Abstract
Introduction:
The European Union Reference Laboratory for alternatives to animal testing (EU RL ECVAM) coordinates the validation of multiple thyroid in vitro assays aiming to set up an in vitro test battery. The validation consists of two independent parts: part 1 (Reproducibility assessment) in which the reproducibility of the method is evaluated by testing a small number of described inhibitors and part 2 (Predictivity assessment) where the predictivity of the method is investigated by testing a blinded set of substances. The deiodinases (DIO) are important regulators of thyroid hormone (TH) metabolism and potential targets for substance-induced thyroid disruption. We previously reported the part 1 results for the Sandell–Kolthoff reaction-based type 1 deiodinase (DIO1-SK) assay and an extensive description of the method. In this study, we report the results of the part 2 testing in the DIO1-SK assay.
Methods:
A set of 22 test substances consisting of known DIO inhibitors in vitro, as well as substances otherwise interfering with the TH system and substances with no such activities were tested in the standardized DIO1-SK assay. Experiments were performed on blinded substances, which were deblinded after experimental completion, statistically evaluated, and compared to literature data. Finally, an in vitro data interpretation procedure (DIP) was generated.
Results and Discussion:
Seven test substances produced a maximum DIO1 inhibition >90% and 11 test substances below 20%; they were regarded as inhibitors and noninhibiting substances, respectively. Two test substances, Ketoconazole and Silichristin, were found to be not applicable based on assay interference. Inhibition data were consistent with results of relevant in vitro and computational models. Using the variation of control data, an in vitro DIP was defined categorizing test substances (i) by efficacy using the maximum inhibition data and (ii) by potency using the IC50 data of the test substance.
Introduction
The thyroid hormone system (THS) can be affected by many substances, potentially leading to endocrine disruption, due to complex neuronal, humoral, and intracellular signaling. Function and regulation of the THS, comprising the hypothalamus, pituitary, and thyroid as well as target organs of thyroid hormone (TH) action, are dependent on numerous specific molecular functions, including various catalytic enzymes, several transmembrane transporters for hormones as well as iodide, humoral transporters/binding proteins for hormones, and hormone receptors (Fig. 1). In particular, the enzymatic deiodination of TH, thyroxine (T4), and triiodothyronine (T3) to more active or less active TH metabolites is a crucial mechanism of pre-receptor of TH action, enabling cell-specific fine tuning of their intracellular fate and potency. This deiodination is catalyzed by the deiodinase (DIO) enzyme family; in humans, three isoenzymes are known. 1 They belong to the family of selenoproteins, having a selenocysteine residue in the catalytic center, which is crucial for DIO function.
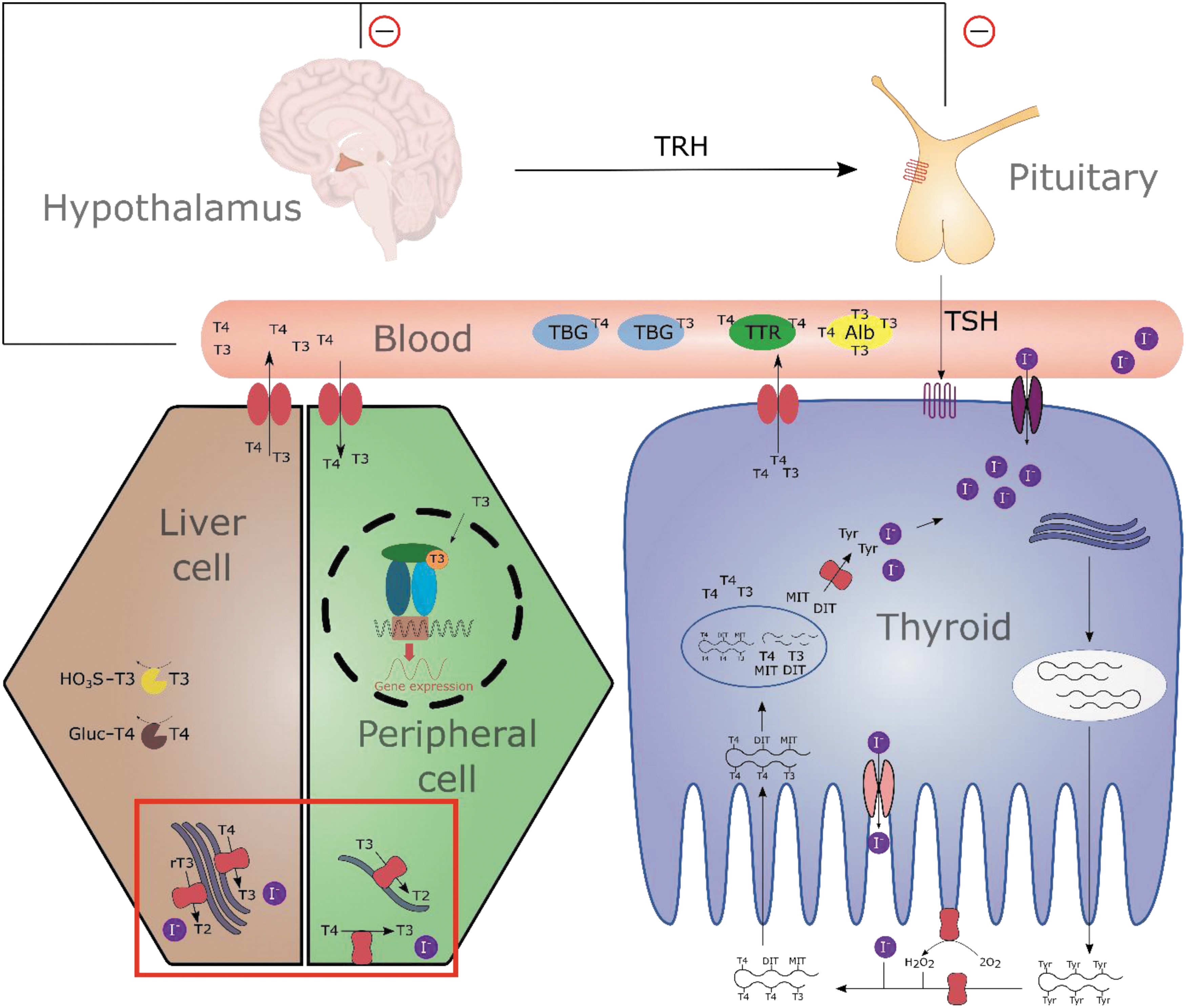
The TH system. The hypothalamus produces TRH that stimulates the anterior pituitary to release TSH. Thyroid activity and growth depend on activation of the TSH receptor, leading to stimulation of TH biosynthesis, including, for example, increased iodide uptake into thyrocytes and production of TH by binding of iodide to Tyr residues of thyroglobulin in the lumen. Bound TH is taken up into the cells by endocytosis and TH (T3 and T4) as well as MIT and DIT is liberated by lysosomal proteolysis. The iodine-containing byproducts of TH biosynthesis, MIT and DIT, are deiodinated by the iodotyrosine deiodinase to retain the released iodine. The THs are released into the blood stream by specific transporters. TH binding proteins (Alb, TBG, and TTR) distribute TH to peripheral cells where they are taken up into the cell by specific transmembrane transporters. In this study, depending on the cell type-specific expression pattern, the three iodothyronine deiodinases exert their function of pre-receptor control of TH action by activation and inactivation through TH deiodination. Enzymatic, selective removal of iodine atoms from, for example, the pre-hormone T4 will lead to the active TH T3 or less active ones like rT3 or T2 (indicated by red box). Finally, TH mediate their effect by binding to the nuclear TH receptors, triggering gene expression of TH-regulated genes. In the sense of homeostasis, TH levels regulate TRH and TSH release through a negative feedback loop. Elimination of THs occurs mainly in the liver by sulfation and glucuronidation. Alb, albumin; DIT, diiodotyrosine; MIT, monoiodotyrosine; rT3, reverse T3; T2, diiodothyronine; T3, 3,3′,5-triiodo-L.thyronine; T4, thyroxine; TBG, thyroid-binding globulin; TH, thyroid hormone; TRH, thyrotropin-releasing hormone; TSH, thyroid-stimulating hormone; TTR, transthyretin; Tyr, tyrosine.
Despite their structural similarity, the isoforms differ in substrate preference, position of deiodination (5- and 5′-deiodination), reaction kinetics, and cellular and subcellular localization, and their physiological function (Table 1). Only a few DIO-inhibiting substances are known. In vitro, various inhibitors were identified,2–4 but in vivo data in animals or humans remain scarce. 6-Propyl-2-thiouracil (6PTU) has been used as a treatment for hyperthyroidism since at least 1946. 5 6PTU specifically inhibits type 1 deiodinase (DIO1), while having little effect on DIO2 and DIO3. 6 Like 6PTU, other thiouracils inhibit DIO1 competitively to the cofactor. 7
Parameters of Human Deiodinase Isoenzymes
6PTU, 6-propyl-2-thiouracil; ATG, aurothioglucose; BAT, brown adipose tissue; CNS, central nervous system; DTT, dithiothreitol; rT3, reverse 3,3′,5-triiodo-L.thyronine; T3, 3,3′,5-triiodo-L.thyronine; T4, thyroxine; TH, thyroid hormone.
In addition, 6PTU inhibits the thyroid peroxidase (TPO), which is a key enzyme of TH biosynthesis within the thyroid gland, responsible for iodide oxidation and coupling. It is unclear which inhibition is responsible for the treatment of hypothyroidism. It was postulated that the therapeutic effect of 6PTU is due to the inhibition of TPO rather than DIO1 6 since the thiouracil methimazole inhibits TPO, but not DIO1 and shows similar therapeutic effects. 8 Nevertheless, DIO1 inhibition is a potential mechanism to cause disturbance of the THS. Robust and reproducible methods are required to investigate DIO1 inhibition in vitro.
We reported the standardization of an in vitro assay to investigate DIO1 inhibition. 9 This method uses human liver microsomes as a source for DIO1 and the Sandell-Kolthoff (SK) reaction to quantify released iodide. This method was originally published by Renko et al. using murine liver microsomes. 13 The method is part of a validation program led by the European Commission's Joint Research Centre (EC JRC) EU Reference Laboratory for alternatives to animal testing (EURL ECVAM). 14 In cooperation with the European Union Network of Laboratories for the Validation of Alternative Methods (EU-NETVAL) and the respective method developer of each method, an in vitro test battery consisting of different THS disrupting methods, should be generated.
This method validation comprises two parts, both adhering to the “Guidance Document on Good In Vitro Method Practices (GIVIMP)” 15 : (1) testing known DIO1 inhibitors in five independent runs to validate the reproducibility of the method (reproducibility assessment, part 1) 9 and (2) testing a set of blinded test substances, including known DIO1 inhibitors as well as negative substances to validate the predictivity (predictivity assessment, part 2). The reproducibility of the DIO1-SK assay has been demonstrated already. 9 In this study, we report the results of the predictivity assessment (part 2) for the DIO1-SK assay.
A set of 22 test substances was blinded (the chemical identity and DIO1 inhibition of the test substances was unknown to the test laboratory) and tested with the DIO1-SK assay. The test substance set included known in vitro inhibitors of DIO, as well as THS-interfering substances with other targets and substances not active in this respect. The set was tested in two additional assays to assess the specificity of observed DIO1 inhibition in the DIO1-SK assay, as described in Weber et al. 9 : (i) testing of the substance in the DIO1-SK assay without microsomes can identify substances that lead to spontaneous iodide release or substances that interfere with the SK reaction and (ii) testing the effect on alkaline phosphatase (ALP), another microsomal enzyme that can indicate global enzyme inhibiting activity unspecific to DIO1.
After completion of the experimental phase and transfer of data to EURL ECVAM, blinded test substances were deblinded. The DIO1 inhibition data were then compared to relevant in vitro and computational data in the literature. Finally, a data interpretation procedure (DIP) was developed based on the distribution of the generated in vitro data to classify the test substances into suggested categories of DIO1 inhibition.
Materials and Methods
Standard operation procedure
Standard operation procedures (SOPs) for the DIO1-SK assay and the ALP specificity testing approach were published. 9 These SOPs were adjusted and revised for this part 2 study to allow more freedom in the choice of material. The SOP:DIO1-SK assay contains information about material, performance, and analysis for the DIO1-SK assay and testing for specificity with and without microsomes during the incubation phase (Supplementary Pdf S1). The SOP:ALP activity testing contains information on how to perform the specificity testing approach that addresses the enzyme ALP (Supplementary Pdf S2). All experimental data of this study were generated using these SOPs.
Material
The required material was previously described 9 and is specified in detail in both SOPs. Materials like the batches of microsomes (provided by EURL ECVAM) or control items remained the same compared to part 1 testing.
Test substances
EC JRC EURL ECVAM selected test substances for the validation of several New Approach Methodologies (NAMs) addressing different THS modes of action. A hybrid approach was used combining different criteria: literature data and expert knowledge on THS disruption covering in vitro, in vivo, and epidemiological expertise as well as information on the availability of substances. Among the eligible substances, test substances that cover the largest possible chemical space were selected. For DIO1 specifically, an appropriate balance between assumed positive and negative substances was aimed.
For the validation of the DIO1-SK assay, EURL ECVAM supplied 22 blinded test substances in brown glass vials. The vials were stored according to their specified storage conditions. The chemical identity of test substances was masked, but relevant safety information and molecular weight were reported for each substance (rounded to 25 Dalton). After completion of the experimental part and providing the generated data to EURL ECVAM, the 22 test substances were deblinded by disclosing the chemical name and CAS number to the testing facility, without providing further information about the expected result in the DIO1-SK assay (Table 3).
DIO1-SK assay
The method is described in detail in the SOP:DIO1-SK assay and Weber et al. 9 and shown in Figure 2. Briefly, test substances were incubated in a 96-well format with human liver microsomes, the substrate reverse (r)T3, the cofactor dithiothreitol (DTT), and phosphate+EDTA buffer for 2 hours at 37°C under constant shaking (assay concentrations: 0.1 M H2KPO4/HK2PO4, 40 mM DTT, 1 mM EDTA, 5 μM rT3, and 1% dimethyl sulfoxide). Ion-exchange resin-filled filter plates were used to separate released iodide from remaining substrate by application of vacuum to elute the samples. The released and eluted iodide was quantified using the SK reaction.
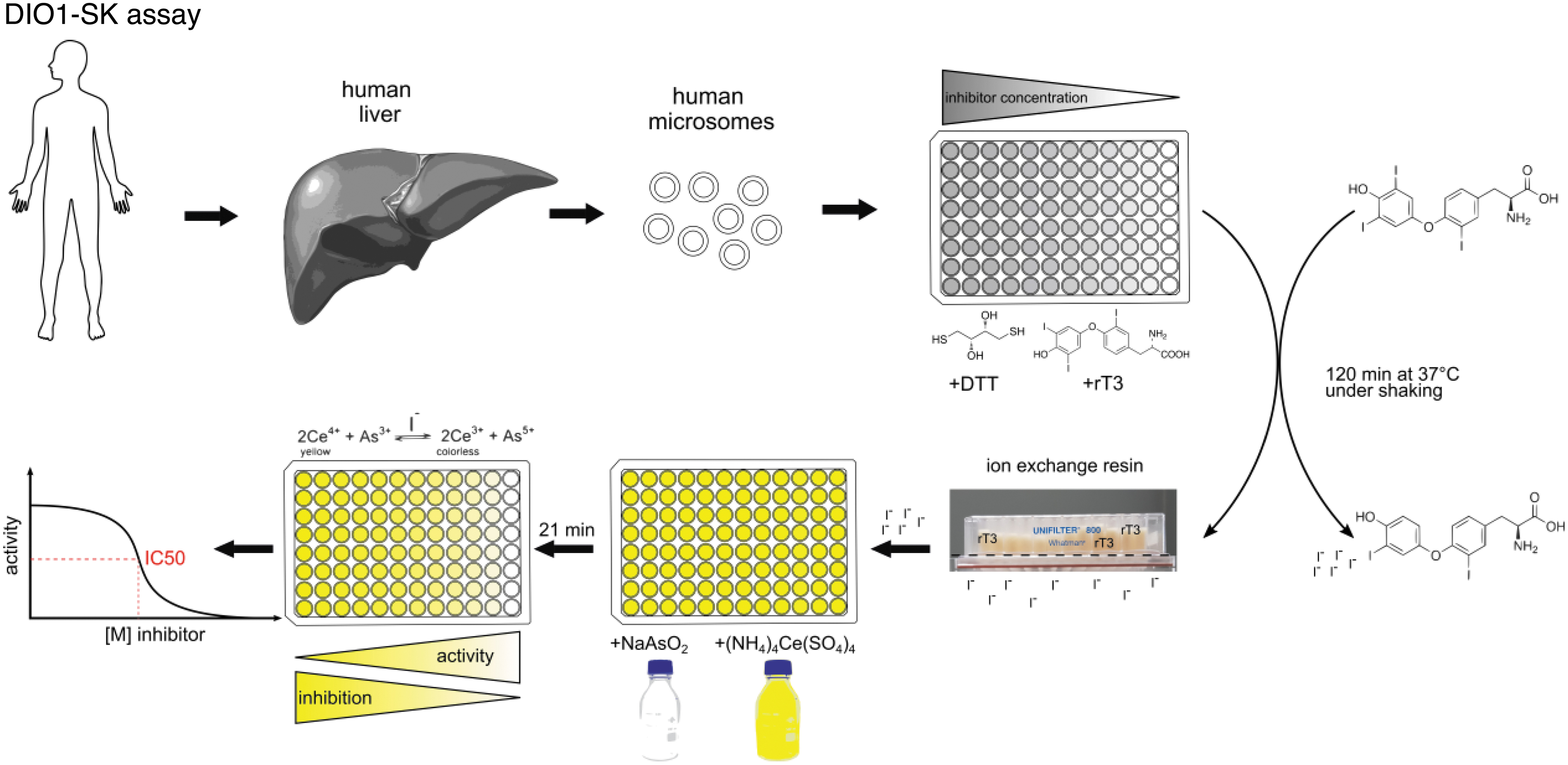
Schematic procedure of the DIO1-SK assay. Human liver microsomes are incubated under pH-buffered conditions with the substrate rT3, an artificial enzyme-regenerating cofactor DTT, and the test and control items. The assay plates are incubated for 2 hours at 37°C under constant shaking. The enzymatically released iodide is separated from the remaining assay constituents, including substrate, by an ion-exchange resin. The iodide concentration of the eluted sample is then quantified by the SK reaction by formation of colorless Ce3+ from yellow Ce4+ using arsenite. The reaction is catalyzed by the available iodide concentration. Nonlinear curve fitting is used to derive inhibition values for the test items. DIO1-SK, Sandell–Kolthoff reaction-based type 1 deiodinase; DTT, dithiothreitol. Figure was adapted from Weber et al. 9
The SK reaction describes the formation of colorless Ce3+ from yellow Ce4+ using arsenite that is catalyzed by the available iodide concentration. 16 A set of control items was previously described 9 and was used to control and monitor assay performance (Table 2). Using the control data for each assay plate, the iodide release activity (IRA) is determined as a normalized value. The IRA is indicative of the amount of iodide released by DIO1 catalysis. A value of 100% IRA is the maximum possible DIO1 activity, while 0% DIO1 activity represents full inhibition by 6PTU. DIO1 inhibition [%] is calculated as 100 − IRA [%].
Used Control Items in the DIO1-SK Assay According to the Concept of Good In Vitro Method Practices 15
6PTU, 6-Propyl-2-thiouracil; ATG, aurothioglucose; DIO1-SK, Sandell–Kolthoff reaction-based type 1 deiodinase; DMSO, dimethyl sulfoxide; NC, negative control; PC, positive control; RI, reference item; SC, solvent control; TGSS, 1-thio-β-
Before testing, the solubility of the test substances was characterized (Table 3); extensive information about the solubility results is given in Supplementary Table S1. Most of the test substances were soluble at assay concentrations between 10−3 and 10−4 M with some of the test substances being less soluble (as low 10−6 M). Stock solutions were prepared on the day of assay performance.
All test substances were tested in three different systems: (i) the DIO1-SK assay, (ii) in DIO1-SK testing without microsomes, and (iii) ALP activity testing.
At least three valid, independent assay runs were performed for each test substance. Testing was performed in three technical replicates for each concentration, resulting in at least nine inhibition values for each concentration. Initially, test substances were tested in a range-finding assay run. In this study, the determined highest soluble concentration and consecutive 10-fold (v/v) dilutions of each test substance were tested.
If any concentration tested inhibited IRA by more than 20%, tested concentrations of following assay runs were adapted, including more concentrations in this concentration range. For these substances, a final set of at least three independent assay runs with adapted concentrations was performed and the range-finding assay run was not used for the final assay run set used for maximum inhibition and IC50 derivation. If the test substance did not show inhibition of >20% in the range-finding assay run, this assay run was included in the final set of at least three valid assay runs.
It was ensured that every test substance was tested in at least two of the three possible positions of the assay plate layout to account for a potential influence of the plate position. Three different laboratory co-workers performed the experiments to ensure intralaboratory reproducibility.
Testing for assay interference without microsomes
Incubations without microsomes were performed according to the DIO1-SK assay SOP. Testing was performed like the DIO1-SK assay, but the microsomal fraction was replaced with deionized water. One initial assay run was performed for all test substances using the highest concentration only. If an increase of IRA >10% was observed in the initial assay run, two additional, independent assay runs were performed for the respective test substances.
ALP inhibition assay
The colorimetric ALP assay is well established and described in the literature.17,18 Testing for ALP inhibition was performed according to SOP:ALP activity. The assay is based on the colorimetric quantification of a metabolite formed by the dephosphorylation activity of ALP in human liver microsomes. One initial assay run was performed for all test substances using the highest concentrations only. Two additional assay runs were performed for test substances that inhibited ALP by more than 15%.
Comparison of data to existing literature data
The data from this study were compared to published in vitro or in silico inhibition data on DIO1 inhibition: (i) the ToxCast database of the U.S. EPA with more than 1800 substances was investigated for DIO inhibition using a comparable method. The method also uses ion-exchange resin to separate released iodide from assay components and uses the SK reaction to quantify iodine, yet recombinant DIO enzymes were used instead of human liver microsomes. 2 (ii) Garcia de Lomana et al. developed a battery of in silico models for different targets of the THS, including DIO1. 19 The models were based on the in vitro screenings of the ToxCast database by the U.S. EPA.
Statistical approaches, machine learning methods, data balancing techniques, and neural networks were used to derive the models. The in silico model uses in vitro data from the U.S. EPA DIO screening and generated predictions are thus not independent. (iii) The U.S. EPA ToxCast screening of the enzyme class iodotyrosine deiodinases (IYDs) uses recombinant IYD to depict inhibitory effects. Compared to DIOs, IYDs represent a different class of enzymes with different inhibitors. However, the method also uses ion-exchange resin to separate released iodide and the SK reaction to quantify iodine.20,21 The IYD inhibition data were used to obtain information on the specificity of DIO1 inhibition by the test substances.
Statistical analysis
R statistical software 22 was used to evaluate inhibition and control data by generating IRA values. GraphPad Prism (version 9.3.1 for Windows; GraphPad Software, San Diego, CA) was used for the graphical representation of data. The DRC package 23 in R was used to fit a four-parameter log-logistic dose–response model (LL.4) and to derive IC50 values [Equation (1)]. The models used in GraphPad Prism and R use the same parameters and function. Only the final set of assay runs was used for curve and IC50 derivation and range-finding assay runs in which the tested concentrations of subsequent runs were changed due to observed inhibition in the range-finding assay run were not used.
Equation (1): the four-parameter log-logistic function “LL.4” was used to derive curve parameters for the test substance and, if possible, to derive IC50 values. “c” represents the lowest response, “d” the maximal response, “b” the slope of the curve, and “e” the IC50 value.
The z′-factor was derived according to SOP:DIO1-SK assay and was previously described. 9 If an assay run did not meet the acceptance criterion of a z′-factor above 0.5, box plot outlier testing was performed. The respective lower limit, defined as lower quartile −1.5 × interquartile range (IQR), and upper limit, defined as upper quartile +1.5 × IQR, were calculated for the replicates that were used for z′-factor derivation (solvent control and reference item) for mentioned assay runs. If replicates were below the lower limit or above the upper limit of the respective control/item, the replicate was defined as an outlier; no more than two outliers were excluded per control/item set. If the assay run did meet a z′-factor of 0.5 after outlier removal, it was considered a valid assay run; otherwise, the assay run was removed from evaluation.
Results
Assay performance, quality control, and comparison of assessment criteria in the DIO1-SK assay
The performance of DIO1-SK assay runs was monitored with a set of acceptance criteria (Table 4); variation of control items per assay run and over time is shown in Supplementary Figures S1 and S2. The variation of control items was low and set acceptance criteria were mostly met. The separation between reference item and solvent control was high, as indicated by a z′-factor, usually above 0.7. Some runs did not meet the acceptance criterion of a z′-factor above 0.5. This might be due to rare cases of the ion-exchange resin becoming impermeable and resulting in high standard deviation (SD) in the controls.
Used Test Substances and Their Highest Test Concentration, Defined by Solubility Testing
Used Acceptance Criteria in the DIO1-SK Assay and the Determined Mean and Variation Data in This Validation Study
CV, coefficient of variation; IRA, iodide release activity; SD, standard deviation.
In this study, outliers were identified by box plot outlier testing and excluded for these cases. Alternatively, the robust z′-factor could be derived and used instead of the z′-factor. The robust z′-factor is less vulnerable to outliers since the mean is substituted for the median and the SD for the median absolute deviation. 24 As such, only 3 assay runs did not meet a robust z′-factor of 0.5 before outlier exclusion (compared to 11 assay runs if the z′-factor is used). After outlier removal, all assay runs reached a z′-factor of 0.5, but two assay runs did not reach a robust z′-factor of 0.5.
Concentration range finding
Every test substance was first tested in a range-finding experiment in the DIO1-SK assay. If any of the tested concentrations led to an IRA decrease of more than 20%, the following assay runs were adapted, including more test substance concentrations in the range of inhibitory concentrations. In total, the concentrations of 11 of the 22 test substances had to be adapted. If no IRA decrease >20% was found in the range-finding experiment, concentrations were not changed and the range-finding experiment was considered the first of the required three biologically independent assay runs. For the final assessment, a cutoff of 25% was selected; 20% as a cutoff for the range finding was selected to not miss concentrations due to variability of range-finding results. The maximum concentrations used to determine DIO1 inhibition are listed in Table 3.
DIO1 inhibition
The results of the DIO1-SK assay for the test substances are shown in Table 5 and exemplary graphs are shown in Figure 3. The results for all test substances are summarized in Supplementary Figure S3. Seven test substances produced a maximum inhibition ≥90%. Derived IC50 values ranged from 2.0 μM for tannic acid and 85.4 μM for bisphenol A diglycidyl ether (BADGE). Eleven test substances produced inhibition below 20% at the highest concentration.
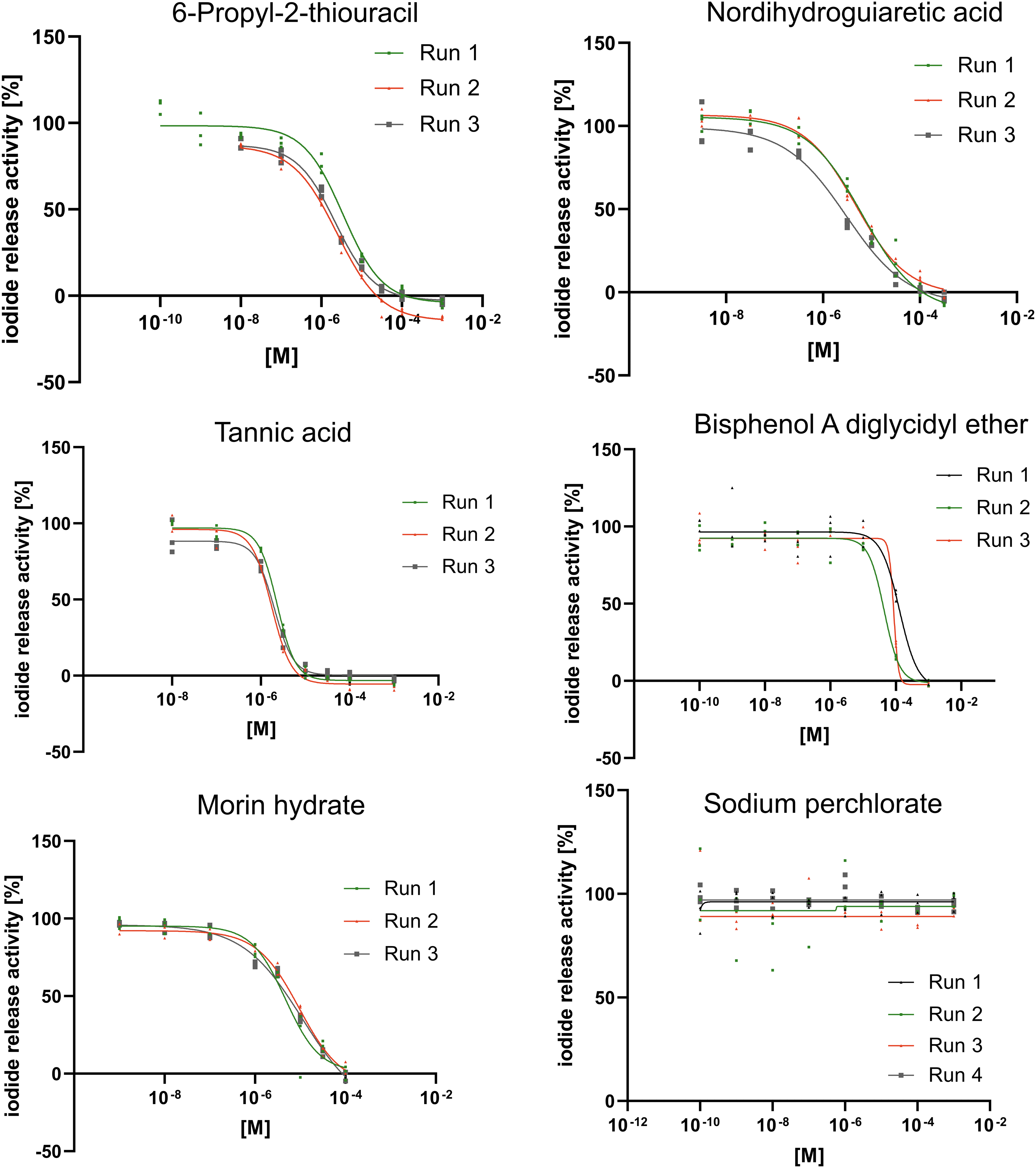
Exemplary DIO1-SK assay results for six test substances. Data are shown as mean iodide release ±SD of three technical replicates per concentration of at least three assay runs. The sigmoidal concentration–response curves were derived using the function “[Inhibitor] versus response—Variable slope (four parameters)” in GraphPad Prism. Range-finding assay runs that led to changed concentrations were not used for the derivation of IC50 values and are not shown. SD, standard deviation.
Summarized Results of the 22 Test Substances in the DIO1-SK Assay
IRA of the used control and reference items, the derived 50% inhibition concentration (IC50) of the reference item 6PTU, derived CV of the estimate of the reference item, and z′-factor of each assay run in the part 2 of the DIO1-SK assay over all performed assay runs.
- : IC50 could not be derived because the inhibition was too low or only ambiguous IC50 values could be derived.
Silichristin and ketoconazole were determined as not applicable in the DIO1-SK assay based on specificity testing.
Results of specificity testing
Two additional assays, (i) the DIO1-SK without microsomes and the (ii) ALP inhibition assay, were performed with each test substance to identify nonspecific interactions with the test system. Results for selected test substances are shown in Figure 4; results for all test substances of specificity testing without microsomes and ALP testing are shown in Supplementary Tables S2 and S3, respectively.
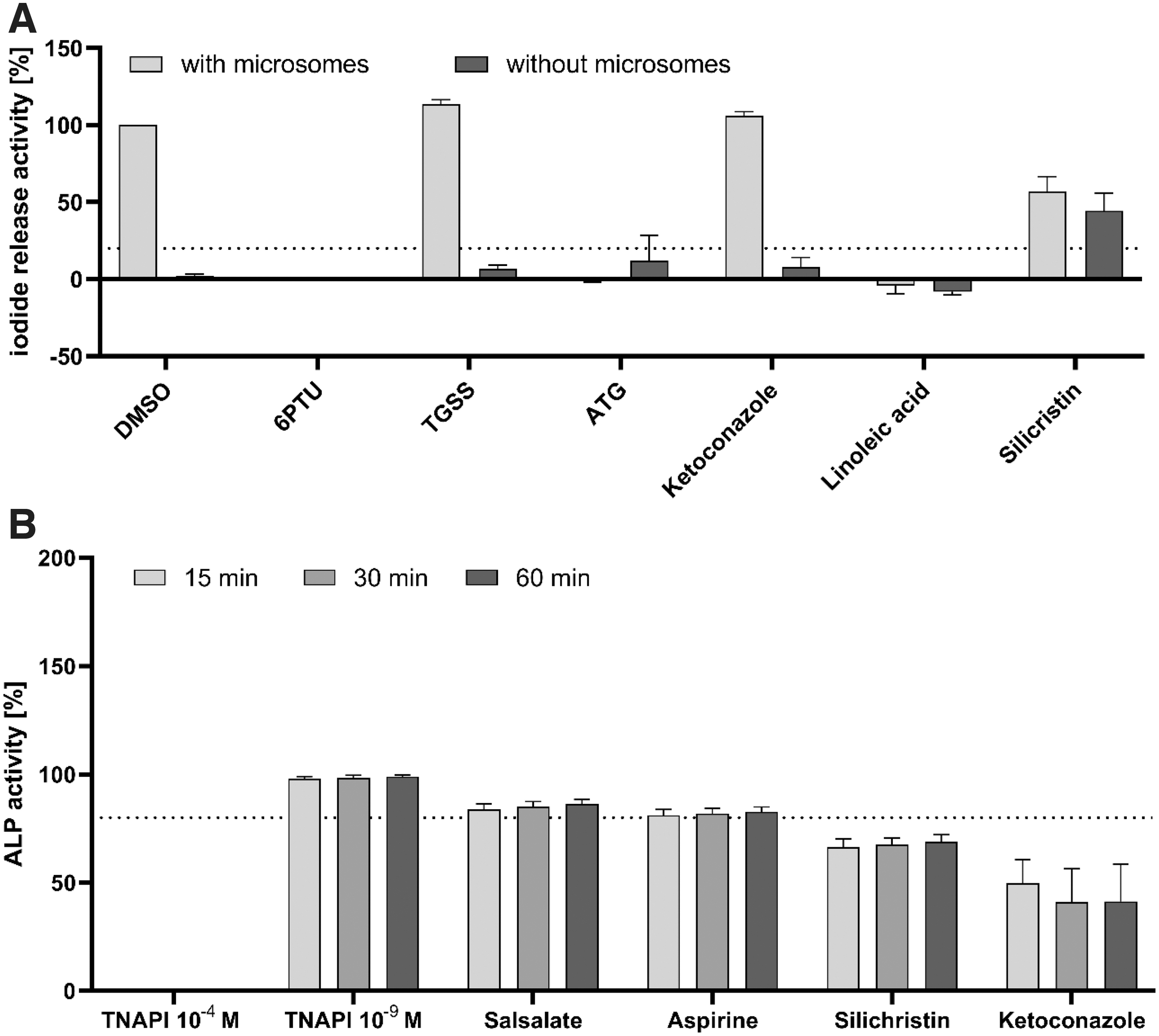
Specificity testing results to identify nonspecific test substances in the DIO1-SK assay for selected test substances.
Testing in the DIO1-SK assay without microsomes was used to identify test substances that can drive the SK reaction without release of iodide from the substrate by DIO1 and/or lead to spontaneous iodide release not catalyzed by DIO1. Silichristin produced an IRA activity around 50% in the presence of microsomes. This IRA activity was still present in the absence of microsomes, indicating interference of silichristin with the assay (Fig. 4A). Silichristin is hence not applicable to the DIO1-SK assay. The results of silichristin do not reflect its DIO1 inhibition and are excluded from further analysis. No other test substance showed considerable IRA increases in the absence of microsome.
Testing the activity of ALP as a second microsomal enzyme was used to identify inhibition of microsomal enzyme activity, which is not specific to DIO1. ALP inhibition greater than 20% after 60 minutes of incubation was regarded as ALP inhibiting. Silichristin was again active in inhibiting ALP activity. In addition, ketoconazole inhibited ALP activity by 59% (Fig. 4B). This may indicate nonspecific interactions of Ketoconazole with the microsomes. Ketoconazole was thus regarded as not applicable to the DIO1-SK assay. Results of the DIO1-SK assay with Ketoconazole were not considered for further analysis regarding DIO1 inhibition.
Some test substances (BADGE, morin hydrate, nordihydroguaiaretic acid, octyl methoxycinnamate, and tannic acid) led to abnormal reaction kinetics in ALP activity testing (Supplementary Table S3). This is likely due to test substance-induced, nonspecific conversion of the substrate, para-nitrophenyl phosphate, to its yellow, quantifiable metabolite, which was also visible in the increase of an initially measured optical density (OD) in the ALP inhibition assay (OD0 minute,415nm). The test substances that had an OD0 minute,415nm of 0.15 or above were excluded from ALP analysis. Still, nordihydroguaiaretic acid showed abnormal reaction kinetics and was removed from evaluation. These test substances were removed from ALP analysis (Table 7), but were not excluded from the DIO1-SK assay. Yet, their applicability to the DIO1-SK assay could only be checked by one of the two specificity assays.
Summarized Information About the 22 Deblinded Test Substances, Including Disclosed Information, DIO1 Inhibition and Specificity Data, As Well As Categorization of This Study, and Relevant In Vitro and In Silico Methods
A threshold of 20% IRA increase in the samples without microsomes was used to classify test substances as nonapplicable in the DIO1-SK assay.
A threshold of 20% inhibition of ALP activity after 60 minutes of incubation time was used to classify test substances as active.
These test substances interfered with the ALP activity test system.
Silichristin and ketoconazole were determined as not applicable in the DIO1-SK assay based on specificity testing and max. inhibition values were put in brackets.
2-Ethylhexyl trans-4-methoxycinnamate (CAS: 83834-59-7) was tested here.
The structural analog N,N′-diethylthiourea (CAS: 105-55-5) was tested here.
ALP, alkaline phosphatase; IYD, iodotyrosine deiodinase.
Data interpretation
As there are only little data about in vivo DIO1 inhibition available, we approached data interpreting by defining a procedure to identify substances that are not active in the assay, that is, have IRA responses like the negative and solvent control. The distribution of the IRA values of controls that can inhibit DIO1 (reference item and positive control) and of controls that have no effect on DIO1 (solvent control and negative control) is shown in Table 6. The data had an approximately normal distribution (Supplementary Fig. S4).
Summary of the Distribution of the Iodide Release Activity Values of Control Data Generated in Part 2 of the DIO1-SK Assay
Ranges that were not used for threshold derivation are shown in brackets.
IRA, iodide release activity; SD, standard deviation.
The ranges of the mean +/−3-fold SD of the control items were determined (Table 6). Based on the lower range of the solvent control (79.2% IRA) and negative control, 1-thio-β-
A second threshold was defined using the upper range of the DIO1-inhibiting controls. A threshold of 10% IRA (≙90% DIO1 inhibition) was defined, below which a test substance has complete inhibitory activity similar to the reference item, 6PTU, at a concentration of 10−3 M, and the positive control, aurothioglucose (ATG). By applying these two thresholds, assay results can be divided into three efficacy categories: (i) category 1: test substances leading to complete inhibition of DIO1 comparable to 6PTU (>90% inhibition), (ii) category 2: test substances with inhibitory activity not resulting in complete DIO1 inhibition (between 90% and 25% inhibition), and (iii) category 3: test substances that do not inhibit DIO1 (<25% inhibition). Using this classification system, 7 test substances were categorized in category 1, 2 test substances in category 2, and 11 test substances in category 3 (Fig. 5A).
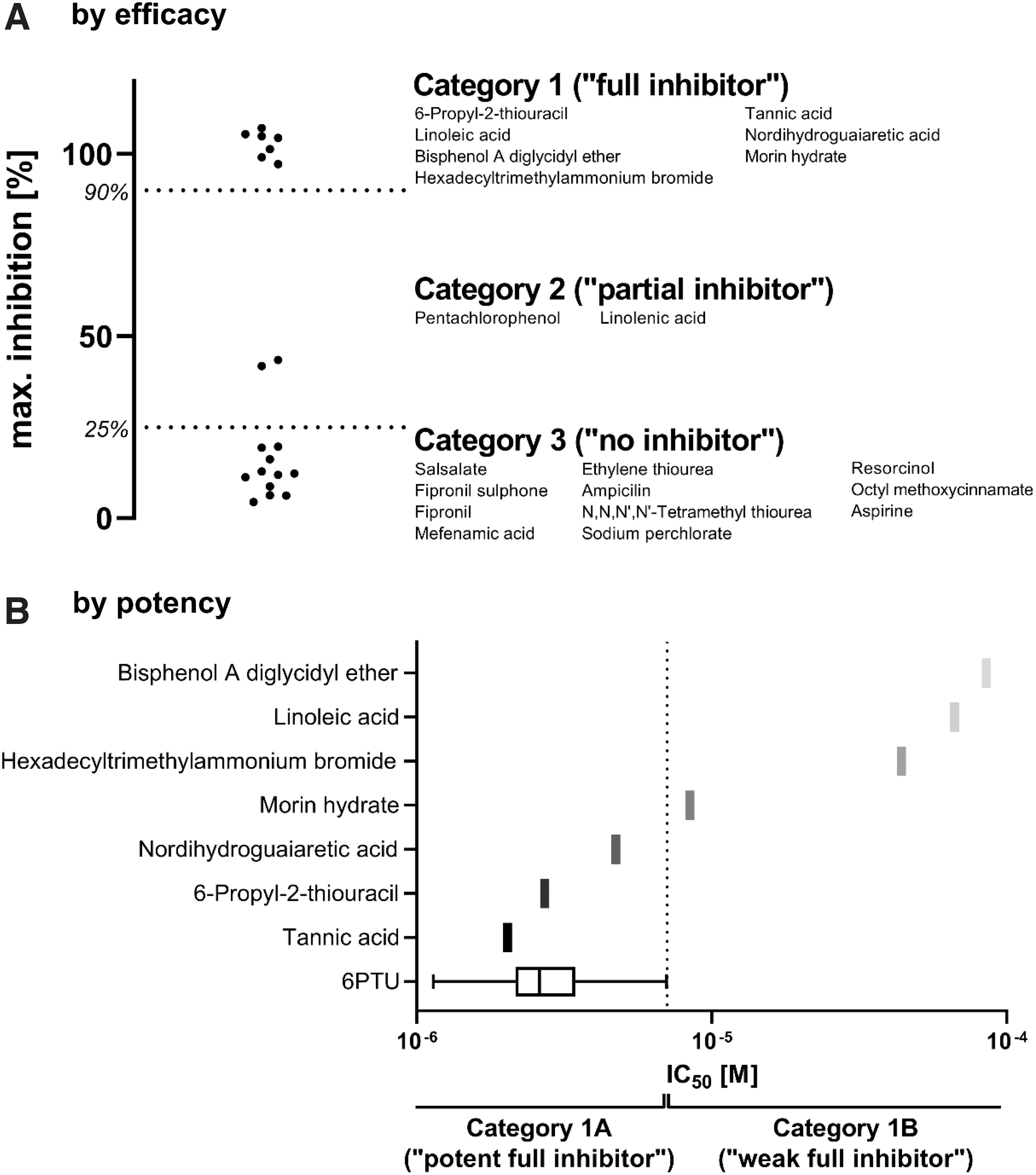
Test substance inhibition data were filtered by inhibition efficacy
In addition to an efficacy estimate, inhibitory activity can be assessed by its potency, characterized by the IC50. In this study, the IC50 values of the reference item 6PTU were used to estimate whether a test substance inhibited DIO1 similarly to 6PTU (Fig. 5B). Again, the mean +/−3 times SD approach was used to estimate the upper range results, resulting in a threshold of 7.1 μM.
Test substances of category 1 that had an IC50 lower than the defined threshold of 7.1 μM were at least as potent as 6PTU and are subcategorized into category 1A. Test substances with a higher IC50 are less potent than 6PTU and are categorized into category 1B. As such, three test substances could be categorized as category 1A (full inhibition, “6PTU like,” high potency) and four test substances as category 1B (full inhibition, lower potency than 6PTU) (Fig. 5B). Substances of category 2 were not subdivided by potency because derived IC50 values tend to be unreliable or could not be derived.
In vivo data regarding effects on DIO of the test substances are rare. Therefore, the correlation of the in vitro DIO1 inhibition data generated here with in vivo data was not applicable to estimate the in vivo predictivity of the DIO1-SK assay. The results of this study can, however, be compared to previously published data. The results of this study are summarized in Table 7 along with previously published data from in vitro screenings and in silico methods.
Discussion
DIO1 inhibition and specificity testing
In this study, a set of 22 test substances was examined in the DIO1-SK assay, as well as additionally checked for nonspecific interactions with the test system. The performance of control items was comparable to part 1. 9 The IC50 of the reference item, 6PTU, ranged from 1.1 to 5.4 μM with a mean of 3 μM. This was a slightly lower IC50 compared to part 1 (4 μM). Usage of the robust z′-factor versus the z′-factor was discussed based on some runs not meeting the set acceptance criterion of a z′-factor above 0.5. If outlier exclusion, for example, by box plot outlier testing is used, the z′-factor can be safely used. Assay runs of the same test substance were generally consistent and produced similar results confirming the reproducibility of part 1. 9
Data of some test items had high variability, which, however, did not affect the final result (classification according to the DIP, see Table 8): linolenic acid (classified as category 2 by the mean inhibition of three runs) inhibited DIO1 by 81% in the first assay run, but markedly less in following runs, possibly due to degradation of the test substance. Nonetheless, inhibition in the first assay run was not sufficient to classify linolenic acid into category 1. Inhibition–concentration curves of BADGE and hexadecyltrimethylammonium bromide were steep so that the tested concentrations resulted in either full or no inhibition. Consequently, the IC50 values varied across the three assay runs. Applying the DIP to the individual assay runs or the mean of all runs assigns BADGE and Hexadecyltrimethylammonium bromide to category 1B.
Data Interpretation Procedure of DIO1 In Vitro Inhibition in the DIO1-SK Assay
The model uses the generated qualitative maximum inhibition data of control data to define categories (“by efficacy”) and the quantitative IC50 values of the reference item 6PTU to subcategorize category 1 test substances (“by potency”).
Testing of substances without microsomes during incubation has proven to be a valuable strategy of identifying potential interferences with the method. Possible causes may be inherent activity of the substances in the SK reaction, through the presence of either iodide or other atoms that drive the SK reaction. Using this approach, false classifications for DIO1 inhibition can be reduced and the confidence in the method can be increased. As such, 1 of the 22 test substances, Silichristin, was found false positive in the DIO1-SK assay with a seeming maximum inhibition of 62%. Silichristin is a natural substance of the fruit of milk thistle and is a described inhibitor of the TH transporter monocarboxylate transporter 8. 25 Silichristin most likely interferes with the SK reaction, by driving the iodide release from substrate or the SK reaction itself. Hence, the DIO1-SK assay results must be disregarded.
In addition to DIO1 inhibition, ketoconazole inhibited ALP activity by 41%. Obviously, any DIO1 inhibition that is observed, in addition to the ALP inhibition, is not strictly specific to DIO1. This is either due to a certain promiscuity of the test substance, making it an inhibitor of two unrelated enzymes, or the test substances interfering with the microsomes, and thus disturbing any activity of its membrane-bound enzymes, for example, by perturbing the lipid membrane or unspecific protein binding. It can, however, never be ruled out that the test substance specifically inhibits both DIO1 and ALP. Ketoconazole is a known inhibitor of CYP3A 26 and produces direct hepatocellular toxicity in humans. 27 Decreased activity of a second liver enzyme that is present in human liver microsomes, the ALP, might hint toward unspecific interaction of the test substance with the microsomal protein. As such, DIO1 inhibition data from ketoconazole was also disregarded.
Comparison to literature data
In vitro data from an U.S. EPA DIO screening 2 is available for 16 of the 22 test substances. Generally, inhibition data are comparable to our study. The maximum concentrations tested are, however, different. While the U.S. EPA largely used fixed upper concentrations of 200 μM in a primary screening run, the upper concentration in our study is determined by solubility pretests and dilutions were tested in all cases. Testing up to higher concentrations might result in higher IC50 values and maximum inhibition, as observed for linolenic acid.
Interestingly, the IC50 of BADGE was 70 times higher in our study compared to the study by the U.S. EPA. The U.S. EPA study used recombinant enzyme as DIO1 protein source, which lacks metabolic competence compared to liver microsomes. Metabolism of BADGE to DIO1-inactive metabolites by microsomal hydrolysis in the DIO1-SK assay is a likely cause for the different IC50s since BADGE is known to be hydrolyzed in vitro by epoxide hydrolases in human, rat, and mouse liver, as well as lung fraction. 28
The DIO1-SK assay results were compared to another screening by the U.S. EPA, which investigated IYD inhibition. 20 IYDs are different from DIO enzymes: they lack the selenocysteine residue in the catalytic center, belong to the NADH oxidase/flavin reductase superfamily, use iodinated single tyrosine residues as substrate, and are inhibited by different substance classes. However, the method as originally developed 21 and used for the screening is very similar to the DIO1-SK methods of this study and the U.S. EPA DIO assay 2 : all three use a buffer system, DTT (or β-mercaptoethanol) as a reductive agent, and ion-exchange separation using resin and the SK reaction for iodide quantification. Interestingly, four test substances (tannic acid, linoleic acid, hexadecyltrimethyl ammonium bromide, and morin hydrate) are active in IYD and DIO assays.
Apart from morin hydrate, the IC50 values are similar in the IYD and the two DIO assays. This inhibitory activity may be due to a common nonspecific effect in all three test systems or concomitant specific effects on DIO and IYD. Whether they interact with DTT, seal the ion-exchange resin, or interfere by another test system-specific effect cannot be clarified. To investigate whether released iodide is prevented from passing through potentially sealed ion-exchange resin, preparations with the test substance and a defined, added iodide concentration that resembles the assay situation could be tested.
The DIO1-SK results of our study were compared to results from computational models by Garcia de Lomana et al. 19 All the test substances that produced more than 40% inhibition in this study were classified as “active” in the DIO1 in silico model. The computational model used the ToxCast database and other scientific literature as training sets. Many of the 22 test substances of this study were tested by Olker et al. and are thus part of the ToxCast database, and hence most likely part of the training set of the computational DIO1 model. There is likely a strong agreement between the experimental and computational datasets because there is little independent comparative data.
Fipronil and the structural analog fipronil sulfone were both classified as “active” in the computational model, but did not produce inhibition in our study. This could possibly be attributed to the tested concentrations in Olker et al. (200 μM). This high concentration resulted in precipitations in our study and therefore lower concentrations were tested. Obviously precipitated test substance is contributing to the inhibitory effect, potentially by continuous dissolution from the solid phase. Then again, the nominal concentration, including the precipitate, is neither the actual effective concentration of the test substance nor is it of relevance in vivo.
There are only few specific in vivo DIO inhibition data available. For 6PTU—one of the substances that was assigned into category 1A by DIO1-SK assay data—effects on DIO1 and TPO are well described in vitro as well as in vivo.5,6,29,30 Two unsaturated fatty acids, linoleic and linolenic acid, were categorized as category 1B and category 2, respectively, by DIO1-SK assay data. Data from rats show increased biliary excretion of thyroid metabolites with iodinated outer rings after treatment with ethanol and linoleic acid, suggesting inhibition of outer ring deiodination by unsaturated fatty acids 31 ; DIO1 is catalyzing the ring deiodination.
Conversion of T4 to T3 was reduced after treatment of rat liver homogenate with fatty acids like linoleic and linolenic acid. 32 There is evidence that linoleic and linolenic acid also effect other modes of action in the THS, as they are able to displace T4 from serum proteins in human serum. 33 The flavonoid morin was categorized into category 1B by DIO-SK assay data. Several flavonoids, including morin, inhibited the DIO1-catalyzed degradation of rT3 in murine thyroid microsomes. 34
Pentachlorophenol (PCP) inhibited DIO1 by 43.5% in the DIO1-SK assay and was classified into category 2. Treatment with up to 30 mg/kg b.w. PCP decreased total and free T4 and T3 levels, as well as thyroid-stimulating hormone levels and T4:T3 ratio in Wistar rats, suggesting interference at pituitary or hypothalamic level. 35 PCP downregulated DIO1 transcription in rat pituitary GH3 cells and—in zebrafish embryos—upregulated the expression of genes along the THS, including DIO1 and DIO2. 36 In summary, there are few data on DIO1 inhibition in vivo and hence classical reference data linking in vitro inhibition directly to in vivo inhibition are sparse.
Disturbance of the THS in vivo, on the other hand, can hardly be traced back to DIO1 inhibition only. Single methods addressing THS disturbance, like many other effects on complex systems, are neither addressed by a single in vitro assay nor validated one-on-one. Rather a battery of assays addressing different steps are employed and are validated by their physiological or mechanistic relevance.37,38 Moreover, if in vitro data are used, one should keep in mind that the DIO1-SK assay is a cell-free test system. In vitro to in vivo extrapolation is needed to extrapolate in vitro DIO1 inhibition data to external exposure to humans.39,40
Data interpretation procedure
For chemical safety assessment, results of the DIO1-SK assay and their relevance toward potential adverse effects on human health need to be clarified. A correlation of generated in vitro DIO1 inhibition data to in vivo data is difficult because (i) information on DIO1 inhibition in vivo is rare, (ii) described in vivo DIO1 inhibitors often target multiple thyroid mode of actions, and (iii) the biological relevance of DIO1 inhibition is unclear. In this study, we propose a DIP to assess DIO1 in vitro inhibition data in relation to the well-characterized reference item 6PTU and used control items (Table 8). A DIP uses a fixed algorithm (e.g., rules or decision criteria) in the context of a defined approach to interpret data from defined methods and is regularly used in the interpretation of data from NAMs, 41 for example, OECD guideline number 497. 42
The proposed DIP expands the DIO1-SK assay and allows the testing of unknown test substances for DIO1 inhibition with subsequent categorization (Fig. 6). This categorization potentially informs follow-up activities, for example, prioritization for further testing.
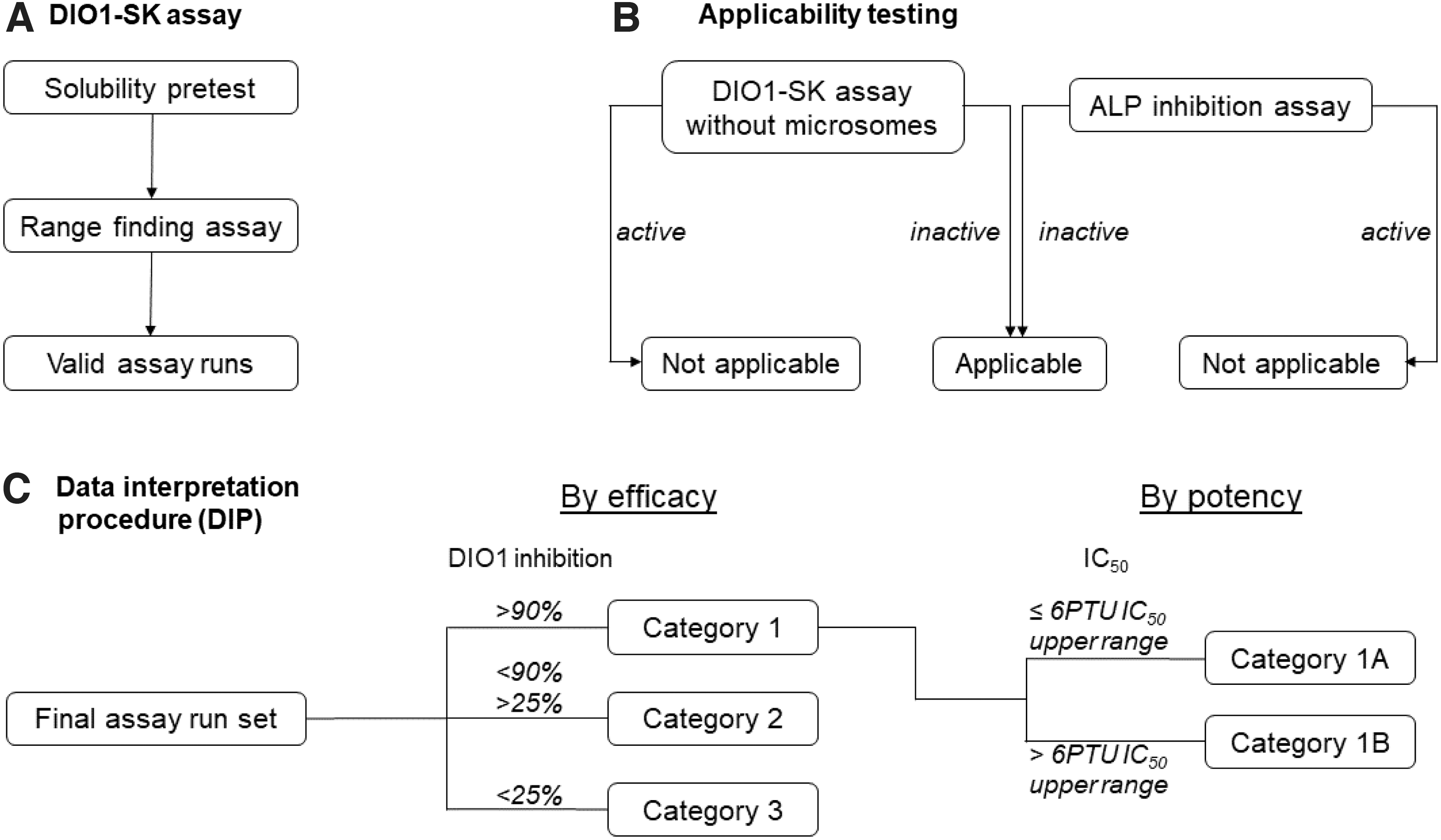
Testing strategy for unknown test substances in the DIO1-SK assay. The highest assay concentration is defined in a solubility pretest and used for testing in an initial range-finding assay. If DIO1 inhibition was observed, concentrations were changed for the following assay runs. In any case, at least three valid assay runs are performed.
Assessment criteria were derived to assess the DIO1 inhibition qualitatively and quantitatively. 6PTU is known as a complete and potent DIO1 inhibitor. 6PTU is used as the reference item in this method to control its performance. It is also used as a reference to classify the results of a test substance regarding its efficacy (complete vs. incomplete inhibition) and potency (inhibition at low, up to 7.1 μM, and high substance concentrations). Likewise, negative and vehicle controls are used to set thresholds to distinguish inhibitors from noninhibitory substances (efficacy only without additional potency classification for partial inhibitors). These thresholds are based on the variability of the control data from this study, which was performed in one laboratory by different technicians. DIO1-SK assays performed in another laboratory may show a different variability, potentially requiring different thresholds. Therefore, each laboratory should derive historical control data.
It might be beneficial for each laboratory to derive their own historical database and derive thresholds for inhibition assessment. Until this historical database is built, a daily derived IC50 value for 6PTU could be derived to estimate DIO1 inhibition. The threshold here could be based on the SD of the derived IC50 value (e.g., mean IC50 +/−3*SD). This means that the threshold values for the respective laboratory conditions are valid, and a lengthy buildup of laboratory data is prevented. The use of the same controls and performance standards across different laboratories helps to adjust for interlaboratory differences.
The proposed DIP is based on data derived from the DIO1-SK assay. Thus, it may not be directly applicable to data generated by other assays. Hence, we are using the DIO1-SK assay results of four test substances previously published and categorizing them according to our DIP 9 : 5-Propyl-2-thiouracil, an isomer of 6PTU, would be classified in 1A. 2-Chloro-N-phenylacetamide fully inhibited DIO1 with an IC50 of 7.4 μM and would hence be classified in category 1B. Tetrabromobisphenol A and genistein were poorly soluble and the maximum tested concentration only partially inhibited DIO1 by 51% and 41%, respectively. Accordingly, both substances would be classified in category 2. Other studies tested both substances at higher, potentially not fully soluble, concentrations, leading to complete DIO1 inhibition.3,5
Obviously, undissolved fraction of the test substances can contribute to the inhibitory effect in vitro, which is probably of no relevance to the in vivo situation. This demonstrates the importance of carefully examining the solubility of test substances and setting relevant upper concentrations. We have also tested gold-containing substances, including organic substances like the positive control ATG, sodium aurothiomalate or auranofin, and inorganic salts [gold(I) chloride, gold(III) chloride] in the DIO1-SK assay. 43 All tested organic and inorganic gold substances fully inhibited DIO1. The least potent gold substance was gold(I) chloride with an IC50 of 0.95 μM. Thus, all tested gold-containing substances would be classified in category 1A.
Conclusion
The DIO1-SK had previously been demonstrated to be reproducible. 9 In this study, the predictivity of the assay has been demonstrated: The results were obtained by testing 22 blinded test substances and, after deblinding, were compared to already published in vitro and in silico data. As the results of the DIO1-SK assay are intended to be used for regulatory decision making, we propose to use inhibitor categories rather than IC50 values. A DIP using maximum inhibition (efficacy) and IC50 (potency) was used to classify test substances into “potent full inhibitors” (category 1A), “weak full inhibitors” (category 1B), “partial inhibitors” (category 2), and “no inhibitors” (category 3).
Inhibition categories as determined by the DIO1-SK assay were in accordance with previously published data. To avoid mistaking unspecific activities of a test substance for specific DIO1 inhibition, two control assays were used (DIO1-SK without microsomes and ALP assay). Based on these, 2 of the 22 test substances were excluded from the assessment of their DIO1 inhibition activity as being nonspecific.
With this, we developed a reproducible DIO1 inhibition assay, including controls for unspecific effects and a DIP to transfer assay results into inhibition categories, which are pertinent to regulatory decision making. Based on previously available in vitro and in silico data, the results of this DIO1-SK assay with its DIP are predictive. In vivo data as reference are, however, not available, and hence the in vivo relevance of in vitro assay results is not yet explored. Obviously, DIO1 inhibition is but one of many events, which could disturb the THS leading to adverse effects. Ultimately, the DIO1-SK assay must be combined with other in vitro and in silico assays addressing other disturbances of the THS. DIO1 inhibition along with other key events of THS disturbance need linking to THS AOP networks 44 to allow valid conclusions on the hazard of a test substance and risk arising from the exposure to this substance.
Footnotes
Acknowledgments
The authors want to thank the team of EURL ECVAM, including Ingrid Langezaal, Roman Liska, Gerard Bowe and Aude Kienzler, for their valuable help during this project. Special thanks go to Chantal Hermann, Svenja Wingerter, and Hans-Albrecht Huener, who helped to generate the experimental data.
Author Disclosure Statement
The authors A.G.W., B.B., V.G., S.S., D.F.-W., and R.L. are employees of BASF SE, a chemical company, which may use the DIO1 assay to develop and register commercial products in the future. S.H., S.C., and K.R. declare no conflict of interests.
Funding Information
This work was funded by BASF SE, Key Technology Capability building Alternative Toxicological Methods. Used human liver microsomes were funded by EC JRC EURL ECVAM. In addition, the blinded test substances were acquired and paid for by EC JRC EURL ECVAM.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
