Abstract
Introduction:
The impurities of the air inside the buildings and the resulting adverse health effects have become an increasing problem. Typically indoor air impurities are mixtures of many chemical substances at relative low concentrations. The toxicity of individual substances may be negligible, the mixture effect being the primary cause for toxicity and the potential adverse health effects. Standardized screening methods for identifying the health hazard are lacking. The aim of this study was, by using conventional cytotoxicity/cell viability assays, to investigate whether indoor air cytotoxicity can be detected from water samples condensed from indoor air.
Materials and Methods:
The cytotoxicity of 712 water samples condensed from indoor air was investigated. First, 24 samples were tested in four different cell types (human bronchial epithelial cells, BJ fibroblasts, Tohoku Hospital Pediatric-1 [THP-1] monocytes, and THP-1 macrophages) using neutral red uptake and water-soluble tetrazolium salt 1 (WST-1) assays. Thereafter, 688 samples were tested using THP-1 macrophage/WST-1 assay. All samples were tested at 10% sample concentration, 56 samples were also tested at 25% concentration to see dose–response effects.
Results:
THP-1 macrophage/WST-1 assay was the most reproducible method for assessing indoor air cytotoxicity. The adverse effects of indoor air samples on THP-1 cells ranged from ca. 33% loss in cell viability to ca. 17% increase in mitochondrial activity (“cell stress”). Indoor air samples from 75% sampling sites where people reported health symptoms caused adverse effects in THP-1 macrophage/WST-1 assay.
Conclusions:
The assessment of indoor air cytotoxicity using water samples condensed from the indoor air and THP-1 macrophage/WST-1 assay provides a novel and practical biological approach for investigating indoor air quality. This method does not replace existing methods, but supplements them and provides a fast and cheap alternative for the first-stage screening for recognizing poor indoor air regardless of its source.
Introduction
The impurities of the air inside the buildings and the resulting adverse effects on human health have become an increasing problem. Since we spend 80%–90% of our time indoors, we are widely exposed to any health hazards present in indoor air.1,2 The most common types of air pollutants encountered indoors are particulate matter (PM), gases such as ozone (O3), nitrogen dioxide (NO2), carbon monoxide (CO), sulfur dioxide (SO2), and volatile organic compounds (VOCs), but also, nonvolatile organic compounds, such as chemical emission from the building and cleaning materials (e.g., biocides and tensides) and microbe-derived toxins (mycotoxins and endotoxins), play an important role.1–3 Impurities of indoor air may cause various symptoms, such as respiratory symptoms, headache, nausea, fatigue, and memory impairment.4,5
The regulatory limits for indoor air impurities are generally quite limited. Present indoor air investigations, for example, in Finland include measurements of VOCs, formaldehyde, CO, tobacco smoke (nicotine), particles (e.g., asbestos), as well as microbiological investigations. 6 However, requirements for assessments of harmful nonvolatile substances, such as mold toxins, biocides, and tensides, are typically lacking.
Indoor air impurities are typically mixtures of many chemical substances at relatively low concentrations. Furthermore, their quality and quantity may vary at different time points. Even if it is known that there are mycotoxins in the indoor air,7,8 and that they are responsible for severe health effects,5,9 it is not known what is the threshold at which mold contamination becomes a threat to health. 10 The toxicity of individual substances may indeed be negligible, the mixture effect being the primary cause for toxicity and the potential adverse health effects. Therefore, there is an urgent need for a first-stage screening method or a battery of methods that are able to assess the biological risk of the indoor air as such (as inhaled) without needing to know, what are the sources, individual components, and their concentrations in the indoor air.
The assessment of the biological risk of the indoor air has been impeded by the lack of suitable sampling methods. Passive collection of settled dust and active collection of aerosolized particles are some of the choices to collect PM.11–15 Dust and aerosols from spaces with poor indoor air quality have been shown to cause immunotoxicity in mouse RAW264.7 macrophages,12,15 and to inhibit boar sperm motility.11,13 Most recently, PM was shown to activate secretion of interleukin (IL)-8 and upregulate genes associated with immune response in a human in vitro airway construct. 14
Here we present a novel approach for determining indoor air toxicity, which consists of collecting water samples from the indoor air by condensing and then testing the cytotoxicity of these water samples. Examples of known substances that could be caught by collecting indoor air vapor include biocides (polyhexamethylene biguanide, MW 185 g/mol; dodecyl-dimethyl-ammonium chloride, MW 362 g/mol), and Genapol X080 (MW 552 g/mol), as they get mobilized into humid air via aerosolization. 16 Moreover, their mobilization increases with increasing relative humidity (RH). 16
Furthermore, there is evidence that various fungi emit mycotoxins to guttation droplets,17,18 which get aerosolized and migrate through air, which in turn may lead to their exposure via inhalation. Mycotoxins detected in these guttation droplets include chaetoglobosins (MW >500 g/mol), communesins A (MW 457 g/mol), B (MW 509 g/mol) and D (MW 523 g/mol), 18 and macrocyclic trichothecenes (MW 484–544 g/mol). 17 Hence, it appears that water can serve as a carrier for nonvolatile (MW >300 g/mol) compounds. Further advantages of collecting water samples from indoor air are that they represent the actual air inhaled at a given moment, unlike, for example, the dust samples that typically accumulate indoor air components over time. Moreover, the water samples are readily available for toxicological applications as the test substance (sample) should usually be dosed to the test system (cells, tissues, animals) in liquid form.
In the present study, we investigated whether the cytotoxicity of indoor air can be detected from water samples condensed from indoor air using conventional cytotoxicity/cell viability assays. First, the effect of 24 indoor air samples on cell viability was investigated in four different cell types, that is, human Tohoku Hospital Pediatrics-1 (THP-1) monocytes, THP-1 monocytes activated to macrophages using phorbol 12-myristate 13-acetate (PMA), human foreskin BJ fibroblasts, and human bronchial epithelial (HBE) cells. Monocytes, macrophages, bronchial epithelial cells, and fibroblasts are all relevant cell types in exposure via inhalation.
Water-soluble tetrazolium salt 1 (WST-1) and neutral red uptake (NRU) assays were used as cytotoxicity/cell viability assays. More precisely, WST-1 assay is an indicator of mitochondrial activity, and hence, an indirect measure of cell viability and proliferation. NRU, in turn, is based on the uptake of neutral red (NR) by lysosomes of living cells, and is hence an indicator of lysosomal activity and membrane integrity.
Based on the results from the first 24 samples, THP-1 macrophage/WST-1 assay was selected for further use. To verify that the THP-1 macrophage/WST-1 assay predicts cytotoxicity correctly, four chemicals known to be cytotoxic [triclosan, nickel (II) sulfate hexahydrate, 1-chloro-2,4-dinitrobenzene (DNCB), and sodium dodecyl sulfate] and one chemical known not to be cytotoxic (
Materials and Methods
All testing was performed in a good laboratory practice (GLB)-compliant laboratory. GLP principles were followed when applicable. The test systems used were commercially available cells of human origin (BJ fibroblasts, THP-1 monocytes and HBEC). The samples subject to this study were condensates of indoor air collected from different buildings. Reasons for sampling were provided anonymously by the building owners or users.
Cells
Human THP-1 monocytes (Cat. No. TIB-202), human BJ fibroblasts (Cat. No. #CRL-2522), and HBE cells (Cat No. # PCS-300-010) were purchased from ATCC (American Type Culture Collection; LGC Promochem AB, Boras, Sweden). Before experiments, the cells were tested for mycoplasma (MycoAlert™), and were mycoplasma-free.
Materials
MycoAlert was from Lonza (Basel, Switzerland). Minimum Essential Medium with Earle's salts (MEM), RPMI 1640 Medium, Airway Epithelial Cell Basal Medium, and Bronchial Epithelial Cell Growth Kit were from ATCC (LGC Promochem AB). Fetal bovine serum (FBS), nonessential amino acids (NEAA),
Indoor air samples
Seven hundred twelve water samples condensed from indoor air (hereinafter “indoor air samples”) were obtained from 366 different buildings in Finland, including private homes, public spaces, offices, and schools. Sometimes several samples were collected from one building. The usual reasons for indoor air sampling were as follows: (1) people living or working in the premises had health symptoms (e.g., headache, fatigue, respiratory symptoms, persistent flu, eye irritation) and wanted to exclude the possible role of indoor air as a cause of the adverse health effects, (2) there were doubts about the indoor air quality due to occurrence of water damage in the building, or (3) the indoor air quality was inspected in a sales situation in connection with the condition inspection. Specifying the reason for sampling was voluntary.
The sampling was performed using the patented E-collector 19 as follows. (1) Dry ice (−79°C) was placed inside the stainless steel E-collector. The lid was closed to facilitate frosting. (2) Temperature near the collector's surface decreased, and when the dew point was reached, the air condensed to liquid water and froze on the collector surface. (3) After collection of the frost, the lid and the dry ice were removed. (4) Frost was melted, trickled on to the receiver, and was then collected into Eppendorf or glass tubes. (5) The tubes were closed and shipped to the laboratory for analysis. (6) Temperature and RH% were recorded.
Temperature ranged between 21.3 ± 3.2°C, and RH% between 36.5 ± 11.0%. Immediately after arrival in the laboratory, the indoor air samples were sterile filtered (using pore size 0.2 μm) and stored at 4°C until tested. According to our stability studies, the stability of the samples was better at 4°C than at room temperature or at −20°C (data not shown). Before exposure, the samples were warmed to 37°C.
First, 24 indoor air samples were tested on THP-1 monocytes, THP-1 monocytes activated to macrophages, BJ fibroblasts, and HBE cells using both WST-1 and NRU assays. Then, an additional 688 samples were tested on THP-1 macrophages using WST-1 assay.
Cell culturing
Cells were grown in standard culture conditions at 37°C and 5.0% CO2, and subcultured at least twice (but maximum three times) before seeding for cytotoxicity assays in 96-well plates as follows:
BJ fibroblasts were seeded in MEM supplemented with 10% FBS, 2 mM
THP-1 monocytes were seeded in RPMI 1640 supplemented with 10% FBS at a density of 10,000 cells/well/50 μL, and grown for 24 hours before exposure.
For activation toward THP-1 macrophages, THP-1 monocytes were seeded at a density of 10,000 cells/well/100 μL in RPMI 1640 supplemented with 10% FBS and 25 nM PMA. Cells were grown in this activation medium for 48 hours, after which the activation medium was removed and replaced with RPMI 1640 supplemented with 10% FBS, but no PMA. Cells were then grown 24 hours before exposure. The activation of THP-1 monocytes to macrophages was confirmed by visual inspection and by measuring their IL-1-beta and tumor necrosis factor (TNF)-alpha secretion after 24 hours of exposure to 20 ng/mL LPS using ProcartaPlex Multiplex Immunoassay according to the manufacturer's instructions. LPS-treatment increased IL-1-beta and TNF-alpha secretion ∼18 and ∼4380-fold, respectively, compared with untreated control.
Human bronchial epithelial cells were seeded at a density of 1500 cells/well/100 μL in Airway Epithelial Cell Medium supplemented with the Bronchial Epithelial Cell Growth Kit according to manufacturer's instructions.
Performance of cell viability/cytotoxicity assays
Exposure to pure chemicals
THP-1 macrophages were exposed to pure chemicals at the following concentrations: nickel (II) sulfate hexahydrate 6–1000 μg/mL, SDS 4.7–201 μg/mL, DNCB 0.5–100 μg/mL, triclosan 1.8–100 μg/mL, and
Exposure to indoor air samples
The indoor air samples were dosed to all cell types at 10% concentration in a cell-specific medium supplemented with 5% FBS and 200 IU/mL Pen/Strep. Ten percent distilled water served as (untreated) vehicle control. For the assessment of dose–response effects, the indoor air samples were dosed to THP-1 macrophages also at 25% concentration. Twenty-five percent distilled water served as (untreated) vehicle control. Ten percent and 25% concentrations were selected because according to our experience, 10% water concentration is safe to the cells and does not affect cell viability compared with cells in 100% cell culture medium. Concentrations <10% would not (based on our preliminary experiments) have caused measurable effects in the assays. Twenty-five percent concentration, in turn, was the highest conceivable concentration as 25% water concentration itself affects cell viability, probably via osmotic stress. The indoor air samples were always tested in six replicates.
When testing the first 24 indoor air samples in all cell types, SDS at eight different concentrations (4.7–201 μg/mL) was used as positive control in NRU assay, and triclosan at eight different concentrations (1.8–100 μg/mL) was used as positive control in WST-1 assay. SDS is a membrane detergent and hence a suitable positive control for NRU assay, which measures membrane integrity, whereas triclosan is a mitochondrial toxin 20 and hence suitable for the WST-1 assay, which measures mitochondrial activity.
As triclosan causes a mild hormetic effect at low concentrations and then a sharp drop in cell viability (Fig. 1), we searched for a more suitable positive control to the THP-1 macrophage/WST-1 assay for the remaining 688 indoor air samples to better match the responses caused by the indoor air samples (based on the first 24 indoor air samples tested, the highest decrease in THP-1 macrophage viability at 10% sample concentration was 13%). Nickel (II) sulfate did not cause hormesis and caused gradual loss of THP-1 macrophage viability (Fig. 1). According to our further optimization, 2.0 and 20.0 μg/mL nickel (II) sulfate hexahydrate concentrations caused 3.59 ± 2.91 (N = 37) and 14.6 ± 4.19 (N = 39) cytotoxicity to THP-1 macrophages, respectively. Therefore, 2.0 and 20.0 μg/mL nickel (II) sulfate hexahydrate was used as positive control when testing the remaining 688 indoor air samples in the THP-1 macrophage/WST-1 assay.
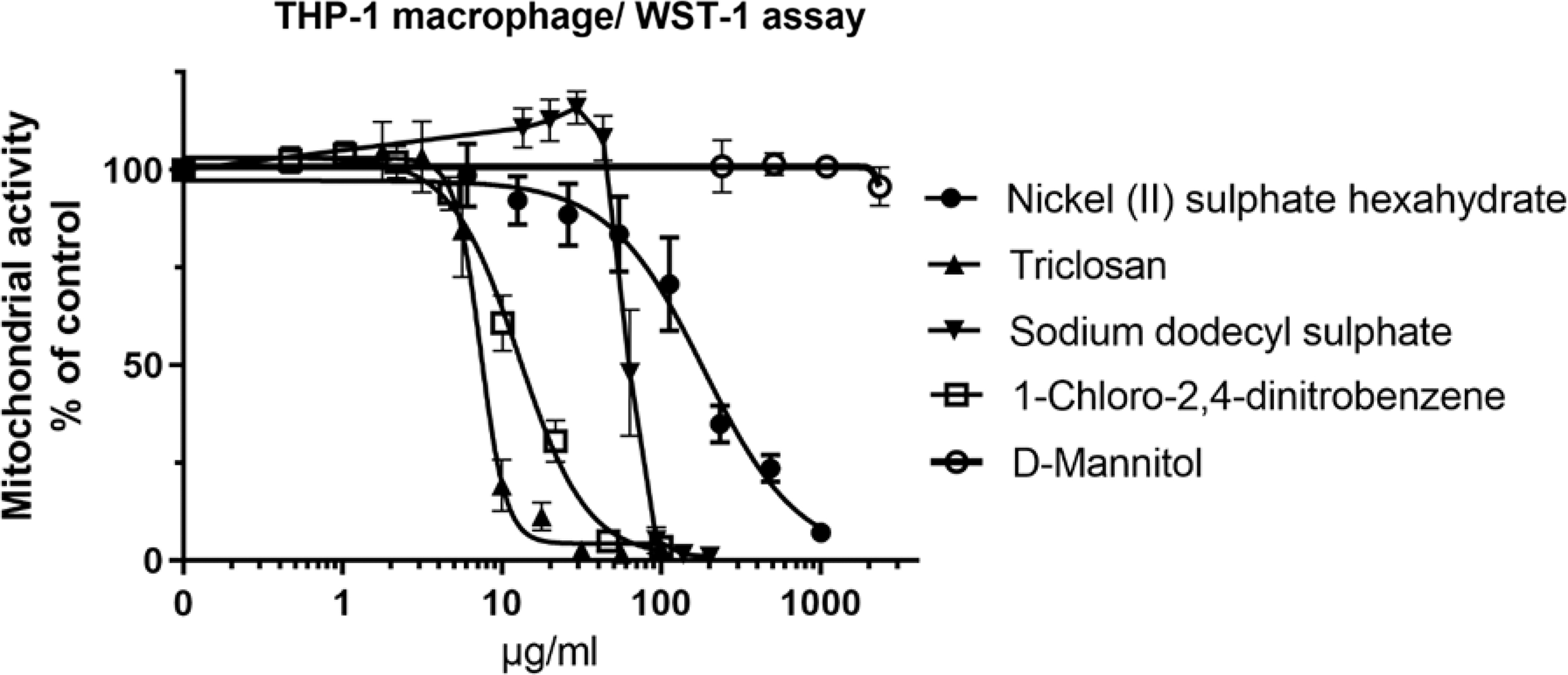
The effects of nickel (II) sulfate hexahydrate, 1-chloro-2,4-dinitrobenzene, sodium dodecyl sulfate, triclosan, and
Cytotoxicity/cell viability assays
Cytotoxicity/cell viability assays used were WST-1 assay (indicator of mitochondrial dehydrogenase activity) in all cell types, and NRU assay (indicator of lysosomal activity and membrane integrity) in BJ and HBE cells. Both WST-1 and NRU assays are photometric methods. For WST-1 assay 10 μL of WST-1 reagent was added to all wells for 1–3 hours at 37°C, 5% CO2. After incubation, the absorbance of the formazan product was read at 450 nm using Spark Multimode reader (Tecan, Switzerland). NRU assay was performed using the standard protocol of Organisation for Economic Co-operation and Development (OECD) GD 12921 and as described by Mannerström et al. 22 The measured absorbances in both assays are directly proportional to cell viability.
Data handling and statistical analysis
The absorbance results were normalized, that is, the viability of the untreated control was always set as 100%, and all other data were calculated relative to the control value. For chemicals, dose–response curves were drawn with GraphPad Prism 8.3 using sigmoidal, four-parameter equation, and the half maximal inhibitory concentration (IC50) values were calculated.
For indoor air samples, results were given in tabular form as either percent decrease in cell viability (negative values) or percent increase in mitochondrial activity (WST-1 assay) or NR uptake (positive values). The statistical significance of the differences between indoor air sample-treated cells and untreated control cells was primarily tested using two-tailed t-test, but if the data of the two groups did not show normal distribution according to the Shapiro–Wilk normality test or their variances were not equal, the Mann–Whitney rank sum test was used (SigmaPlot 14.0). When p < 0.05, the difference was considered statistically significant. Coefficient of variation (CV%) values were calculated for the parallel absorbance measurements.
Results
The measured effects on cell viability were (1) no change in viability, that is, no effect on NR uptake or mitochondrial activity (WST-1 assay), and (2) decreased NR uptake or decreased mitochondrial activity (WST-1 assay), which both indicate cytotoxicity, that is, decrease (loss) of viability or (3) increased NR uptake or increased mitochondrial activity or proliferation (in WST-1 assay), which were also referred to as “cell stress.” It is noteworthy that the numbering of the samples in Tables 1 and 3is independent of each other, that is, the same number does not refer to the same sample.
A Summary of the Results Using Different Cells and Cytotoxicity/Cell Viability Assays to Investigate the Changes in Cell Viability Caused by Water Samples Condensed from Indoor Air
The results are normalized against (untreated) control and expressed as percent change in cell viability, mean ± standard deviation, compared with control (0% change). Negative values refer to decrease in viability, and positive values refer to increased mitochondrial activity (WST-1 assay) or increased NRU. Both decreased viability and increased activity are considered adverse effects. Each sample was tested at six replicates. All samples were tested at 10% sample concentration.
, **, and *** refer to statistically significant change in cell viability compared with (untreated) control; * p < 0.05; **p < 0.01; and ***p < 0.001.
THP-1, Tohoku Hospital Pediatric-1; WST-1, water-soluble tetrazolium Salt 1; SEM, standard error of the mean; NRU, neutral red uptake; HBEC, human bronchial epithelial cells.
Comparison of the effects of indoor air samples on cell viability in different cell types and assays
Table 1 summarizes the effects of 24 indoor air samples on the viability of different cell types. All samples were collected from the premises with reported health effects.
THP-1 macrophages were most sensitive to indoor air samples in terms of the number of indoor air samples (12 of 24) that affected cell viability (in a statistically significant manner). The highest decrease in THP-1 macrophage viability was 13.01%. The highest effect altogether was a 45.96% decrease in viability and was measured in HBE cells. Four to five indoor air samples (depending on the assay) affected BJ fibroblast viability: In BJ fibroblast/NRU assay, two samples decreased BJ viability (highest decrease 9.07%), and two samples increased NRU (at highest 8.82%). In BJ fibroblast/WST-1 assay, five samples decreased cell viability (highest decrease 8.60%). Two samples were cytotoxic to THP-1 monocytes (at highest 8.83%), other samples did not affect THP-1 monocytes.
In terms of well-to-well variation, that is, CV% of untreated controls, THP-1 macrophages showed least well-to-well variation (CV% 2.40%) followed by BJ fibroblasts (CV% 4.56% in NRU and 4.09% in WST-1 assay), monocytes (CV% 5.80%), and HBE cells (CV% 9.98% in NRU and 7.73% in WST-1 assay).
The effects of pure chemicals and 688 indoor air samples on cell viability in THP-1 macrophage/WST-1 assay
The effects of nickel (II) sulfate hexahydrate, SDS, DNCB, triclosan, and
A summary of the effects of 688 indoor air samples on THP-1 macrophage viability tested at 10% sample concentration is presented in Table 2. The results of 56 indoor air samples (collected from 25 sampling sites) tested at two different concentrations, 10% and 25%, are shown in Table 3. Twenty of 56 samples did not affect THP-1 cell viability. Eight samples of 56 were cytotoxic at 10% exposure. The same samples were also cytotoxic at 25% exposure, but caused higher loss in cell viability than at 10% exposure. An additional 17 samples were cytotoxic at 25% exposure, but not at 10% exposure. Eleven samples increased mitochondrial activity at 10% exposure. At 25% exposure, two of these samples increased mitochondrial activity as well, two were cytotoxic and seven did not affect THP-1 macrophage viability.
A Summary of the Effects of Indoor Air Samples on Tohoku Hospital Pediatric-1 Macrophages Investigated with Water-Soluble Tetrazolium Salt 1 Assay
All samples were tested at 10% sample concentration.
The Effect of Indoor Air Samples on Tohoku Hospital Pediatric-1 Macrophage Viability (Tohoku Hospital Pediatric-1/Water-Soluble Tetrazolium Salt 1 Assay) When Tested at Two Concentrations, Ten Percent and Twenty-Five Percent
The results are normalized against (untreated) control and expressed as percent change in cell viability, mean ± standard deviation, compared with control (0% change). Negative values refer to a decrease in viability, and positive values refer to increased mitochondrial activity. Both are considered adverse effects. Each sample was tested at six replicates.
Quality check means inspection of indoor air quality.
, **, and *** refer to a statistically significant change in cell viability compared with (untreated) control; * p < 0.05; **p < 0.01; and ***p < 0.001.
Indoor air samples from 12 sampling sites (building) of the 16 sampling sites with reported health effects (e.g., headache, fatigue, respiratory symptoms, persistent flu, eye irritation) showed cytotoxicity or increased mitochondrial activity in THP-1 macrophage/WST-1 assay (at least one indoor air sample per sampling site).
The osmolality of THP-1 cell culture medium containing no water was 284 mOsm/kg, containing 10% distilled water was 256 mOsm/kg, and containing 25% distilled water was 214 mOsm/kg. By visual inspection, the morphology of THP-1 macrophages (untreated controls) exposed to 10% and 25% distilled water did not differ. The absorbances of the untreated cells with 10% distilled water were 0.780 ± 0.080 and with 25% distilled water 0.745 ± 0.006. Hence, the viability of cells exposed to 25% water was ∼4.5% less compared with the viability of cells exposed to 10% water. Indoor air sample-treated cells were always compared with the respective control to consider the effect of the water itself.
Discussion
Collecting water samples from indoor air by condensing is a novel approach for sampling indoor air. 19 Water vapor mobilizes and serves as a carrier for potential indoor air pollutants, including large molecular-size (>300 g/mol) nonvolatile substances such as mycotoxins, biocides, and Genapol X080. 16 The adverse health effects of biocides 23 and mycotoxins5,9 are well known. Genapol X080, a substance used in industrial and institutional cleaning agents, is found to be toxic at least to human THP-1 monocytes (IC50 0.0027%) and THP-1 macrophages (IC50 0.0035%) (Mannerström M and Heinonen T). Despite the risks posed by these nonvolatile compounds, most obviously present in the indoor air, they are not harvested, nor is their risk assessed in current indoor air investigations. 6
Conventional cytotoxicity assays, such as BALB/c 3T3/NRU assay, 21 were developed for chemicals that can be solubilized, tested at several concentrations to obtain dose–response effect, and to calculate IC50.
Testing of mixtures of environmental samples, such as indoor air samples, is not that straightforward. The composition and stability of the indoor air samples, as environmental samples in general, are not known. In theory, the same sample can be tested once, next time the composition may have changed due to breakdown or reactions between the different components. In addition, obtaining IC50 values may be challenging as the environmental samples may be too dilute to make further dilutions for the testing. The amounts of individual components in mixtures may be below the threshold of toxicological concern, but in mixture, the components with common adverse outcome pathways may act jointly to produce combination effects, that is, additive effects, synergism, or antagonism. 24 Hence, even if each component in the sample would be known and tested separately, it might result in incorrect conclusion on its toxicity. Therefore, methods for assessing the risk of whole mixtures are needed.
The aim of present study was, indeed, to investigate whether cytotoxicity can be detected from water samples condensed from indoor air by using conventional in vitro cytotoxicity/cell viability assays. Testing the effects of indoor air samples on cell viability represents a whole mixture approach, as indoor air samples represent the air as it is inhaled. The toxicity of indoor air samples was investigated using four different cell types: HBE cells, THP-1 monocytes, THP-1-macrophages, and BJ fibroblasts.
HBE cells lining the bronchi were a natural choice as lung is the organ that the indoor air impurities come first into contact with. HBE cells have been used, for example, in research of respiratory diseases.25,26
THP-1 cells, shown to be suitable to study monocyte and macrophage functions, 27 were used as such (monocytes) and as differentiated toward macrophages, to represent pulmonary immune cells, which also belong to the frontline cells exposed to inhaled air. Both monocytes and macrophages are essential in the immune defense. 28 Upon external stimuli, monocytes increase proliferative activity and differentiate toward macrophages, 29 which in turn phagocytose foreign material and initiate protective immune responses. 30 THP-1 cells were activated to macrophages using 25 nM PMA treatment for 48 hours followed by a 24-hour recovery, which has been shown to be optimal for studies on inflammatory responses under various conditions. 31 Using this protocol, THP-1 cells expressed the morphology typical of macrophages, and showed induction of IL-1-beta and TNF-alpha secretion in response to LPS.
Fibroblasts were the fourth cell type used as they are the most common cells in the connective tissue, and widely present in the body. Fibroblasts (mouse BALB/3T3) are also used, for example, in OECD guidelines for acute toxicity, 21 and phototoxicity 32 assessments. In the present study, we used human BJ fibroblasts instead of, for example, BALB/3T3 cells, to improve prediction of human effects.22,33
The cell viability/cytotoxicity assays used were WST-1 (indicator of mitochondrial dehydrogenase activity)34,35 and NRU assays (indicator of lysosomal uptake and cell membrane integrity). 36 Both WST-1 and NRU assays were used with BJ fibroblasts and HBE cells; WST-1 assay was used with THI-1-monocytes and THP-1-macrophages. WST-1 is suitable for suspension and adherent cells, whereas NRU is suitable for adherent cells only. Even if during PMA differentiation THP-1 cells become adherent, 31 they tended to get washed away to some degree during the multiple washing steps included in the NRU test protocol (data not shown). Therefore, NRU assay was not used with THP-1 macrophages.
The highest changes in viabilities (decrease in cell viability or increase of mitochondrial activity or NRU) were measured in HBE cells. These changes were, however, rarely statistically significant probably due to the heterogeneity of HBE cells. 25 In terms of THP-1 monocytes, we expected to observe either cytotoxicity or induced proliferation due to activation by foreign stimuli, 29 but the changes were rarely statistically significant due to large well-to-well variation. Hence, we concluded that HBE and THP-1 cells were too heterogenous to be used in the toxicity assessment of the indoor air samples.
Comparing the effects of indoor air samples on BJ fibroblasts and THP-1 cells, THP-1 macrophages were more sensitive than fibroblasts: 12 of the 24 samples were cytotoxic to THP-1 macrophages, while 2–5 (depending on the assay) were cytotoxic to BJ fibroblasts, and 2 increased NRU.
Phagocytosis is an efficient uptake mechanism for foreign particles, and may explain why THP-1 macrophages were generally more sensitive than fibroblasts: Macrophages are “professional phagocytes” with high phagocytic activity, whereas fibroblasts express lower levels of phagocytic activity. 37 Mouse macrophages have been shown to display several endocytotic uptake mechanisms depending on the particle size. 38 The higher uptake and sensitivity of macrophages compared with fibroblasts were also demonstrated in our previous study comparing nanoparticle toxicity between rat macrophages and BALB 3T3 fibroblasts, 39 as well as in a study by Lanone et al. 40 comparing nanoparticle-treated THP-1 macrophages with the pulmonary cell line A549.
Some indoor air samples induced NR uptake by BJ fibroblasts. One disadvantage of NRU assay is that the results can be interfered by weak basic molecules that accumulate in lysosomes causing swelling, which promotes further NR uptake by the lysosomes. 41 As we investigated mixtures, they may contain a large spectrum of different substances, including weak basics.
Three indoor air samples that were toxic to BJ cells (in either NRU or WST-1 assay) were toxic to THP-1 macrophages as well, whereas five indoor air samples that were toxic to BJ cells were not toxic to any other cell types, suggesting that there are cell-specific differences in the responses to indoor air samples. Hence, taken into account the results of both THP-1 macrophages and BJ fibroblasts, 17 of 24 (70.8%) indoor air samples (all from sites with reported health effects) were cytotoxic.
Using both THP-1 macrophages and BJ cells might thus improve the prediction of indoor air toxicity. However, similarly to HBE cells and THP-1 monocytes, the disadvantage of BJ fibroblasts was their large well-to-well variation (CV% of untreated controls), which was almost two times bigger than that of THP-1 macrophages. This explains why in several cases (samples 3, 20, 22, and 24, see Table 1) the effects of indoor air samples on BJ fibroblasts in WST-1 and NRU assays seemed to be the same, but there was no statistical significance in either assay. Consequently, THP-1 macrophage/WST-1 assay was selected for further evaluation, because it exhibited least well-to-well variation (in terms of CV% of untreated control cells), and hence the best reproducibility.
Before embarking on the large-volume testing of indoor air samples, the performance of THP-1 macrophage/WST-1 assay to predict cytotoxicity in general was verified using four chemicals known to be toxic, that is, nickel (II) sulfate hexahydrate (an electroplating substance), SDS (a surfactant), DNCB (an immunoinductive medicine for the treatment of wart), and triclosan (an antimicrobial agent). All these chemicals, tested six independent times, were reproducibly toxic in THP-1 macrophage/WST-1 assay.
A larger number (688) of indoor air samples were then investigated using THP-1 macrophage/WST-1 assay to find out the prevalence of indoor air toxicity in general, and to get the knowledge about the expected changes in cell viability, that is, what is mild and what is severe effect, in this context, and what is the limit of detection of cytotoxicity. Thirty-six percent of all 688 samples investigated (at 10% sample concentration) were cytotoxic, ∼8% increased mitochondrial dehydrogenase activity and the rest did not affect cell viability. It is noteworthy that the samples were not collected from randomly selected sites, but usually focused on spaces where people suffered from health symptoms.
The measured effects ranged from ca. 20% decrease to 20% increase in THP-1 macrophage viability. Forty-two percent of the samples showed mild toxicity (3%–5% cell death, toxicities <3% were usually not statistically significant), and 58% caused moderate toxicity, that is, 5%–15% cell death. Toxicity higher than 15% was considered severe, and was observed rarely.
Eight percent of the samples increased mitochondrial activity in the WST-1 assay compared with the control. This may be due to increased mitochondrial respiration caused by mitochondrial uncoupling substances. Uncoupling of the electron transfer system from the ATPase leads to accelerated respiration to maintain energy homeostasis.48,49 For example, many microbe toxins are mitochondrial uncouplers. 50 Increased mitochondrial activity can also reflect increased cell number. Although PMA-activated THP-1 cells are not expected to proliferate, 31 there is evidence that macrophages proliferate in pathological conditions. 51 There may also be undifferentiated monocytes left in the THP-1 macrophage culture that were induced to proliferate. Increased monocyte number, that is, monocytosis is indeed a marker of several inflammatory diseases in vivo. 36
Both increased mitochondrial activity and cell proliferation reflect perturbations in cellular homeostasis, that is, cell stress, which cannot be considered serious as cell death, but is all the same an adverse reaction caused by components in the indoor air samples.
As the effects of indoor air samples tested at 10% concentration caused no more than maximum 20% changes in THP-1 viability, we tested 56 indoor air samples also at higher concentrations, that is, at 25%, in an attempt to (1) measure higher changes in cell viability and (2) to see whether the effects are dose–responsive (as toxicology is based on the relationship between dose and the effect).
As expected, the use of these two concentrations improved the sensitivity of the method; 27 indoor air samples were toxic when tested at 25% concentration, whereas only 8 were toxic at 10% concentration. The observed changes in cell viability caused by 25% indoor air sample concentrations were generally higher than those ever measured at 10% concentrations being 33% cell death at highest; furthermore, the effects were always dose-dependent.
In some cases, there was increased proliferation/mitochondrial activity at 10% concentration, but toxicity or no effect at 25% concentration. It appeared that the low concentrations caused cell activation (cell stress), which often preceded cell death. (There is a phase where cell proliferation and cell death cancel each other resulting in 0% effect.) All in all, the use of two sample concentrations, that is, 10% and 25%, improved the reliability and certainty of the results.
When water content is >10% in the cell exposure, it is important to consider the potential osmotic stress caused to cells. The osmolality of THP-1 cell culture medium containing 25% distilled water was 214, the recommended osmolality for most cell cultures being 260–320 mOsm/kg. 52 Hyperosmotic (+200 mOsm/kg) conditions have been shown to evoke inflammatory responses. 53 There are no reports in literature (to our knowledge) on such effects caused by of hypo-osmolality. In terms of cell viability, the −46 mOsm/kg hypo-osmolality present in the 25% exposures had only a minor effect (<5%) on cell viability, and was always considered as the water content of the respective control was the same.
Indoor air samples from the majority (75%) of the sampling sites where people reported health symptoms (e.g., headache, fatigue, respiratory symptoms, persistent flu, eye irritation) caused adverse effects (either cytotoxicity or cell stress) in THP-1 macrophage/WST-1 assay (tested at two indoor air sample concentrations, 10% and 25%), suggesting a connection between poor indoor air and adverse cellular effects. It is noteworthy that even if the reported health symptoms were likely, they were not necessarily related to indoor air. Other reasons may be seasonal cold, flu, and so on.
Finally, it should be emphasized that the method presented here reflects the indoor air quality as it is at the time of sampling. Hence, as there is a direct correlation between the RH% and substance mobilization in the air, 19 increased humidity increases the amount of water inhaled, and also the toxicity of the water vapor.
Conclusions
To conclude, the assessment of indoor air toxicity using water samples condensed from the indoor air and THP-1 macrophage/WST-1 assay provides a novel and practical approach for investigating indoor air quality. This biological method does not replace existing methods, but supplements them, and provides a fast and cheap alternative for the first-stage screening for recognizing bad indoor air quality regardless of its source. At its best, it might help to identify toxic spaces or rooms within “unhealthy” buildings, and can help to target further indoor air investigations and reparation work.
Footnotes
Acknowledgments
We thank the field workers for collecting the indoor air samples, and the occupants and property owners for providing us with the useful information on the sampling sites and potential health effects. Ms. Mirja Hyppönen and Ms. Maaret Vaani are acknowledged for their skillful technical assistance throughout the laboratory experiments. We are grateful to Prof. Emer. Mirja Salkinoja-Salonen for valuable advice during the preparation of this article.
Author Disclosure Statement
No competing financial interests exist.
Funding Information
Part of the funding of this this study was from a commissioned testing service for Sisäilmatutkimuspalvelut Elisa Aattela OY.
