Abstract
Introduction:
Tobacco harm reduction (THR) represents one of the most promising public health policies with continued product innovations crucial to making THR a reality. Tobacco-free nicotine pouches (TFNPs) are a recent THR product innovation available in a growing number of countries as a potentially less harmful alternative to traditional tobacco products.
Materials and Methods:
In this study, we compared the in vitro biological activity of two commercially available TFNPs, one snus product, and combustible cigarette smoke in three in vitro toxicological assays (neutral red uptake [NRU], in vitro micronucleus [IVM], and Ames test). An extraction method for TFNPs and snus in phosphate-buffered saline based on the International Organization of Standardization 10993-12 standard to assess the in vitro biological activity is also presented.
Results and Discussion:
Combustible cigarette smoke total particle matter (TPM) induced a statistically significant positive response in all three in vitro assays. By contrast, the TFNPs and snus extracts were negative in both the Ames and IVM assays, and demonstrated weak cytotoxicity in the NRU assay compared with TPM, under the conditions of test. When using the HepG2 cell line in the NRU assay, it was possible to differentiate between TFNP and snus extracts, with only snus extracts resulting in a measurable EC50 response.
Conclusion:
This initial preclinical in vitro toxicity assessment of TFNPs compared with both tobacco snus and combustible cigarette smoke indicates that the tested TFNPs have a substantially reduced in vitro toxicity activity compared with traditional tobacco products. Given this initial data, both further scientific exploration and public health discussion on these products are warranted.
Introduction
Smoking is a cause of serious diseases in smokers, including lung cancer, heart disease, and emphysema. There is general scientific consensus that it is the toxicants generated from burning tobacco and inhaling the smoke, and not nicotine, which is the primary cause of smoking-related diseases.1,2 Nicotine is an addictive substance and is not considered to be risk free.3,4 The undisputed best action adult smokers can take to improve their health is through complete cessation of all tobacco and nicotine use. However, many adults are either uninterested or unwilling to quit and continue to smoke combustible cigarettes. With this in mind, transitioning adult smokers, who would otherwise continue to smoke, to alternative nicotine-containing products, which are likely to be substantially less harmful than smoked tobacco is posited as the next best option.1,2 This is the basis of tobacco harm reduction (THR).
Central to THR is a recognition that not all nicotine-containing products are as equally harmful as combustible cigarettes, and that alternative nicotine products are legitimate and substantially less harmful tools to reduce smoking-related morbidity and mortality. 5 For example, a comprehensive and established scientific evidence base, including long-term epidemiological studies confirm that traditional Scandinavian snus, an oral tobacco-containing product, is an effective THR product that is substantially less harmful than combustible cigarette smoking to human health. 6 Moreover, recent scientific review found that the health risks associated with snus use, where nicotine is decoupled from harmful tobacco smoke, are considerably lower than those associated with smoking cigarettes. 7
Recent advances in technology have, and continue to, facilitate the creation of a range of new and innovative alternative nicotine products, which either do not combust tobacco or do not contain tobacco leaf, and have the potential to be substantially less harmful for adult smokers than continued combustible cigarette smoking. Tobacco-free nicotine pouches (TFNPs) are a recent product innovation available in a growing number of countries, adding to the repertoire of potentially reduced harm alternatives for adult smokers who would otherwise continue to smoke. TFNP has a similar appearance and oral route of use to that of traditional tobacco-containing Scandinavian snus. Typically, TFNPs are small pouches containing high-purity pharmaceutical-grade nicotine sprayed onto a plant fiber-based substrate, and contain other high-quality ingredients, including flavorings, humectants to retain moisture, and additives to ensure product stability.
Pouches are placed under the consumers' lips, where nicotine is efficiently released into the bloodstream through the oral mucosa (and not swallowed). 8 A number of TFNPs are available commercially, including Skruf Superwhite and ZoneX from Imperial Brands, Zyn from Swedish Match, On! from Altria, and Velo from British American Tobacco. The relative nascency of the TFNP category is reflected in the state of the accompanying published science.
Studies characterizing TFNPs are now beginning to appear in the scientific literature. Due to TFNPs being a relatively new product, there is very limited in vitro toxicological data in the published scientific literature, and an approach for assessing these products has not yet been standardized. For example, a recent study by East et al. evaluated a range of TFNPs with different nicotine strengths and flavors using real-time cell analysis rather than use of classic regulatory toxicology assays. 9 Another study by Bishop et al. utilized a series of contemporary in vitro high-content screening and mechanistic reporter assays to detect changes in cell viability, health markers, oxidative stress, and genotoxicity in human oral fibroblast and human lung epithelial cells (H292) after 24 hours' exposure to TFNP extracts. 10
For traditional smokeless tobacco products that contain tobacco leaf, including snus and chewing tobacco, the U.S. FDA has identified nine harmful and potentially harmful constituents (HPHCs) of interest in smokeless tobacco products for reporting purposes. 11 These constituents represent several different chemical classes (e.g., tobacco-specific nitrosamines, carbonyl compounds, aromatic amines, metals, and volatile organic compounds) with established analytical methods widely available. There is currently no specific regulatory guidance for the reporting of HPHCs or other toxicants of notable public health interest in TFNPs. Recent chemical analyses of TFNPs have demonstrated substantial reductions in the levels of toxicants when compared with both Swedish-style snus, 12 and combustible cigarettes. 13
In this study, we present an extraction method for preclinical in vitro testing, and determine the suitability of assessing TFNP and snus extracts in three established regulatory in vitro toxicology assays: neutral red uptake (NRU) for cytotoxicity, Ames test for mutagenicity, and in vitro micronucleus (IVM) for genotoxicity. To provide a thorough risk characterization of these products as an alternative to combustible tobacco for current adult smokers, these endpoints were compared with that of combustible smoke from a 1R6F reference cigarette. Since nicotine is the main component in TFNPs, it is also tested in the NRU assay as a control. Data on Scandinavian snus, a proven less harmful alternative to combustible cigarettes, 7 were also included in this study to allow for a better positioning of TFNPs on a relative risk scale and thus their THR potential.
Materials and Methods
Reagents
All reagents and equipment were purchased from Sigma-Aldrich (St. Louis, MO) unless otherwise stated.
Test products
All TFNPs were commercially available at the time of the study and obtained from the manufacturer before study initiation (Skruf Snus A.B., Sweden is a wholly owned subsidiary of Imperial Brands PLC). Two strengths of TFNPs were tested in this study: Skruf Superwhite Fresh #2 (5.8 mg nicotine/pouch) and Skruf Superwhite Fresh #3 (10.1 mg nicotine/pouch). The Scandinavian tobacco snus product was Skruf Slim Fresh White portion (10.9 mg nicotine/pouch). The products were selected to best match their flavor and nicotine strength. The TFNPs and Scandinavian tobacco snus both had the same flavor profile of mint. The chemical composition of the mint flavor was the same for both the TFNPs and Scandinavian tobacco snus test samples. All ingredients in this flavor have undergone a toxicological risk assessment by in-house professional toxicologists and other specialists, to determine their suitability in products, using techniques described by others.14,15 For biological evaluation, TFNPs and snus were stored frozen at −70°C before extraction.
The 1R6F reference combustible cigarette (Lot No. 273165) was used as a comparator in the biological evaluation (University of Kentucky, Kentucky). Before smoking and total particle matter (TPM) generation, 1R6F cigarettes were conditioned at 22°C and 60% relative humidity for at least 48 hours, according to the International Organization of Standardization (ISO) standard 3402. 16
Further information on the products tested are detailed in Table 1.
Test Article Information
From combusted cigarette data.
TFNP, tobacco-free nicotine pouch.
Extract generation for in vitro assessment
TFNPs and snus were extracted based on ISO 10993—Biological evaluation of insoluble medical devices, specifically ISO 10993-1217 (part 12: Sample preparation and reference materials). In brief, 6 g of test article was covered with 20 mL phosphate-buffered saline (PBS) as extraction medium to obtain an extraction ratio of 300 mg/mL and agitated at 600 rpm and 37°C for 1 hour. After centrifugation and filtration through 0.45 and 0.2 μm sterile filters, aliquots of 500 μL per extract were frozen at −80°C. Three independent extracts for each test article were generated. Nicotine was used as a marker for evaluating extraction efficiency. The average extraction efficiency in this study was 66%–75% (similar to the extraction efficiency in East et al. 10 ). Nicotine analyses of extracts were performed using liquid chromatography with tandem mass spectrometry with an AB Sciex API 6500 QTRAP (SCIEX, Framingham, MA) and nicotine-d4 as the internal standard. After 1:200,000 methanol dilution, data were collected over a 5-minute solvent gradient with 0.05% acetic acid in water and methanol, using a Gemini NXC18 Column (Phenomenex, CA). Analysis was performed in duplicate.
Cigarette smoke collections were performed with an ISO harmonized Borgwaldt RM-20 D smoking machine. TPM from 1R6F reference cigarettes was generated according to ISO 20779:2018 (55 mL/2 second duration/2 puffs/min). 18 TPM was collected on a 92-mm Cambridge filter pad. After collection, the Cambridge filter pad was cut radially with a scalpel in six identical parts by using a special cutting device guide. Two opposite sections were used for nicotine and water evaluation of the TPM. The TPM from the remaining four parts of the filter pad were extracted in 13.3 mL dimethylsulfoxide (DMSO) by shaking for 20 minutes at room temperature. The extracts were centrifuged through sterile PP-filter (0.45 μm) in Maxi-Spin Filter Tubes and frozen at −70°C aliquoted in 0.75 mL portions. The TPM concentration in DMSO was 25–30 mg of TPM/mL.
Cytotoxicity: NRU assay
Cytotoxicity of 1R6F TPM, TFNPs, and snus extracts was determined using the NRU method.
19
BEAS-2B cells (European Collection of Authenticated Cell Cultures [ECACC]) were cultured with epithelial cell growth medium, supplemented with the corresponding Supplement Mix (Promocell). HepG2 cells (American Type Culture Collection) were cultured with MIS medium (75% Minimum Essential Medium alpha medium and 25% Weymouth's medium with 2 mM
For testing, 100 μL of HepG2 (2 × 105/mL) or BEAS-2B (0.8 × 105/mL) in serum-free medium was seeded into each of the 60 inner wells of a 96-well tissue culture plate and preincubated at 37°C, 5% (v/v) CO2 for 20 ± 3 hours. Following removal of culture medium, cells were exposed to 200 μL of increasing concentrations of extracts diluted in cell medium, up to a maximum concentration of 10 mg/mL (mg extract/mL medium) for 65 ± 2 hours. The corresponding nicotine concentration for the top dose were: 57 (TFNP #2), 84 (TFNP #3), and 86 (Snus) μg nicotine/mL, these nicotine concentrations were similar to the nicotine levels found in the saliva of moist snuff users (73 μg nicotine/mL).20,21 TPM was tested up to a maximum concentration of 30 μg/mL in BEAS-2B and 50 μg/mL in HepG2, for 65 ± 2 hours.
Sodium dodecyl sulfate (SDS) (1–15 μg/mL) and nicotine (0.01–1.0 mg/mL) were used as positive controls.
Following exposure, the medium was replaced with 200 μL neutral red staining solution in culture medium (supplemented with 20 mM HEPES and 10% fetal bovine serum [FBS]) and incubated at 37°C, and 5% CO2 for 3 hours. After staining, the cells were washed and lysed over 30 minutes at room temperature. The neutral red, which is retained in the lysosomes of viable cells, was released and quantified by measuring the absorbance at 540 nm on a microplate reader (TECANSunrise).
The experiments were performed in duplicate (two plates per cell line), and each experiment was repeated on three independent days (replicates). A nonlinear regression model was applied to determine the concentration of test article, which resulted in 50% cytotoxicity (EC50) and 20% cytotoxicity (EC20).
Mutagenicity: bacterial reverse mutation assay (Ames)
The in vitro mutagenicity of 1R6F TPM, TFNPs, and snus extracts was determined using the Ames test, in general accordance with the Organization for Economic Co-operation and Development (OECD) guideline number 471. 22 The induction of reverse mutations with each test article was tested with five bacterial strains (Salmonella typhimurium TA98, TA100, TA102, TA1535, and TA1537; MOLTOX®) with and without S9 (MOLTOX, Lot No. 4246), using the preincubation method as previously described. 23 In brief, 50 μL of extracts (1–5 mg/plate) or TPM (25–120 μg/plate), 100 μL of bacterial culture, and 0.5 mL of S9 mix (+S9) or buffer (0.2 M sodium phosphate) (−S9) were mixed and incubated at 37°C for 20 minutes. After incubation, 2 mL of Top-Agar (45°C) was added, mixed, and then poured on top of a Vogel–Bonner agar plates. After 48–72 hours of incubation at 37°C, the number of revertant colonies was counted using the Synbiosis ProtoCOL SR—Automatic Colony Counter (Meintrup-DWS). The number of revertants in TA1535 and TA1537 strains was determined by manual counting.
The test article was considered mutagenic if: it produced a two-fold increase in the number of revertants when compared with the negative control (solvent, or solvent plus S9-Mix) in strains TA98, TA100, and TA102 and a three-fold increase in TA1535 and TA1537; revertant numbers of three or more test substance concentrations are significantly higher than the negative control; linear dose–response is achieved; and a positive response of a test article was reproducible. Mutagenic activity was calculated from the slope of the dose–response (fold increase in revertants) curve using a nonthreshold model and Dunnett's test (p < 0.05).
Genotoxicity: IVM assay
The IVM assay was used to determine the genotoxic potential of TFNP extracts, snus extracts, and 1R6F TPM, in accordance with OECD test guideline number 487. 24 Genotoxicity was assessed using Chinese hamster lung fibroblast V79 cells (obtained from ECACC). Twenty-four-well multititer, collagen-coated plates were filled with 500 μL of V79 cell suspension (7.5 E4/mL) in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% FBS and 1% antibiotic/antimycotic mixture. The plates were preincubated for 20 ± 2 hours at 37°C and 5% CO2 to allow adherence of the cells to the well bottom. On the next day the cell medium was discarded, cells were exposed to increasing concentrations of test article in serum-free DMEM for 3.5 ± 0.25 hours in the presence or absence of S9-mix or for 22 ± 2 hours without further wash in 10% serum containing exposure medium. Following the short-term treatment, the cells were washed and allowed to recover and divide for 18–22 hours in complete serum-supplemented (10%) medium. The highest doses applied for TFNPs were based on product weight equivalents calculated from the extraction procedure.
Dose levels were applied to reach a maximum concentration of 5 mg/mL as recommended for low toxic test substances in the OECD guideline 487. Nicotine concentrations were calculated following nicotine determination in the different extracts. Dosing of TPM was limited by its high toxicity and did not exceed 140 μg/mL (ST+S9). Positive controls, bleomycin (3 μg/mL) and cyclophosphamide A (short-term treatment: 15 μg/mL; long-term treatment 5 μg/mL), were tested in duplicate. The IVM tests were performed in 24-well plates without Cytochalasin-B.
After recovery, cells were detached using cell detachment solution (Accumax™) and counted using a handheld cell counter (Scepter™ Cell Counter; Millipore). Cell suspensions were centrifuged onto microscopic slides for 5 minutes in a Cytospin™ centrifuge at 380 g. The supernatant was removed, and the preparations dried by one repetition of centrifugation. The cells were then chemically fixed (150 mL methanol/18.5 mL glacial acetic acid/1 mL 37% formaldehyde/30.5 mL water) onto the slides, washed once in methanol then air dried. The cells were stained using 1 μg/mL 4′,6-diamidino-2-phenylindole dihydrochloride (DAPI) in mounting medium (VECTASHIELD, H-1000). Slides were evaluated microscopically using the fully automated Metafer imaging system coupled to a fully automated Microscope (Imager, Z2; Zeiss), with at least 1000 interphase cells per replicate dose level (2 replicates), positive and negative controls.
The criteria for assessing micronuclei included: did the cytoplasm remain intact; were any micronuclei present separated in the cytoplasm micronuclei or just touching the main nucleus; was the main nuclei of the cell of approximate equal size; and was the diameter of the micronuclei smaller than 1/3 of the main nucleus?
For an IVM response to be considered positive, there must be a clear dose-dependent increase in micronucleus frequencies, the increase in micronucleus frequencies should reach statistical significance compared with negative control and the historical data of the laboratory. The cytotoxicity associated with the statistically significant increase should not exceed 60%. Relative population doubling (RPD) was used for the assessment of cytotoxicity in this study. Data on relative cell count and relative increase in cell count were also collected (data not shown).
The micronucleus values for each test article concentrations were compared pairwise to those from the corresponding negative controls using chi-square analysis for a qualitative analysis. A Cochran–Armitage trend test was performed over the increasing dose ranges to check for a dose–response relationship.
Statistical analyses
All statistical analyses were performed using GraphPad Prism version 8.4.3.
Results
Nicotine quantification of PBS extracts
The concentration of nicotine in the test article extracts is shown in Table 2, displayed as milligrams of nicotine per product or milliliter extraction solvent (PBS or DMSO). Snus extracts contained the highest level of nicotine per milliliter, whereas 1R6F TPM in DMSO contained the lowest.
Nicotine Content of Total Particle Matter from 1R6F, Tobacco-Free Nicotine Pouch, and snus Phosphate-Buffered Saline Extracts
Nicotine concentrations are displayed as a mean of three replicates.
DMSO, dimethylsulfoxide; PBS, phosphate-buffered saline; SD, standard deviation; TPM, total particle matter.
Cytotoxicity
The cytotoxicity potential of 1R6F TPM, TFNPs, and snus extracts was evaluated in the NRU assay using BEAS-2B and HepG2 cells. Cytotoxicity was assessed on the basis of the concentration of test article, which resulted in an EC50, or if not achieved, an EC20. The data are represented as both mg/mL and nicotine μg/mL in Figure 1. EC50 and EC20 values with corresponding confidence intervals are reported in Table 3. EC50 values of the positive control SDS and nicotine were consistent with the historical data (not shown) for both cell lines.
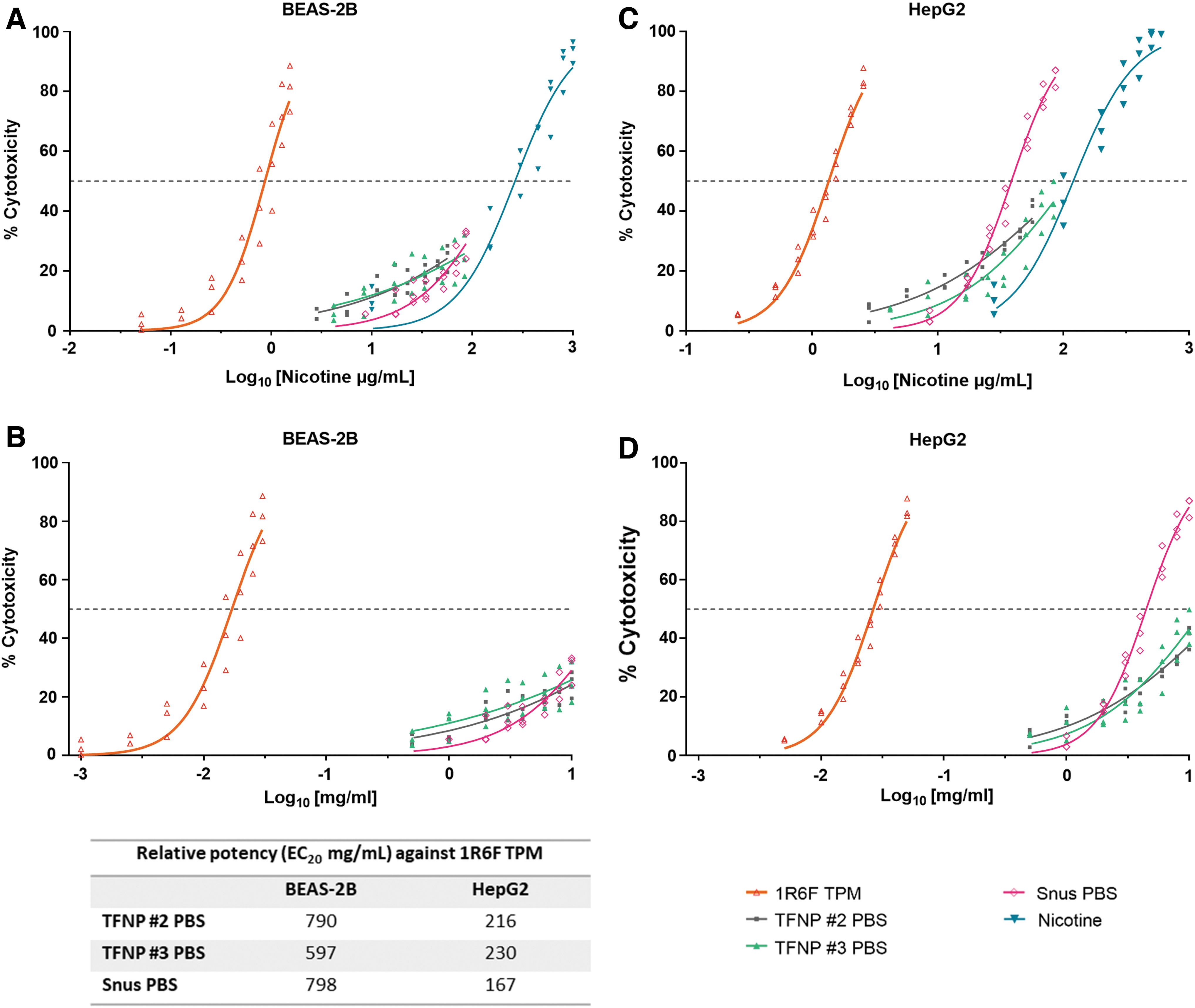
The percent cytotoxicity induced by increasing concentrations of 1R6F TPM, TFNPs, and snus extracts in the NRU assay with
BEAS-2B and HepG2 Cytotoxicity (EC20 and EC50) Data for Tobacco-Free Nicotine Pouches, Snus Extracts, 1R6F Total Particle Matter, and Nicotine
95% CI, 95% confidence interval.
The 1R6F TPM, TFNPs, and snus extracts all showed statistically significant cytotoxicity, as determined by achievement of >20% cytotoxicity, in both cell lines in the tested concentration range. The 1R6F TPM induced a strong cytotoxic response in both cell lines. Extracts from both TFNPs did not achieve EC50 at the top testing concentration (10 mg/mL) in BEAS-2B or HepG2 cells, whereas the snus extract achieved EC50 in HepG2 cells only. In terms of EC20, 1R6F TPM was more than 150 times more cytotoxic than TFNP #2, TFNP #3, and snus PBS, in both cell lines. The relative potency data (EC20) are detailed in Figure 1. Overall, 1R6F TPM was clearly cytotoxic in both test conditions, whereas TFNPs demonstrated a weak cytotoxic response, orders of magnitude less than 1R6F TPM (with fold reductions in cytotoxicity ranging from 167- to 798-fold when snus and TFNPs extracts are compared with 1R6F TPM), under the conditions of the test.
Mutagenicity
No signs of toxicity (thinning of the bacteria colony background lawn or presence of pinpoint nonrevertant colonies) were observed following exposure to either snus, TFNP extracts, or 1R6F TPM. Exposure to 1R6F TPM caused a significant increase in the number of revertants, which were more than twofold higher in strains TA98 (+/−S9), TA100 (+/−S9) (p < 0.05) as shown in Figure 2, and a more than threefold higher in TA1537 (+S9) (p < 0.05). By contrast, no dose-dependent or statistically significant increases in revertant frequencies were observed for TFNPs and snus extracts in any of the five S. typhimurium strains (TA98, TA100, TA102, TA1535, and TA1537) with or without S9 metabolic activation, when compared with the negative controls. Therefore, TFNPs and snus extracts were not mutagenic within the specificity and sensitivity of the method applied in this study, under the test conditions. For detailed results for all test articles in the five S. typhimurium strains see Appendix Table A1.
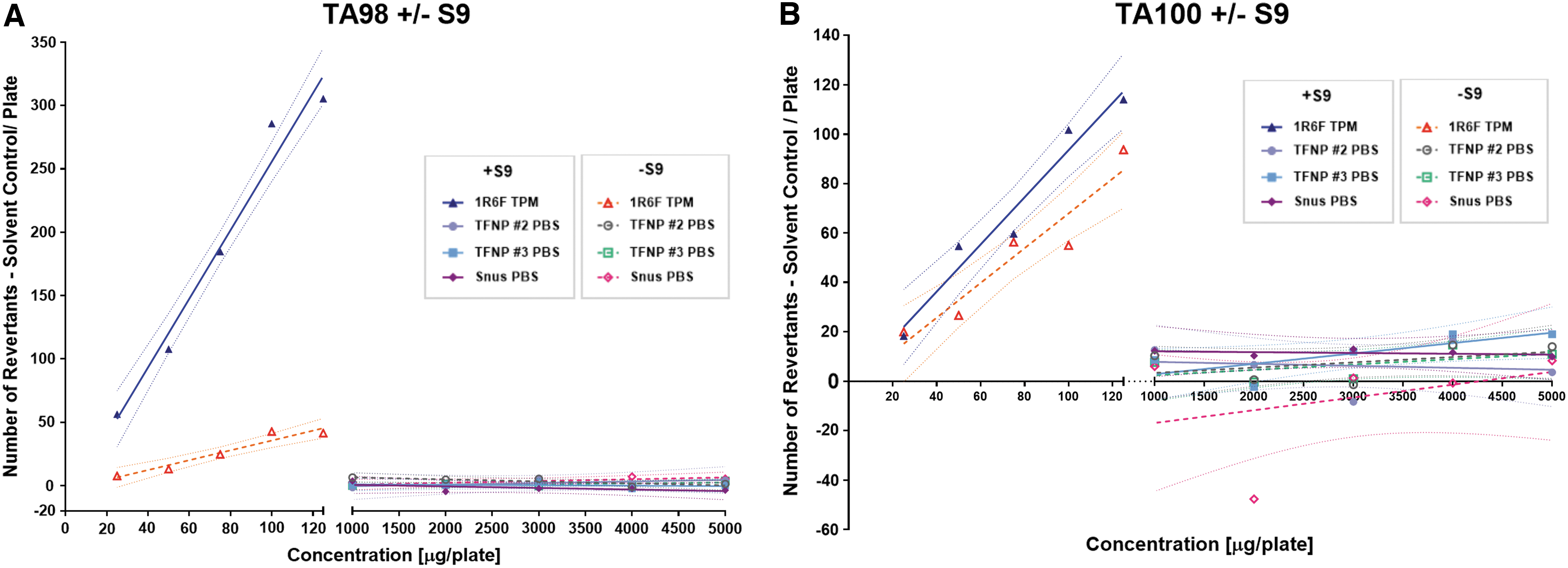
Dose-specific mutagenicity in Salmonella typhimurium in
Genotoxicity
V79 hamster lung cells were exposed to two TFNP extracts, snus extracts, and 1R6F TPM under short-term conditions in the presence and absence of a metabolic activation system (S9), and under long-term treatment conditions in the absence of S9. The response to positive controls (Cyclophosphamide A and Bleomycin) was in accordance with historical data (data not shown). At the top testing concentration of 1R6F TPM, cytotoxicity levels between 45% (ST+S9), 47% (LT-S9), and 88% (ST−S9) (RPD) were obtained. The increase in toxicity was dose dependent for 1R6F TPM. 1R6F TPM caused a dose-dependent, statistically significant (p < 0.0001) increases in micronucleus frequencies in all three treatment conditions when compared with the negative controls, indicating a strong genotoxic response (Fig. 3).
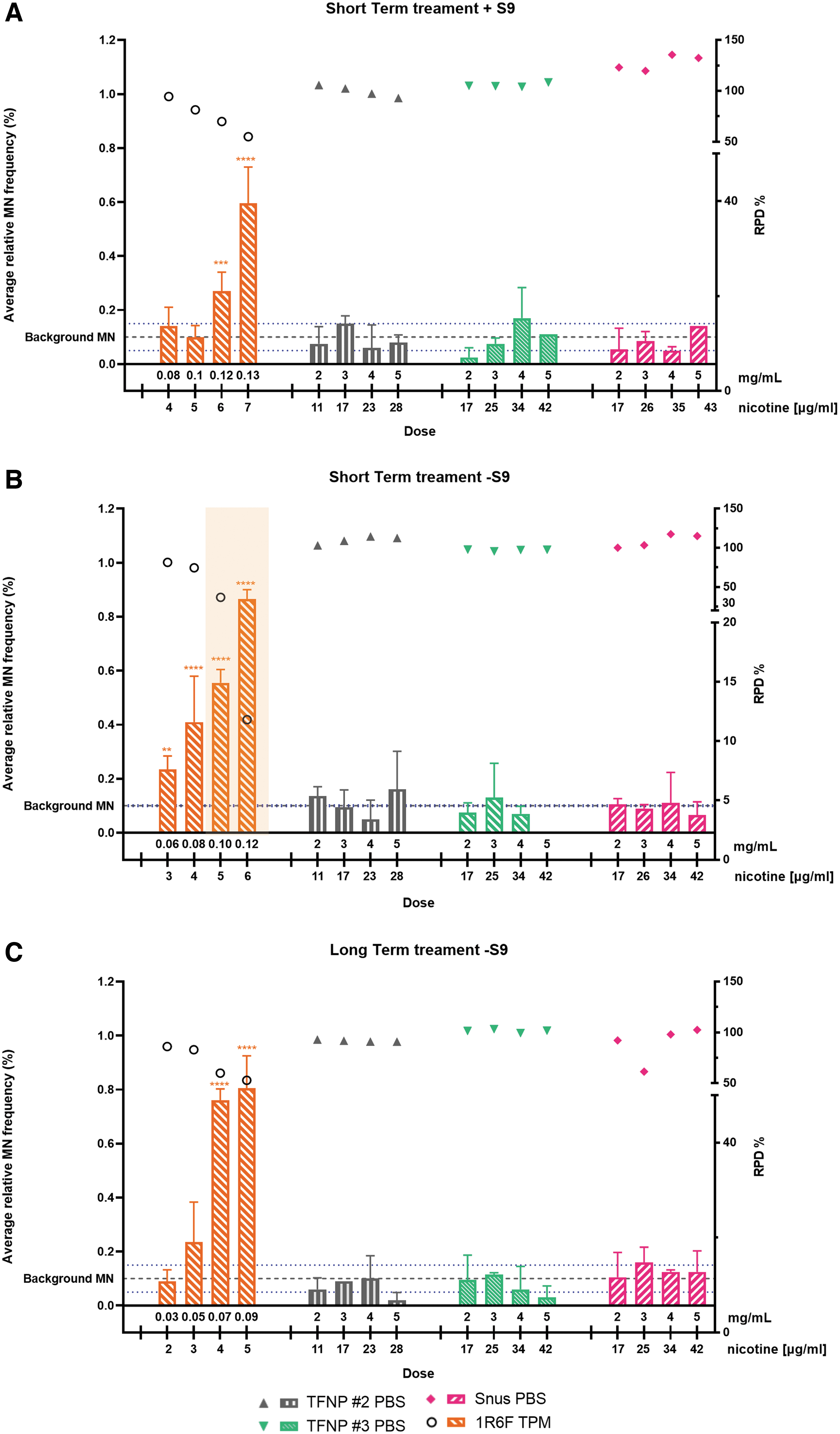
The genotoxicity assessment of test articles in the IVM assay for
Using the short-term treatment in the absence of S9, the toxicity exceeded the limit of 60% maximum toxicity, but even at acceptable toxicity levels the increase in micronucleus frequencies reached high significance and allows a classification as genotoxic. No signs of cytotoxicity (RPD) were observed for the two TFNPs and snus extracts. By contrast, no dose-dependent or statistically significant increases in micronucleus frequencies were observed for any of the TFNPs or snus extracts independent of the treatment applied, when compared with the negative controls.
Discussion
The objective of this study was to assess the cytotoxicity, mutagenicity, and genotoxicity of two commercially available TFNP and one snus extracts in three established regulatory in vitro assays, and compare the responses to combustible cigarette smoke TPM. The NRU, 19 IVM, 24 and the Ames assay 22 are routinely used for product assessments, including the assessment of tobacco and nicotine-containing products. 23 The combustible cigarette smoke induced strong responses in all three assays, whereas TFNP and snus extracts demonstrated little to no response in the in vitro toxicological assays under the assessed conditions. To enable the exposure of the in vitro systems to oral nicotine delivery products, such as TFNPs and snus, these were extracted using PBS to enable the trapping of water-soluble compounds. A range of extraction techniques has been reported for the testing of oral tobacco products (such as snus), including PBS, DMSO, cell media, and complete artificial saliva, with their own advantages and limitations.10,21,25–29 Artificial saliva and cell media are highly variable between batches and suppliers, making it challenging to generate a uniformed exposure system.
The chemical simplicity of PBS allows for the generated extract to be quantified for nicotine without any potential confounding interference due to cell medium constituents, for example, protein binding, and can be used in multiple, independent assays (different assays will require different cell medium).30–32 Zanetti et al. similarly employed PBS extraction over artificial saliva for the assessment of Scandinavian snus; with the authors rationalizing the use of this extraction medium due to its similar properties to artificial saliva but contains none of the biological additives, which could interfere with the cell culture. The extracted concentrations of nicotine found in PBS extracts by Zanetti et al., were at comparable levels to those determined in the present study, which in turn were equivalent to nicotine concentrations measured in the saliva of moist snus users.20,21
The trapping of cigarette smoke aerosol in PBS has been reported, although limited data are available on the impact of these in regulatory toxicity assays, 33 therefore TPM was selected as an appropriate delivery mechanism. The cytotoxic response of products was assessed in BEAS-2B and HepG2 cell lines through the NRU assay. BEAS-2B lung cells have a history of use in the cytotoxic assessment of inhaled products, including reference cigarettes and electronic cigarettes.23,34,35 Although the BEAS-2B cell line may not be relevant to adult smokers' exposure to oral nicotine delivery products, the cell line is well established and can give an indication of relative cytotoxicity, which could be used as an internal control and as part of future comparison studies. Similar approaches for the screening and assessment of TFNP extracts using well-characterized, established cell lines has been reported by Bishop et al., and East et al.9,10
A difference in sensitivity between the two cell lines was observed in this study. HepG2 cells appear to detect cytotoxicity at a lower concentration than BEAS-2B cells for all the test items. It should be noted that BEAS-2B cells have limited metabolic capacity, whereas HepG2 cells, which are widely used to screen the cytotoxic potential of new chemical entities, retain some metabolic capacity making these more appropriate in the context of protoxicant assessment.36,37 In addition, within the published literature, it has been reported that different subtypes of nicotinic acetylcholine receptors (nAChR) are expressed on each cell line: HepG2 express α7nAChR38,39; BEAS-2B express muscle-type nAChR (α1/β1/δ/ɛ).40,41 From published data, 0.1 mM of nicotine has been reported to reduce the cell viability of HepG2 to 45%–58%,38,39 whereas 2.5 mM of nicotine has been reported to reduce cell viability of BEAS-2B to ∼50%. 42 An EC50 of 0.27 mg/mL (1.65 mM; BEAS-2B) and 0.12 mg/mL (0.7 mM; HepG2) have been derived for pure nicotine in this assay, which is in line with the trends reported in the literature.
The TFNP, snus extracts, and 1R6F TPM demonstrated statistically significant cytotoxicity responses, as determined by the achievement of >20% cytotoxicity, in both cell lines. However, TFNPs and snus extracts demonstrated substantially reduced cytotoxicity when compared with combustible cigarette TPM, despite the higher nicotine content. In addition, when using the HepG2 cell line, it is possible to differentiate between TFNP and snus extracts, with only snus extracts resulting in a measurable EC50. As both TFNP and the snus product contained the same flavor, it may be anticipated that the tobacco-related constituents may have driven this response in the tobacco-containing snus. The Ames and IVM assay were used to assess the mutagenicity and genotoxicity of the four products assessed in this study. As expected, 1R6F TPM resulted in a clear mutagenic and genotoxic response in both assays, in line with previous literature.23,43
By contrast, TFNPs and snus extracts demonstrated no mutagenic or genotoxic effect in these assays, under the conditions of test. Bishop et al. used the higher throughput ToxTracker assay also demonstrated no genotoxic effect from TFNPs and reference Swedish-style snus pouch extracts, although DNA damage (gamma-H2AX a marker for double-strand breaks in DNA) was reported for the snus extract in the high-content screening assay. 10 The biological responses observed for TFNPs reflect the chemical characterization findings from Azzopardi et al., where HPHCs levels in nicotine pouches were lower compared with those in combustible cigarette and tobacco-containing snus. 12
Study Limitations and Future Perspectives
The results of this study should be considered within the context of the study limitations. Different extraction solvents were used for the TFNP extracts and cigarette TPM, which should be considered when comparing between product categories. Nicotine was used as a marker of dosimetry, but further characterization of extracts as dosimetry markers is recommended, to ensure that any other toxicants of notable public health interest transfer at the same rate as nicotine. It is important to note that the TFNP category is not homogeneous in construction and therefore future studies should assess a range of TFNPs from different manufacturers to further enhance the biological understanding of these products.
Future studies could consider the use of commercially available, human oral epithelial cell lines, or more physiologically relevant three-dimensional models to assess the relative toxicity of TFNPs to combustible cigarettes.10,21 Additional technologies, such as high-content screening and transcriptomics, have the ability to yield more in-depth information on cellular changes and mechanisms following test article exposure. 44 These techniques could be incorporated to further differentiate the biological impact between snus and TFNPs at subcytotoxic levels of exposure to determine any early mechanistic effects. In addition, quantitative in vitro to in vivo extrapolation may be useful in the determination of human exposure levels from in vitro datasets.45,46
Conclusion
To our knowledge this is the first study to assess the preclinical in vitro responses of TFNP extracts in OECD established in vitro assays. The data add to the weight of evidence that snus is a less harmful alternative to combustible cigarettes. The inclusion of snus and combustible cigarette data allows for a better estimation of the harm reduction potential of TFNPs on a relative risk scale. The data from the present study indicate that the assessed TFNPs offer promising THR potential, even more than the already established harm reduction profile of snus, when used by adult smokers as an alternative to smoking combustible cigarettes. The results presented here add to the growing body of scientific evidence supporting a positive role for TFNPs in THR strategies; however, further chemical characterization, preclinical, clinical, and perception and behavior studies are required for a fuller scientific substantiation.
Footnotes
Acknowledgments
The authors would like to acknowledge the staff at Imperial Brands PLC Laboratory Network and Scientific Research & Harm Reduction teams for their contribution in conducting these experiments and data analysis. The authors would also like to acknowledge Maleek Salawu for his technical support. The authors would finally like to thank Imperial Brands PLC internal reading committee for their critical review of the article.
Author Disclosure Statement
All authors were employees of Imperial Brands PLC or subsidiaries at the time of this study. Imperial Brands PLC is the sole source of funding and sponsor of this project. Skruf Snus A.B., the manufacturer of the products used in this study, is a wholly owned subsidiary of Imperial Brands PLC.
Funding Information
This work was funded solely by Imperial Brands PLC.
Appendix
Linear Nonthreshold Model of Tobacco-Free Nicotine Pouch, Snus Extracts, and 1R6F Total Particle Matter for the Ames Assay
| Strain | S9 metabolic activation | TFNP #2 PBS extract | TFNP #3 PBS extract | ||||
|---|---|---|---|---|---|---|---|
| Linear nonthreshold model | Linear nonthreshold model | ||||||
| Slope of the dose-response curve | 95% Confidence interval | Does slope demonstrate significance over negative control? (p < 0.05) | Slope of the dose-response curve | 95% Confidence interval | Does slope demonstrate significance over negative control? (p < 0.05) | ||
| TA98 |
|
0.0012 | 0.0043 | 0.5466 | −0.0003 | 0.0020 | 0.7297 |
| − | −0.0016 | 0.0015 | 0.0446 | 0.0007 | 0.0012 | 0.2612 | |
| TA100 | + | −0.0008 | 0.0061 | 0.7811 | 0.0042 | 0.0043 | 0.0525 |
| − | 0.0021 | 0.0044 | 0.3136 | 0.0022 | 0.0042 | 0.2874 | |
| TA102 | + | 0.0022 | 0.0088 | 0.5985 | 0.0015 | 0.0074 | 0.6703 |
| − | −0.0036 | 0.0114 | 0.5059 | 0.0043 | 0.0096 | 0.3524 | |
| TA1535 | + | 0.0001 | 0.0010 | 0.7772 | 0.0011 | 0.0014 | 0.0991 |
| − | 0.0003 | 0.0009 | 0.4674 | 0.0001 | 0.0009 | 0.809 | |
| TA1537 | + | 0.0000 | 0.0015 | 0.9612 | −0.0008 | 0.0013 | 0.2073 |
| − | −0.0003 | 0.0031 | 0.8549 | 0.0019 | 0.0022 | 0.0873 | |
| Strain | S9 metabolic activation | Snus PBS extract | 1R6F TPM | ||||
|---|---|---|---|---|---|---|---|
| Linear nonthreshold model | Linear nonthreshold model | ||||||
| Slope of the dose-response curve | 95% Confidence interval | Does slope demonstrate significance over negative control? (p < 0.05) | Slope of the dose-response curve | 95% Confidence interval | Does slope demonstrate significance over negative control? (p < 0.05) | ||
| TA98 | + | −0.0012 | 0.0028 | 0.3554 | 2.7040 | 0.3600 | <0.0001 |
| − | 0.0014 | 0.0017 | 0.1057 | 0.3880 | 0.1278 | <0.0001 | |
| TA100 | + | −0.0003 | 0.0042 | 0.8654 | 0.9533 | 0.2480 | <0.0001 |
| − | 0.0052 | 0.0113 | 0.3410 | 0.7027 | 0.2524 | <0.0001 | |
| TA102 | + | 0.0026 | 0.0059 | 0.3558 | −0.2480 | 0.4166 | 0.2209 |
| − | 0.0103 | 0.0072 | 0.0085 | 0.3147 | 0.2832 | 0.032 | |
| TA1535 | + | −0.0001 | 0.0012 | 0.8612 | 0.0200 | 0.0376 | 0.2712 |
| − | 0.0008 | 0.0010 | 0.1200 | 0.0373 | 0.0385 | 0.0566 | |
| TA1537 | + | −0.0001 | 0.0015 | 0.8547 | 0.3560 | 0.1146 | <0.0001 |
| − | −0.0012 | 0.0020 | 0.2315 | −0.0387 | 0.0582 | 0.1748 | |
PBS, phosphate-buffered saline; TFNP, tobacco-free nicotine pouch; TPM, total particle matter.
