Abstract
Introduction:
Identifying substances which can disturb thyroid hormone (TH) signaling pathways is gaining importance and is soon to be a regulatory requirement in the EU. The European Commission Joint Research Centre's (EC JRC) European Union Reference Laboratory for alternatives to animal testing (EURL ECVAM) is coordinating the validation of multiple in vitro methods focusing on different thyroid modes of action (MoA) resulting in an in vitro test battery. Deiodinases (DIO) are regulators of TH signaling by activating or inactivating TH via deiodination. Inhibiting DIO activity is one potential MoA of substance-induced TH signaling disruption.
Objectives and Results:
Here, the DIO1-SK assay using human liver microsomes as a DIO1 enzyme source and the Sandell-Kolthoff (SK) reaction as a colorimetric readout to quantify released iodide was standardized and optimized according to Good In Vitro Method Practices (GIVIMP). Potential pitfalls are described, and possible solutions are provided. The reproducibility of the method was demonstrated by testing a set of six described DIO1 inhibitors in five independent assay runs. The results of this study were used to amend the testing protocol: a set of acceptance criteria was defined to assess the validity of assay runs and strategies to verify the specificity of observed DIO1 interaction were implemented.
Conclusion:
This work completes the first part of the DIO1-SK assay validation and defines a robust assay which provides results that can be used with confidence.
Introduction
The impairment of thyroid hormone (TH) homeostasis has been associated with several adverse outcomes: that is, hypothyreosis of mothers can impair the neuronal development of their children.1–3 Xenobiotic substances can disturb the different functions of the hypothalamic–pituitary–thyroid (HPT) axis. Such disturbances are key events (KE) that can be linked to adverse outcomes in adverse outcome pathways (AOPs).4,5 Several thyroid-related AOP networks have been published during the last years.6–8
Inhibition of the deiodinases (DIOs) is one KE (Event: 1002—AOP Wiki 5 ) linked to several thyroid-related AOPs, i.e., Adverse Neurodevelopmental Outcomes in Mammals (AOP8). The DIOs, a family of selenocysteine-containing enzymes, consist of three isoforms that regulate TH signaling through the deiodination of iodothyronines, resulting in the formation of metabolites with differing activity. DIO1 contributes to the triiodothyronine (T3) production in the thyroid and facilitates the recycling of iodide from TH metabolites in excreting organs such as the liver and the kidney. DIO2 and DIO3 regulate local TH signaling in peripheral tissue by activation of thyroxine (T4) to T3 (DIO2) or inactivation (DIO3) of THs. The expression of DIO enzymes varies in different tissues and in different stages of fetal development. 9
The DIO activity can be measured by quantifying TH and its metabolites in DIO-containing incubations using liquid chromatography–tandem mass spectrometry.10,11 This is, however, a demanding procedure that often provides more information than needed. Classically, DIO activity was investigated by using a radioactively labeled tracer substrate and quantifying the released radioactive iodide.12–15 Instead of using a radiolabeled substrate, the released iodide can be quantified by the Sandell-Kolthoff (SK) reaction. The reduction of yellow-colored cerium(IV) to colorless cerium(III) by arsenite is dependent on the concentration of iodide that is released from the substrate. The color change is quantified by light absorption at 415 nm. 16
The SK reaction has been used to quantify iodide for various investigations, such as the determination of urinary iodide concentration, 17 the uptake of iodide through the sodium–iodide symporter,18,19 cellular TH uptake by the TH transporter, 20 and released iodide from iodotyrosine DIOs21,22 as well as DIOs.23–25 The SK method is universally accessible and rather simple to perform, but is susceptible to interferences. Several ions and molecules are known to directly affect the reaction including iodide containing compounds, 26 nitrite and ferrous ions, 16 copper, chromium, Ni2+, Hg2+, Al2+, or thiocyanate. 27
The method to measure the effect of a test substance on DIO1 activity in murine liver microsomes was originally published by Renko et al. 23 This method was listed by the OECD scoping document that summarized available in vitro and ex vivo thyroid assays. 28 The method was further developed and evaluated as part of a validation study for 18 in vitro methods covering different AOPs of the HPT axis by the European Union Network of Laboratories for the Validation of Alternative methods (EU-NETVAL) coordinated by the European Union Reference Laboratory for alternatives to animal testing (EURL ECVAM). The method validation is conducted in two parts: assessing the method's reproducibility in part 1 and reliability and relevance in part 2. High standards regarding reproducibility, standardization, and documentation are essential to use methods with confidence—especially those being proposed for regulatory application. 29
The development and validation of this method adhered to the “Guidance Document on Good In Vitro Method Practices (GIVIMP)”,29,30 to define a standardized in vitro method that can reproducibly show DIO1 inhibition in human liver microsomes. In this study, we report the results from part 1: further development and assessment of transferability and reproducibility (“Reproducibility Assessment Study”). The method of Renko et al was adapted to human liver microsomes, standardized, and optimized for robust and reproducible performance (Fig. 1). The method was tested regarding its reproducibility and performance (Reproducibility Assessment) by testing six described DIO1 inhibitors in five valid assay runs.
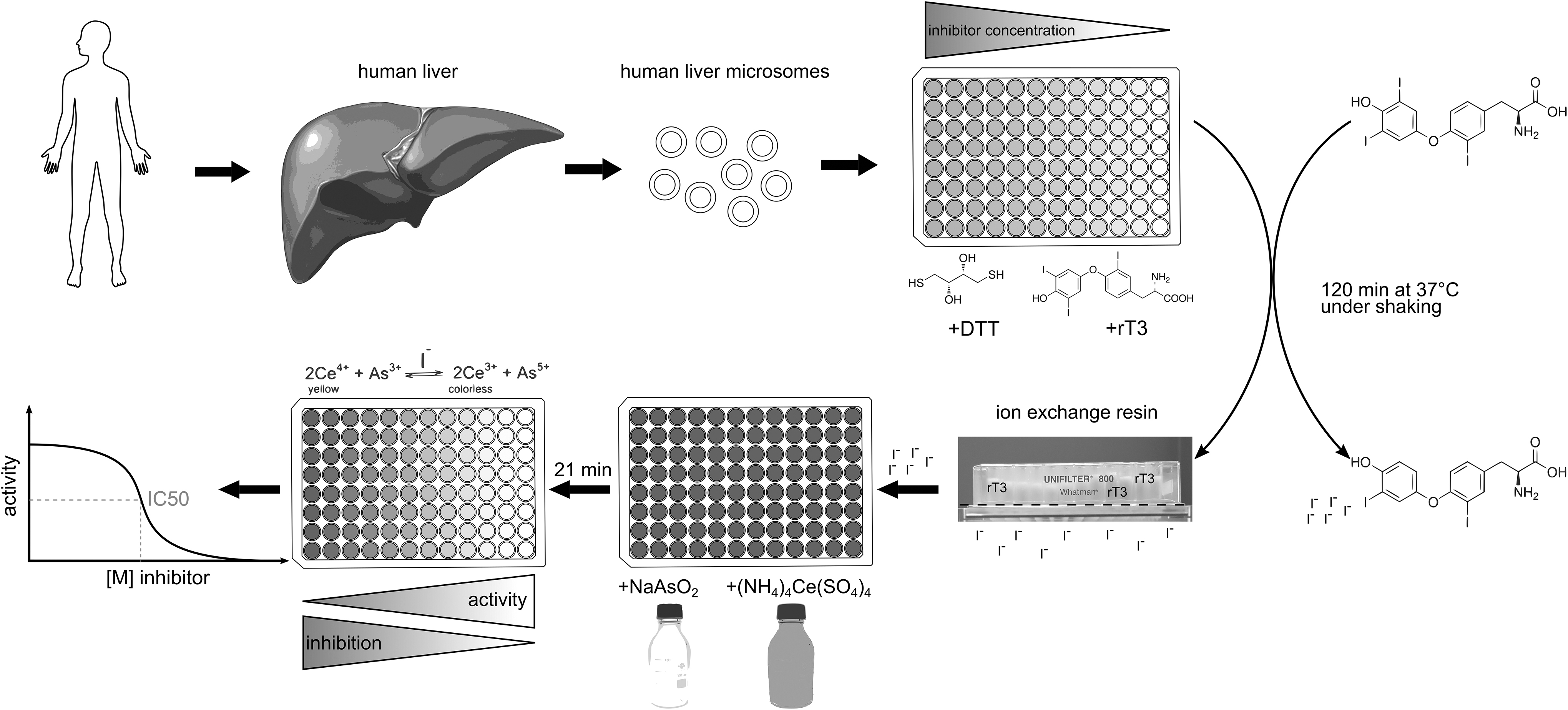
Schematic procedure of the DIO1-SK assay. Human liver microsomes are incubated with the substrate rT3, an artificial enzyme-regenerating cofactor DTT, and the test items under pH-buffered conditions. The released iodide is separated from the remaining assay constituents including substrate by an ion exchange resin. The iodide concentration of the eluted sample is then quantified by the SK reaction using the formation of colorless Ce3+ from yellow Ce4+ by arsenite that is catalyzed by the available iodide concentration. Curve fitting is used to derive inhibition values for tested items. DIO1, deiodinase I; DTT, dithiothreitol; rT3, reverse triiodothyronine; SK, Sandell-Kolthoff.
Testing strategies were introduced to exclude nonspecific effects of the test substance: test items were tested without microsomes to reveal potential interference in the SK reaction, and inhibition of a secondary microsomal protein, the alkaline phosphatase (ALP), was addressed. This strategy follows the concept of cell viability readouts as an indication of nonspecific effects in cell-based in vitro methods. A detailed description of the optimized and standardized method including its challenges is provided and will enable other laboratories to obtain reliable data on DIO1 inhibition using human microsomal preparations.
Materials and Methods
Standard operation procedures
The method standardization and optimization led to two standard operation procedures (SOPs) providing information on the test system, material, method, and data evaluation. The first SOP “SOP: DIO1-SK assay” addresses the DIO1-SK assay and was approved by the EURL ECVAM (Ispra, Italy) as well as the original method developer, Renko, on September 29, 2020 (Supplementary Pdf S1). This SOP was used during this thyroid validation study. The second SOP “SOP: ALP testing” provides a method to assess another enzyme activity in microsomes, which is independent of DIO1 activity and can hence be used to identify nonspecific DIO1 inhibition relying on general denaturation or protein binding (Supplementary Pdf S2). This SOP was developed in parallel to the thyroid validation study.
Material
The chemicals and materials used in the DIO1-SK assay are listed in the “SOP: DIO1-SK assay” (Supplementary Pdf S1). Chemicals and material that were obtained from other sources or used in addition to those listed in the SOP are explicitly stated in this article. Reverse T3 (rT3) was ordered from Cayman (Ann Arbor), Santa Cruz Biotechnology (Dallas) and Sigma-Aldrich. Ninety-six-well filter plates were purchased from VWR (microplates, 96-well, clear polystyrene, 800 μL, DNA Binding; Whatman) or Sigma-Aldrich (800 μL, clear polystyrene, filter bottom with long drip; Whatman). DOWEX ion exchange resin was purchased from Thermo-Fisher (50WX2-200; ACROS Organics) or Merck (AmberChrom® 50WX2 hydrogen form, 100–200 mesh).
Human liver microsomes were purchased from Sigma-Aldrich (pooled), Thermo-Fisher (pooled, 50 donors) or BioIVT (Westbury, NY). The used batch #QQY of human liver microsomes for the Reproducibility Assessment was distributed by the European Union Reference Laboratory for alternatives to animal testing (EURL ECVAM) and originally ordered from BioIVT; the batch was derived from 150 donors of mixed gender. The other used microsome batches met the minimum requirements that were set in the SOP (Supplementary Pdf S1).
Control and test item preparation
The control and test item preparation is described in the SOP (Supplementary Pdf S1, SOP: DIO1-SK assay). In accordance with GIVIMP, positive, negative, and solvent controls as well as a reference item were used (Table 1). 6-Propyl-2-thiouracil (6PTU), a specific DIO1 inhibitor, was used as a reference item; aurothioglucose (ATG), an inhibitor of all DIO isoforms, was used as a positive control; a structural analog of ATG lacking the gold moiety, TGSS (1-thio-β-
Used Control Setup and Control Items in the Deiodinase I-Sandell-Kolthoff Assay in Accordance with Good In Vitro Method Practices
6PTU, 6-propyl-2-thiouracil; ATG, aurothioglucose; DIO1, deiodinase I; DMSO, dimethyl sulfoxide; TGSS, 1-thio-β-
Control item and test item stock solutions as well as their respective predilutions were prepared according to SOP: DIO1-SK assay.
DIO1-SK assay
The incubation of human microsomes with the test and reference items and the positive, negative, and solvent control, as well as the separation by ion exchange and the SK reaction was performed according to SOP: DIO1-SK assay (Supplementary Pdf S1); plate layouts are also presented in this SOP. In short, human liver microsomes were incubated with the test item, the substrate rT3, the cofactor dithiothreitol (DTT), and a phosphate/diethylene diamine tetraacetic acid (EDTA) buffer. The final concentrations were as follows: 0.1 M H2KPO4/HK2PO4, 40 mM DTT, 1 mM EDTA, 5 μM rT3, and 1% dimethyl sulfoxide (DMSO). The reactions were constantly shaken for 2 hours at 37°C. Released iodide was separated using ion exchange resin-filtered plates and subsequently quantified using the SK reaction.
Data processing
Data were processed according to “SOP: DIO1-SK assay” (Supplementary Pdf S1) deriving multiple values describing the result of the assay (Table 2). The sigmoidal dose–response model “log(Inhibitor) versus response − Variable slope (four parameters)” (GraphPad Prism version 9.3.1 for Windows; GraphPad Software, San Diego, CA), was used to visualize a concentration–response relationship of calculated iodide release activities (IRAs) and used test item concentrations [Eq. (1)]. This was used to estimate the concentration causing 50% inhibition (IC50) of the test item.
Calculated Values in the Data Processing of the Deiodinase I-Sandell-Kolthoff Assay
BG, background; IRA, iodide release activity.
Equation 1: The sigmoidal dose–response model “log(Inhibitor) versus response − Variable slope (four parameters)” was used to visualize concentration–response relationships of test items. “Top” represents the maximal response, “Bottom” represents the lowest response, and “HillSlope” describes the steepness of the curve.
Assessing assay performance
Various control parameters were used to check the proper function of the assay: the IRA of the positive and negative control, the coefficient of variation (CV) of the log IC50 estimate for the reference item, and the z′-factor that is widely used to assess quality of assays. 33 A z′-factor above 0.5 is considered sufficient to demonstrate the significance of an assay. 34 The z′-factor was calculated using the standard deviation and mean of ΔOD21 minutes of the nine replicates of the reference item and the solvent control on each assay plate [Eq. (2)].
Equation 2: calculation of the z′-factor. “RI” represents the reference item 6PTU, “SC” represents the solvent control, “σ” represents the standard deviation, and “μ” represents the mean.
Microsome batch-specific IRA
For each microsome batch, a microsome batch-specific IRA of the solvent controls was determined according to “SOP: DIO1-SK assay” (Supplementary Pdf S1) and the defined microsome concentration, as well as the defined dilution factor of samples in the SK reaction, was used for testing of this microsome batch in the DIO1-SK assay.
Reproducibility assessment
The literature was screened for potential DIO1 inhibitors as potential test items. Six test items were chosen based on their inhibitory activity and tested in the DIO1-SK assay (Table 3).
Summarized Information About the Used Batches of the Test Items 6-Propyl-2-Thioruracil, 5-Propyl-2-Thiouracil, Aurothioglucose, Genistein, 3,3′,5,5′-Tetrabromobisphenol A, and 2-Chloro-N-Phenylacetamide
2CPA, 2-chloro-N-phenylacetamide; 5PTU, 5-propyl-2-thiouracil; 6PTU, 6-propyl-2-thioruracil; ATG, aurothioglucose; GEN, genistein; TBBPA, 3,3′,5,5′-tetrabromobisphenol A.
6PTU and ATG were used as a reference item and as a positive control, respectively. Both substances were also used as test items. On each plate, the reference item and the solvent controls were tested in nine replicates, and the positive and negative controls in three replicates. Each test item was tested in eight concentrations in triplicates. For each test item, five valid assay runs were performed. The assay runs were performed by three different laboratory staff members to check the intralaboratory reproducibility.
ALP activity testing
The ALP is located in human microsomes, and its activity is independent of DIO1 activity. In this study, ALP activity is used to identify nonspecific inhibition of DIO1. Testing for ALP activity was performed according to “SOP: ALP activity testing” (Supplementary Pdf S2) with one exception: only the highest concentration of each test item was used for ALP activity testing.
ALP activity testing is based on the formation of yellow (405 nm) para-nitrophenol from para-nitrophenyl phosphate. The tissue-nonspecific alkaline phosphatase (TNAP) inhibitor [2,5-dimethoxy-N-(quinolin-3-yl) benzene sulfonamide, CAS No. 496014-13-2] is described as a specific TNAP inhibitor 35 and was used as the reference item for ALP activity testing.
Number of repetitions
All experiments were carried out in at least three technical replicates; if more replicates were conducted, the number of technical replicates was specified for this experiment.
Experiments that were carried out for the standardization and characterization of the method were usually performed as one biological replicate. While independent biological replication is preferred, assay optimization decision points can be derived from a single experiment.
Concentration–response testing and specificity testing of test items were performed in at least three biological replicates.
Results
This section describes the results of the method development, modifications, and improvements, the results of the Reproducibility Assessment Study concerning performance and reproducibility, as well as the test results obtained with the six test items.
Initial method development
To confidently identify DIO1 inhibitors among the test items, a high resolution of the response, characterized by the calculated ΔOD-BG (BG: background), is essential. The response depends particularly on (i) the maximum SK-reaction obtained with the solvent controls and (ii) the minimum background SK-reaction obtained with the maximum concentration of the reference item.
The maximum SK-reaction (i) may be influenced by a variety of factors, mainly the IRA of the used microsome batch and the microsome concentration, incubation time, and used concentrations of reagents. The background reaction (ii) should be kept at a minimum. Increases in background could either result from assay constituents or impurities that are active in the SK reaction itself; this can be outlined by the ΔOD21 minutes values of the reference item. Precipitations can be formed from the test item or reactions of the test item with assay constituents; these precipitations can influence the initial measured OD415nm.
Role of assay constituents in the method
The DIO1 activity of murine liver microsomes is known to be inhibited by the reference item of the method, 6PTU, as well as heat inactivation. 23 In the present study, the DIO1-SK assay was performed with untreated, heat-inactivated, 6PTU-inhibited, or heat-inactivated as well as 6PTU-inhibited human liver microsomes. 6PTU only or in combination with heat-inactivated microsomes led to ΔOD21 minutes comparable with those of heat-inactivated microsomes. This is indicating full inhibition of DIO1 activity by the reference item 6PTU in human liver microsomes (Fig. 2A).
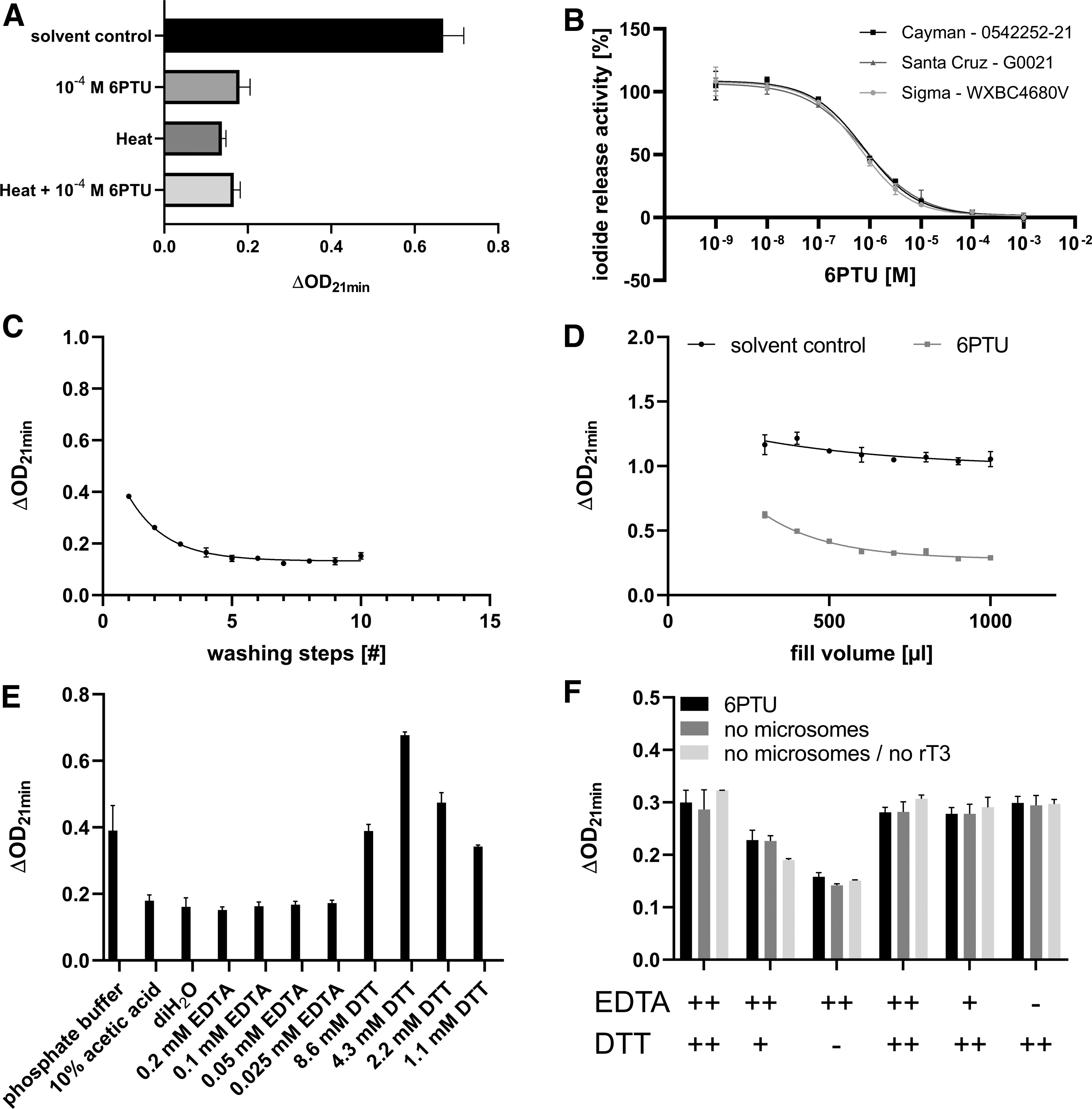
Characterization of assay constituents and ion exchange separation.
Multiple batches of different suppliers of the substrate, rT3, were compared regarding their activity. Concentration–response curves with the reference item 6PTU were derived for each substrate batch to exclude potential impact of the substrate, for example, through impurities. All rT3 batches produced curves that produced comparable 6PTU concentration–response curves (Fig. 2B).
This method uses ion exchange resin to separate organic-bound iodide molecules from the released iodide products. The ion exchange resin needs to be prepared as an aqueous suspension where it forms a reddish, cloudy, and viscous suspension. Before filling into filter plates, the resin suspension is washed several times with 10% acetic acid for 10 minutes to remove present dyes and exclude interference in the SK reaction. Samples of the supernatant of each washing step were tested in the SK reaction. The supernatants of the first washing steps led to a decrease of the OD415nm after an initial measurement (data not shown) and an increase of the background reaction. After five washing steps, these effects reached a plateau; additional washing steps did not further reduce the background reaction (Fig. 2C).
Due to the physical properties of the resin, some separation of the aqueous part from the resin cannot be fully avoided, even under constant stirring. Resin suspensions with different viscosities were prepared and casted into a filter plate. The wells that were filled with viscous resin suspension resulted in overall higher reaction values compared with the samples that were prepared with more liquid resin suspension (Supplementary Fig. S1), further showing the importance of uniform resin distribution over the plates.
In addition to separating substrate and free iodide, the resin filtration step may also retain other assay constituents that would drive the SK reaction. Microsome incubations with solvent control and reference item were filled into a filter plate that was prepared with different volumes of resin suspension. After the ion exchange, optical densities (ODs) were measured in the SK reaction. ODs of the solvent control as well as reference item were dependent on the resin volume. A minimum volume of 600 μL of resin suspension kept the influence of assay components in the assay to a minimum (Fig. 2D). Since the resin itself had no effect on the SK reaction, an increased leakage of assay constituents from the incubation step through the ion exchange separation into the SK reaction was assumed as the cause for background increases. The individual constituents of the assay were tested in the SK reaction without prior ion exchange.
The cofactor DTT increased the background reaction as well as the initial measured OD. Increasing concentrations of EDTA led to a decrease of initial measured OD while having no effect on the background reaction. Interestingly, phosphate buffer at the used concentration also influenced both the initial measured OD and the overall reaction (Fig. 2E and Supplementary Fig. S2A). The ion exchange separation step likely acts as a barrier preventing leakage of assay constituents into the SK reaction. Different concentrations of EDTA and DTT were tested in the DIO1-SK setup, including microsome incubation and ion exchange separation, with additional controls lacking rT3 or microsomes or both. Still, DTT was able to drive the SK reaction in a concentration-dependent manner in all tested controls, while EDTA had no effect on the initial measured OD anymore (Fig. 2F); the ion exchange separation step likely prevented EDTA from leaking into the eluted sample.
Different concentrations of phosphate buffer and the HEPES buffer as a replacement for the phosphate buffer 24 were tested regarding potential background increases. Phosphate buffer showed little reduction of initial measured OD at used assay concentrations (0.1 M) in both solvent controls and reference item samples. A reduced concentration of the phosphate buffer (0.025 M) or replacement of the phosphate buffer by the HEPES buffer negated the observed effect on an initial OD parameter (Supplementary Fig. S2C). To ensure that the reduced phosphate buffer concentration is not affecting substance-induced DIO1 inhibition, concentration–response curves with the reference item, 6PTU, were performed using 0.1, 0.05, 0.025, and 0.01 M phosphate buffer concentrations. 6PTU response curves were comparable with different phosphate buffer concentrations (Supplementary Fig. S2D).
In summary, DTT itself is active in the SK reaction, it is not retained in the ion exchange separation, and is the main driver of the observed background in the SK reaction.
Microsome activity
Human liver microsome batches show differences in their deiodination activity of rT3. This is resulting in different maximal ΔOD-BG values (as a measure of enzyme activity). A microsome–response curve with the batch QQY from BioIVT was performed in the DIO1-SK assay, and different dilutions of eluted samples after the ion exchange separation step were tested in the SK reaction (Fig. 3). Increasing microsome concentrations led to a higher overall reaction in the solvent controls up to 10 μg of protein per reaction volume (Fig. 3A1). Concentrations higher than 10 μg led to increased background reaction as observed in the reference item-treated samples (Fig. 3A2). Higher dilution of samples in the SK reaction overall led to a decrease of both solvent controls and reference item-treated samples and prevented higher achievable ΔOD-BG.
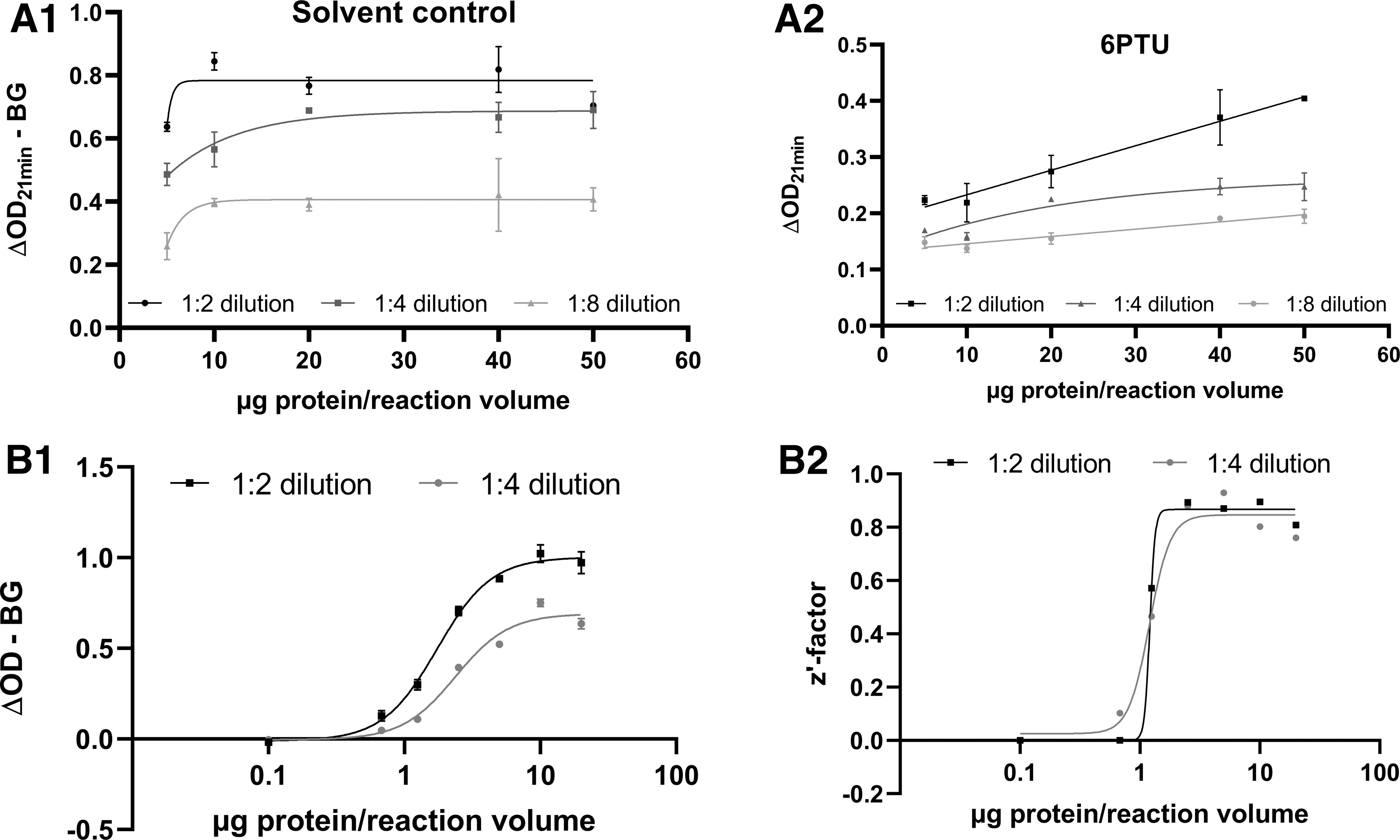
characterization of microsome activity and sample dilution in the SK reaction.
Based on background increases at high microsome concentrations observed in several microsome batches, 20 μg was defined as the highest microsome concentration for future testing of individual microsome batches.
The generation of a microsome batch-specific enzyme activity curve is used in this method to assess the IRA and to determine a microsome batch-specific protein concentration to be used for assay runs. The aim is to determine the microsome concentration per reaction volume and the 10% acetic acid dilution in the SK reaction that offers the highest OD resolution between solvent controls and the reference item. As an example, a concentration–response curve was derived using different concentrations of the microsome batch QQY with solvent controls and reference item samples for each microsome concentration. The respective z′-factors were derived (Fig. 3B). The ΔOD-BG obtained in the SK reaction after incubation with different microsome concentrations reached a plateau at a protein concentration of 10 μg of protein per reaction volume (Fig. 3B1). The calculated z′-factors plateaued at 2.5 μg of protein (Fig. 3B2).
For this microsome batch, 5 μg of microsome per incubation volume and a subsequent 1:2 dilution offered a high OD difference (represented by a high z′-factor).
Iodide quantification using the SK reaction
Increasing the cerium(IV) ion concentration might be one tool to compensate for increased background activity, provided the photometer used is still able to linearly quantify the resulting OD. In addition, increasing the incubation time might facilitate the maximum difference between solvent control and reference item. To check this, solvent control and reference item-treated samples were incubated from 1 to 4 hours and the released iodide content was measured in the SK reaction using solutions with increasing cerium concentrations. Both the increase of cerium concentration and the increased incubation times led to higher ΔOD-BG values in the solvent controls (Fig. 4A). Reference item samples only increased with higher cerium concentrations and were not affected by longer incubation times.
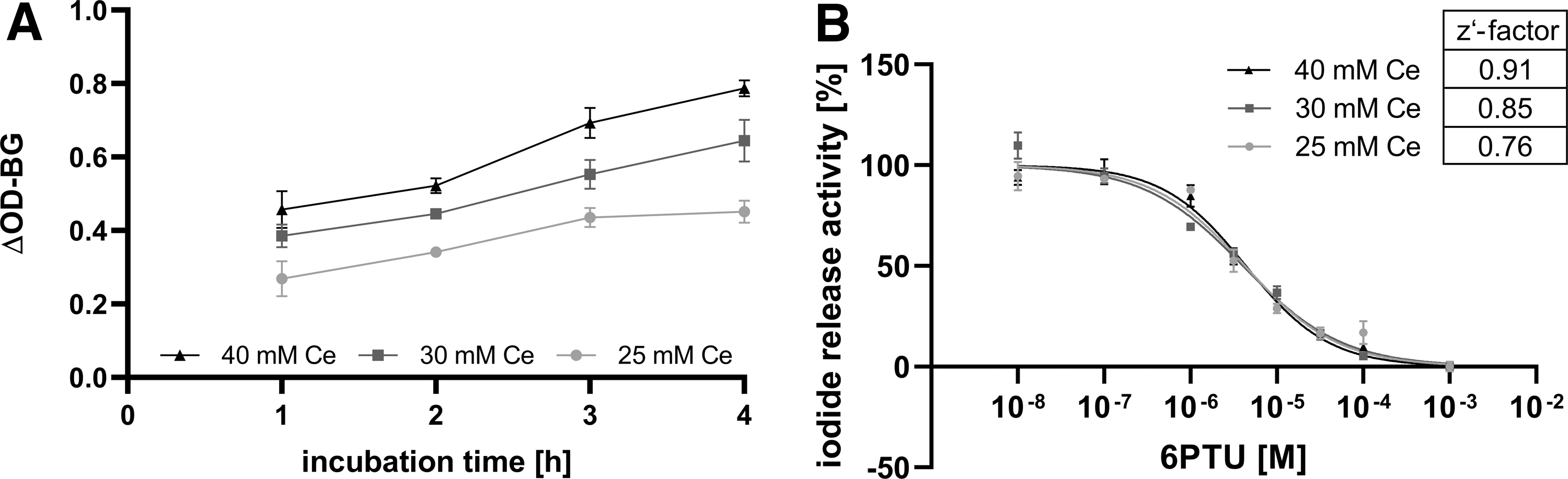
Impact of microsome incubation time and cerium concentration in the SK reaction on the performance of the method.
A concentration–response curve of the reference item was derived using 2 hours of incubation time, and different cerium concentrations were used in the SK reaction (Fig. 4B). The increase of cerium in the SK reaction had no effect on the reference item concentration–response curve, but increased the z′-factor.
Additional assay procedures were addressed to further optimize the method. Initially, released-iodide containing samples were eluted in the ion exchange separation step by the application of vacuum to ion exchange resin-filled filter plates. This elution process led to differences in the signal of wells as a function of their distance to the vacuum source, and sometimes failed to fully elute samples that were far away from the vacuum source. As an adaption, plates were eluted using centrifugation with free swinging buckets, which resulted in a more uniform elution. The centrifugation speed that was needed to fully elute the entire volume of samples varies between different filter plates; 150 g was generally sufficient to elute all resin-filled filter plates.
Based on the characterization and standardization efforts, the following setup was defined for the Reproducibility Assessment: The highest possible filling volume of 600 μL of ion exchange resin was used for the preparation of filter plates; the incubation was prepared with the defined microsome-batch-specific concentration using a 0.1 M phosphate buffer and was incubated for 2 hours; the SK reaction was performed with the defined microsome-batch-specific dilution factor in 10% acetic acid and 25 mM cerium(IV) ion solution.
Reproducibility assessment
The six test items in this Reproducibility Assessment were tested regarding their solubility in the stock solution in DMSO, the predilution in deionized water, and their final concentration in the incubation. The highest soluble concentrations in the stock solution and the final concentrations are shown in Table 3; detailed information about the solubility of each test item is shown in Supplementary Figure S3.
Five assay runs were performed for each test item in eight concentrations in the DIO1-SK assay, using information of pretests for the determination of final concentrations. 6PTU, 5PTU, ATG, and 2-chloro-N-phenylacetamid (2CPA) fully inhibited DIO1 activity and showed sigmoidal concentration–response curves with IC50 values of 4.12, 1.52, 0.50, and 7.39 μM, respectively (Fig. 5A–C, E and Table 4). Tetrabromobisphenol A (TBBPA) and genistein (GEN) only showed inhibition up to about 50% at the highest tested concentration and did not produce full concentration–response curves (Fig. 5D, E). No discerning IC50 values could be derived.
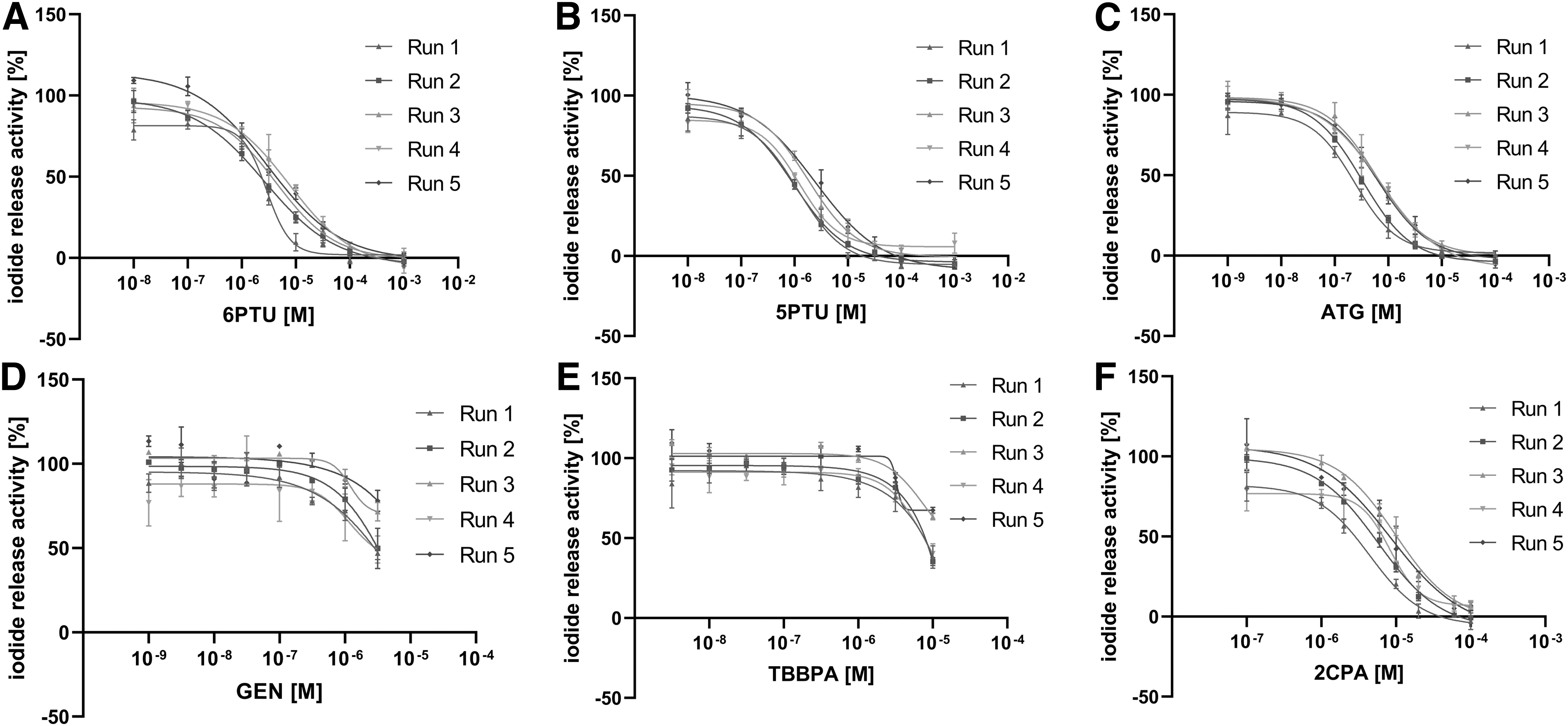
Reproducibility Assessment of the DIO1-SK assay. Iodide release activity in human liver microsomes after incubation with six known DIO1 inhibitors was determined (microsome batch #QQY, 5 μg protein/reaction volume, incubated for 2 hours at 37°C). Five independent assay runs with three technical replicates per concentration and test item were performed by three different laboratory staff members (data shown as mean iodide release activity ± SD of three technical replicates per concentration). A sigmoidal concentration–response curve was derived using the function “log(Inhibitor) versus response − Variable slope (four parameters).” 6PTU
Summary of the Concentration–Response Screenings of Six Test Items in the Reproducibility Assessment and Available Literature Data on Inhibition Values of Each Test Item
The model “log(Inhibitor) versus response − Variable slope (four parameters)” was used to derive inhibition curves and concentrations causing 50% inhibition (IC50) values for the six tested items.
2CPA, 2-chloro-N-phenylacetamide; 5PTU, 5-propyl-2-thiouracil; 6PTU, 6-propyl-2-thiouracil; ATG, aurothioglucose; GEN, genistein; SD, standard deviation; SK, Sandell-Kolthoff; TBBPA, tetrabromobisphenol A.
Performance of the method
The performance of control items was monitored for each assay plate (Fig. 6). The IRAs of the negative control and positive control were close to 100% and 0%, with low standard deviation (Fig. 6A, B). The IC50 values of the reference item, 6PTU, ranged from 2.65 to 7.60 μM (Fig. 6C) and the CV of the log IC50 estimate was always below 2%. z′-Factors were consistently over 0.5 (Fig. 6D); one run with a z′-factor below 0.5 had an apparent outlier in the reference item replicates, determined using boxplot outlier testing. After removal of this outlier, the run met the acceptance criterion.
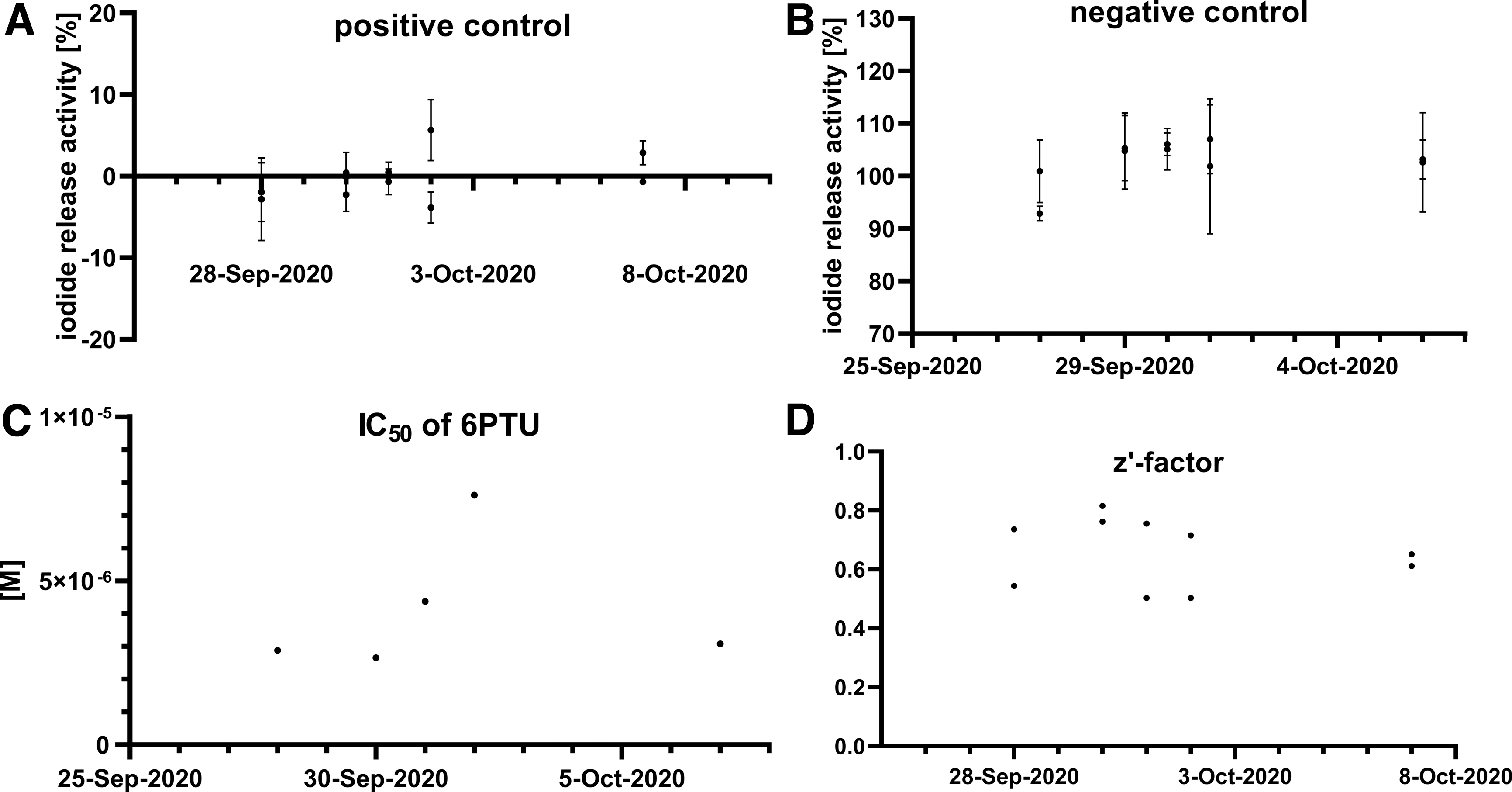
Performance of control values in the Reproducibility Assessment. The iodide release activity values for the positive control, ATG
To assess the variability between assay plates, the number of triplicates with a CV of 20% was determined. Among all triplicates of the 10 assay runs, only four triplicates showed a CV above 20%.
The results of the control items in the Reproducibility Assessment resulted in the selection of acceptance criteria as well as quantitative descriptors to assess the validity of assay runs (Table 5).
Proposed Acceptance Criteria for the Deiodinase I-Sandell-Kolthoff Assay
Shown are measured values of derived acceptance criteria in the Reproducibility Assessment and suggested cutoff values for future runs of the DIO1-SK assay. Mean and SD were calculated from the corresponding values of the 10 valid assay runs; criteria assessing reference item–response curve are only based on the 5 performed runs.
6PTU, 6-propyl-2-thiouracil; CV, coefficient of variation; IRA, iodide release activity.
Assessing specificity of DIO1 inhibition
In vitro assays often lack controls to account for potential nonspecific interactions. Many ions or iodide-containing substances are known to drive the SK reaction. To identify potential interference, the highest tested concentration of each test item and reference item, as well as positive and negative control of the reproducibility assessment, was incubated with and without a microsome. In the absence of microsomes, none of the tested items was active in the SK reaction (Fig. 7A).
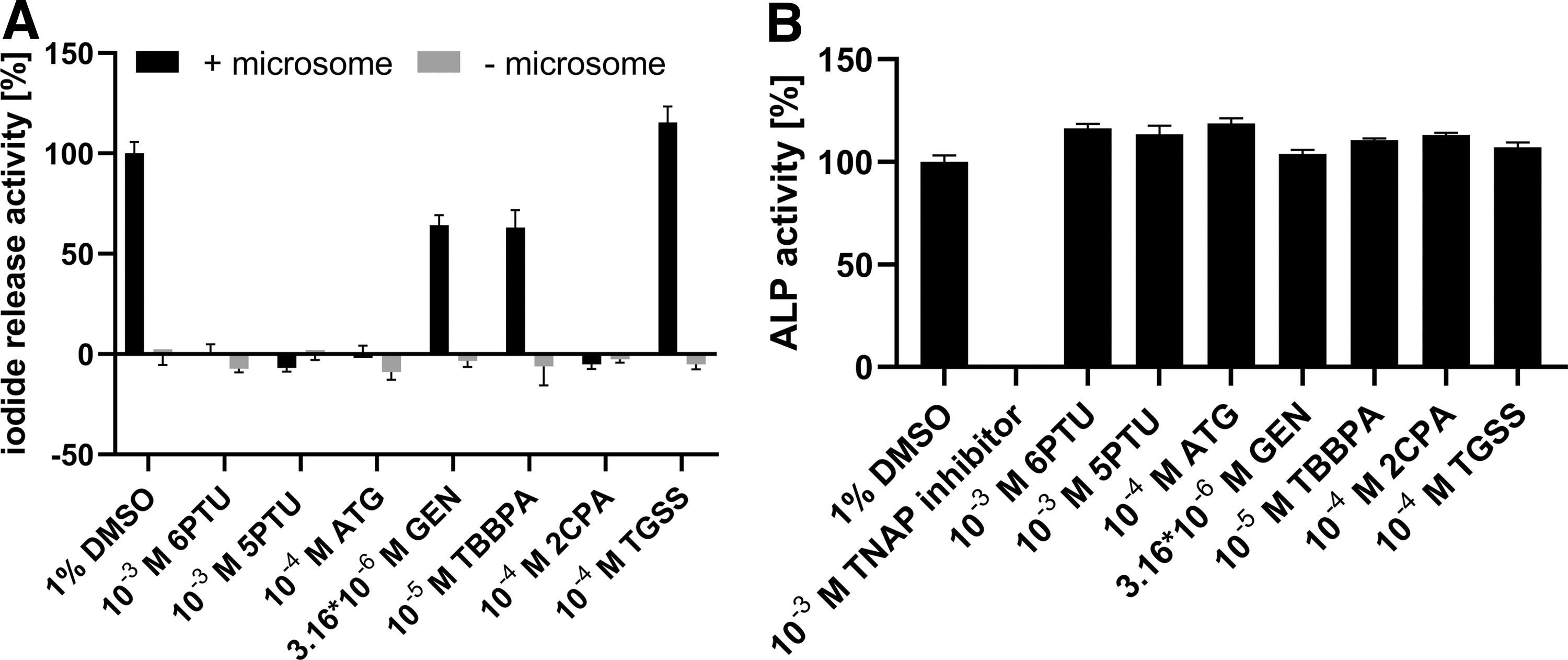
Specificity testing of control and test items of part 1.
The DIO1-SK assay is a cell-free assay using microsomes, and cell viability testing as an indication of nonspecific effects cannot be performed. Hence, the TNAP was used as an indicator of nonspecific inhibition, for example, due to nonspecific protein binding and modification, leading to structural denaturation. TNAP is one of the many isoenzymes of the ALP family. It has a main function in bone mineralization and is expressed in various tissues, including skeletal tissues, kidney, gut, and the liver. 36 A protein–response curve using human liver microsomes was linear up to 20 μg of protein per incubation volume (Supplementary Fig. S4A).
A specific TNAP inhibitor, 2,5-dimethoxy-N-(quinolin-3-yl) benzene sulfonamide (CAS No. 496014-13-2), 35 was used as a reference item to show ALP inhibition. TNAP inhibitor concentration–response curves were reproducible and produced comparable ALP inhibition curves in three independent runs with IC50 values of 0.19, 0.12, and 0.08 μM (Supplementary Fig. S4B). The six test items of the reproducibility assessment as well as the positive and negative control and the reference item of the DIO-SK assay did not inhibit ALP activity in human liver microsomes (Fig. 7B).
Discussion
Method development, optimization, and standardization
The influence of the assay conditions on the outcome of the assay was investigated and described in detail. The results were used to define the optimized assay conditions and to give advice to laboratories establishing the DIO1-SK assay. A process to establish the method is shown in Table 6.
Proposed Actions for Establishing the Deiodinase I-Sandell-Kolthoff Assay to Another Laboratory
diH2O, deionized water; DMSO, dimethyl sulfoxide; EDTA, diethylene diamine tetraacetic acid; OD, optical density; rT3, reverse triiodothyronine.
The SK reaction has been used by different groups to investigate iodide release as a function of DIO1 activity.23,24 Little information on the influence of the materials and assay conditions on the results has yet been investigated and published.
Human liver microsomes
Higher microsome concentrations up until 20 μg of protein per incubation volume led to a better resolution in the assay and increased the z′-factor; even higher microsome concentrations had no benefit. The microsome concentration that offers the highest OD difference between the reference item and solvent control that did not plateau in the microsome–response curve should be used.
Other materials used in the SK
It is highly recommended that batches of the substrate rT3 are tested once supplied to prove their performance and, upon availability of respective methods, identity. One out of the five batches used in this study did not show activity (data not shown).
The OD should linearly increase with the cerium(IV) ion concentration. Some cerium(IV) ion solution batches caused higher background in the SK reaction, likely due to iodide contaminations (Supplementary Fig. S5). These batches should be excluded.
The cofactor DTT can drive the SK reaction without iodide. DTT is used in the method to regenerate the DIO1 enzyme from its oxidized nonfunctional form. 37 The ion exchange resin can retain DTT and prevent it from leaking into the final SK reaction solution. A minimum fill volume of 600 μL of resin suspension per well is advised to significantly reduce the observed background reaction of DTT.
Phosphate buffer and EDTA decreased the measured OD parameters when added directly to the SK reaction solution. This is most likely caused by precipitations. Again, this is avoided by passing the solution through the ion exchange resin. Other groups have successfully replaced phosphate buffer by HEPES buffer. 24
Once the ion exchange resin is in aqueous suspension, it forms a reddish cloudy suspension that affects the measured OD. In addition, water-soluble components of the resin were shown to be active in the SK reaction. These observations varied among resin batches. The standardized washing procedure is sufficient to avoid this (Fig. 2). When filling plates with resin, the viscosity of the resin suspension might influence the amount of resin per well. This will impact the assay results.
Reading the OD of the SK reaction solution is time critical. Hence, the order and speed of measurements might influence the results. A plate reader that can simultaneously measure all wells of the plate is preferred. Otherwise, the time lag between measurements of different wells of a plate must be accounted in the study protocol.
Method improvements
The SK reaction was generally performed using a 25 mM cerium(IV) ion solution. Increasing the cerium concentration improved the z′-factors by increasing the ΔOD21 minutes of all samples, while only marginally increasing standard deviation. The use of 40 mM cerium(IV) ion solutions in the SK reaction might be a valuable adaption of the method leading to more robust results, especially under conditions with increased background reaction.
Increasing the time of microsome incubation might increase the dynamic range of the method. Two hours of incubation was sufficient to release enough iodide.
During assay development, additional assay procedures were addressed to further improve reproducibility of the method. Centrifugation is preferred over suction to elute samples from the ion exchange resin. The rapid addition of arsenite solution is crucial for a simultaneous start of the SK reaction. Arsenite should be added to all wells of a plate within a maximum of 15 seconds. Using pipetting tools such as a liquidator pipette (Steinbrenner, Germany) or an automated multichannel pipette is advisable.
Reproducibility assessment
Six test items were investigated in 10 assay runs to estimate the reproducibility of their concentration–response curves. A standardized control setup according to GIVIMP, using reference and control items and a set of acceptance criteria, was used to monitor the performance of the DIO1-SK assay and confirm the validity of results. Inhibition values of the respective controls and reference item were reproducible. Derived z′-factors generally met the acceptance criterion of 0.5. Future data evaluation might include outlier testing or robust statistics such as mean absolute deviation or the robust z′-factor. The reproducibility assessment was carried out by three different laboratory coworkers and no variation by the performing personnel was observed. In addition to this intralaboratory reproducibility study, an interlaboratory ring-trial should be performed to assess the reproducibility across different laboratories.
The results of the Reproducibility Assessment led to the adjustment of the initial acceptance criteria. There may be a need for further adaptations after gaining experience with the routine use of the assay in different laboratories.
The reference item, 6PTU, has shown full inhibition of IRA, comparable with the heat-inactivated microsome protein. As 6PTU is a specific DIO1 inhibitor 38 and no activity was observed in 6PTU inhibited samples, DIO2 or DIO3 activity contributing to the observed IRA can be ruled out, with the used substrate rT3. Concentration–response curves were reproducible in multiple biologically independent runs and comparable with those previously published (Table 4). 6PTU is a suitable reference item for this method. Little is known about the inhibition of DIO1 by the structural analog of 6PTU, 5PTU. Visser et al investigated the inhibitory potency of thiouracil analogs and found 5PTU to be a twice as potent as 6PTU. 39 This is consistent with our result indicating a 2.7-fold higher potency of 5PTU compared with 6PTU.
The positive control ATG is known to fully inhibit DIO isoforms, including DIO125,38 based on the affinity of its gold ligand to the selenocysteine-containing catalytic center of DIO. 40 Inhibition curves for ATG were reproducible, and the IC50 value of 0.5 μM was similar to the previous results of 0.64 μM. 32
The test item 2CPA was identified as an inhibitor of recombinant human DIO1 and showed only marginal DIO2 and 3 inhibition. 25 The DIO1 IC50 in this study, using human liver microsomes, was 7.4 μM. The IC50 determined in recombinant enzymes was 2.5-fold higher. Liver microsomes possess xenobiotic-metabolizing enzymes that may either inactivate or attenuate substrates of DIO1, or generate metabolites that are more or less active DIO1 inhibitors. 41 Many xenobiotic-metabolizing enzymes such as cytochromes P450 or uridine glucuronyl transferases require cofactors, whereas hydrolases such as esterases or epoxide hydrolases catalyze their reaction independent of cofactors.
Various aromatic amines, including 2CPA, were shown to be hydrolyzed in vitro when incubated with human liver microsomes, 42 potentially leading to more potent DIO1 inhibiting metabolites. Also, a different disposition of the test item in the in vitro systems (recombinant enzyme vs. microsomes) might lead to observed differences in inhibition potency. 43 The isoflavone GEN and flame retardant TBBPA were described as potent substances fully inhibiting DIO1.25,38 In these studies, GEN and TBBPA were tested up to 200 μM and 190 μM, respectively. In this work, a strong focus was placed on fully dissolving tested substances. This might result in lower concentrations tested for poorly soluble substances compared with other publications and might impact derived inhibition values. As such, the highest tested concentrations of GEN and TBBPA were 3.16 and 10 μM, respectively. Observed DIO1 inhibition of these concentrations was not high enough to derive reliable IC50 values.
Testing substances above the solubility limit can result in higher inhibitory potency if the insoluble components form a depot that provides a steady supply of disposable molecules. However, testing of precipitated substances could also introduce artifacts that are not related to the disposable concentrations. For further tests, the testing of concentrations that lead to homogeneous solutions might be included.
Improving specificity of the assay
The readout of this method is based on the SK reaction and a colorimetric readout of the redox reaction: the loss of absorption at 415 nm. Such loss-of-signal assays are susceptible to nonspecific interactions 44 that could be caused by various conceivable effects such as test items that are active in the SK reaction itself, interaction of the test item with the substrate or the cofactor, spontaneous iodide release, or nonspecific protein binding/modification. Testing of test items with and without an iodide-releasing enzyme can reveal test items that are able to release iodide without DIO1 activity. It can also reveal test items that are active in the SK reaction, provided they pass the ion exchange separation.
Both modes of interference would per se lead to a virtual change in iodide release. Testing for interference of the test item with the cofactor DTT was not further investigated. It was suggested to replace DTT with weaker reducing agents such as cysteine or glutathione since DTT was shown to be active in the SK reaction.
The testing of another enzyme present in liver microsomes independent of DIO1 activity provides information about the functionality of the microsomes. Inhibition of ALP and DIO activity in microsomes can indicate interactions of the test item with microsomes, which are not specific to the DIO1 activity. If the secondary analyzed enzyme is affected in addition to the DIO1 readout, this hints toward nonspecific interaction of the test item with the microsomes, ultimately leading to an inhibition of the enzyme activities. Obviously, this does not generally rule out the DIO inhibiting effects. The homodimeric protein ALP was used since its activity can be quantified by light absorption, and a photometer can be used for ALP and DIO1 testing.
In comparison with the DIOs, the ALP uses Zn2+ and Mg2+ as cofactors and more alkaline conditions are needed for enzymatic activity. 36 Neither of the six test items of the Reproducibility Assessment led to changes in absorbance without microsomes present nor inhibited ALP activity in human liver microsomes, indicating that their effects on DIO1 activity are specific. The ALP specificity testing approach increases the specificity of the DIO1-SK assay and will be included and challenged in part 2 of the assay validation.
Conclusion
In this work, the DIO1-SK assay was amended and improved making it a robust and standardized assay. SOPs were issued and are available as supplements, and further improvements are proposed. The different steps and conditions of the assays were investigated, and the effect of variations is described to inform the assay user and to ensure the robust performance of the assay. The reproducibility of the DIO1-SK assay was tested using a set of known DIO1 inhibitors. In addition, ALP activity tests were introduced to exclude nonspecific interactions of the test items. The DIO1-SK assay proved to be a reliable method in this intralaboratory Reproducibility Assessment Study (part 1). Xenobiotic-metabolizing competence of the method could be introduced by the addition of necessary cofactors. The results of the relevance study (part 2) will be published before long.
Footnotes
Acknowledgments
The authors thank Ingrid Langezaal, Roman Liska, and Gerard Bowe from EC JRC EURL ECVAM for their valuable help in the standardization of the method and review of the article.
Author Disclosure Statement
The authors A.G.W., B.B., C.H., H-A.H., S.S., D.F.-W., and R.L. are employees of BASF SE, a chemical company, which may use the DIO1 assay to develop and register commercial products in the future. S.C. and K.R. declare no conflict of interest.
Funding Information
This work was funded by BASF SE, Key Technology Capability building Alternative Toxicological Methods. Used human liver microsomes were funded by EC JRC EURL ECVAM.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
