Abstract
Introduction:
The Vitrocell® Ames 48 whole-aerosol exposure module offers a high-throughput platform for air-agar-interface exposures that mimic human exposure conditions for assessing potential mutagenic activity of a test article. The aim of this study was to characterize the Vitrocell Ames 48 exposure module for assessment of aerosolized tobacco products utilizing multiple dosimetry techniques and nicotine deposition as determined by chemical analysis.
Materials and Methods:
Characterization was performed using the Vitrocell VC10® smoking machine and three nicotine containing tobacco product test articles: Kentucky Reference cigarettes (3R4F), a commercially available electrically heated tobacco product (eHTP), and an Electronic Nicotine Delivery System (ENDS). The following dosimetry tools were used: Aerosol photometers (using area under curve values, ∑AUC), free glycerol deposition in phosphate-buffered saline (PBS), fluorescence of anhydrous dimethyl sulfoxide (DMSO)-captured smoke particulate matter, and analytical determination of nicotine in PBS and DMSO. Characterization of 3R4F Kentucky Reference cigarettes was examined over a diluting airflow range 8 to 0.5 L/min; eHTP and ENDS were examined over a diluting airflow range of 4 to 0 L/min (undiluted).
Results:
Results from the dosimetry techniques showed that whole smoke from 3R4F cigarettes and whole aerosol from eHTP and ENDS demonstrated reproducible and consistent delivery, with results being consistent between experiments and within each airflow.
Conclusion:
These results show that this exposure module is fit for purpose using these three tobacco product types.
Introduction
Cigarette smoke is a complex aerosol consisting of both gaseous and particulate fractions, and containing more than 7000 compounds. 1 Smoking is one of the leading causes of preventable death and is associated with cardiovascular disease, chronic obstructive pulmonary disease, stroke, and cancer. 2 In recent years, Electronic Nicotine Delivery Systems (ENDS) and heated tobacco products (HTP) 3 have become increasingly popular,4,5 resulting in regulatory bodies having to implement premarketing requirements and guidance of nonclinical toxicological data necessary to assess the potential health effects of tobacco and next-generation tobacco products (NGTPs).6–10 To meet regulatory requirements to bring NGTP to market, manufacturers will need to assemble extensive toxicological data sets; therefore, new technologies and equipment will be required to increase understanding of the biological effect of these products and in vitro assay throughput to make appropriate assessments of potential toxicological effects.
Traditional testing methods, such as those published by regulatory agencies 9 have been recommended for genotoxicity and cytotoxicity testing of aerosolized tobacco products using gaseous and particulate fractions or a combination thereof. These methods have been used historically for product assessment.11–15 In vitro studies were initially designed to evaluate single compounds treated in solution and added to test systems in submerged conditions. However, as demonstrated in the above studies, these in vitro testing approaches designed to assess single chemical compounds have regularly been performed for more complex mixtures of particulate and gaseous phase components of aerosols. Adapting methods to allow in vitro exposures to unfractionated whole aerosol has led to the development of exposure systems that allow testing at the air–liquid interface (ALI).16–20
Performing in vitro exposures at the ALI maintains interactions between the particulate and gaseous phases while the aerosol is delivered directly to the cell surface, creating an exposure that is more comparable to that of an adult tobacco user. Although the application of separate particulate and gaseous phase components of tobacco product aerosols has been widely accepted and promoted by regulatory agencies, there are some limitations. For example, the interactions that occur between the two phases separated during collection are not assessed, loss of volatile compounds particularly from the gaseous vapor phase may occur before application in the test system, and trapping procedures may not represent the complete chemical composition of the aerosol due to selective capture of aerosol constituents. For these reasons, there has been movement into whole-aerosol exposures at the ALI.
However, exposures at the ALI introduce a level of complexity in terms of aerosol generation, handling, dosimetry, and delivery that is not observed in standard in vitro testing batteries.
There are a wide variety of smoking machines available from various companies, including: Vitrocell® Systems GmbH (Waldkirch, Germany; e.g., Vitrocell VC10®),21–23 Borgwaldt KC (e.g., Borgwaldt RM20S, LM4E),24–26 Burghart Messtechnik GmbH (SAEVIS smoking machine),27,28 and some in-house manufactured examples.29,30 These smoking engines are attached to cell exposure modules via a dilution system; examples of which are available from Vitrocell Systems GmbH,20,31–33 Cultex® modules, 34 and bespoke examples.35,36 Although there are many combinations of materials available, the concept of delivering fresh whole aerosol to the cell cultures at the ALI (or equivalent) is consistent. A recently published survey 37 pointed out differences across whole-aerosol exposure systems used to assess the cytotoxicity of tobacco product aerosols, emphasizing the importance of method and dosimetry harmonization, but not necessarily system harmonization. Most important is that the systems being used are understood through characterization, qualification, and validation.
The aim of this study was to characterize the Vitrocell Ames 48 exposure module system using a Vitrocell VC10 smoking machine. 3R4F (University of Kentucky, Kentucky) combustible cigarettes were used to characterize the system using three dosimetry techniques: aerosol photometers (using area under curve [AUC]), fluorescence of anhydrous dimethyl sulfoxide (DMSO) as previously described, 23 and analytical determination of nicotine in DMSO. The HTP used in this study was a commercially available, electrically heated tobacco product (eHTP). CORESTA has classified HTPs based on their heating source (CORESTA 2020). For example, eHTP use a battery powered device to heat a consumable containing an aerosol-generating tobacco substrate, while carbon-heated tobacco products use smoldering carbon as the heat source. For eHTP and ENDS, commercially available products were used for characterization using three dosimetry techniques: aerosol photometers, determination of free glycerol in phosphate-buffered saline (PBS), and analytical determination of nicotine in PBS.
Materials and Methods
All chemicals and reagents were obtained from Merck KGaA (Darmstadt, Germany) unless otherwise stated. PBS was obtained from Gibco® via Thermo Fisher Scientific (Waltham).
Test article
3R4F Kentucky Reference cigarettes were obtained from the University of Kentucky. The eHTP was commercially available and purchased in the United Kingdom. The ENDS, a puff-activated ENDS device, was commercially available and purchased in the USA. Before smoking, cigarettes were conditioned for at least 48 hours and no more than 10 days at 22°C ± 1°C and 60% ± 3% relative humidity. 38 Before smoking, eHTP consumables were conditioned for at least 48 hours and no more than 5 days at 22°C ± 1°C and 60% ± 3% relative humidity, 38 but were conditioned in packs with the cellophane wrapper intact and not as individual consumables. ENDS consumables were stored at room temperature sealed in their original packaging until transferred to the laboratory for use.
Vitrocell VC10 smoking robot and Ames high-throughput module
Cigarette smoke was generated using a Vitrocell VC10, serial number VC10/210311 (Vitrocell Systems GmbH), and eHTP/ENDS aerosol was produced using a Vitrocell VC10, serial number VC10/200814. Different concentrations of whole smoke or aerosol were achieved by altering the diluting airflow using mass flow controllers (Analyt-MTC GmbH, Mülheim, Germany). For all experiments, concentrations were expressed in terms of air flow as liters per minute. The vacuum rate was fixed at 5 mL/min. Operation and confirmation of functionality, including installation, operational, and performance qualification, were performed as previously described. 33
All cigarettes were smoked according to the Health Canada Intense (HCI) smoking regimen (55 mL puff, 2 second duration, 30 second frequency, with 100% vent blocking).39,40 All eHTP consumables were smoked according to a modified HCI smoking regimen (i.e., no vent blocking as the consumables lack ventilation holes). All ENDS consumables were vaped according to a modified CORESTA recommended method No. 81 (i.e., a 60 second pause was taken after every 10 puffs).41,42
This study utilized a Vitrocell Ames 48 exposure module climatic chamber. The Vitrocell Ames 48 module uses the same technology as previously described17,20,21,33; however, this equipment allows higher throughput compared to the standard Vitrocell exposure modules (i.e., up to seven doses and one control, and up to six replicates per dose; Fig. 1). Additional individual dosimetry modules were located on each of the seven dose rows to allow assessment of delivered dose.

Vitrocell® Ames 48 exposure module. Image supplied by Vitrocell Systems GmbH (Waldkirch, Germany).
Photometers
The first dosimetry measure used for assessing both combustible and NGTP aerosols in this characterization exercise was the laser photometer. Photometers have the capacity to characterize the particle density of aerosols in a system by measuring the amount of light scattered as a laser passes through the aerosol stream. As the aerosol enters the scattering chamber, a laser diode (wavelength 680 nm) is shone through the sample and the scattered light is detected by an offset photodetector and reported as voltage over time.
The signal is amplified to an output voltage between 0 and 5 V. The signal is independent of the flow direction and velocity through the photometer. In this application, mass concentration readings cannot be obtained because the aerosol is not composed of a single particle type, and particle size is inconsistent. Thus, the photometers are harmonized for each test article product aerosol being measured. The photometers are connected to the Vitrocell VC10 smoking robot at an appropriate point to allow the aerosol to pass through the photometer.
Photometers were purchased from Vitrocell Systems, GmBH and harmonized for each tobacco product type used in this study. Harmonization of the photometers was performed as previously described. 23 In brief, photometers for combustible cigarettes were harmonized to whole smoke using the HCI regimen and a diluting airflow of 1.5 L/min. For eHTP consumables, photometers were harmonized to whole aerosol using a modified HCI regimen and a dilution airflow of 0.25 L/min. For ENDS consumables, photometers were harmonized to whole aerosol using a modified CRM81 puffing regimen and a dilution airflow of 0.25 L/min.
A set of eight photometers were harmonized for 3R4F and seven photometers were harmonized for the market eHTP and ENDS products. Photometers are not suitable for the undiluted aerosol (0 L/min) dose because photometers require a positive pressure of air to avoid the occurrence of damage to the photometer. All photometers were harmonized so that the voltage output for each photometer was ∼4.0 V. Photometer data were output as both comma-separated value (CSV) and virtual-compact-disk (VCD) file formats.
DMSO fluorescence
The second dosimetry measure comprised assessment of 3R4F smoke constituents captured in DMSO contained in the liquid traps within the exposure module. It is known that smoke constituents fluoresce (or emit; Em) blue (485 nm) when exposed (Ex) to longwave ultraviolet radiation (355 nm). Dose quantification then follows via the fluorescence intensity assay.16,43 It has been demonstrated that there is a linear relationship between fluorescence and aerosol production.23,44 This fluorescent property enables the estimate of deposited smoke constituents via extrapolation from a standard curve of known concentrations.
3R4F pad-collected total particulate matter (TPM) dissolved in DMSO at 24 mg/mL was serial diluted (twofold dilutions) in DMSO (0.0024–5 mg/mL). Fluorescence readings were taken from these dilutions and calculated as Ex355/Em485 to establish a standard curve of relative fluorescence unit (RFU) (i.e., the emission at 485 nm) versus TPM concentration (mg/mL). Immediately postexposure, all DMSO samples were retained and triplicate 100 μL aliquots from each well were transferred into 96-well plates. Analysis was performed using a VERSAmax™ Microplate reader (Molecular Devices, San Jose, CA). The mean values for each smoke exposure (represented in RFU485) were subtracted from the mean RFU for the air control to give relative fluorescence per smoke dose. An equivalent TPM concentration was determined using the standard curve.
Free glycerol determination assay
The third dosimetry measure was determination of free glycerol. A major component of the aerosols generated by the ENDS and eHTP used in this study is glycerol.45,46 Thus, glycerol serves as a relevant marker of aerosol deposition and supports exposure system characterization. A free glycerol determination assay is used for the quantitative enzymatic determination of glycerol in PBS, which was contained in liquid traps within the exposure module. Glycerol is phosphorylated by adenosine-5′-triphosphate forming glycerol-1-phosphate (G-1-P) and adenosine-5′-diphosphate in the reaction catalyzed by glycerol kinase. G-1-P is then oxidized by glycerol phosphate oxidase to dihydroxyacetone phosphate and hydrogen peroxide (H2O2).
Peroxidase catalyses the coupling of H2O2 with 4-aminoantipyrine and sodium N-ethyl-N-(3-sulfopropyl) m-anisidine to produce a quinoneimine dye that shows an absorbance maximum at 540 nm. The increase in absorbance at 540 nm is directly proportional to the free glycerol concentration of the sample. The ENDS and eHTP products contain glycerol in the aerosol; therefore, this method was used to determine the deposition of the glycerol found in the aerosol into the PBS in the module.
Free glycerol reagent (F6428-40ML) was reconstituted with 40 mL of ultrapure water. Glycerol standard (G7793-5ML) solutions were prepared using a serial dilution from 130 to 2.03125 μg/mL, 100 μg/mL was also included in PBS. Twenty-five microliters of the prepared standards were transferred in triplicate into the appropriate wells of each 96-well plate. Samples from undiluted and 0.25 L/min airflow were diluted 1:4 with PBS and samples from 0.5 to 1 L/min were diluted 1:1 to ensure the optical density (OD) values were within the range of the standard control before transferring, in triplicate, 25 μL of the whole aerosol-exposed samples into the appropriate wells of the 96-well plate. One hundred microliters of the reconstituted free glycerol assay reagent were added to each well. The 96-well plates were incubated at room temperature for 15 minutes. Analysis was performed using a VERSAmax Microplate reader (Molecular Devices) at a wavelength setting of 540 nm.
Free glycerol absorbances were expressed in terms of absolute OD540. The mean OD540 obtained from the air controls were subtracted from the mean OD540 values for each aerosol dilution to give free glycerol absorbance per aerosol dose. Using this value and the glycerol standard curve, an equivalent glycerol concentration (μg/mL) was determined.
Nicotine
The fourth and final dosimetry measure was the analytical determination of nicotine from the smoke or aerosol exposed liquid trap: DMSO for combustible cigarettes and PBS for eHTP and ENDS.
Samples in either PBS or DMSO were separated using a binary gradient and a Phenomenex Kinetex 2.6 μm EVO C18 HPLC column. Detection and analysis of nicotine were performed using a triple quadrupole tandem mass spectrometer utilizing electrospray ionization and multiple reaction monitoring. The linear range of the method was ∼5 to 500 ng/mL with an limit of quantification defined as the nominal concentration of the lowest level calibration standard and an limit of detection defined as 1.5 ng/mL.
Whole-smoke exposures
In each whole-smoke and whole-aerosol exposure experiment, each well within the module (as well as the dosimetry modules exposed to the test article or air control) contained a 35 mm Petri dish (391-3205; Greiner Bio-One) containing 2 mL of DMSO (combustible) or PBS (eHTP, ENDS). Trumpet (aerosol conduit) heights were confirmed to be 2 mm above the surface of PBS. One “in-line” photometer was placed between the dosimetry module and dilution system (Fig. 2).
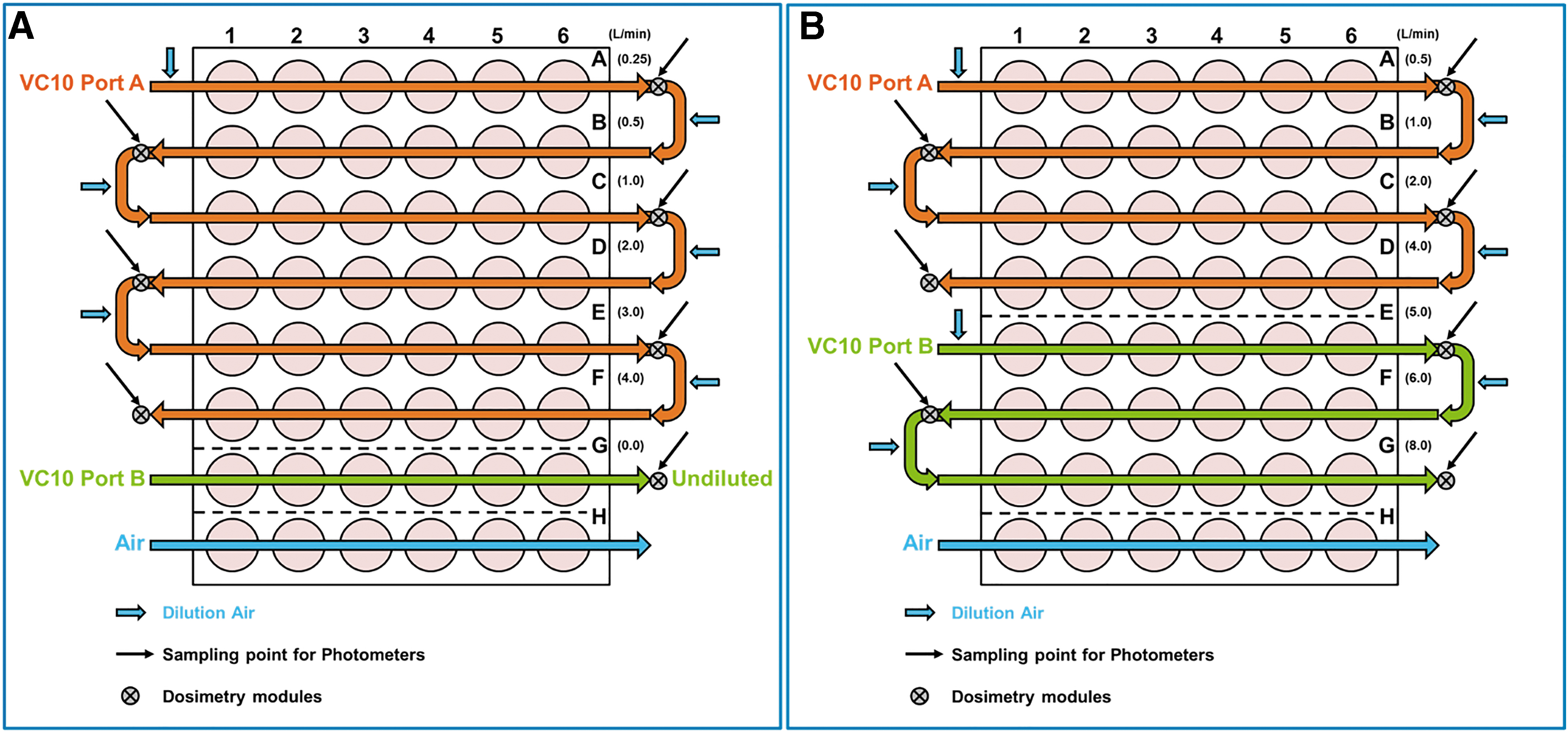
3R4F studies were completed on two Ames 48 systems and each study consisted of three main experiments, each of which included seven whole-smoke dilutions generated from eight cigarettes using the HCI smoking regimen. The smoke was serially diluted via the addition of diluting air through the air inlets to achieve the desired concentration for the diluted smoke doses (Fig. 3).
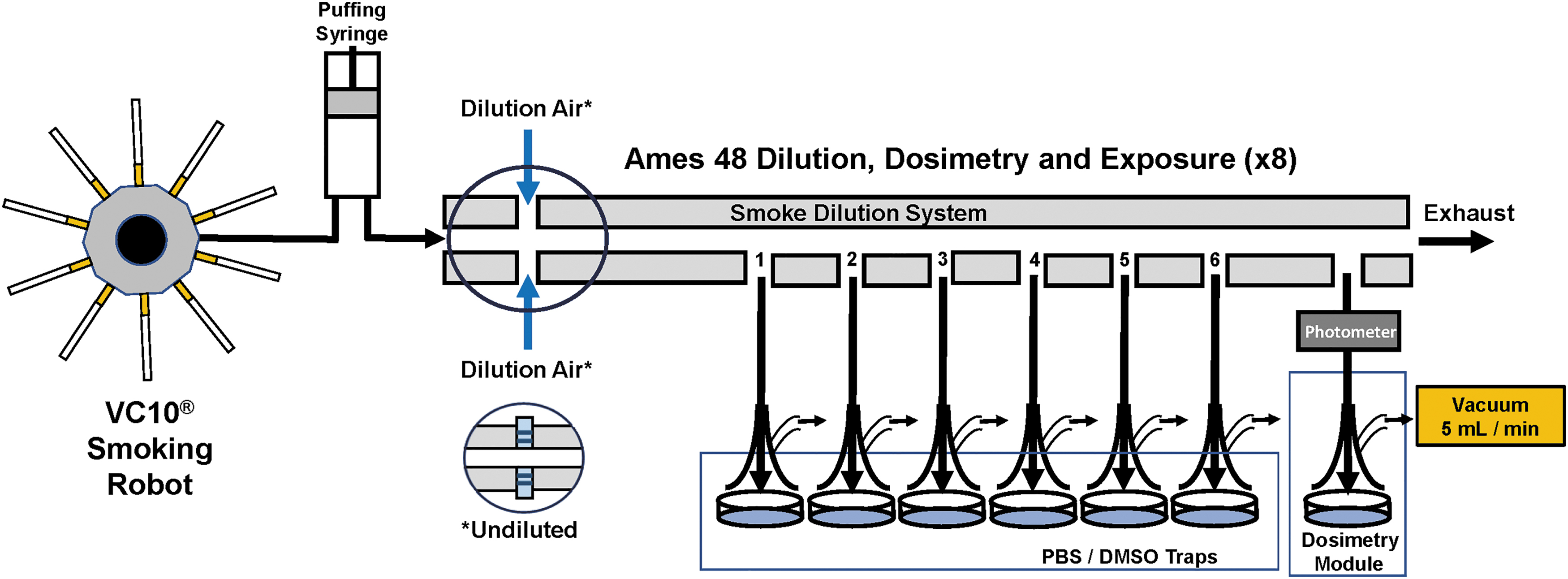
Vitrocell VC10® and Ames 48 High-throughput module schematic.
eHTP/ENDS studies were completed on the same two Ames 48 systems. Each study consisted of 3 main experiments, which included 1 undiluted and 6 whole-aerosol dilutions generated from 10 consumables using the modified HCI smoking regimen (eHTP), or alternatively, 120 puffs under a modified CRM81 puffing regimen (ENDS). The whole aerosol was exhausted from two ports of the VC10 and directed to the dilution system. The whole aerosol directed to the first row of the Ames 48 exposure module was subsequently serially diluted before entering each following row via the addition of diluting air through the air inlets to achieve the desired concentrations for the diluted aerosol doses (Figs. 2 and 3).
Data analysis
Photometer data were output as both CSV and VCD file formats. Voltage for each airflow was recorded every second and from this an AUC value was calculated for each experiment. For DMSO-captured material an equivalent TPM concentration was calculated using the Ex355/Em485 readings against a standard curve of known 3R4F HCI TPM concentrations (0.0024 to 5 mg/mL). For Free glycerol deposition in PBS, an equivalent glycerol concentration (mg/mL) was calculated using the corrected OD540 against a standard curve (130–2.03125 μg/mL, with 100 μg/mL included to increase the linear portion of the standard curve). Nicotine values were reported in ng/mL.
Before analysis, results were subject to an Iterative Grubbs' test (significance α = 0.05) to identify outliers in DMSO-captured material, free glycerol deposition, and nicotine analysis. For photometer data, a simple (noniterative) Grubbs' test (significance α = 0.05) was used due to the smaller sample size. From the results obtained, linear regression analysis was used to make pairwise comparisons between the two Vitrocell Ames 48 exposure modules.
Results
Photometers
AUC data from the photometers are presented in Table 1. Data are presented as the mean of values from three individual experiments. AUC data from eHTP was consistent between experiments as demonstrated by the low coefficient of variance (%CV), with all values less than 15%. In the case of AUC data from 3R4F cigarettes, good consistency is observed at the lower airflows; however, at airflows higher than 4 L/min the increased airflow appears to have some influence on the AUC values, as highlighted by the 58%CV value at 8 L/min in module 1. For AUC data from ENDS, the coefficient of variance was generally increased versus the other two product categories. This can be attributed to the product variability. In all cases, except for 8 L/min for 3R4F in Ames 48 module 2, there is a concentration-related increase in measured dose.
Photometer Area Under Curve Values from Ames 48 Modules 1 and 2 Using 3R4F Kentucky Reference, Electrically Heated Tobacco Product, and Electronic Nicotine Delivery System (Seven Replicates per Airflow, n = 3 Experiments)
Photometers are not suitable for the undiluted airflow.
AUC, area under curve; CV, coefficient of variance; HCI, Health Canada Intense; eHTP, electrically heated tobacco product; ENDS, Electronic Nicotine Delivery System; mHCI, modified Health Canada Intense.
DMSO fluorescence
DMSO fluorescence values from all experiments are shown in Table 2. An equivalent TPM concentration was calculated using the corrected RFU485 extrapolated against a standard curve of known TPM concentrations. In all experiments and in both modules, there was a concentration-related increase in deposited mass. The deposited mass was consistent between the two modules. Although there was higher variance observed for Ames 48 module 2 at the higher airflows, this can be attributed to the lower TPM deposition at these airflows.
Dimethyl Sulfoxide Fluorescence Values from Ames 48 Modules 1 and 2 Using 3R4F Cigarettes (Seven Replicates per Airflow, n = 3 Experiments)
TPM, total particulate matter.
Free glycerol determination assay
Free glycerol deposition values from all experiments are shown in Table 3. An equivalent glycerol concentration (mg/mL) was calculated using the corrected OD540 against a standard curve, from which the values were extrapolated. Overall, for both Ames 48 modules, the free glycerol deposition from the eHTP increased in a concentration-related manner from 4 to 1 L/min. For Ames 48 module 1, there is good consistency between experiments, although the coefficient of variance increases as the airflow increases. This can be attributed to lower deposition at these airflows. For Ames 48 module 2, there is larger variation at all airflows with the exception of the undiluted airflow.
Free Glycerol Values from Ames 48 Modules 1 and 2 Using Electrically Heated Tobacco Product and Electronic Nicotine Delivery System (Seven Replicates per Airflow, n = 3 Experiments)
This is attributed to the fact that one of the three experiments included in the analysis was not identified as an outlier by the Iterative Grubbs' test (significance α = 0.05) and these data are included in the results. For ENDS, in both Ames 48 modules, the free glycerol deposition increased in a concentration-related manner from 4 L/min to undiluted airflow. Higher variation is observed in Ames 48 module 1 than module 2, with no outliers being identified using the Iterative Grubbs' test (significance α = 0.05).
Nicotine
Nicotine concentrations from all experiments are shown in Table 4. In all conditions, a concentration-related increase was observed in deposited nicotine demonstrating consistency in dilution and vacuum rates of the Ames 48 system. Overall, there is higher variability in 3R4F cigarettes than in the eHTP. This can be attributed to the combustion process associated with this product type. In addition, there is a higher variability in ENDS Ames 48 module 1. Nicotine deposition between modules is consistent and the uniformity comparable.
Nicotine Values from Ames 48 Modules 1 and 2 Using 3R4F, Electrically Heated Tobacco Product, and Electronic Nicotine Delivery System Test Articles with Heath Canada Intense, Modified Health Canada Intense, mCRM81 Regimens, Respectively (Seven Replicates per Airflow, n = 3 Experiments)
For eHTP using mHCI from Ames 48 module 1 n = 2 experiments were analyzed for 3, 1 and 0.5 L/min.
Comparison of dosimetry techniques
To evaluate the multiple dosimetry techniques, pairwise comparisons were made between the two Vitrocell Ames 48 exposure modules for each of the three test articles. Linear regression (GraphPad Prism v8.0.0) was used to determine the slopes of the dose-response curves, and these were compared to determine if any significant differences were observed. Y values were transformed [Y = >Log(Y)] before linear regression analysis. The following comparisons were made for each test article: equivalent TPM (mg/mL) or free glycerol (mg/mL) versus photometer (∑AUC), nicotine (ng/mL) versus photometer (∑AUC) and nicotine (ng/mL) versus equivalent TPM (mg/mL) or free glycerol (mg/mL) (Fig. 4). No statistical differences were observed in any of the comparisons; p-values ranged from p = 0.3748 to p = 0.9030.
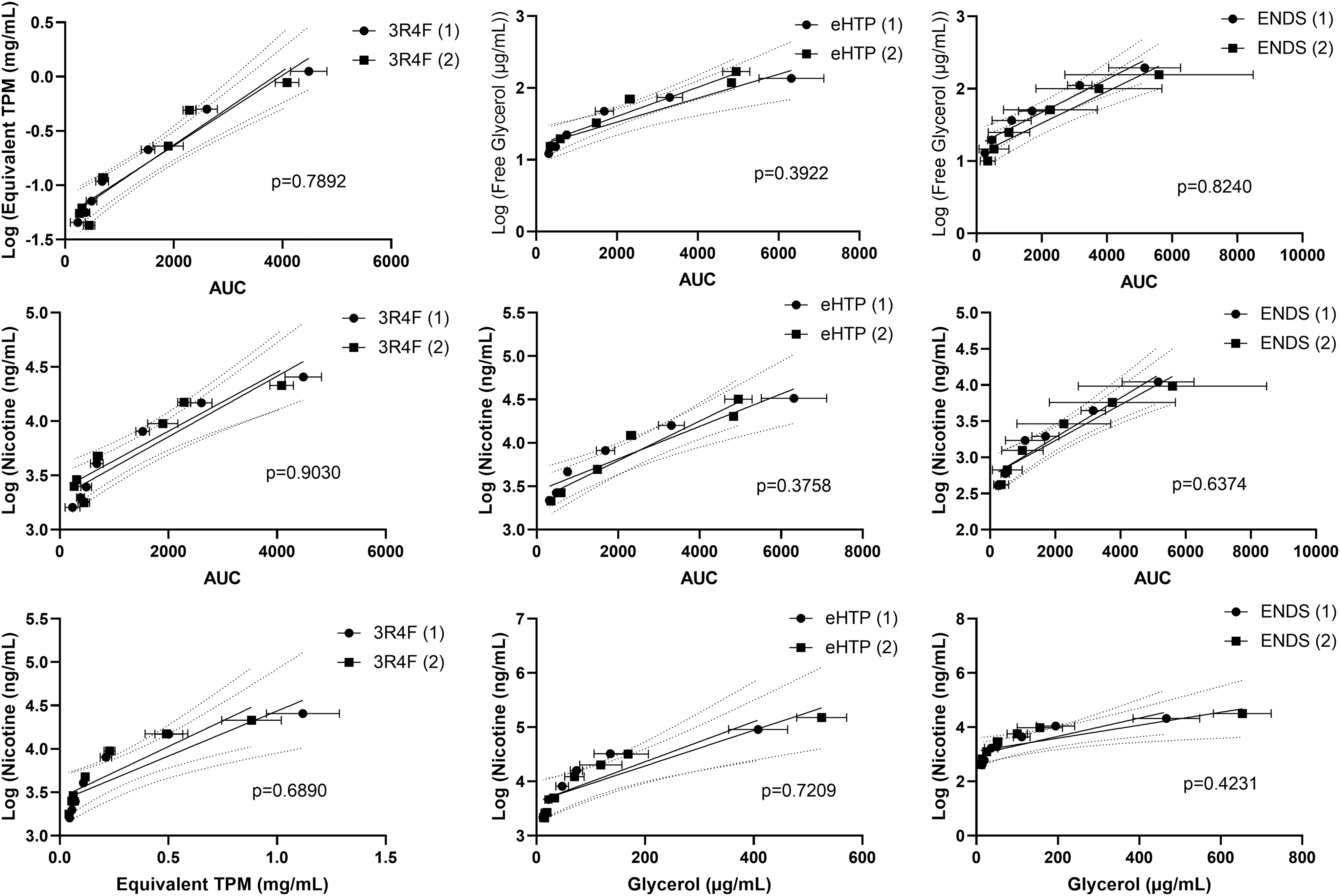
Linear regression comparing dosimetry measurements between two Vitrocell Ames 48 exposure modules using all three test articles.
Discussion
The Vitrocell Ames 48 module in vitro exposure system was developed to increase the number of airflows or doses per experiment from four, as per the standard Vitrocell module system as previously described,17,20,31–33 to seven. This allows further discrimination to fully elucidate toxicity or mutagenic activity. The addition of three airflows/doses also allows further compatibility of the whole-aerosol Ames to the Organization for Economic Co-operation and Development (OECD) guidelines, 47 which recommends at least five different analyzable concentrations. The Vitrocell Ames 48 module allows increased throughput of the assays with the simultaneous exposure of three replicate plates of both treatments, that is, in the absence and presence of exogenous sources of metabolic activation. The advantages therefore, at minimum, are to reduce by half the total number of exposures required and/or closer alignment with the OECD guidelines. 47
Understanding and characterizing a technically complex aerosol exposure system is crucial to decipher any results in terms of toxicological assessments. In the case of tobacco products, it is also critical to characterize new exposure systems with multiple test article types due to the known differences in aerosol composition between a combustible cigarette and NGTPs. 48 For this study, dosimetry techniques, which have previously been demonstrated as useful in the characterization of exposure systems with cigarettes, eHTPs, and ENDS, were utilized.
Photometers were used to characterize the particle density of aerosols through the measurement of the scattered light as a laser beam (of appropriate wavelength) passes through the aerosol stream. The scattered light was then detected via an offset photodetector. The photometer is an important dosimetric tool as it can be used across product types. One caveat to use of photometers we determined was that a direct comparison in the output voltage cannot be made as each set of photometers is harmonized to the specific aerosol to be measured. The harmonization is constant as long as the size distribution of the aerosol does not change, but since the size of the particles in the aerosol changes over time, the photometers cannot give a mass concentration in this application.
In contrast, photometers are suitably able to demonstrate aerosol movement throughout a system and have been used for different aerosol classes.49–53 Enzymatic determination of glycerol and equivalent TPM concentrations via DMSO fluorescence offer an efficient and accurate assessment of aerosol deposition. 23
The data generated from the three tobacco product types used in this study (combustible reference cigarette, eHTP, and ENDS), as analyzed via the dosimetry techniques mentioned, demonstrate the ability of the Vitrocell Ames 48 module to differentiate aerosol deliveries among different dilution airflows. In all experiments, a concentration-related increase was observed using photometers, nicotine, and either glycerol or equivalent TPM deposition dosimetry methods. The data illustrate a correlation between total whole smoke/aerosol delivered to the module as determined via the photometers and deposition in the module. As demonstrated by the results generated from each dosimetry technique, there is a large degree of consistency between experiments and between the two Vitrocell Ames 48 modules assessed. The highest variability was observed with ENDS, which can be ascribed to the lower number of consumables used per exposure. 1
There is also higher variability associated with ENDS products, which added to the experiment-to-experiment variability. Overall, there is slightly higher variability in the results from the 3R4F reference combustible cigarette compared to eHTP. This can be attributed to the fact that the aerosol from this product is generated via pyrolysis and heating of tobacco leading to variability from stick to stick.12,54 Variability is also observed to be slightly greater at the higher airflows, which is not unexpected due to the lower deposition at these concentrations. 22
Statistically comparing the deposition of the aerosols from the three tobacco product types in both Vitrocell Ames 48 exposure modules resulted in no statistical differences. All comparisons revealed p > 0.05, with p-values ranging from p = 0.3748 to p = 0.9030 across all nine comparisons. 3R4F cigarettes resulted in the highest concordance with p = 0.6890 to p = 0.9030. This is not unanticipated and highlights the need to standardize reference products within the eHTP and ENDS categories.
This study has demonstrated the importance of characterizing and understanding aerosol delivery and deposition within an ALI exposure system. It has also established the necessity of tailoring dosimetry techniques to the product being used and the analyte being examined.55–57 The suitability and accuracy of the measures have been shown for glycerol and equivalent TPM deposition. It is also noted that for a true comparison, having an analyte across product types is useful for comparisons. Nicotine is ideal for these tobacco product test matrices. Photometers are a useful real-time measure of aerosol distribution throughout the test system; however, without the ability to calibrate the photometers to a known standard prevents them from being used in a quantitative manner.
Overall, the experimental conditions with the dosimetry techniques applied demonstrated reproducible and consistent delivery of whole aerosol generated from 3R4F Kentucky Reference cigarettes and commercially available eHTP and ENDS to both of the Vitrocell Ames 48 modules used in this study. Further assessment is required to move beyond the characterization of aerosol deposition within this system to include the associated biological responses (i.e., cytotoxicity, genotoxicity) within this system.
Footnotes
Acknowledgments
The authors acknowledge Hsiao-Pin Liu for his assistance in the statistical analysis of the data and Stephen Sears, PhD, and Patrudu Makena, PhD, for their critical review of the article.
Author Disclosure Statement
The data presented in this article was generated and analyzed in studies commissioned by RAI Services Company and conducted under contract at Labcorp Early Development Laboratories Ltd. Brian M. Keyser, Robert Leverette, and Kristen Jordan are full-time employees of RAI Services company. Randy A. Weidman and Carlton J. Bequette are full-time employees of RJ Reynolds Tobacco Company. No competing financial interests exist.
Funding Information
There was no grant for this study.
