Abstract
Introduction:
The current toxicological registration package for the contact fungicides Captan and Folpet relies exclusively on animal tests for acute endpoints. The actives rapidly degrade and are not systemically available in toxicologically relevant amounts, which may pose challenges in in vitro scenarios.
Materials and Methods:
To investigate the predictiveness of New Approach Methods (NAMs) for Captan and Folpet hazard assessments, a set of 18 in vitro studies was conducted, comprising eye and skin irritation and skin sensitization assays compliant with Organisation for Economic Co-operation and Development (OECD) test guidelines. Further performed were GARD®skin sensitization assays and rat EpiAirway™ acute airway toxicity assays, the latter with single and repeated exposure. The results were compared against publicly available and confidential information from the legacy vertebrate dataset for each fungicide, and also against current European health classifications and the proposed revision of these classifications.
Results:
The NAM data package appropriately identifies Captan and Folpet as contact irritants with predicted effects in eyes and the respiratory system and identifies the skin sensitization properties correctly for each fungicide. However, the assays under-classify eye irritation hazard, compared with animal data.
Discussion and Conclusion:
The results may indicate species sensitivity differences, which are plausible owing to morphological tissue differences. Hence, the suitability of animal test data for the assessment of human irritation risk from Captan and Folpet may be questioned as being overly protective.
Introduction
The active ingredients Captan and Folpet are contact fungicides and have been used in pesticidal products for >50 years. They react with biomembranes either directly or indirectly, through the in situ release of thiophosgene. 1 Thiophosgene is generated by their common trichloromethylthio-moiety; the primary degradation products are 1,2,3,6-tetrahydrophthalimide for Captan and phthalimide for Folpet. This reaction occurs quickly and results in fungicidal activity when used as a pesticide. The subsequent degradation and metabolism profile of the substances differ owing to the aromatic stability of phthalimide.
As Captan and Folpet are very reactive, they do not enter the systemic compartment in toxicologically relevant amounts. However, even if the fungicides could reach systemic circulation in significant quantity, their degradation half-time is <10 seconds and any released thiophosgene is sequestered within 1 second in (human) blood (Unpublished data; Gordon E, Williams M, 1999). 2
Any systemic effects observed in animal studies are thus most likely either the result of initial local irritation at the site of first contact, for example, in the gastrointestinal tract with related food reduction and associated body weight loss, or the respiratory tract with edema, or are not treatment related. Although systemic effects may be associated with the systemic metabolites of Captan and Folpet, their primary metabolites do not show clear or prominent toxicity (see Materials and Methods section for direct links to the European registration dossiers).
In mice, continuous dietary exposure to Captan and Folpet results in gastrointestinal tumors, when the concentrations are high enough to induce gastrointestinal irritation. However, tumors do not occur in rats or repeated dose studies in dogs even at concentrations irritating the gastrointestinal lining. The etiology of the effect is well investigated and driven by subsequent local irritation following repeated oral exposure and not genotoxicity.3–7
Acceptance of this etiology for carcinogenicity led to a cancer classification in the United States as “not likely to be carcinogenic to humans” for Captan8,9 and subsequently also adopted for Folpet, 10 which is clearly different from the current harmonized (up to 2022) European classification, according to the Classification, Labelling and Packaging Legislation (EU CLP). 11 An Adverse Outcome Pathway (AOP) was proposed 12 for small intestinal tumors upon very high dietary exposure involving chronic cytotoxicity and regenerative hyperplasia based on earlier work with Captan, Folpet, and hexavalent chromium as lead substances.7,13,14 This AOP is not considered to be relevant for human exposure. 12
The current toxicological registration package for the contact fungicides Captan and Folpet relies exclusively on animal tests for acute endpoints. Hence, it was investigated how New Approach Method (NAM) studies predict the known acute endpoints. This newly generated data may allow for a targeted use of these methods in regulatory packages of Captan and Folpet products, providing more human relevant toxicology hazard data with the side effect of contributing to a reduction of animal testing. The latter is a focus of the United States Environmental Protection Agency (US EPA). 15
Materials and Methods
Legacy dataset
Some key information on the proprietary legacy vertebrate dataset for both Captan and Folpet are given in Table 1. The vertebrate studies were conducted owing to legal registration data requirements to assess the toxicity of active ingredients for pesticide use; the studies were generated over a long historic time frame and in a range of different laboratories globally. When nationally required, the studies were approved by an institutional ethics committee. Most of the presented legacy dataset is the basis of the currently harmonized European classification.
Acute Inhalation Toxicity, Eye and Skin Irritation, and Skin Sensitization Legacy Registration Data
As the LC50 is estimated from the treatment concentrations the MMAD is stated that corresponds to the nearest treatment concentration to the LC50.
F, female; LC50, median lethal concentration; M, male; MMAD, Mass Median Aerodynamic Diameter; OECD, Organisation for Economic Co-operation and Development; RAR, Risk Assessment Report; US EPA, United States Environmental Protection Agency.
Exhaustive information on the available vertebrate studies or each fungicide can be found in their respective European Risk Assessment Reports (RARs) that were generated with the intention to register the fungicides as pesticidal active substances in Europe; the 2018 versions of Captan (efsa.europa.eu/de/consultations/call/180417) and Folpet (efsa.europa.eu/de/consultations/call/180417-0) RARs were published for public consultation. The relevant information for toxicology is summarized in Volume 1 of the RAR, with more detailed information in Volume 3, B.6. Reference to the relevant page numbers of Volume 3, B.6 is made in Table 1. Please note, however, that the RARs were subsequently updated after public consultation but are not yet publicly available.
For Folpet, six additional acute studies generated for registrations outside Europe were identified after 2018 as being relevant for the fungicides' assessment; however, their summaries are not yet available in the public domain and are, therefore, provided as Supplementary Material S3. The information contained therein is the basis for the currently proposed and revised European harmonized classification by the dossier submitter for Captan (echa.europa.eu/de/registration-dossier/-/registered-dossier/12775) and Folpet (echa.europa.eu/de/registry-of-clh-intentions-until-outcome/-/dislist/details/0b0236e186004927). The future harmonized classification, however, is not final and may deviate from the current European Chemicals Agency (EChA) Risk Assessment Committee (RAC) classification. The classification dossier (i.e., the “harmonised classification and labelling report”) is also not yet in the public domain or available to the authors but will eventually become available at the indicated links.* Legacy Dataset section indicates the rationale supporting the proposed classification.
The provided study information was gathered to give the reader a better understanding of the reasons for classification and the proposed revised classification, and to support comparison of the NAM data to existing in vivo study outcomes. Unfortunately, the study reports cannot be shared as supplementary information for this article owing to commercial interests of the Sponsor; however, the available summaries allow an assessment of the studies' relevance.
Newly conducted in vitro tests
NAM studies have been developed (and shown to be fit for purpose) to predict human hazards. This is in contrast to virtually all vertebrate studies, which have not been formally validated nor demonstrated to be fit for purpose. Vertebrate studies, however, are generally considered to be conservative even if they may not necessarily predict human hazards well. Hence, the NAM study package aims to:
evaluate the hazards more appropriately for humans, and elucidate whether there are options to replace required vertebrate data for Captan and Folpet products.
A total of 18 new studies were conducted to investigate the inhalation, eye, and skin effects of Captan and Folpet (Table 2) (note, studies were generated separately for each fungicide and the experimental phase for the Direct Peptide Reactivity Assay [DPRA] had to be terminated). All studies were Good Laboratory Practice-compliant except the EpiAirway™ studies for acute airway toxicity. All studies followed Organisation for Economic Co-operation and Development (OECD) test guidelines, except the GARD®skin and EpiAirway studies for which there are currently no formal guideline tests available. The tests are briefly described hereunder, and detailed summaries of the reports are available as Supplementary Material 1. Unfortunately, it is not possible to provide more detail on the individual assays in the main article because of size restrictions. In addition, the original reports cannot be made publicly available because of commercial interests of the Sponsor.
Conducted In Vitro Study Package
BCOP, bovine corneal opacity and permeability; DPRA, Direct Peptide Reactivity Assay; GLP, Good Laboratory Practice; RhCE, reconstructed human cornea-like epithelium; RhE, reconstructed human epidermis; TG, test guideline.
The same test material was used in all NAM studies. Captan technical (CAS No.: 133-06-2), batch number M2009042, was an off-white solid with a chemical purity of 94.6%. The expiry date is September 4, 2025. Folpet technical (CAS No.: 133-07-3), batch number 99138337, was an off-white solid with a chemical purity of 95.8%. The expiry date is May 4, 2024. Both Captan and Folpet test material were stored at room temperature and were not adjusted for purity in the assays.
Direct skin effects
Direct skin effects refer to reversible and irreversible damage to the skin, skin irritation, and corrosion, respectively, which are a result of cellular toxicity following direct contact with a test item. The effects can be evaluated in a single in vitro model test system evaluated according to two different OECD test guidelines, which differ only in the experimental procedure. The assay uses artificial tissue, reconstructed human epidermis (RhE), grown from human-derived nontransformed epidermal keratinocytes, which resembles the human epidermis.
The model tissue is prepared in inserts with a porous membrane to ensure nutrient supply to the cells. A cell suspension is seeded into the insert in specialized medium. After an initial period of submerged culture, the medium is removed from the top of the tissue so that the epithelial surface is in direct contact with the air. This allows the test item to be directly applied to the epithelial surface reflecting an in vivo exposure scenario or representing an overexposure. The tissue is commercialized in six-well assay kits. The surrogate endpoint in the assays is tissue viability in combination with exposure duration. The specific viability thresholds differ by the used test system; here, the EpiSkin™ Small model was used.
OECD 431: RhE—corrosion
The skin tissue was moistened with Milli-Q water (25 μL) and test item (at least 25 mg) was applied directly on top of the skin tissue for 3 or 60 minutes of direct exposure. The exposure was terminated by thoroughly rinsing the tissue surface with phosphate-buffered saline to remove the test item. The cytotoxic (corrosive) effect was then determined. Cytotoxicity is expressed as the reduction of mitochondrial dehydrogenase activity measured by formazan production from MTT in a plate reader at the end of the treatment.
OECD 439: RhE—irritation
The skin tissue was moistened with Milli-Q water (5 μL) and test item (at least 10 mg) was applied directly on top of the skin tissue for 15 minutes. After a 42-hour postincubation period, determination of the cytotoxic (irritancy) effect was performed. Cytotoxicity is assessed as in the corrosion assay.
Direct eye effects
Direct eye effects, like direct skin effects, refer to reversible and irreversible damage to the eye, that is, ocular irritation and damage, respectively.
Two assays were used here utilizing either an isolated cornea from bovine eye or an artificial tissue, reconstructed human cornea-like epithelium (RhCE), grown from human normal human keratinocytes, which models human corneal epithelium with progressively stratified, but not cornified cells. The bovine corneal opacity and permeability (BCOP) assay uses an in vitro irritancy score (IVIS) as the endpoint, which is based on corneal opacity induced by the test item. The RhCE assay is based on tissue viability, like the skin irritation tests.
OECD 437: BCOP
Isolated corneas were mounted in a corneal holder (one cornea per holder) with the endothelial side against the O-ring of the posterior half of the holder. The anterior half of the holder was positioned on top of the cornea and tightened with screws. The compartments of the corneal holder were filled with medium and incubated for the minimum of 1 hour at 32°C. Then the baseline opacity was determined. After removal of the medium from the endothelial side, the test item (303.0–397.2 mg) was applied to completely cover the cornea for 240 minutes. To determine the effect of moisture on the study outcome, a second experiment was performed in which physiological saline was added resulting in a test item concentration of 20% (w/v) on the cornea. After exposure, the cornea was washed with medium and assessed for opacity by measurement of the diminution of light passing through the cornea and corrected against baseline values.
OECD 492: RhCE
The EpiOcular™ (RhCE) tissue is similarly generated as the RhE tissue. The RhCE model was dampened with Ca2+Mg2+-free-
Skin sensitization
In vitro assays are used to identify the potential of a test item to induce certain key events in the skin sensitization AOP (see https://AOP-wiki.org).
The DPRA assay indicates key event 1 “Covalent interaction with skin proteins.” It is an in chemico method and investigates the binding of the test item to a cysteine- or lysine-containing peptide. The antioxidant/electrophile response element-nuclear factor-erythroid 2-related factor 2 (ARE-Nrf2) Luciferase (KeratinoSens™) assay indicates key event 2 “Keratinocyte responses,” which is characterized by inflammatory responses as well as gene expression associated with specific cell signaling (stress) pathways such as the ARE-dependent pathways. The U-Sens™ assay indicates key event 3 “Dendritic cell responses,” which is characterized by the expression of specific cell surface markers, chemokines, and cytokines. Key Events 4 and 5 are tested in the typical in vivo assays but are not considered to be required to predict the sensitization potential of a chemical (OECD Guideline No. 497).
OECD 442C: DPRA
Following a 24-hour incubation with the test item at 25°C, the concentration of cysteine- or lysine-containing peptide is assessed. The synthetic peptides contain phenylalanine to aid in the detection. The relative peptide concentration is measured by high-performance liquid chromatography with gradient elution and spectrophotometric detection at 220 and 258 nm. Cysteine and lysine peptide percent depletion values are calculated and used in a prediction model, which assigns the test item to one of four reactivity classes used to support the discrimination between sensitizers and nonsensitizers. Because no suitable assay compatible solvent could be found to dissolve the test items at the desired concentration, the assays were terminated, in compliance with this test guideline.
OECD 442D: KeratinoSens
The KeratinoSens test quantifies the induction of luciferase expression in an ARE-Nrf2 luciferase reporter assay. The test item was dissolved in dimethyl sulfoxide (DMSO) to create stock solutions. From those stocks, twofold dilutions series were derived and tested for luciferase expression in two assay repetitions, experiments 1 and 2, with cells cultured in 96-well plates.
OECD 442E: U-Sens
The U-Sens assay quantifies the change in the expression of a cell surface marker associated with the process of activation of monocytes and dendritic cells (i.e., CD86), in the human histiocytic lymphoma cell line U937. The test item was dissolved in DMSO to create stock solutions. From those stocks, 2 dilution series were derived for two assay repetitions, experiments 1 and 2, with cells cultured in 96-well plates. Surface marker expression was measured by antibody staining for CD86 and flow cytometry.
GARDskin
The GARDskin assay16–18 evaluates, with a machine learning algorithm, 19 test item–induced transcriptional patterns of genomic biomarker signatures in the SenzaCell cell line (ATCC Depository PTA-123875), that is, the messenger RNA (mRNA) expression of 200 genes. Based on the gene expression pattern for the test item, a decision value is calculated. A decision value >0 indicates skin sensitization potential.
Acute airway toxicity
The rat EpiAirway assay is a three-dimensional (3D) cell model of the rat mucociliary airway epithelium (MatTek Corp., Ashland, MA), derived from cells collected from the airways of healthy rats and cultured at the air–liquid interface to achieve a physiologically relevant morphology. The human tissue model has previously been described. 20 The commercial human versions, human EpiAirway and human MucilAir™ have been evaluated in prevalidation testing 21 and used extensively in air pollution, 22 pharmaceuticals, 23 chemicals, 24 and in crop protection. 25 This is the first formal description of the use of the rat EpiAirway model other than in poster publications.26–28
Following a dose range finding experiment, rat EpiAirway tissues (n = 4) were exposed to eight dilutions of the test item for either a single 24-hour period (single exposure), or for three continuous 24-hour periods (rinsing with prewarmed phosphate-buffered saline in between but no recovery periods so a repeated exposure scenario). The following analytical measurements were carried out.
Measurement of transepithelial electrical resistance (TEER) as a measure of the integrity of tight junctions between cells (predose and following each 24-hour exposure period).
Analysis of lactate dehydrogenase (LDH) release into the culture media as a measure of tissue damage (predose and following each 24-hour exposure period).
Histopathology (following the single or final exposure period [n = 2]).
Benchmark dose modeling
To quantify and compare the results in the rat EpiAirway studies, the results were submitted for a benchmark dose (BMD) analysis (Supplementary Material S2). This approach has been used previously in regulatory respiratory toxicology evaluation (US EPA 29 ).
BMDs and their lower (BMDL) and upper (BMDU) 90% confidence limits were derived for rat EpiAirway studies using the R 30 extension bmd.31,32 A benchmark response of 10% was generically used for deriving the critical effect size, that is, BMD was derived using a relative definition. The model selection was based on Akaike's Information Criteria (AIC) and visual inspection of model fit.
Results
Legacy dataset
The available in vivo acute toxicity registration packages for Captan and Folpet are briefly summarized in the sections that follow. The individual studies are given in Table 1, except those through oral and dermal route, where no effects were associated with test item exposure.
Acute toxicity
Captan and Folpet are not acutely toxic through the oral or the dermal route, that is, the results from the OECD test guideline compliant studies do not require classification under the European classification regulation.11,33 However, both Captan and Folpet are acutely toxic through the inhalation route, with harmonized classifications of Acute Tox Cat. 4 and Acute Tox Cat. 3, respectively. For Folpet, the proposed classification is revised to Acute Tox Cat. 2, owing to the availability of novel data for the classification process.
There are several acute inhalation toxicity studies in the rat available for both fungicides, because of the ownership of these substances by different companies over time, their registration in different countries, and their production at different plants. Some studies are not compliant with current test guideline requirements, with particle sizes exceeding the required limits. Nevertheless, and importantly, all studies show exposure of the respiratory system, evidenced by observed toxicity. The observed mortality is summarized in Figure 1. Overall, Folpet appears to be slightly less acutely toxic than Captan (Fig. 1A), except for the results from one study (Study 000040833, 1991), which gives a lower median lethal concentration (LC50) value and, thus, will be used for European hazard classification purposes. Figure 1B, where the results are stratified by study and sex, further indicates that the mortality is partly driven by particle size, with smaller particles resulting in increased mortality, which is a common observation for irritative particles. 34
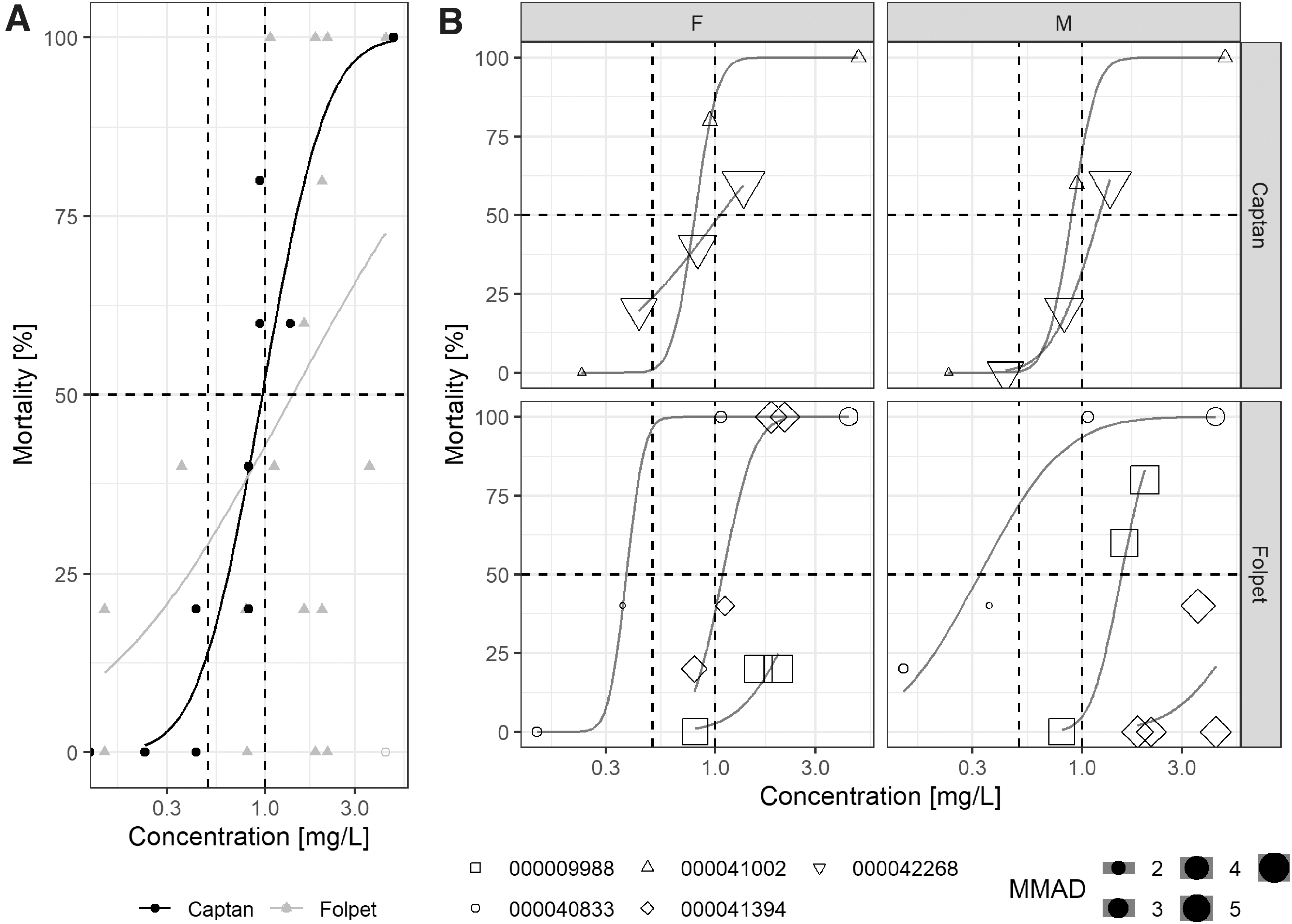
Results of the nose-only acute inhalation nose-only toxicity studies of Captan and Folpet
All available acute inhalation toxicity studies reported clinical observations in line with acute irritation, for example, gasping, labored and noisy respiration. The gross necropsy findings in each study also support the presence of acute irritation, for example, hemorrhage and swollen lungs, fluid in the respiratory system, and increased lung weights. Repeated inhalation exposure studies in rat (data not shown, however, available in RAR evaluations as referenced previously) indicate exposure toward irritant particles: mortality because of edema, influx of inflammatory cells, increased lung weights, and fibrosis, especially in the larynx, which is a typical finding after irritation and inflammation in the respiratory system of rats.35,36
Another study (000041392, 1993) tested Folpet in a nonmicronized form (as placed on the market), which indicates that this material is not acutely toxic through the inhalation route. Using Folpet in nonmicronized form may be more representative for products available on the market. This observation could be potentially used in refined inhalation risk assessments, as it considers a more realistic human exposure scenario.
The difference of acute oral and inhalation toxicity is probably driven by the exposure scenario. In the respiratory tract, rapidly degraded material is quickly replenished by newly inhaled and deposited material, whereas a bolus gavage dose, as used in the acute oral studies, has relatively limited contact to a specific part of the gastrointestinal tract. Furthermore, the diffusion distance within the lower respiratory tract is also shorter than in the intestine, and there is no potential to interact with residue diet. Indeed, a functional disturbance in the gastrointestinal tract is of less short-term toxicological concern than in the respiratory system, as the latter is associated with lethality. Together, the specific “acute” exposure scenarios are different between the respiratory and the gastrointestinal tract because there is a difference in exposure kinetics at target organs, which is relevant for irritants.
Skin and eye effects
Both Captan and Folpet show irritation effects in skin and eye and are skin sensitizers, with harmonized classifications as Eye Dam. 1 and Skin Sens. 1 for Captan and Eye Irrit. 2 and Skin Sens. 1 for Folpet. For Folpet, the proposed classification is revised to Eye Dam. 1, owing to the availability of novel data for the classification process.
Although some irritation to the skin was observed for both Captan and Folpet, in acute corrosion/irritation studies in rabbits, the effect is not potent enough to warrant a classification according to EU CLP. Skin irritation is also observed in the skin sensitization assays using guinea pigs. For Folpet, significant but reversible skin irritation was observed in a 4-week dermal toxicity study in rats (Folpet RAR 2018, p. 112), which could be the underlying reason for the classification revision proposal. The animals showed scabs, dryness/flakiness, redness, and swelling in the low-dose group and thickening and sloughing in the high-dose group. The rat study with Folpet might indicate a difference of species sensitivity between rabbit (less sensitive) and rat and guinea pig (more sensitive). Because both Captan and Folpet rapidly react and degrade, it is possible that stratum corneum thickness may drive the differences in sensitivity. Humans have a thicker and more effective stratum corneum than all the tested laboratory species.
Newly conducted in vitro tests
Direct skin effects
The assays indicated no skin irritation potential (Table 3). Although tissue viability decreases to some extend with exposure time, the effect is small and not sufficient for classification. The positive control sufficiently decreases viability, below the threshold score, and accordingly demonstrates proper assay performance.
In Vitro Results for Direct Skin Effects
The results are concordant with the acute skin irritation assays in rabbit. However, skin irritation is observed in studies with rats and guinea pigs. The in vitro model uses human-derived tissue, similar to human epidermis, and this provides further evidence for species-specific sensitivity and generally points to an increased sensitivity of rodents toward the fungicides' irritation potential.
Direct eye effects
For the BCOP assays, the test item was initially tested dry, as this application reflects the in vivo testing conditions. These initial BCOP experiments did not indicate any irritation potential of Captan and Folpet (Table 4), which is not concordant with in vivo results that predict a serious eye damage hazard potential. The hypothesis was made that a certain amount of water is needed to facilitate an interaction with cell membranes. Accordingly, the in vitro assay was repeated and conducted according to OECD recommendation of testing in a suspension at 20% (w/v) in water. The addition of water resulted in an increased IVIS, however, still at relatively low levels: Folpet 2.9-14; Captan 3.9-6.8. Based on the predictive model, the assay does not allow a prediction; however, it does indicate some irritation potential. Similarly, the RhCE assay did not predict “no classification,” which also indicates some irritation potential. The positive controls demonstrated appropriate assay performance. Based on this, a weight-of-evidence assessment can be applied37,38 and a category 2 can be proposed for eye irritation for both Captan and Folpet based on the in vitro results.
In Vitro Results for Direct Eye Effects
IVIS, in vitro irritancy score.
Skin sensitization
The DPRA assay was terminated owing to unsuitable solvents for the test item. KeratinoSens and U-Sens results are given in Table 5.
KeratinoSens, U-Sens, and GARDskin Endpoint Summary
Experiments 1 and 2 are technical replicates.
The calculated EC1.5 value is 0.05 μM. However, owing to a plateau effect at the lower concentrations, the EC1.5 value is more likely to be between 7.8 and 16 μM, where a clear dose related increase is observed.
NA, not applicable; S.I., stimulation index.
The KeratinoSens luminescence fold induction from two experiments is given in Figure 2. Both Captan and Folpet showed a dose-dependent luminescence increase, which is limited by cytotoxicity at the higher concentrations for Captan experiment 2.
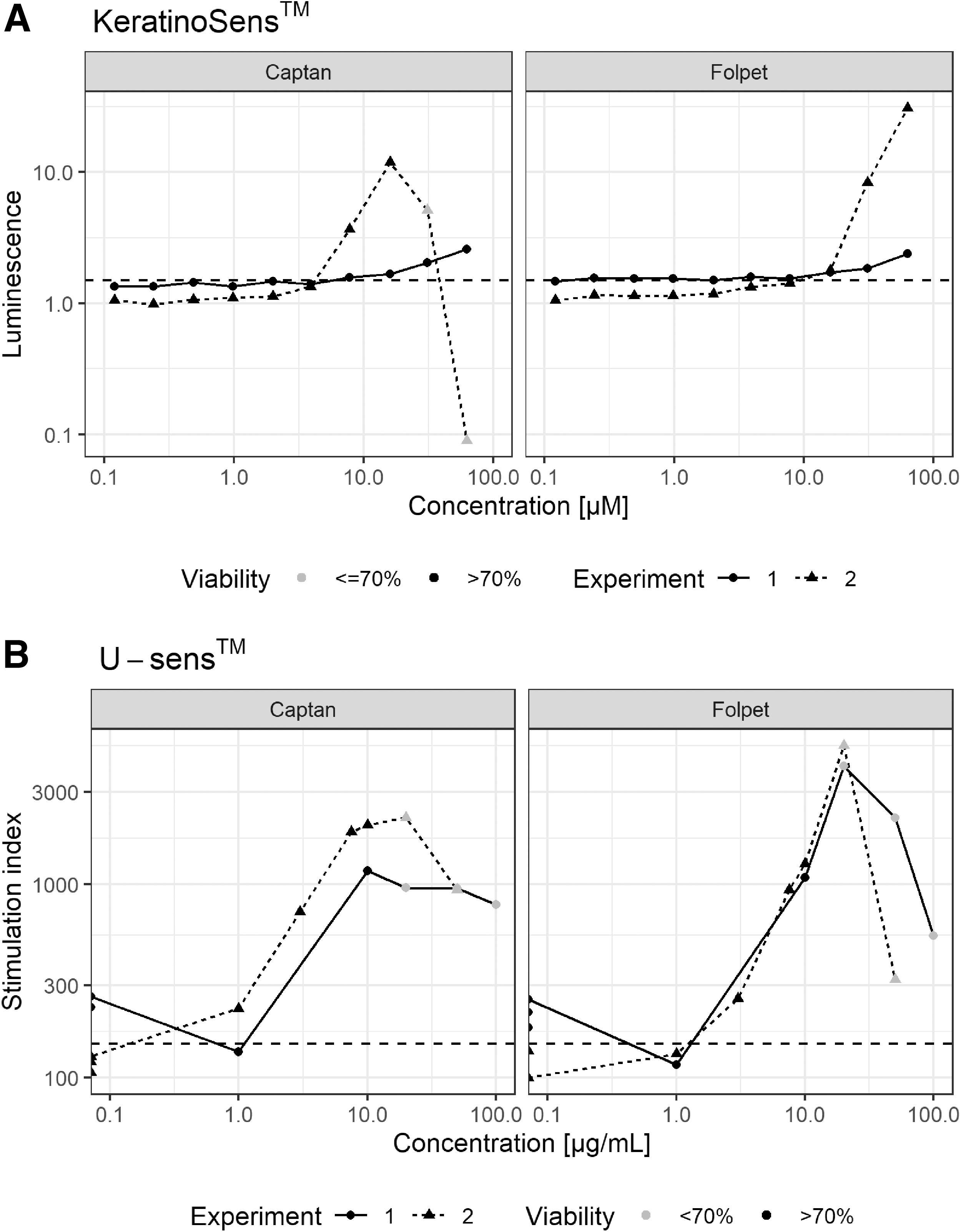
The test items are positive in the assay, as they increase luminescence at concentrations <1000 μM over 1.5-fold induction with a cell viability of >70% compared with the vehicle control. The positive control, ethylene dimethacrylate glycol, used in concentrations from 7.8 to 250 μM (final concentration DMSO of 1%), induced the luminescence sufficiently.
The expression of CD86, the primary endpoint in the U-Sens assay, is given in Figure 2B. The increase of CD86 cell surface marker expression was assessed by measuring the amount of fluorescent cell staining of the CD86 cell surface marker compared with the vehicle control. In addition, the viability was assessed with propidium iodide. Both Captan and Folpet showed a dose-dependent increase that is limited by cytotoxicity at higher concentrations.
Captan experiment 2 again showed a slightly higher potency (increase <1 μg/mL) of the response compared with experiment 1 and the Folpet responses (increase between 1 and 10 μg/mL), potentially for the same reasons as for the differences observed in the KeratinoSens assay. The test items were positive in the assay, as they increased CD86 expression >150% compared with vehicle control with a cell viability >70%. The positive control, TNBS, used at a concentration of 50 μg/mL induced the stimulation index (SI) sufficiently, with SIs of 1873% and 1213%, whereas the negative control, lactic acid, used at a concentration of 200 μg/mL did not stimulate the expression of CD86, SIs of 127% and 138%, in experiments 1 and 2, respectively, which are assay repetitions.
In the GARDskin assay, both Captan and Folpet showed a gene expression pattern that leads to a decision value >0 and are thus considered to be positive in this assay. Both negative and positive controls led to appropriate gene expression patterns.
A more detailed analysis of the expression pattern (Fig. 3), investigating the t-statistic of test item versus negative control, shows that the Captan and Folpet expression profiles are highly concordant and generally induce the same genes. The assay is optimized to detect sensitizers by assessing ∼200 genes. Hence, a more detailed gene expression analysis should presumably be conducted using microarray analysis to identify potentially affected signal pathways, for example, owing to cell membrane interaction. However, the genes with the most extreme expression patterns, based on t-statistic, are given in Figure 3B.
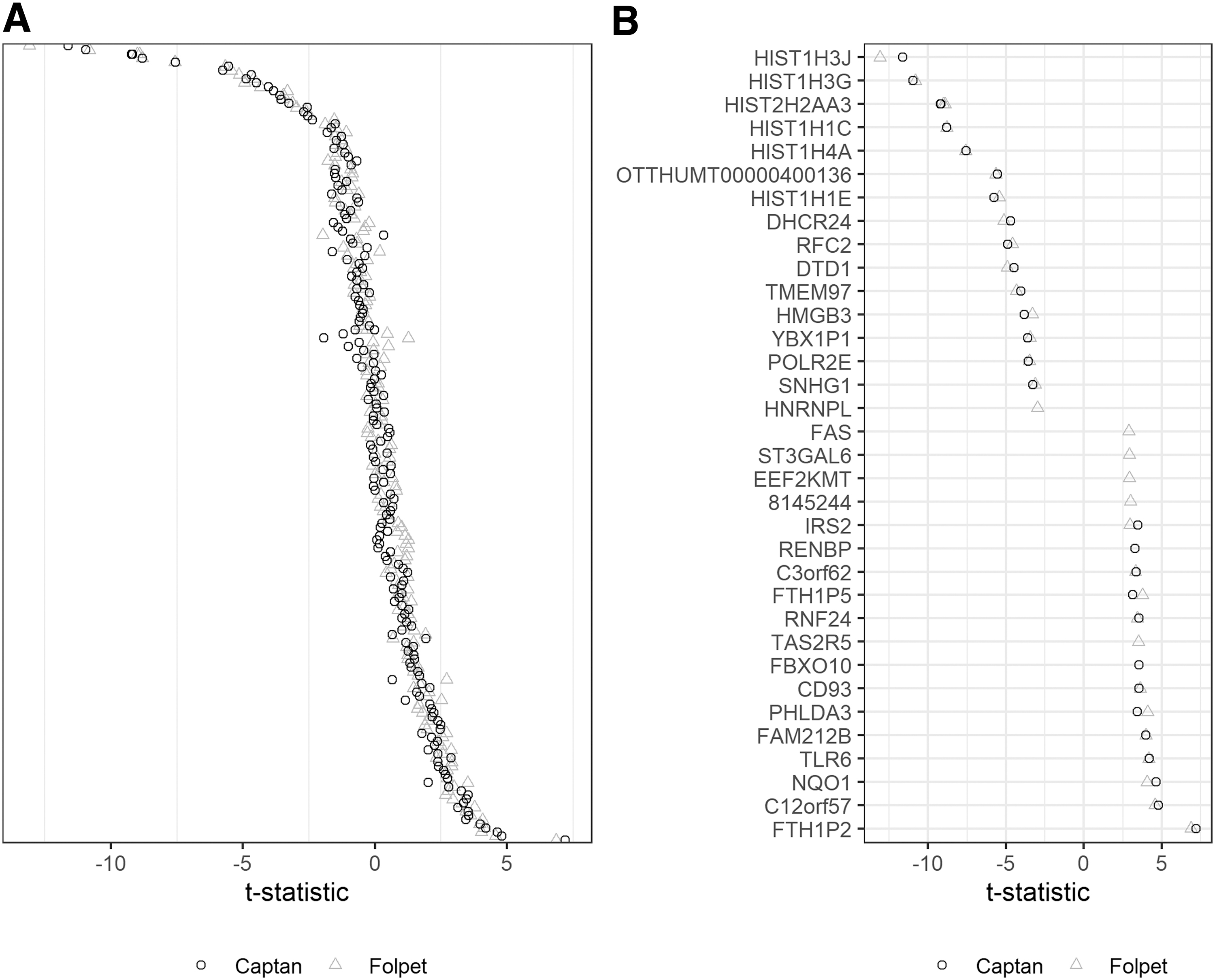
GARD®skin assay gene expression profile as t-statistics.
The fungicides form different primary metabolites after the loss of the trichloromethylthio-moiety to the release of thiophosgene, that is, phthalimide for Folpet and 1,2,3,6-tetrahydrophthalimide for Captan. However, it is unknown how the parent molecules are metabolized in the GARD cell test system. Based on the parents' reactivity, it is, however, very unlikely that they can translocate into the cells without reacting with membrane-associated thiol-groups or damaging the membranes. Therefore, the similar response patterns may indicate that the gene expression pattern is driven by extracellular events and secondary mechanisms.
Acute airway toxicity
The results of the rat EpiAirway assays with single exposure are given in Figure 4A. Both LDH and TEER are affected by treatment by both Captan and Folpet, with pronounced effects starting at ∼100 μg/mL. It is plausible that both endpoints are affected because there is a direct relationship of cytotoxicity and membrane integrity. The higher concentrations, 250, 400, and 600 μg/mL, almost all show a maximum response at 24 hours for Captan, but only the highest concentration shows a maximal response for Folpet.
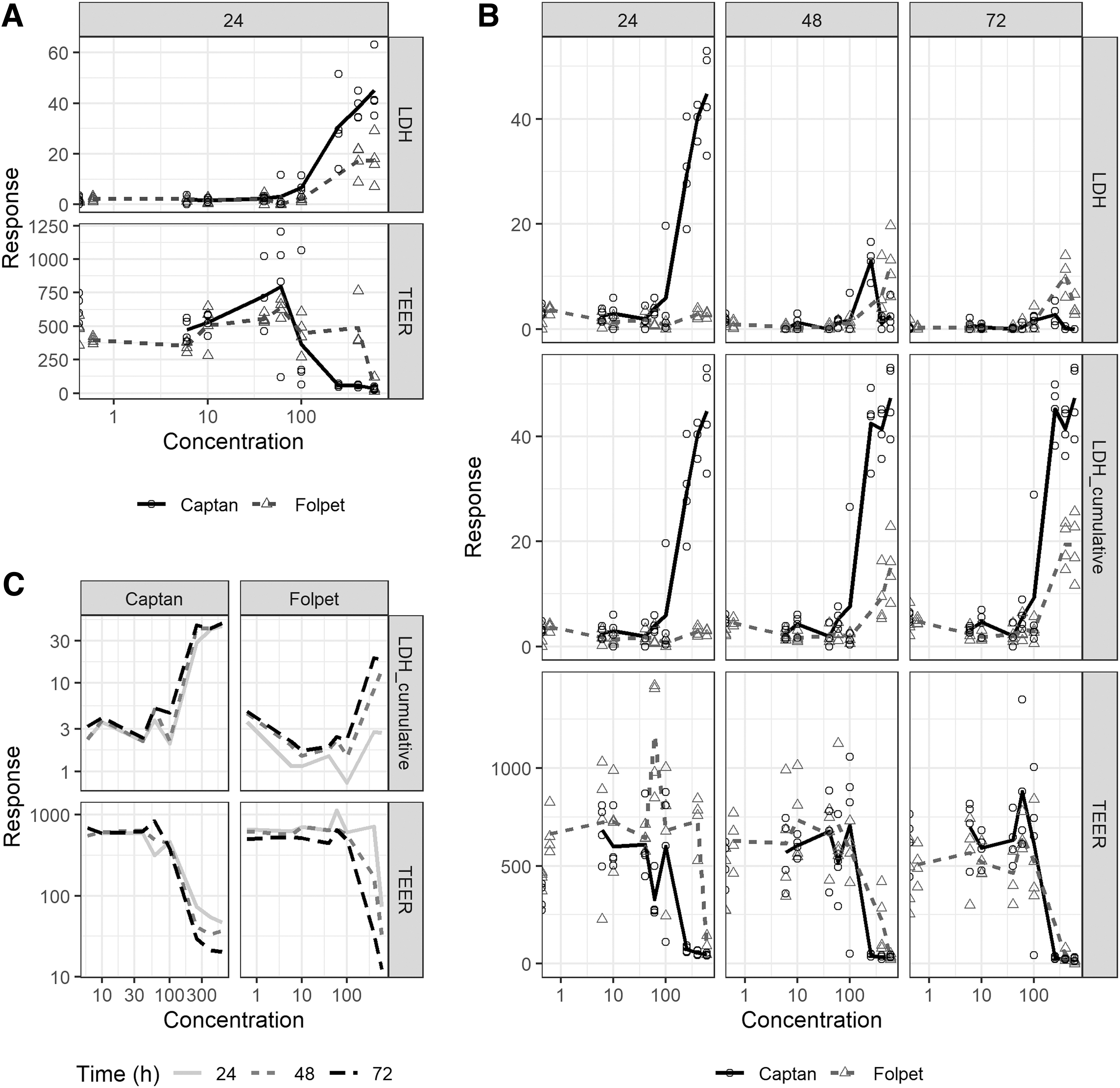
The repeated exposure experiment was conducted to investigate whether repeated dosing increased toxicity (Figure 4B). Because the assay system has no regenerating potential and because there are washings between treatment, LDH is decreasing over time, that is, the effect of the already dead cells is hidden by the experimental method. This can be ameliorated by assessing the cumulative LDH. Both cumulative LDH and TEER are affected by time, with increasing exposures resulting in a stronger effect, which indicates direct cumulative toxicity. This is seen more clearly when the mean responses are added to the plot (Figure 4C), where there is a left shift of the mean responses with increasing time.
Because 100 μL were applied, the 100 μg/mL concentration corresponds to an applied amount of ∼10 μg; for an area of 0.6 cm2 the dose is ∼16.7 μg/cm2.
For Captan, at concentrations ≥100 μg/mL degenerative changes were of a severity greater than those seen in control samples with total or near total epithelia loss in samples exposed to ≥250 μg/mL. In general, samples treated with ≤60 μg/mL exhibited changes of a similar severity as controls except for one of the samples dosed with 60 μg/mL, which exhibited minimal necrotic cells, and 10 μg/mL, which exhibited intercellular separation of similar severity to the formaldehyde-positive control.
In the repeated exposure treatment scenario, there was a higher severity of degenerative findings in untreated and control-treated samples with greater squamous differentiation (larger areas affected with more pronounced keratinization) and higher numbers of necrotic cells. As untreated samples exhibited a similar response to control-treated samples, it is likely that these changes indicate a shift to a squamous phenotype over time in culture. In the treated samples, those receiving ≥250 μg/mL of Captan showed more severe degenerative changes compared with the vehicle-treated controls with total epithelial loss. In samples dosed with ≤100 μg/mL of Captan, degenerative changes were of a similar severity as control samples.
For Folpet, concentrations of ≥100 μg/mL degenerative changes were of a severity greater than those seen in control samples. Samples treated with 400 and 600 μg/mL exhibited marked changes, whereas those treated with 100 μg/mL showed only slightly more severe changes than the controls. In the repeated exposure scenario, there was a higher severity of degenerative findings in untreated and control-treated samples with greater squamous differentiation (larger areas affected with more pronounced keratinization), greater epithelial thinning, and higher numbers of necrotic cells. As untreated samples exhibited a similar response to control-treated samples, it is likely that these changes indicate a shift to a squamous phenotype over time in culture. The treated samples receiving ≥400 μg/mL of Folpet showed more severe degenerative changes compared with the vehicle-treated controls with greater epithelial thinning and epithelial loss.
Derived BMDs are given in Table 6. The BMDs decrease with increasing treatments for Folpet as expected and correspondingly to the mean responses, described previously. For the LDH 24-hour timepoint, no BMD could be derived; it is possible that residue mucus in the test system interfered with test item-induced biomembrane toxicity (a protective response from the tissues). For Captan, the BMD does not decrease with increasing treatments, which is different from the mean responses. The reason might be relative high toxicity (and low BMD) of the first treatment. A finer dose spacing may resolve this issue.
Estimated Benchmark Dose and its Lower and Upper Confidence Limits (in μg/μL) of the Rat EpiAirway Studies, Each Timepoint Separately
“—” Means no dose–response is observed for the data.
BMD, benchmark dose;
For both Captan and Folpet, 24-hour LDH and cumulative LDH give lower BMDs than TEER. This indicates that LDH is more sensitive than TEER to identify test item-associated toxicity at this acute timepoint. Hence, epithelial integrity disruption may occur owing to primary cell toxicity, which aligns with the known cytotoxic properties of the test items.
The assay further indicates that Captan is slightly more potent than Folpet, which aligns with the acute inhalation toxicity data, where Captan has overall a slightly lower LC50 than Folpet. Both the effects in the in vitro assay and the in vivo studies may be associated with differences in solubility, because Captan is more soluble than Folpet in water, that is, 4.9 mg/L versus 0.8 mg/L in water (25°C), which may affect their in situ bioavailability.
Discussion
The results of the NAM assays are mostly concordant with in vivo data (Table 7). Of interest is the nonconcordance of the BCOP assay with in vivo results and the GARDskin and rat EpiAirway studies outcome as they indicate nuances of Captan's and Folpet's toxic properties.
Conducted In Vitro Study Package Results Overview
MoA, Mode of Action.
Captan and Folpet result in almost identical results in the assays. Differences in physicochemical properties, for example, solubility, may however affect some assay outcomes.
The GARDskin tests show essentially identical gene expression patterns, which in combination with the prior knowledge of rapid degradation and reaction, indicates that the substances' toxic effects may occur mainly, or are initiated by, effects on the biomembranes of the exposed cells. This would be plausible as Captan and Folpet have the same toxicophore, the trichloromethylthio-side chain, which also drives their fungicidal activity.
Although the results in the dedicated rabbit acute skin corrosion and irritation assays in the legacy regulatory toxicity package do not require a classification for skin irritation, skin irritation is clearly observed in various in vivo studies and a skin irritation hazard can be associated with the fungicides in animal models. The in vivo skin sensitization studies and repeated exposure dermal study in rats indicate substantial irritation and even skin damage (see RARs as referenced in Table 1 and Materials and Methods section), which may have triggered the revised classification proposal, as stated previously. Hence, it is curious that the in vitro assays do not indicate any skin irritation potential for Captan and Folpet, arguably demonstrating the more sensitive nature of the rat skin than human skin confirming the animal test's greater conservatism.
A key problem for regulatory toxicologists working with pesticides is whether animal models accurately predict human health hazards. This may be especially true for skin irritation, because human skin structure differs substantially from that of animal species typically used in acute toxicity assays. Humans have different skin structures than laboratory animal species, particularly with regard to hair or fur and hair follicle density, complexity, and effectiveness of the stratum corneum barrier function, which may explain differences in assay outcomes. Of note, human skin has the best barrier function because of low hair follicle density and much thicker layers of stratum corneum among other physiological differences, which should be able to protect the underlying tissue more efficiently against direct reactivity.
Hence, the in vitro irritation assays, using a human 3D skin model, may indicate that humans are more resistant to the irritation hazard from Captan and Folpet than guinea pigs and rats and that the acute studies in rabbits better predict human hazard. There is only limited evidence in humans following exposure to Captan or Folpet39–44 with low incidences and it is unclear in all studies how irritation and sensitization were distinguished if truly occurring in those studies. However, considering the long use of Captan and Folpet products, without reports of apparent skin effects in primary users, the observed skin effects are toxicologically probably not relevant for human skin exposure in practice.
The results of the in vitro eye damage and irritation assays were most surprising, as it was expected a priori that the in vitro assays would be more sensitive than the available in vivo studies in rabbits, because of missing tear fluid, mechanical intervention (blinking), and a smaller pool in thiol groups for detoxification. It was shown 45 that the rabbit in vivo Draize test is the most robust test for clearly nonirritant (no classification) or irreversibly damaging chemicals (category 1), which is per se not surprising, as classification reduces the amount of the available information by design and favors extreme effect sizes. Chemicals with a continuous spectrum of potency are allocated into classes by generic thresholds.
Hence, the results of the in vivo studies conducted with the fungicides might indicate a potency at the lower end of chemicals that fall into category 1. The BCOP used with dry material did not indicate any irritation potential and would even support a no classification proposal for the substances. Based on the hypothesis that an aqueous environment is needed to elicit irritation potential, the BCOP was modified by testing the chemicals as 20% (w/v) solutions. Although this indicated some limited irritation potential, the assay is not validated to predict a category 2 irritation classification. The results of the modified BCOP together with the RhCE assay may allow a category 2 classification proposal by a weight-of-evidence assessment or applying Integrated Approaches to Testing and Assessment (IATA).
However, the failure to detect eye damage potential, as indicated in the available rabbit in vivo studies, is curious. There are species differences between human, rabbit, and bovine cornea. 46 Therefore, it is unclear whether the difference is an underprediction of the BCOP assay or increased sensitivity of the rabbit eye as compared with the used models. Hence, it is proposed to add water when testing Captan or Folpet granular or powdered products in BCOP assays for hazard assessment purposes.
The results from the skin sensitization assays concur with the predictions observed in the guinea pig assays in the older regulatory toxicity package. However, both the KeratinoSens and the GARDskin tests are reporter gene assays, without a functional skin barrier. It is unclear how a direct interaction with the cell membrane, as expected and indicated by the gene expression profile in the GARDskin assays, affect assay outcome. The KeratinoSens assay specifically investigates ARE-dependent pathways. It was reported 47 that glutathione depletion induces CD86 overexpression and glutathione depletion is a known effect for both Captan and Folpet.3,4 Therefore, it is unclear how this affects the KeratinoSens and potentially the GARDskin assays. Similarly, the species differences in the irritation tests may indicate that intact Captan and Folpet do not penetrate efficiently into human skin.
Hence, human relevance of the fungicides skin sensitivity potential should presumably be investigated in more realistic human 3D tissue models, as compared with animal models and reporter gene cell lines, which have thinner or absent stratum corneum, respectively. The experimental repeats in the KeratinoSens assay show that luminescence increase is limited by cytotoxicity for Captan experiment 2. It is unclear why this did not occur in experiment 1 or for Folpet; it might be associated with solubility differences between assay repetitions; however, precipitate was not observed. Experiment 2 luminescence fold increase occurs at lower concentrations (<10 μM) than Captan experiment 1 and those for Folpet (all increase between 10 and 100 μM). Another potential reason for the difference would be slight differences in, for example, incubation or test item preparation times, as both Captan and Folpet can degrade and react quickly in the presence of protein residue and when not stabilized by acidic conditions and low temperature.
The rat EpiAirway studies indicate cumulative toxicity of Captan and Folpet because of repeated acute toxic irritation insults, which is typical for inhalation toxicity of irritant particles. The studies show that the severity of histopathological effects is dose-dependently increased and more pronounced with subsequent treatments. Previously, data were presented48,49 showing that Captan produced similar effects as the positive control, sodium dodecyl sulfate, on toxicity estimates and breathing mechanics owing to development of cellular toxicity-related pulmonary edema in an air–liquid interface cell culture model and an isolated perfused rat lung model, respectively. The results are fully in line with the rat EpiAirway studies conducted here. Canal-Raffin et al previously showed in an in vitro model that cytotoxicity can occur rapidly in bronchial epithelial cells upon contact with Folpet, which would be a plausible mode of action and relates to the AOP presented for gastrointestinal tumors. 12
It is notable that thiols are known to be protective against chemically induced pulmonary edema 50 and other lung injury, 51 which is of relevance because both Captan and Folpet are detoxified by thiols. 3 This may explain variation in the rat EpiAirway studies, because the model epithelia produce “mucus,” which may interfere with Captan and Folpet tissue toxicity, for example, if tissue washing varies between repetitions. Furthermore, the use of a nonaqueous vehicle may have affected the fungicides' potency. Compare the results from the ocular BCOP assay, where the addition of water increased irritation potency. In the rat EpiAirway studies it was considered necessary to use a nonaqueous vehicle to ensure sufficient stability of the fungicides.
The presented NAM data would underclassify eye irritation hazard compared with the animal data for Captan and Folpet. However, the animal data are inconsistent in predicting the hazards as well: rodents are more susceptible to Captan and Folpet irritation than other species. Hence, the NAM data might indicate that the results of the animals are oversensitive (conservative) for human health assessment purposes in this case. This is plausible regarding skin irritation potential owing to known differences in stratum corneum thickness between species. Thus, it might be helpful to explore species differences with respect to irritation also in vitro; however, appropriate and comparable assays using rat tissue are missing.
The rat EpiAirway assay correctly predicts inhalation hazard using rat tissue. It might be interesting to compare the results obtained using human EpiAirway tissue, although these models have been shown to be similar or the rat being more sensitive. 27 The issue with this approach is that rat and human respiratory geometry needs to be considered if realistic hazard and risk predictions are intended, as discussed previously, which is not appropriately modeled when using the EpiAirway assay with human tissue alone. There are numerous challenges in extrapolation of animal data to predict human outcomes owing to physiological, anatomical, and metabolic differences across species (e.g., dissimilar airway physiology including types and composition of cells and enzymes composition). The rat has a more complex nasal turbinate system than the human, and rats, unlike humans, are obligate nose breathers with different nasal and laryngeal airway anatomy. Furthermore, the data highlight the practical difficulty for any testing strategy for Captan and Folpet, as they are very reactive and act locally, which is prone to bias toxicological assays, both in vitro and in vivo.
The derived BMDs from the rat EpiAirway studies can be used to derive a human equivalent dose with dosimetry and measured or predicted inhalation exposure doses of operators handling Captan and Folpet products, which is not explored in this article. This may, however, offer a tool to generate hazard and risk assessment data resulting in a reduction in the animal use for acute toxicity through the inhalation route, for which there are not many alternative approaches available. 52 A decision tree 52 prescribed a more human relevant prediction of toxicity through the inhalation route considering target site exposure, physicochemical properties of inhaled agent, toxicity alerts while using simple two-dimensional cellular, complex 3D/air–liquid interface, ex vivo human precision-cut lung slices and computational models. Such an approach has been evaluated for the pesticide, chlorothalonil by US EPA. 29 In this case, computational fluid dynamics, in vitro tests (MucilAir), BMD modeling, and particle size distribution were brought together to identify human equivalent concentrations and doses for the respiratory component of the formal risk assessment.25,29
Evaluation of the in vitro endpoints (TEER, LDH, and resazurin) using MucilAir has been shown to predict in vivo (patient) clinical respiratory toxicity in pharmaceuticals. 23 The respiratory irritant, sodium dodecyl sulfate, has also been evaluated using the human MucilAir model. 24 It shows a similar hazard profile as Captan and Folpet in the rat EpiAirway studies.
Conclusion
The NAM data package appropriately identifies Captan and Folpet as contact irritants and indicates their skin sensitization potential. However, the assays underclassify eye irritation hazard, when compared with animal data. This may indicate species sensitivity differences, which are plausible owing to morphological differences. Hence, the suitability of animal test data for human irritation risk from Captan and Folpet may be questioned as being overly protective.
Footnotes
Acknowledgments
The authors thank the study directors and technicians responsible for the studies at Charles River Laboratories, here specifically Hazel Paulo and Joanne Wallace for their support with the rat EpiAirway studies. The authors thank Lisa Theorin and Andy Forreryd of SenzaGen and Walter Westerink from Charles River Laboratories for their support in data interpretation and in-depth analysis of the GARDskin gene expression data.
Authors' Contributions
All authors actively contributed to the article. F.M.K.: Conceptualization, writing—original draft, writing—review and editing, resources, investigation, methodology, visualization, formal analysis, supervision. C.S.R.: Methodology, writing—review and editing. S.M.J.: Writing—review and editing, visualization, formal analysis. C.M.K.: Writing—review and editing.
Author Disclosure Statement
F.M.K. and C.M.K. work as regulatory toxicologists for ADAMA, which registers, produces, and markets Captan, Folpet and their products. C.S.R. and S.M.J. worked as paid consults for ADAMA.
Funding Information
This research did not receive any specific funding; it was conducted within a commercial registration project.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
