Abstract
Abstract
The exposure of the skin to consumer products, drugs, and environmental chemicals can result in their penetrating the skin barrier and entering systemic circulation, potentially resulting in adverse effects in the skin and other organs. The assessment of dermal penetration and bioavailability (including penetration, metabolism, and entry into the systemic circulation) is therefore an important consideration in the risk assessment of chemicals. The skin is a heterogeneous organ with a multilayer structure. Based on its architecture and physiology, substances can penetrate through three major ways but can also be blocked in the different skin layers and in the skin appendages, which act as reservoir. In addition to that, as the skin is a metabolically competent organ, substances can undergo metabolism. After a brief description of the skin architecture, this review will focus on the skin penetration mechanisms and skin metabolic capacities. The skin absorption has traditionally been tested in vivo on animals. However, with the new legislation (i.e., Registration, Evaluation, Authorisation, and Restriction of Chemicals Regulation or Cosmetics Regulation), alternatives to animal testing have to be implemented. In a second part, this review will provide a description of the main in vitro and in silico or computational models available to study skin absorption and skin metabolism (i.e., ex vivo skin models, artificial membrane barriers, primary cells and cell lines, Quantitative Structure–Activity Relationship [QSAR], simulators for the prediction of skin metabolism).
Introduction
O
Human skin is a complex organ that acts as the first protecting barrier of the body. However, some substances can be transported across the skin, mainly by passive diffusion, and taken up into the living tissue of the body: after contact, the chemical penetrates the stratum corneum and then, reaches the viable epidermis and dermis where the vascular system is hosted. Thus, it may cause local reactions or enter systemic circulation. Like other extra-hepatic organs, the skin is metabolically competent, and during the absorption process, certain chemicals may undergo biotransformation; however, this metabolic capacity is less important than that of the liver.
Human dermal absorption assessment is required by the Plant Protection Products Regulation if toxicity following dermal exposure is of concern compared with that following oral exposure, 4 by the new Biocides Regulation before conducting an acute dermal toxicity test in order to assess the likely magnitude and rate of dermal bioavailability, 5 and by the Cosmetics Regulation. 6 It is also recommended by the Scientific Committee and Consumer Products, as it can then be taken into consideration to calculate the margin of safety. 7 Skin absorption has traditionally been tested in vivo in animals, 8 but an in vitro test guideline based on the use of excised skin is also available. 9 Even though human skin is the ideal system to study dermal toxicity, this is limited by numerous legal and ethical issues. Furthermore, for animal welfare reasons, the use of animal models is limited or banned, depending on the regulatory sector. With the implementation of Registration, Evaluation, Authorisation and Restriction of Chemicals in June 200710 and the ban of the use of animals for testing of cosmetic ingredients and products, 6 the development and the validation of alternative (non-animal) methods such as in vitro and in silico models is a priority.
After a brief description of the skin structure and skin penetration mechanisms and a review of the metabolic capacities of the skin, this paper will focus on the existing in vitro and in silico models available to study dermal absorption and the metabolism of chemicals entering in contact with the skin.
Skin Structure
The skin is the primary barrier to systemic absorption of topically applied chemicals, including cosmetics and pharmaceuticals. Dermal exposure occurs also in a wide variety of occupational settings (e.g., for most pesticides the skin is the primary occupational exposure route). It is a heterogeneous organ (Fig. 1), comprising a number of cellular layers: the epidermis (83.7 μm thick) including the stratum corneum (14.8 μm thick) and the viable epidermis (or cellular epidermis, 68.9 μm thick), 11 the dermis (between 300 and 4000 μm thick), and the hypodermis (thickness up to several millimeters). 12
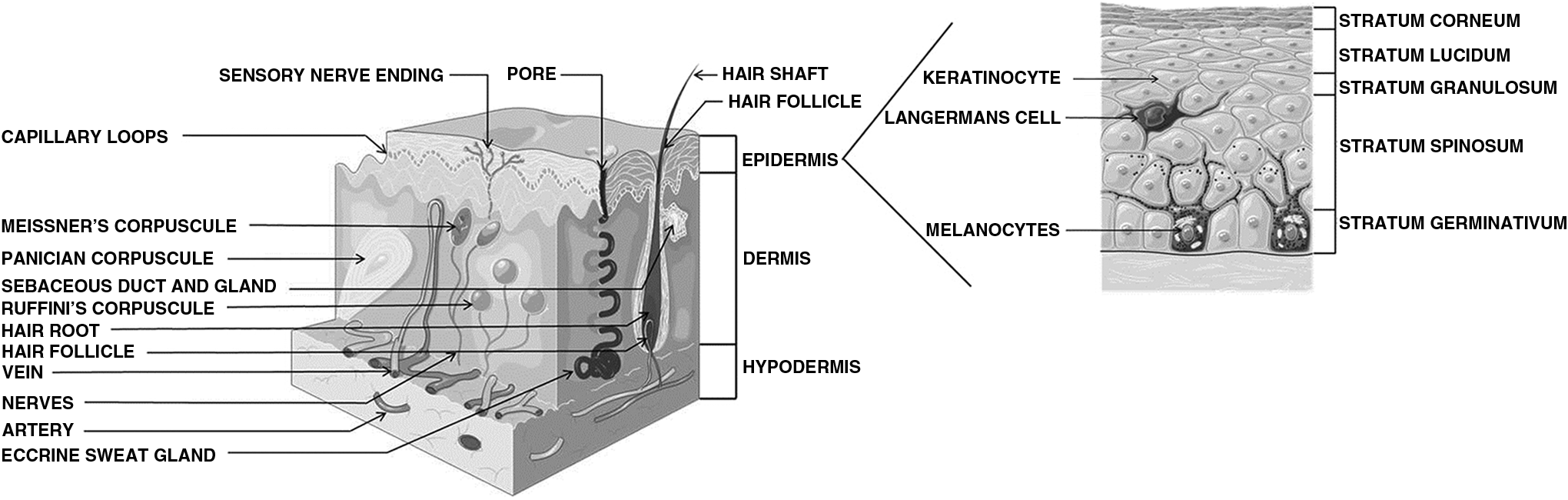
Structure of the skin. Source: Adapted from Servier Medical Art available at /www.servier.co.uk/content/servier-medical-art. Servier Medical Art is licensed under a Creative Commons Attribution 3.0 Unported License (http://creativecommons.org/licenses/by/3.0/).
The epidermis consists of several sublayers starting from the non-viable stratum corneum and as follows: stratum lucidum (clear layer), stratum granulosum (granular layer), stratum spinosum (spinous or prickle layer), and stratum germinativum (basal layer). Described as a “brick and mortar” structure, the stratum corneum contains corneocytes (the “bricks”) and an intercellular lipid matrix (the “mortar”), 13 which is mainly responsible for the major skin barrier function. The corneocytes, which do not contain nuclei or cytoplasmic organelles, are embedded in keratin and surrounded by a protein-rich envelope composed of highly cross-linked proteins, forming a resistant barrier protecting the body from external attacks and water loss. The stratum corneum is continuously replaced through epidermal turnover (i.e., new cells generated at the base of the epidermis and desquamation). The typical time to completely replace the stratum corneum is approximately 14 days. The viable epidermis contains mainly keratinocytes in an avascular environment and in addition, two dendritic cell types, the melanocytes, and the Langerhans cells. It is composed of approximately 40% water, 40% protein, and 20% lipids. 14
The second layer, the dermis, contains blood vessels, lymphatics, nerves, and various skin appendages. It is composed of two layers: the upper papillary dermis and the reticular dermis. The third skin layer is the hypodermis, a subcutaneous fat tissue. Also housing blood vessels, lymphatics, and nervous system, its role is to insulate the skin from below and to connect the skin to underlying structures. 15
Another component of the skin is skin appendages. They set in the dermis and are mainly the sweat glands and the hair follicles. The appendages are estimated to represent up to 0.1% of the skin surface. 16 Sweat glands and hair follicles are present in most of the anatomic sites with an average number of 200–250 sweat ducts and 40–70 hairs per square centimeter, respectively. However, the number of each varies significantly by sites. 17
Skin Penetration
The absorption into the skin occurs predominantly by passive diffusion, which depends on the physicochemical properties of the chemicals. Furthermore, based on the skin architecture and its physiology, the skin penetration can occur in three major ways: (1) through the keratin-packed corneocytes by partitioning into and out of the cell membrane; (2) by transfer around the corneocytes in the lipid-rich extracellular region; and (3) by the shunt pathway through the sweat glands, sebaceous glands, and hair follicles (transappendageal).17,18
Penetration through the epidermis
In the stratum corneum the permeation of a compound through intercellular or intracellular routes depends on its partitioning behavior. As the stratum corneum is a lipophilic barrier, the physicochemical properties of a chemical are important determinants of skin penetration. Lipophilic compounds are thought to pass mainly via the lipid-rich intercellular pathway (around the corneocytes) or “lipid pathway.” This route is the most common. 19 Hydrophilic compounds are thought to pass mainly via the transcellular route (crossing into and out of the corneocytes) or “polar pathway.” For many compounds, the lipophilic stratum corneum represents the primary or rate-limiting barrier. Indeed, highly hydrophilic compounds will not pass this first barrier.
The third way, the shunt transport, is the penetration of substances through the appendages (sweat glands, sebaceous glands, and hair follicles) into the dermis and thus the systemic compartment. With the small relative surface area of the appendages (0.1–1.0% of the total area), this pathway is not generally considered to play an important role in the absorption of chemicals in humans. However, this is a controversial point since the density and size of the hair follicles increase considerably in some areas of the body (e.g., scalp), thus increasing the skin surface area. Moreover, the penetration through the appendages may be important at early time points in the absorption process. 17 In the assessment of dermal penetration, the shunt pathway could be considered using human skin (viable or cadaver) from the appropriate origin (e.g., forearms, scalp, etc.).
Penetration through the dermis
The permeation of chemicals through the viable epidermis can be regarded as a partition from the lipidic stratum corneum to a mainly aqueous environment. For this layer, the high percentage of water constitutes a more effective barrier against lipophilic compounds. 15
Permeation through the dermis is considered to be similar to the viable epidermis, through a mainly aqueous medium, the main difference being the presence of blood vessels and the lymphatic and nervous systems. That contributes significantly to drug transport and distribution in the skin. Substances applied to the skin surface and able to permeate into deeper tissue layers may reach the body's systemic circulation by entering lymphatic or blood vessels in the dermis. 18 Lying immediately below the epidermis and with its extensive vascularization, substances found in the dermis can be considered as potentially bioavailable and having passed into the systemic compartment.15,18
Reservoir effect
The reservoir effect of the stratum corneum, the viable epidermis, and the dermis can play an important role in the bioavailability of a substance through the skin. This means that a substance can remain partly in the skin and can be released (or not) at later time.20,21 For the stratum corneum, the evidence of a reservoir effect was first shown by Vickers in 1963 for topical corticosteroids. 20 The finding was then confirmed for a number of other chemicals (e.g., nicotine, 22 caffeine, 23 cationic β-blocking drugs, 24 sunscreen agents, 25 and chlorpyrifos 26 ). Several parameters determine the duration of the reservoir effect, such as the nature of the permeant, the vehicle used, skin temperature, and the relative humidity to which the skin is exposed. 20 In addition, the role of keratin, the major protein in the stratum corneum, has recently been demonstrated by Seif and Hansen as the fast absorption and slow desorption of chemicals from this protein may be an important contributor. 27 It has been generally assumed that the reservoir is continuously emptied by desquamation. 16 In support of this, Reddy et al. (2000) showed that significant amount of highly lipophilic and very large solute (>350 Da) can be removed if the epidermal turnover was fast relative to the rate of diffusion through the stratum corneum. For other chemicals, they concluded that epidermal turnover is not fast enough and chemicals reach the body. 28 Thus, desquamation will only affect the reservoir effect of the stratum corneum when the penetration rate of the solute is very slow. 21
In 1988, Walter and Kurz showed the binding of 10 drugs to both epidermis and dermis. 29 A more recent study measured in vitro the retention of dihydroxyacetone (DHA), 7-(2H-naphtol[1,2-d]triazol-2-yl)-3-phenylcoumarin (7NTPC) and disperse blue (DB1). After penetration in the skin and at the end of 24 hr, significant proportions of the chemicals were found remaining in the skin and with different partitioning: 10% and 12% of the applied dose of DHA was measured in stratum corneum and viable epidermis/dermis, respectively; 80% of the remaining DB1 was localized in the stratum corneum; 7NTPC diffused also in the deeper layers of the skin. An extended absorption study (after 72 hr) showed that DB1 remaining in the skin did not appreciably move into the receptor fluid beyond the extent measured after 24 hr. These results show that the amount of systematically available chemical can be affected by the reservoir effect. 30 In another study, Jung et al. (2003) showed that the catechol reservoir effect formed in skin during a 24 hr study with catechol, did not decrease (in vivo) or only partially decreased (in vitro) in a 72 hr extended study. 31
Finally, it could be noted that hair follicles (mainly the infundibulum, the upper segment of the hair follicle) can also act as a reservoir.32,33 For example, a study on the antifungal drug isoconazole nitrate, has shown that one week after the last application, approximately equal amounts of the substance were recovered from the hair follicle and from the stratum corneum. 34
Skin Metabolism
As described above, human skin is an important protective barrier of the physical and chemical exposures of the body. However, skin is also a metabolically competent organ. The first-pass effect of the skin has largely been neglected even though the metabolism of many compounds has been highlighted (e.g., parabens, 35 methyl salicylate, 36 benzo[a]pyrene 37 ). Some chemicals are absorbed across the skin and metabolized into active compounds. A good example is the biotransformation of cinnamic alcohol to the protein-reactive cinnamaldehyde. 38
Enzymes in the skin are able to metabolize both endogenous and exogenous compounds. Even though the main role of xenobiotic metabolism is to detoxify potentially reactive chemicals, this can also lead to the activation of the compound. In addition, dermal metabolism can have consequences on the rate of absorption and vice-versa. The physicochemical properties of a penetrating compound can change due to metabolism. The dermal absorption of lipophilic compounds has been shown to increase as these lipophilic compounds are converted into more polar, water-soluble compounds. 39 For example, benzo[a]pyrene penetrates hairless guinea-pig skin more readily than non-viable skin. The explanation could be that more polar metabolites diffuse more readily through the aqueous viable tissue. 40 In addition, it has been shown that the potential for first-pass dermal metabolism during absorption is reduced if the physicochemical properties of the chemical enable rapid penetration. 41
Skin versus liver
Like other extra-hepatic organs, the skin is metabolically competent, but the metabolism is much less extensive than liver metabolism. There are both qualitative and quantitative differences between skin and liver metabolism. The major enzymes important for systemic metabolism in the liver have been also identified in the skin, but with lower activities: 0.1–28% of the activities in the liver for phase I, and 0.6%–50% for phase II. 17 In Van Eijl et al., 42 expression of a total of 36 xenobiotic-metabolizing enzyme (XME) proteins (oxidoreductase, hydrolase, transferase, antioxidant) were detected by proteomic profiling in both the skin and liver, with comparative levels in the skin 4- to 10-fold lower than in the liver (Table 1). Forty-six XMEs, including thirteen cytochrome P450 (CYP) proteins were detected in the liver only. Xenobiotic metabolizing pathways in the skin, previously identified in liver, have been reviewed by Sartorelli et al. 49 Phase I reactions are represented with oxidation (e.g., hydroxylation, epoxidation), reduction (e.g., azo reduction, nitroxide reduction), and hydrolysis (e.g., phosphate ester hydrolysis, epoxide hydrolation) pathways. Concerning phase II reactions, glucuronidation, sulfation, glutathione conjugation, acetylation, amino acid conjugation, and methylation pathways are reported. However, in addition to the activity of metabolic enzymes, the area of the skin exposed should be taken into account when evaluating the importance of skin metabolism; the only place that metabolism will be important for most substances will be the actual area that is exposed to the chemical. 17
When available, the skin/liver ratio is indicated.
Much less studied than in other organs, information on the expression of XMEs in skin is mainly obtained from experiments using ex vivo viable skin or normal human keratinocytes, which represent 90–95% of cells in the epidermis. 50 A non-exhaustive list of XMEs detected in human skin is given in Table 1. The constitutive levels of enzymes can be below the limits of detection, so exposure to an exogenous inducer is sometimes needed.51,52 Some studies reported below and in Table 1 have measured mRNA expression. Even if this is not the most accurate method (compared with protein-level expression and activity), some authors claim that a correlation between mRNA levels and the physiological functions of corresponding genes is generally observed. 43
Phase I enzymes
The expression of various CYPs has been demonstrated in normal human skin keratinocytes, using reverse transcription–polymerase chain reaction, immunohistochemistry, and catalytic assays. The main enzymes are CYP1A1, CYP1B1, CYP2B6, CYP2E1, and CYP3A. 51 Among 26 CYPs mRNA levels (not involved in the steroidogenic pathway) measured by Luu-The et al., 43 the two highest expression levels were observed for CYP4B1 and CYP26B1. However, using proteomic profiling to measure metabolic enzyme expression, Van Eijl et al. 42 concluded that with levels at least 300-fold lower than those of the liver, the rate of oxidative biotransformation of a xenobiotic catalyzed by any CYP enzyme in the skin will be very low. These low activities at a basal level are expected to limit the contribution of CYP metabolic pathway to overall dermal metabolism. This finding is in accordance with Götz et al., 53 who reported a very low basal activity in whole human skin. However, it has been shown that the activity of the enzymes can be considerably induced. Gelardi et al. 54 have measured, in the human keratinocyte cell line NCTC 2544, the phase I activities 7-ethoxycoumarin O-deethylase (ECOD), 7-ethoxyresorufin O-deethylase (EROD), and 7-pentoxyresorufin O-depenthylase (PROD) after incubation with the cytochrome p450 inducers β-naphtoflavone, 3-methylcholanthrene, and phenobarbital. They observed a considerable increase in all the three activities. Thus, it is also essential to note that these enzymes, even though expressed at low basal level, are highly inducible.
According to their mRNA expression, other phase I enzymes were detected in the skin but at lower levels than in the liver: flavin-containing monooxygenases 1, 3, 4, and 5 in the epidermis, sebaceous glands, and hair follicles; alcohol dehydrogenases 1 and 3; and aldehyde dehydrogenases 1 and 3.43,55 Conversely, several enzymes are expressed at higher levels in the skin than in the liver: alcohol dehydrogenase 1, epoxide hydrolases 1 and 2, hydroxyacyl-coenzyme A dehydrogenase, and aldo-keto reductase 1C2 and 1C1. 38 In addition, it can be noted that the enzymatic activities of esterases (high activity) have also been reported in normal human skin. 56
Phase II enzymes
Concerning phase II enzymes, most studies have shown that they are present and highly active in the skin compared with phase I enzymes. A uridine 5′-diphospho-glucuronyltransferase (UGT) enzyme activity has been reported in human skin, with 50% of the UGT rate found in human liver microsomes.57,58 This activity measurement is in accordance with the one reported by Eilstein, which found also a very low sulfotransferase (SULT) activity (except for steroid sulfation) in normal human skin. 56
Glutathione S-transferase (GST) was studied by Götz et al., 57 who reported a noticeable total enzyme activity in human skin. Among the five GST isoforms, the pi isoform seems to be the more expressed one, 59 with a protein expression level two-fold higher than in the liver. 42 According to Reilly et al. 60 (measuring enzyme activity), the N-acetyltransferase 1 (NAT1) isoenzyme is expressed in the skin, but NAT2 is not. The protein activity of NAT family was also detected by Eilstein. 56 However, Van Eijl et al. 42 claimed that the proteomic approach they used failed to detect any NAT isoform in skin or liver. This is not in accordance with Eilstein et al., 56 but these authors reported that a high variability was observed for this activity in human skin. This could explain the differences observed between the studies. In Luu-The et al., 43 and measuring mRNA expression, NAT5 is the more expressed NAT, compared with NAT1 (low expression) and NAT2 (not detected). The mRNA expression of other phase II enzymes like sulfotransferases (SULT) genes (SULT1A1, 1A3, 1E1, and 2B1a/b) has been reported by Dooley et al. 47 and by Luu-The et al. 43 for SULT2B1b, 1A1, and at a low level, 1E1.
Inter-individual and intra-individual variations
It is important to note that inter-individual variations may account for some of the differences in relative expression or activity levels. For example, Janmohamed et al. 55 quantified several flavin-containing monooxygenase (FMO) and CYP mRNAs in human skin samples. FMO1 was found in 5 of 9 samples (with inter-individual variation of about 18-fold), FMO3 in 5 of 9 (with inter-individual variation of 2.5-fold), FMO5 in 7 out of 8 (with inter-individual variation of 5.6-fold), CYP2A6 in 5 of 9 (with inter-individual variation of 27-fold), and CYP3A4 in 3 of 7 (with inter-individual variation of 4-fold). 55
Moreover, the different enzymes detected may be localized in different cell types and skin layers. Lipase, protease, phosphatase, sulfatase, and glycosidase activities have been identified in stratum corneum, while alcohol dehydrogenase and aldehyde dehydrogenase have also been shown to be present in epidermal basal layers. 17 In general, higher amounts of enzymes have been detected in keratinocytes in comparison with lymphocytes, monocytes and fibroblasts (e.g., CYP1A1, CYP2B6, CYP2E1, and CYP3A). 51 As shown by Luu-The et al., 43 mRNA of some metabolizing enzymes are more selectively expressed in the dermis (e.g., CYP4F8, CYP27A1, CYP46A1, CYP21A2, ADH1B, EPHX1, FMO3, SULT1A1, and GSTM5), whereas others are more expressed in the epidermis (e.g., CYP39A1, CYP1A1, CYP2C18, CYP3A5, EPHX2, and SULT2B1b).
In addition to its influence on the absorption of applied compounds, skin metabolism can affect local and/or systemic toxicity in three different ways: activation of inert compounds (e.g., benzo[a]pyrene), detoxification of active compounds (e.g., paraoxon), or conversion of active chemical to active metabolites (e.g., estradiol). Thus, skin metabolism should be considered as part of the percutaneous absorption process. 49
One element for future consideration about skin metabolism could be the presence of skin bacteria on the skin surface. Recently it has been shown that azo dyes or benzo[a]pyene can be transformed by skin bacteria. That could lead to prolonged skin exposure to any intermediates products or metabolites. The metabolic capacities of skin microbiota could have an impact on the risk assessment process and should be considered in the future.61,62
In Vitro Models for Dermal Absorption
Guidance documents
Several documents are available for dermal absorption studies and all of them encourage a harmonized approach to the conduct these studies. In order to facilitate this harmonization, in 2011 the Organisation for Economic Cooperation and Development (OECD) published the Guidance Notes on dermal absorption (No. 156). 63 This publication was intended to complement other sources from other organizations, among them the OECD Test Guidelines 427 (in vivo methods) 8 and 428 (in vitro methods), 9 the OECD Guidance Document 28 for the conduct of skin absorption studies, 64 the World Health Organization International Programme on Chemical Safety (WHO/IPCS) Environmental Health Criteria 235, Dermal Absorption, 17 and different guidance documents from Europe65,66 and the United States. 67 Documents by others are not cited in these OECD Guidance Notes, such as the European Centre for Ecotoxicology and Toxicology of Chemicals (ECETOC) Monograph 20, 68 the United States Environmental Protection Agency (USEPA) report on dermal exposure assessment 69 and the European Food Safety Agency (EFSA) Guidance on dermal absorption for plant protection products (PPR). 70
Some of these documents are general and describe the different methods available for dermal absorption in vivo and in vitro: the OECD Guidance Notes 156 on dermal absorption, 63 the OECD Guidance Document 28 for the conduct of skin absorption studies, 64 the WHO/IPCS document on dermal absorption, 17 the European Commission Guidance Document on dermal absorption, 65 and ECETOC Monograph 20. 68 Some of the documents present or allow only in vivo approaches (OECD Test Guideline 4278 and EPA guideline on dermal penetration 67 ) or in vitro approaches (OECD Test Guideline 4289 and the Scientific Committee on Consumer Safety (SCCS) Basic Criteria for in vitro assessment of the dermal absorption of cosmetics ingredients 66 ). The EFSA guidance on dermal absorption for PPR describes tier approaches based on in vitro and in vivo assays. 70 The EPA document is more focused on the different EPA approaches for dermal exposure assessment. 69
In most of these documents, criteria are described to perform in vitro dermal absorption assays. For five of them (OECD TG 428, ECETOC Monograph 20, EFSA PPR, WHO/IPCS, and SCCS), the main criteria are reported in Table 2.
“—” means no information available.
ECETOC, European Centre for Ecotoxicology and Toxicology of Chemicals; EFSA, European Food Safety Authority; OECD TG, Organisation for Economic Co-operation and Development Test Guideline; SCCS, Scientific Committee on Consumer Safety; WHO, World Health Organization.
Mammalian skin
For in vitro methods, skin from human donors is the gold standard, since excised skin retains its barrier properties. The skin comes from different sources such as plastic surgery, amputation, or cadaver, 71 and the skin samples should be breast or abdominal skin. 72 In addition to ethical questions, two main limitations are known for the use of human skin samples: the availability and the variability. Variability can arise between the specimens taken from the same or different anatomical sites of the same donor but are increased between the specimens from different subjects or different age groups.17,73 To overcome these drawbacks, alternatives to human skin are needed. In the OECD Test Guidelines 428, 9 it is indicated that animal skin could be used to assess dermal absorption. Pig skin (especially pig ears or skin from the flanks) has been shown to be a suitable surrogate for human skin, since its permeability is similar. It is the recommended model for cosmetics, for example. Rat skin is 2 to 10 times more permeable than human skin and, therefore, is not recommended.17,66 However, rat is often used, as this species is the model of the majority of regulatory toxicity studies. Furthermore, even though viable skin is preferred, since skin penetration occurs mainly by passive diffusion, it is possible to use non-viable (e.g., cadaver) skin, provided that its integrity can be demonstrated. However, this is not the case if there is a need to study or account for metabolism.9,66
Reconstructed skin models
Some recent developments have been based on reconstructed skin models, such as reconstructed human epidermis (RhE; stratum corneum and viable epidermis) and full-thickness skin models (stratum corneum, viable epidermis, and dermis equivalent). Compared with the use of excised skin, these methods offer the advantage of better quality control of the test system, and in the case of models derived from human keratinocytes, improved species relevance. RhE includes models such as Episkin™, Epiderm™, SkinEthic™, EST-1000, and StratiCell® EPI/001 or Mel/001. Full-thickness models include models such as Phenion™, EpidermFT™, AST-2000, and StrataTest®. One of the current drawbacks of reconstructed human skin models is that these systems generally have a considerably weaker barrier function compared with human skin, 74 which complicates the accurate estimation of skin absorption/penetration. Permeability 10-times superior for RhE and 3–4 times superior for full-thickness models to human cadaver control skin for caffeine, tamoxifen, and hydrocortisone, were measured. 75 A difference in lipid structure was shown by Tfayli et al. 76 between human skin and the Epiderm™ model. These authors showed that in this RhE model, lipids are present either in droplets or in separate zones and concluded that this could be a reason for the reported higher permeability of skin models. 76 Recently, human tissue-engineered skin trilayers were developed and proposed to be used as model for dermal absorption studies. 77
Some reconstructed human skin models have already been validated for skin irritation and corrosion (i.e., Episkin and EpiDerm). 78 However, validation studies are still required to use them for dermal absorption studies.
The majority of these skin equivalents are based on static culture systems, missing or limiting some physiological properties important for toxicity testing. Methods in bioengineering are now used for the implementation of adipose tissue, hair follicles, and functional vascular network. In a recent paper, the use of a multi-organ-chip bioreactor platform was reported to improve the spatiotemporal control of the cellular microenvironment compared to traditional in vitro assays. 79
Artificial membranes
Another type of in vitro permeability method, suitable for assessing passive diffusion, is based on the use of artificial membrane barriers, including non-lipid-based models or lipid-based models. Among the non-lipid-based models, the silicone model membranes have been used for decades. In 2006, Ottaviani et al. 80 described a parallel artificial membrane permeability assay (PAMPA) constituted of a pure solvent membrane (composed of 70% silicone oil and 30% isopropyl myristate coated on a hydrophobic polyvinylidene fluoride filter). Recently, a poly(dimethylsiloxane)/poly(ethylene glycol) 6000 copolymer-impregnated membrane was synthesized. A good correlation was shown between its permeability and that of the human skin. 81 Another example is the Strat-M™ membrane composed of polyether sulfone, creating the morphology of the skin. It includes a very tight surface layer supported by a porous substructure bound to a nonwoven fabric support. 82
Among the lipid-based models, the modified skin PAMPA, an artificial membrane designed to mimic the human stratum corneum, contains a synthetic analogue of ceramide along with other major lipid matrix components (cholesterol and free fatty acids) coated onto polyvinylidene fluoride. 83 Another example, predating the PAMPA surrogates, is provided by silicone surrogates, which are membranes, composed of polydimethylsiloxane. 84 Artificial membrane barriers have also been developed by using liposomes. An example is the phospholipid vesicle-based permeability assay 85 , in which the lipid composition is designed to mimic that of the stratum corneum. This membrane includes ceramide, free fatty acids, cholesterol, and cholesterol sulphate.
These artificial membrane barriers do not require any human or animal tissue. Thus, they can be used for high throughput screening. However, they may have different permeability characteristics than skin, and their applicability domains may not be clearly defined. Further work is needed to characterize such methods and compare them with in vitro models.
Diffusion cells
For in vitro testing of skin penetration, the models described above are used in diffusion cells. Briefly, the test sample is applied to the surface of the skin model separating two chambers of the diffusion cells: the donor chamber and the receptor chamber containing the receptor fluid. The percutaneous absorption is then evaluated after sampling from the receptor chamber. Two types of diffusion cells are available: the static diffusion cell and the flow-through system. The essential difference is the continuously replaced receptor fluid in the flow-through test system. The flow-through system provides an environment more similar to real physiological conditions, since the continuous replacement of the receptor fluid mimics the systemic uptake of the chemical in the blood vessels. This presents an advantage when the chemical tested is poorly soluble in the receptor fluid (see below). Furthermore, it is easier to maintain skin conditions for longer time periods. However, the static cell has other advantages: it has a simpler design, lower cost, and a larger area of absorption, which provides for a more sensitive indication of absorption potential as well as a more accurate mass balance assessment. 86
Several parameters have to be taken into account for the use of the diffusion cells (Table 2). The major drawback is that measurement of some chemicals (i.e., very lipophilic compounds) can be difficult because of their low solubility in the receptor fluid. According to the OECD Test Guideline 428, 9 the receptor fluid must be compatible with the skin preparation and meet the key parameter of solubility of the test substance. The barrier integrity of skin must not be damaged. For non-viable skin, saline solutions with pH of 7.4 are recommended for water soluble test compounds. For lipophilic test substances, the receptor fluid can contain organic solvents (e.g., 1:1 ethanol:water or 6% polyethylene glycol 20 oleyl ether in water).64,68,87 For metabolically active skin preparations, a physiological-based receptor fluid (e.g., tissue culture medium) should be used. The potential inadequate solubility of the test substance in the receptor fluid should be addressed by the addition of a modifier to the physiological fluid, such as 6% polyethylene glycol 20 oleyl ether 88 or 5% bovine serum albumin. 89 In any case, it is imperative that solubility in the receptor fluid is not a rate-determining step in skin absorption. The type of vehicle used to expose the skin to the chemical can have an important effect depending on the relative affinity of the chemical for the vehicle in which it is applied. This is defined by the vehicle/stratum corneum partition coefficient: the more soluble the chemical in the vehicle, the more likely that it will be retained. If some components of the vehicle interact with the stratum corneum, the interaction could result in enhancement or retardation of permeation. 90 Ideally, the chemical should be applied in the same vehicle as used in the real exposure scenario. Concerning the chemical concentration, it is known that the flux increases in proportion to the concentration of the dissolved chemical. The non-solubilized part of the compound can act as a chemical reserve, maintaining a constant level for a prolonged time. 18 According to the OECD Test Guideline 428, testing a range of typical formulations should be considered. 9 The exposure time should reflect the in-use conditions. Temperature is another parameter to consider, as it can affect the passive diffusion of chemicals and, therefore, the skin absorption. A temperature close to the normal skin temperature (i.e., 32±1°C) should be maintained. 9
Concerning the use of RhE and artificial models, the tape-stripping step required for regulatory purposes is not applicable. This step is needed to calculate the amount of substance not absorbed due to desquamation of stratum corneum cells.
All of these parameters can lead to variability in the data obtained. Indeed, different study designs can be used due to the lack of standardization of the guidelines.
In silico methods
In vitro models are not the only models used to assess dermal penetration; in vitro and ex vivo tests are usually the sources/references from which quantitative structure–activity relationship (QSAR) models for skin penetration are derived. Since the 1990s, there has been a considerable effort to develop QSARs for skin penetration (sometimes referred to as quantitative structure-permeability relationships), as described in many review papers.91–95
Lipophilicity and molecular weight are among the most important factors determining the ability of compounds to permeate the skin. 96 In addition, the density, viscosity, and lipophilicity of vehicles have a huge impact on permeation rates, which is why much research in pharmacokinetics is focused on finding better vehicles for drug delivery. Most models use the quantity of test compound that passively diffuses through the skin of a donor as the measure of penetration (i.e., most models predict the permeation rate constant [Kp]). The simplest QSARs (e.g., Potts and Guy, 1992, 97 and Moss and Cronin, 2002 98 ) are simple linear regression models for Kp based on partitioning behavior (typically the octanol–water partition coefficient, logKow) and molecular size (expressed by molecular weight or volume). Other (multiparametric) models have incorporated additional descriptors, typically describing molecular shape, solubility, polarity, and hydrogen-bonding capacity. In addition to simple and multiple linear regression approaches, non-linear modelling has also been applied, including relatively simple approaches, such as parabolic models (e.g., Lien and Gao, 1995 99 ) and bilinear models (e.g., Zhang et al., 2009 100 ) to more complex ones, such as artificial neural networks (e.g., Fatemi and Malekzadeh, 1995 101 ). Most QSARs are published in the scientific literature, although some are implemented in freely available software, such as Episuite, or commercial tools, such as Schrodinger's QikProp. In general, available QSARs are based on the assumption that the intercellular lipid pathway is the main route of permeation and are applicable to hydrophobic compounds, with the skin permeability of hydrophilic compounds being underpredicted, typically by 2–6 orders of magnitude. 102
In addition to QSARs, classification models have also been developed, using, for example, Classification And Regression Tree (CART) methodology. 103 Simple rules of thumb to distinguish between “good” and “bad” penetrants on the basis of readily available physicochemical properties (e.g., molecular weight, melting point) have been proposed by Magnusson et al. (2004). 104
Another approach, although not widely explored, is membrane-interaction QSAR (MI-QSAR), in which some of the descriptors are derived from molecular dynamics simulations of the transport of organic compounds through a phospholipid monolayer or bilayer. This approach is not tailored for routine screening purposes, as it implies demanding computations, but it could be useful for elucidating mechanisms of permeation. For example, it has been used to illustrate how penetration enhancers change the fluidity (disorganization) of lipid membranes. 105
Some models 106 take a closer look at the skin structure107,108 and determine skin permeation as function of the stratum corneum permeation coefficient (KSC). The KSC is extrapolated to Kp using “effective” diffusivity and “effective” path lengths, which correspond to the diffusivity and thickness of an equivalent homogenous membrane having the same properties as the stratum corneum.
A limitation of most available QSARs is that they assume infinite dose conditions, whereas in reality, dermal exposure mostly occurs under finite dose conditions. A simple model to predict dermal absorption under finite dose conditions was developed by Buist et al. (2010). 109 In addition, Kasting and coworkers have developed a finite-dose model 110 that is implemented in a freely available web-based program (www.cdc.gov/niosh/topics/skin/finiteSkinPermCalc.html). A few studies have attempted to develop models for multicomponent mixtures (e.g., Ghafourian et al., 2010, 111 and Riviere and Brooks, 2011 112 ).
In Vitro Models for Skin Metabolism
The formation of reactive chemicals is an important mechanism in systemic toxicity, and as described above, the skin is an important site of extra-hepatic metabolism, including phases I and II enzymes. Even if the skin metabolism is not the main first-pass effect involved in systemic toxicity, it could still play a role in local toxicity (sensitization, irritation). In vitro models for dermatotoxicity have thus been studied for their metabolic capacities. These systems are mainly based on human excised skin, human cell lines, and human in vitro skin models.
To study dermal metabolism, human skin is the ideal system. Critical variables are mainly the inter-individual differences in skin permeability and the source of the skin (e.g., sex, age of donor, body site, skin thickness). 113 Moreover, there are a number of legal and ethical issues. To evaluate metabolism, it is especially important to use viable skin. Even if it is possible to maintain metabolic activity in an in vitro model for experimentally useful periods, 114 some enzymes (e.g., alcohol dehydrogenase/aldehyde dehydrogenase [ADH/ALDH] 114 ) and cofactors (e.g., glutathione 115 ) deteriorate quickly ex vivo even in fresh specimens.17,116
The metabolic capacity of in vitro models has been shown in different studies measuring the mRNA level or the protein level expression and activity. Even if the measurement of mRNA expression is not the most accurate method compared to protein level expression and activity, it has been shown that a correlation between mRNA levels and the physiological functions and corresponding genes is generally observed. 43
Primary cells and cell lines
Among all models, primary cell keratinocytes represent the most common skin cells used to assess skin metabolism of drugs and chemicals. 117 Hirel et al. 118 concluded that the normal human epidermal keratinocytes model is suitable for investigating the biotransformation of drugs and other chemicals, based on the analysis of the expression and induction of several drug metabolizing enzymes involved in either phase I or phase II reactions. However, to overcome certain limitations (donor dependencies, cell isolation, cost), human primary keratinocytes are often replaced by immortalized keratinocytes like HaCat or human monocytic cell line (THP-1). Concerning phase I metabolism, HaCaT cells have shown an expression pattern of cytochrome p450 isoenzymes close to normal cultured keratinocytes, but also expression of CYP2B6 mRNA, which is not found in the epidermis or in cultured normal keratinocytes (NE Fusenig, unpublished data, as cited in Van de Sandt et al., 2009 119 ). This is in contradiction with Janmohamed et al., 55 who reported mRNA expression of CYP2B6 in HaCat cells at an amount more similar to that in human skin compared with human keratinocytes. CYP3A basal enzyme activities were detected in HaCaT by Götz et al. 53 but not by Rolsted et al. 120 or by Janmohamed et al., 55 who failed to detect CYP3A4 mRNA. This discrepancy could be due to different HaCaT subclones. CYP1A1 activity could be significantly enhanced by 3-methylchloranthrene. 121 The expression of CYP2S1 122 and CYP4F11 123 mRNA has also been demonstrated. Concerning FMO, the amounts of mRNA of FMO3 and FMO5 were similar to those measured in human skin, and FMO4 was three-fold more than in human skin samples. FMO1 mRNA was not detected. 55 Concerning phase II enzymes, Götz et al. 57 have shown that HaCaT cells have the closest NAT enzyme activity to human skin with an expression approximately four-fold higher than in normal human epidermal keratinocytes. This is in agreement with the findings of Bonifas et al. 124 Moreover, it has been demonstrated that routine culture might result in significant changes in proliferation and phase II metabolism. 125
Concerning THP-1, the dendritic cell model for the h-Cell Line Activation Test (h-CLAT) for skin sensitization testing, 126 mRNA expression and activity (proteins) (after differentiation by phorbol 12-myristate 13-acetate) of cytochrome P4502J2 has been demonstrated. 127 In a more recent study, the cell line failed to detect pro-haptens without exogenously added Aroclor 1254-induced rat liver S9 homogenate. On this basis, the authors concluded that endogenous CYP activity in THP-1 cells is insufficient for pro-hapten screening. 128 In the study of Fabian et al. 129 it has been shown that CYP (investigating alkylresorufin O-dealkylase activities—EROD, PROD, and benzyloxyresorufin O-debenzylase [BROD]), FMO, and UGT1 basal protein activities are below the level of detection and that ADH and ALDH are below the level of quantification. Only esterase activity and observable and quantifiable NAT1 activity were highlighted.
Reconstructed human epidermis
The metabolic capacities of RhE (e.g., EpiSkin®, SkinEthic®, EPI-200, and Epiderm®) or full-thickness skin models have also been assessed. Most studies measuring activity42,53,57,130,131 or mRNA130–132 have concluded that RhE are suitable models for dermatotoxicity testing. For example, Jäck et al., 52 measuring enzyme activity, confirmed the xenobiotic metabolizing capacity of Epiderm™ and full-thickness skin model phenion®FT, supporting their use for toxicology testing.
Hu et al. 130 compared the expression of 139 genes encoding xenobiotic metabolising enzymes in the EpiDerm® model and human skin. Using microarray analysis, they determined that 87% of gene expressions were consistent between EpiDerm® model and human skin. On this basis, they concluded that similar metabolic pathways are present in the two systems.
Comparing EPI-200 with monolayer cultures, Götz et al.53,57 measured phase I and II enzyme activities and concluded that EPI-200 is superior in mimicking human skin xenobiotic metabolism.
Similar mRNA expression levels of 61 phase I and II metabolizing enzymes were shown between EpiSkin™, a full-thickness model derived from EpiSkin™, and ex vivo epidermis. 43 The main discrepancies were CYP P450 and flavin monooxygenases expression, which were detected at low levels, while phase II enzymes were detected at much higher levels, especially GSTP1, COMT, SULT2B1b, and NAT. The authors concluded that EpiSkin™ and the full-thickness model derived from EpiSkin™ are good in vitro human skin models for studying the function of phase I and II metabolizing enzymes in xenobiotic metabolisms. 43 This is confirmed by the results of Neis et al., 132 which showed mRNA expression of CYP type 1A1, 1B1, 2E1, 2C, and 3A5 in organotypic skin models.
To avoid using biological models, Bergstrom et al. 133 developed a skin-like P450 cocktail to activate pro-haptens in studies of cutaneous CYP-mediated metabolism of contact allergens. After measurement of skin content of 18 human skin samples, they developed an enriched skin-like recombinant human CYP cocktail using CYP1A1, 1B1, 2B6, 2E1, and 3A5. They demonstrated the usefulness of this method by using a pro-hapten, (5R)-5-isopropenyl-2-methyl-1-methylene-2-cyclohexene.
In silico models
In vitro models are not the only way to predict skin metabolism. Several software tools include simulators for the prediction of skin metabolism [e.g., the OECD QSAR Toolbox, TIMES-SS (LMC-Bourgas), Meteor Nexus (Lhasa Ltd.), Percepta (ACD/Labs), ADMET Predictor (Simulation Plus, Inc.) MetaSite (Molecular Discovery Ltd.), and StarDrop (Optibrium)].
The OECD QSAR Toolbox, developed by the OECDs, was made freely available to promote its use by industry, governments, and other stakeholders. The Toolbox is especially designed for chemical grouping and read-across. Toxicity predictors are available for several endpoints including acute toxicity, carcinogenicity, mutagenicity, eye irritation/corrosion, protein binding, skin irritation/corrosion, and skin sensitization. The software includes different metabolic and transformation simulators, including a skin metabolism simulator.
The simulator was donated by Laboratory of Mathematical Chemistry (LMC)-Bourgas and is basically a simplified version of the one found in TIMES-SS. It uses a hierarchical list of spontaneous, enzyme-mediated, and protein-binding reactions that the query compounds can undergo. These reactions were defined on the basis of empirical and theoretical knowledge and are used in an iterative process—parent compound and subsequent generations of metabolites are considered—to determine the formation of metabolites. Probabilities of formation are determined on the basis of structural alerts for the query compound as well as on the neighboring groups. The iterative process continues until a constraint for propagation is found (i.e., until the metabolite is found not to be reactive, to undergo a phase II reaction, to react with a protein, or to be transformed into a hydrophilic derivative. When the iterative process is finished, a set of metabolites and their corresponding metabolic pathways with associated probabilities is obtained. The Toolbox outputs the list of metabolites, which can later be profiled for toxicologically relevant properties, such as protein binding alerts to determine their skin sensitization potential. TIMES-SS is a commercial software package developed by LMC that offers toxicity predictors for several endpoints as well as some metabolism simulators. 134 In contrast to the OECD QSAR Toolbox, TIMES-SS uses some quantum mechanics descriptors such as HOMO–LUMO gaps and electronegativity, which are calculated at the semi-empirical level with the embedded MOPAC software. The skin simulator also includes extra features with respect to the one on the OECD QSAR Toolbox that make it a more powerful tool and the data output is more verbose. For instance, the metabolism pathway with their associated probabilities and reliabilities can be explored, the training sets used to derive the transformations can also be explored, and an estimation of protein-adduct formation is provided.
Meteor Nexus is a commercial software package developed by the not-for-profit organization Lhasa Limited. It can be used alongside the Derek Nexus software for toxicity prediction. Meteor Nexus is not a specific simulator for skin metabolism, but rather, is a full metabolism/biotransformation simulator. It is a knowledge-based system that uses a database of possible biotransformations to determine the metabolic fate of the query compound. 135 The user can select from a list of possible biotransformations the ones to be used in the prediction. The transformations are separated into phase I and phase II groups and subcategorized in chemically based groups like redox, epoxidation, decarboxylation, glucuronidation, acetylation, etc. Another option is to select an enzyme from a drop-down list, which basically corresponds to predefined combinations of the above-mentioned biotransformations. There are no predefined combinations of reactions accounting for just skin metabolism but the user can define the set of transformations to be applied. When a query compound is submitted, it is processed by the transformations database and a list of first-order metabolites is generated. The list of metabolites is based on lipophilicity, presence or absence of structural features that modulate the reactions, and the likelihood of competing reactions. 136 The first-order metabolites are subsequently processed for further metabolism. The results are graphically displayed as an interactive tree with associated probability measures. Each metabolic path can be neatly explored and links to supporting information on the biotransformations are provided.
Percepta is a commercial software package developed by ACD/Labs. It is a platform mainly tailored for industry to design and synthetize new chemicals with desired properties. The platform allows the prediction of physicochemical, absorption, distribution, metabolism, excretion, and toxicity properties. The metabolism model was built from experimental data from scientific publications only and is specific not for skin metabolism, but for CYP.137,138 It calculates how compounds will interact with the five cytochrome P450 (CYP) isoforms: 3A4, 2D6, 2C9, 2C19, and 1A2 that are responsible for the majority of phase I metabolic reactions. The model consists of three modules: (1) P450 substrates estimates if a compound is metabolized by a particular CYP isoform; (2) P450 inhibitors estimates if a compound is a CYP inhibitor or an efficient CYP inhibitor; and (3) P450 regioselectivity predicts sites of metabolism by human liver microsomes. The prediction of the model is based on a predefined set of fragmental descriptors that is modified by a subset of similar compounds found on the training set or provided by the user.
MetaSite (Molecular Discovery) is another CYP-based metabolism simulator that predicts metabolic transformations related to cytochrome and flavin-containing monooxygenase mediated reactions in phase I metabolism.139,140 The model is an automated docking system with reactivity correction (i.e., reactivity of the different atomic parts toward the heme are used to determine the preferred sites of attack). The software predicts the preferred sites of attack of the query compound by the enzyme, and provides the user sets of modifications of the query compound to reduce the CYP mediated metabolism. In addition, a list of most likely metabolites is provided.
StarDrop is a commercial platform developed by Optibrium tailored for drug discovery. It includes a metabolism module, namely P450, that predicts the relative proportion of metabolite formation at different sites on a molecule by different CYP 450 enzymes. 141 The model uses an adapted version of the Austin Method 1 (AM1) 142 to calculate (MOPAC97) the activation energies of possible oxidation reactions (i.e., the limiting step in product formation) and the orientation of the query compound in the active site to correct the estimated liability.
Conclusions
In this paper, we describe the biology of dermal penetration and metabolism, and review the availability of in vitro and in silico methods for assessing these processes, which are important determinants of dermal bioavailability.
Following dermal application, chemicals can remain in the skin. Since the different reservoirs (stratum corneum, epidermis, dermis) could impact the bioavailability, they should be taken into account in the exposure assessment of dermally applied chemicals, along with the likelihood and extent of metabolism, which also affects the kinetics of absorption (transforming permeable substances into impermeable ones) and the nature of systemically available chemicals. Reservoir effect can complicate the assessment of diffusive transfer, as chemicals remain in the different layers of the skin. Different organizations may take different approaches for the determination of in vitro dermal absorption of the chemicals. For the ECETOC, the measurement is based only on receptor fluids. 68 According to the OECD Test Guideline 428, the analysis of receptor fluid and of the distribution of the test chemical in the test system has to be performed. Skin absorption may sometimes be expressed using receptor fluid data only, but if the chemical remains in the skin at the end of the study, it may need to be included in the total amount absorbed. 9 For SCCS, the possible retention of the test chemical in the viable epidermis and in the dermis must be taken into account for the results interpretation. 66 For measurement on the receptor fluid only, all other parameters should be carefully controlled, as for example, the nature of the receptor fluid and solubility of the chemical in the receptor fluid, as it must not limit the penetration.
It is clear that the first-pass metabolic effect of the skin is much less extensive than liver metabolism at a basal level, even though some enzymes can be induced. The major difference seems to be the phase I enzyme activities. However, it is important to bear in mind that the area of skin exposed and time of exposure are also important. The skin is the largest organ for humans. Thus, even with weak metabolic capacities, the surface exposed can affect the importance of the chemical metabolism. Furthermore, metabolism can modify the physicochemical properties of the chemicals and, thus, may affect the cutaneous absorption of the considered compound.
Both dermal penetration and metabolism are complex, interacting processes. Currently available in vitro models are suitable for the screening and prioritization of chemicals based on expected dermal penetration and also to estimate skin penetration for humans, but validation studies are still required. The main question concerning the validation of skin penetration models concerns the reference data. As a result of different kinds of experimental setup (e.g., use of radiolabeled compounds), analytical work, and protocol use, it is often difficult to compare and aggregate these data. Thus, further standardization of the dermal absorption studies is needed to generate data suitable for use in the validation process. A wide variety of in vitro methods expressing key metabolic pathways can be used for more qualitative determinations of metabolic fate even if the quantification is more difficult. In vitro models for assessing both the skin metabolism and dermatotoxicity of chemicals include immortalized cell lines (e.g., HaCat cells) and reconstructed human epidermis (e.g., EpiSkin®, SkinEthic®). Epidermal models mimic whole skin more closely, although monolayer cultures may also be useful for metabolism studies, depending on the expected metabolic fate of the compound of interest (for example, as predicted by in silico methods).
In addition to the in vitro approaches, in silico or computational models are being developed: QSAR, classification models, membrane-Interaction QSAR (skin penetration), and simulators for the prediction of skin metabolism.
Another alternative in vitro approach, which avoids differences observed between cell lines and epidermis models, is to use enzyme cocktails that mimic whole skin metabolism. This merits further development.
Footnotes
Acknowledgments
This review was partly carried out under the terms of a contract between Cosmetics Europe and the European Commission's Joint Research Centre (No. 32485-COLIPA-2011-T1CD ISP).
Author Disclosure Statement
No competing financial interests exist.
