO Smoking Regimen (ISO:3308 2012) with n = 6 and n = 3 Independent Experiments, Respectively
ALI, air–liquid interface; HTP, heated tobacco product; ISO, International Organisation for Standardisation; PC, positive control.
For all six experiments, the positive control resulted in <50% relative survival. The mean airflow inducing a 50% inhibition in Neutral Red Uptake (IC50) was calculated at 5.6 ± 2.3 L/min for HTP-1 and 3.8 ± 1.6 L/min for HTP-2. The response was concentration related and reproducible.
The responses from the mean % Survival were compared between the two Vitrocell Mammalian 48 high-throughput modules using an unpaired t-test with Welch's correction (Fig. 3), nicotine was used as the dosimetry marker (Table 2). No significant differences in the mean % Survival responses were observed with p-values of 0.5385 and 0.6782, respectively, for ISO intense smoking regimen 53 and ISO smoking regimen. 54 For ISO intense smoking regimen 53 IC50 values were estimated at 344.7 ± 22.77 and 449.7 ± 18.07 ng/mL nicotine for HTP-1 and HTP-2, respectively. For ISO smoking 54 IC50 values were estimated at 996.1 ± 64.38 and 872.0 ± 77.28 ng/mL nicotine for HTP-1 and HTP-2, respectively.
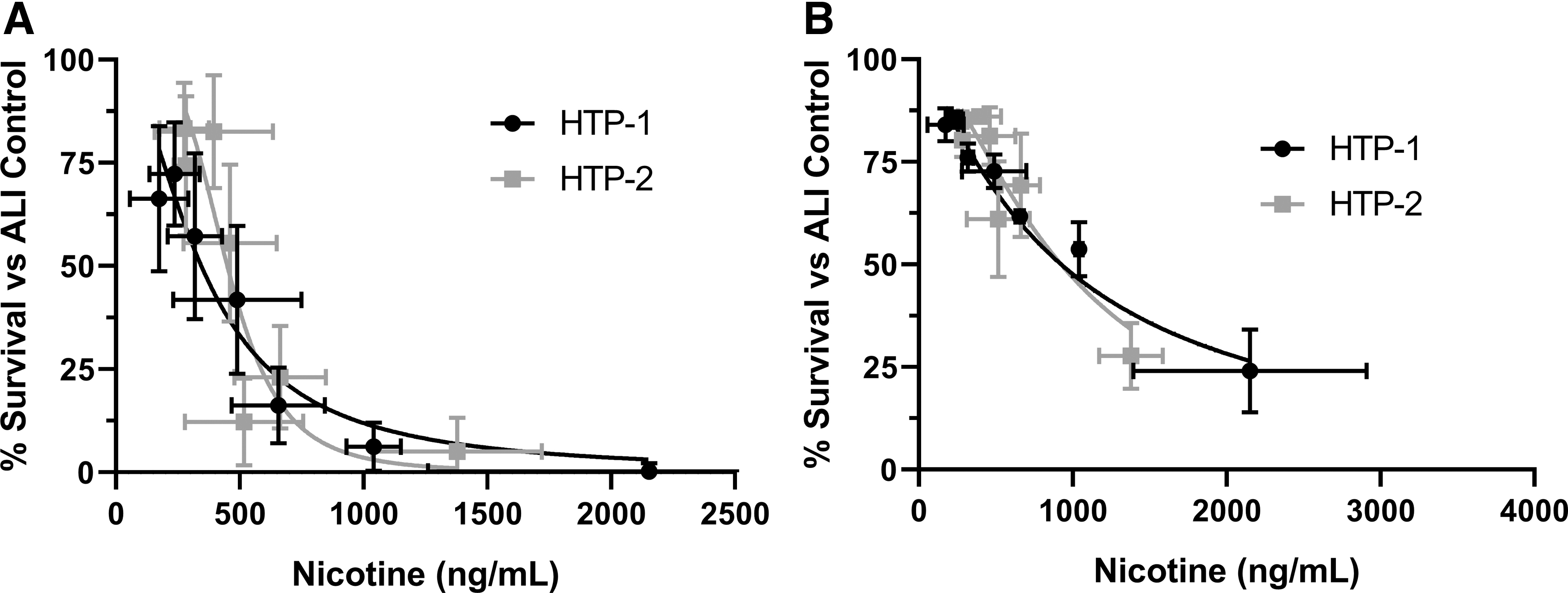
CHO cytotoxicity presented as % Survival versus ALI control using nicotine (ng/mL) for Mammalian 48 high-throughput modules HTP-1 and HTP-2.
Nicotine Values from Vitrocell Mammalian 48 Modules 1 and 2 and Vitrocell 12/4 Modules Using 3R4F Kentucky Reference Cigarettes Using the ISO Intense (n = 6 Heated Tobacco Product Modules) and ISO (n = 3) Smoking Regimes
n = 4 for each airflow.
Values <LOQ, LOQ used for analysis.
LOQ, limit of quantification.
Exposures were performed using the ISO intense regimen 53 in the Vitrocell 12/4 modules to compare the cytotoxicity response in the Vitrocell Mammalian 48 High-Throughput module, data are shown in Table 3 (airflow) and presented graphically in Figure 4 (TPM). Data from both Vitrocell Mammalian 48 High-Throughput modules were pooled for the comparison. The responses from the mean % Survival were compared between the two Vitrocell exposure systems using an unpaired t-test with Welch's correction (Fig. 4). IC50 values were estimated at 18.07 ± 1.64 and 15.53 ± 3.12 μg/mL TPM equivalent for HTP modules and standard 12/4 modules, respectively, with no observed significant differences (p = 0.5523).
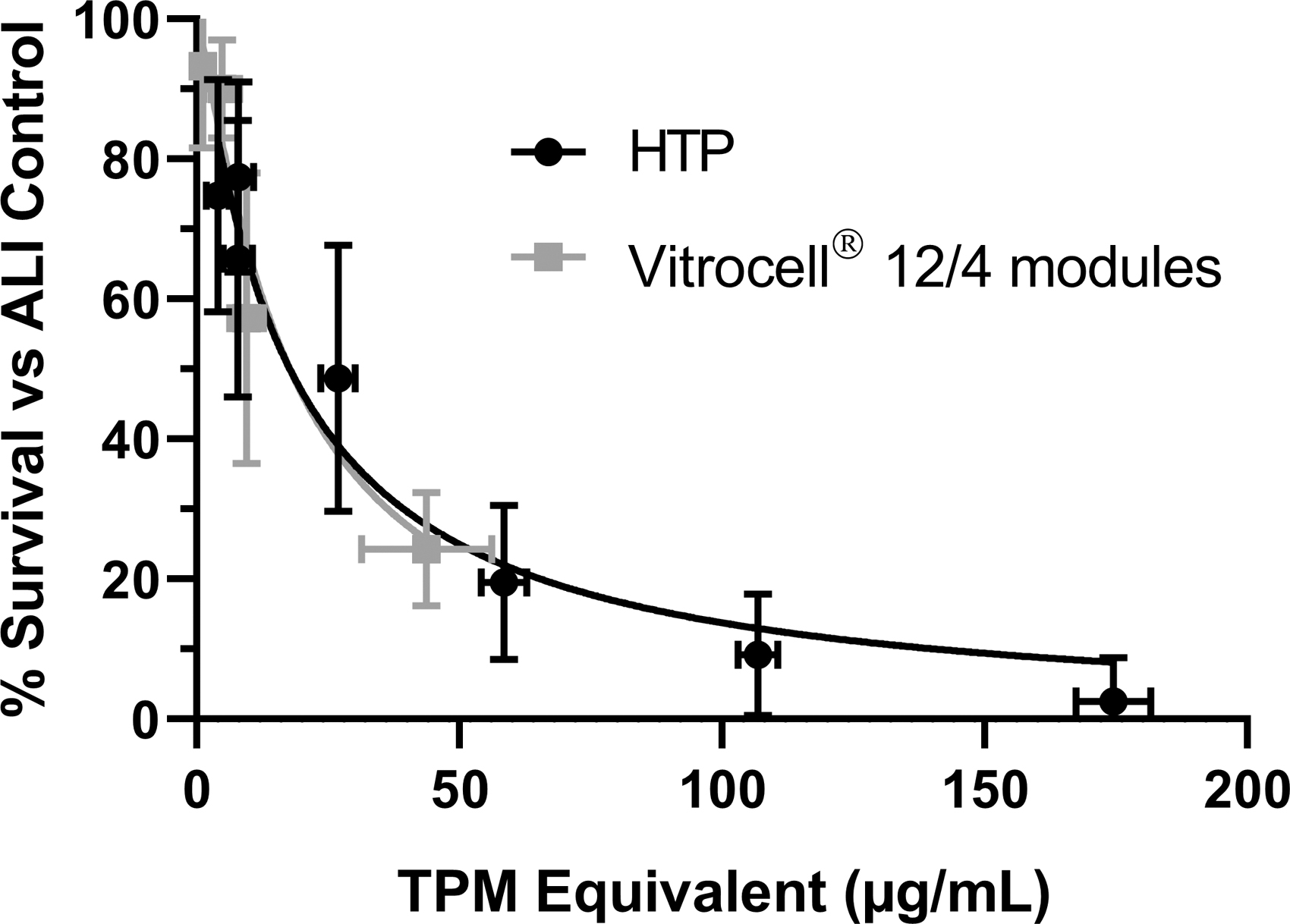
CHO cytotoxicity presented as % Survival versus ALI control using reciprocal of airflow rates for both the Mammalian 48 high-throughput modules (averaged response from HTP-1 and HTP-2) and the Vitrocell 12/4 modules.
Heated Tobacco Product and 12/4 Module Mean % Survival with Standard Deviation (n = 12 Experiments from Both HTP-1 and HTP-2, n = 3 Experiments 12/4 Modules) from Chinese Hamster Ovary Cells Exposed to Whole Smoke from 3R4F Kentucky Reference Cigarettes Using ISO Intense Smoking Regimen
Source: ISO. 53
Exposures were also conducted using the ISO smoking regimen to compare the response with previously published data; 21 data are shown in Table 4. Three independent (n = 3) experiments were conducted on both Vitrocell Mammalian 48 modules, no significant differences in the mean % Survival responses were observed with a p-value of 0.5644. Data from both Vitrocell Mammalian 48 High-Throughput modules were pooled for the comparison with the historical % Survival data set. The responses (IC50) were compared between the Vitrocell Mammalian 48 high-throughput module and standard dilution system using an unpaired t-test with Welch's correction (Fig. 5). No significant difference was observed with p-value of 0.3875.
Mean % Survival (n = 3 Experiments Each for HTP-1 and HTP-2, n = 6 Experiments Historical) from Chinese Hamster Ovary Cells Exposed to Whole Smoke from 3R4F Kentucky Reference Cigarettes Using ISO Smoking Regimen
Historical data taken from Fields et al. 21
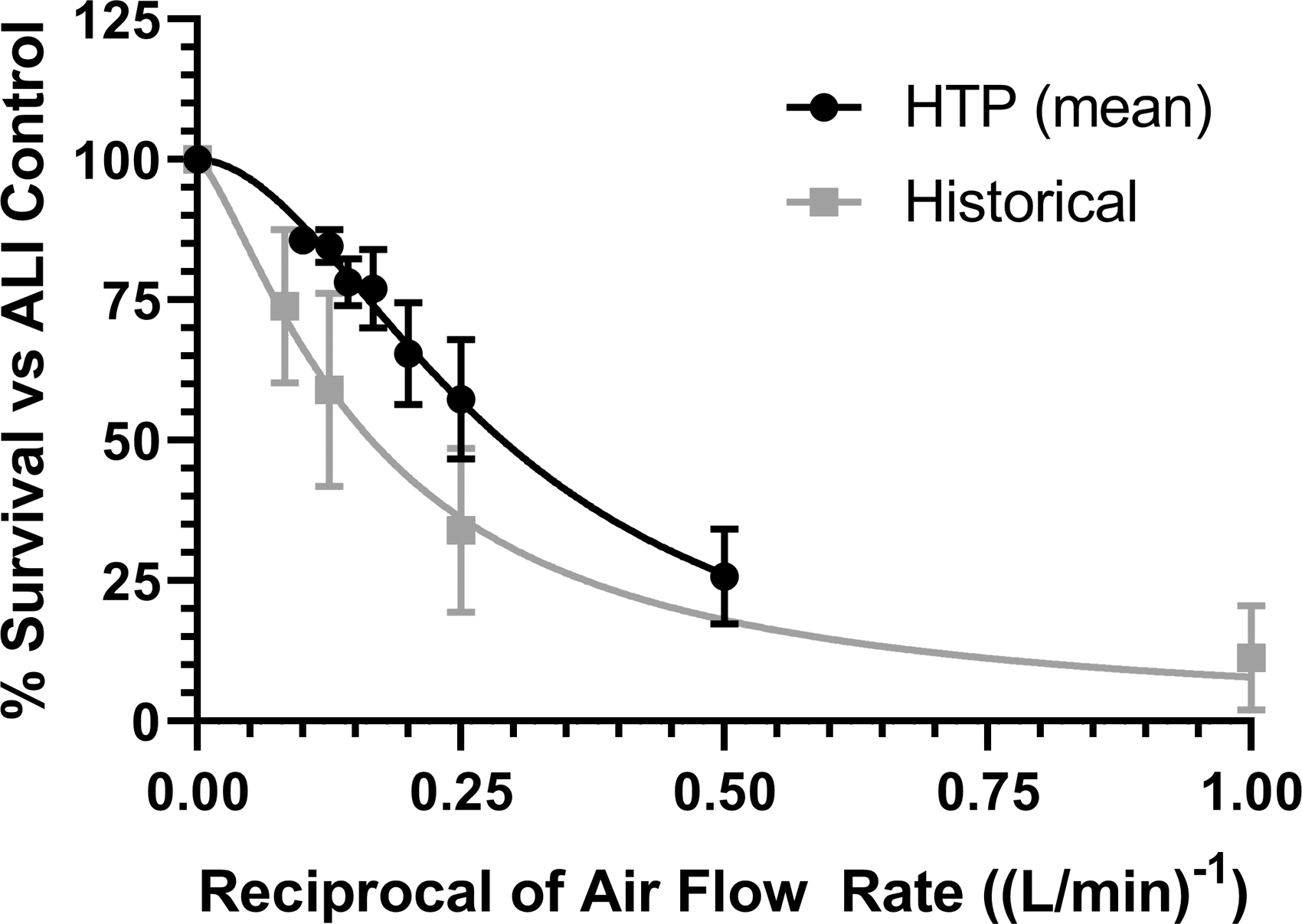
CHO cytotoxicity presented as % Survival versus ALI control using reciprocal of airflow rate for Vitrocell Mammalian 48 high-throughput modules and historical data from Vitrocell 12/4 modules (Fields et al. 21 ).
DMSO fluorescence
DMSO fluorescence from all experiments is shown in Table 5. An equivalent TPM concentration was calculated using the corrected RFU485 extrapolated against a standard curve of known TPM concentrations. In all experiments and in both modules there was a concentration-related increase in deposited mass. The deposited mass was consistent between each module for the ISO intense 53 and ISO 54 smoking regimens.
Dimethyl Sulfoxide Fluorescence Values from Vitrocell Mammalian 48 Modules 1 and 2 and Vitrocell 12/4 Modules Using 3R4F Kentucky Reference Cigarettes Using the ISO Intense (n = 6 Heated Tobacco Product Modules) and ISO (n = 3) Smoking Regimes
n = 3 Experiments due to values being below standard curve.
n = 2 Experiments.
SD, standard deviation; TPM, total particulate matter.
The deposition was comparable between different exposure systems between the high-throughput modules and the 12/4 module system at the higher airflows, although some differences were observed at the lower airflows. Note that the values for 3R4F ISO Intense 12/4 module differ at the lower airflows, and this is due to the different volumes of DMSO in the stainless steel insert, smaller module size and are, therefore, not directly comparable.
Nicotine
Nicotine concentrations from all experiments is shown in Table 2. A concentration-related increase in nicotine was observed from 8 to 0.5 L/min for the high-throughput modules. It should be noted that 10 L/min was higher than the subsequent lower airflow. Nicotine deposition was comparable between the different high-throughput modules. Nicotine deposition is lower in the 12/4 module compared with the corresponding regime in the high-throughput modules, and this is due to the different volume of DMSO and smaller surface area of the stainless steel insert and are, therefore, not directly comparable.
Photometers
AUC data from the photometers are presented in Table 6. In all cases there is a concentration-related increase in measured dose.
Area Under Curve Values from Mammalian 48 Modules 1 and 2 Using 3R4F Kentucky Reference Cigarettes Using the ISO Intense (n = 6) and ISO (n = 3) Smoking Regimes
n = 4 Due to fault with photometer connection.
AUC, area under curve.
Discussion
The Vitrocell Mammalian 48 module advanced in vitro exposure system was developed and characterized to increase the number of airflows per experiment from four as per the standard Vitrocell module system as previously described,19–22,34 to seven doses allowing further discrimination and to fully elucidate the toxicity of whole smoke. In addition, the number of replicates can also be increased, which adds further confidence to the % Survival and allow the option of multiple dosimetry measures to be able to quantify the dose received by the biological system.
This study was initiated to validate and characterize the biological response of two Vitrocell Mammalian 48 modules using the ISO intense 53 smoking regime. In addition, it was also a requirement to assess the magnitude of the response to the ISO smoking regimen 52 and compare with previous published data. 21 Additional data were generated as per historical data set using the ISO intense smoking regimen utilizing the Vitrocell 12/4 module system to perform a full assessment of both regimens with both exposure systems. 3R4F Kentucky Reference cigarette was chosen as the test article to facilitate comparing with historical data and the requirement to test a product that can elicit a response.
The data presented in this publication demonstrate that the Vitrocell Mammalian 48 module coupled with the Vitrocell VC10 smoking machine was able to elicit a cytotoxic response in CHO cells using the NRU assay after exposure to whole smoke from 3R4F Kentucky Reference cigarettes using the ISO intense regimen. All six experiments from both Vitrocell Mammalian 48 modules elicited a cytotoxic response with IC50 values obtained 344.7 and 449.7 ng/mL, respectively, for modules (1) and (2).
Further analysis demonstrated that there was no significant difference (p = 0.5385) in the cytotoxic responses between the two Vitrocell Mammalian 48 modules. Data were pooled from both Vitrocell Mammalian 48 modules to compare with the response to whole smoke from 3R4F Kentucky Reference cigarettes using the ISO intense regimen with the Vitrocell 12/4 modules. IC50 values were calculated as 18.07 ± 1.64 and 15.53 ± 3.12 mg/mL equivalent TPM, respectively, for the Vitrocell Mammalian 48 module and Vitrocell 12/4 modules.
Analysis demonstrated that there was no significant difference (p = 0.5523) in the cytotoxic response. The p-value is comparable with that obtained for the comparison between the two high-throughput modules. It should be noted that the comparison was based on equivalent TPM (mg/mL) as opposed to on a nicotine basis, and this is due to the lower than expected nicotine delivery, only the lowest airflow is comparable with the high-throughput modules 1 L/min. However, it should be noted that there are some mechanical differences between the modules validated in this study compared with those in previous work.
The Vitrocell Mammalian 48 modules have a longer smoke path (distance between smoke generation and delivery to the ALI surface), use a serial dilution of the smoke versus single smoke dilution in the standard modules and the distance between the ALI surface and the sampling point of the smoke path is also shorter when compared with the Vitrocell 12/4 modules. Another contributing factor is the different module sizes; the Vitrocell 12/4 modules utilize a 12 mm diameter Transwell with a surface area of 1.12 cm2, the Vitrocell Mammalian 48 modules utilize a 24 mm diameter Transwell with a surface area of 4.67 cm2 resulting in an ∼4.2-fold increase in surface area. The authors note that further investigation is needed to address these observations.
To perform a true comparison with the previously validated Vitrocell VC10 coupled with the 12/4 module setup, 21 exposures were conducted on the Vitrocell Mammalian 48 module with the ISO regimen 54 to generate comparable data. Airflows were matched where appropriate; however, the highest airflow for the Vitrocell Mammalian 48 module was set at 10 L/min compared with 12 L/min for the Vitrocell 12/4 modules and the lowest at 2 and 1 L/min, respectively.
Comparisons with the Vitrocell Mammalian 48 module data and historical data set show no significant differences (p = 0.3875) between the calculated IC50 values, demonstrating that data generated using both systems are comparable. Note that comparisons were conducted using reciprocal airflow due to this being the unique dosimetry marker between the modules.
It is necessary to quantify the dose received by the biological system; therefore, dosimetry measures were included in the exposures to better understand the biological response and allow comparisons between exposure systems. The dosimetry approaches utilized in this study included an AUC value generated by inline aerosol photometers, fluorescence of smoke particulates trapped in DMSO and nicotine determination in smoke-exposed DMSO confirm reproducible and concentration-related delivery of whole smoke to the ALI surface.
Equivalent TPM (mg/mL) doses determined through extrapolation of the smoke-exposed DMSO fluorescence values from a standard curve of known TPM concentrations demonstrated the smoke delivery between experiments is consistent, although higher variability was observed at the lowest dilution airflows or highest smoke concentrations. This observation has been previously demonstrated and seems to occur particularly with smoke generated from combustible cigarettes. 27
This may be partially explained by inconsistent or nonhomogeneous mixing of the cigarette smoke and diluting air that may occur at the lower dilution airflows versus more sufficient mixing with the higher dilution airflows. Photometer AUC values and nicotine determinations also confirm the concentration-related delivery of smoke to the ALI cultures. The use of multiple dosimetry approaches demonstrates their utility and the importance of the implementation of multiple dosimetry measures to improve the overall confidence at the lower dilution airflows and higher concentrations of whole smoke. 28
Under the experimental conditions employed in this study, the Vitrocell Mammalian 48 module when used with CHO cells in the NRU assay was able to generate and deliver whole cigarette smoke from a 3R4F reference cigarette, resulting in cytotoxic responses that are consistent with previously published data. In addition, we have demonstrated that the cytotoxic responses are consistent between two Vitrocell Mammalian 48 systems allowing meaningful comparisons to be made between data sets generated from both systems.
We have also demonstrated that data generated on the Vitrocell Mammalian 48 module are comparable with data generated on the smaller Vitrocell 12/4 modules, enabling comparison with historical data sets if required. Overall, the data generated in this study demonstrate and support the characterization and validation of the Vitrocell Mammalian 48 exposure module for use in the assessment of whole smoke and aerosol cytotoxicity and the importance of the determination of the delivered smoke dose to allow the comparison of results generated on different whole smoke exposure systems.
Footnotes
Acknowledgment
The authors appreciate the valuable comments by Dr. Eric Scott.
Author Disclosure Statement
B.M.K., R.L., R.A.W., C.J.B., and K.J. are full-time employees of RAI Services Company.
Funding Information
The data presented in this article were generated and analyzed in studies commissioned by RAI Services Company and conducted under contract at LabCorp Early Development Laboratories Ltd.
