Abstract
Identification of kinase, especially protein kinase, modulators through high-throughput screening (HTS) has become a common strategy for drug discovery programs in both academia and the pharmaceutical industry. There are a number of platform technologies that can be used for measuring kinase activities. However, there is none that fits all criteria in terms of sensitivity, ATP tolerance, robustness, throughput, and cost-effectiveness. Therefore, development of a homogeneous and robust HTS assay for some kinase targets is still challenging. We recently evaluated the ADP-Glo assay from Promega. This is a homogeneous, signal increase assay that measures ADP production from a kinase reaction by coupled enzymes that first convert ADP to ATP and subsequently quantifies ATP using luciferase in the presence of luciferin. Since the unused ATP in the reaction is depleted prior to ADP to ATP conversion, this assay shows excellent sensitivity over a wide range of ATP concentrations. We demonstrate that ADP-Glo™ assay can be used for 2 kinase targets that belong to different classes, and compare the results of compound profiling with SPA and FP assay technologies.
INTRODUCTION
Protein kinases play a pivotal role in all aspects of cellular physiology such as growth, differentiation, and metabolism. Their involvement in pathological conditions such as cancer, inflammatory diseases, neuronal disorders, and metabolic disorders make kinases important targets for drug discovery.1 , 2
Since the discovery of Gleevec for the treatment of chronic myelogenous leukemia in 2001,3 , 4 several small molecular kinase drugs as well as protein therapeutics have been approved.5 These early successes have ignited interest in identifying kinase inhibitors in various disease areas, particularly in oncology.2 , 6 , 7 As a result, identification of protein kinase modulators through high-throughput screening (HTS) has become a common strategy for kinase drug discovery in both academia and the pharmaceutical industry.
To meet this increasing demand, many companies have commercialized biochemical assays that are readily available to users. In general, kinase assays can be roughly categorized into 3 groups based on their detection modality: Group I: Detection of phosphopeptide; Group II: Measurement of ATP consumption; Group III: Quantification of ADP production. In the past few years, more attention has been given to the measurement of either ATP consumption or ADP accumulation as generic approaches for kinases. Compared with phosphopeptide detection in Group I, assays in Groups II and III are applicable to all kinases including tyrosine and serine/threonine kinases. In Group II, Kinase-Glo™ has been widely used for HTS campaigns owing to its homogeneity and antibody-free nature among others.8 , 9 However, this assay measures the remaining ATP in a kinase reaction and a significant consumption of ATP (at least 20%) is required to achieve a statistically robust signal. Therefore, higher enzyme concentration may have to be used, which would result in higher cost and lower sensitivity of this assay. In addition, this assay cannot tolerate high ATP concentration, which makes it difficult to perform compound mechanistic studies.10 In Group III, ADP generated from a kinase reaction is detected by either ADP-specific antibody11 or coupled enzyme systems.12 The former is reported as Transcreener™ (Bellbrook Labs, Madison, WI), which has 2 readouts: FP and TR-FRET. The principle of these 2 formats is that the ADP produced from a kinase reaction competes with tracer-ADP, which causes assay signal decrease. In the latter, ADP-Quest™ (DiscoverX, Fremont, CA) measures ADP produced from a kinase reaction by a coupled enzyme system.12 The advantages of these ADP detection assays are that they are universal, and are potentially applicable to most ADP production reactions. The ratiometric nature of either FP or TR-FRET formats of Transcreener™ is not sensitive of assay volume, making this assay highly miniaturizable without loosing sensitivity or robustness. However, the major drawbacks include nonlinear response to product ADP and low range of usable ATP concentrations due to antibody’s cross-activity with ATP. Thus, an ATP–ADP standard curve is required to quantify how much ADP is produced. For ADP-Quest™ assay, the nature of assay signal increase and homogeneous format make it easily adapted to automated HTS platforms. However, low tolerance of high ATP concentration limits its use for compound mechanistic studies.13
In spite of the large number of available assays, there is still a need for an universal assay that is ideal for both HTS and MOA studies. For an HTS assay, the robustness, throughput, and cost per well are top priorities when choosing a preferred format. However, for a MOA assay, sensitivity and broad range of ATP concentrations are top concerns. In addition, assay development for kinases with weak activity is still a challenge.
To address all of these needs, Said Goueli and colleagues (see previous article in this issue) have developed the ADP-Glo™ assay (Fig. 1). The assay contains 2 simple “mix-and-read” cocktails. In the first step, Reagent I is added, which contains an enzyme to quench the target kinase by removing unrelated ATP. In the second step, Reagent II is added, which contains a mixture of an enzyme that converts ADP produced as a product in the primary kinase reaction back to ATP, and luciferase that converts ATP to AMP in a 2-step reaction that results in luminescence, which is proportional to the amount of ATP consumed. Reagent II also contains an inhibitor to the enzyme in Reagent I preventing it from degrading newly converted ATP from ADP.
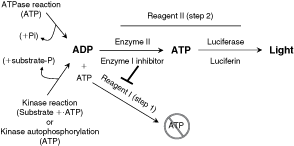
The assay principle of ADP-Glo™ technology. At the end of the primary reaction (such as kinase and ATPase), the remaining ATP is depleted by adding Reagent I; after that the primary reaction product ADP is converted into ATP by adding Reagent II, which also contains a Reagent I inhibitor. The so-produced ATP is then quantified by the luciferase/luciferin reaction.
In this article, we evaluate ADP-Glo assay by presenting assay development and profiling data for 2 kinase drug targets as examples, one was a protein tyrosine kinase (referred as Kinase I hereafter) and the other was a small molecule kinase that is complicated in metabolic disorders (referred as Kinase II hereafter). The main focus of our evaluation is to determine the sensitivity of ADP-Glo assay, the dynamic range of ATP concentrations that can be used, and to compare the generality of this assay for protein and nonprotein kinase targets.
MATERIALS AND METHODS
Reagents and devices
Kinase I was expressed and purified in-house, aliquoted, and stored at −80°C in 50 mM Tris, pH 8.0, 150 mM NaCl, 0.1 mM TCEP, and 10% glycerol. Kinase II was expressed and purified in-house, aliquoted, and stored at −80°C in 25 mM phosphate, pH 8.0, and 500 mM NaCl. For the ADP-Glo assay, the reagents were supplied by Promega (pre-marketing product, Madison, WI). For the fluorescence polarization assay of Kinase I, the fluorescent ligand 5-({[2-({[3-({4-[(5-hydroxy-2-methylphenyl)amino]-2-pyrimidinyl}amino)phenyl]carbonyl}amino)ethyl]amino}carbonyl)-2-(6-hydroxy-3-oxo-3H-xanthen-9-yl)benzoic acid (referred as FP ligand hereafter) was synthesized in-house. For the scintillation proximity assay of Kinase I, biotinylated antibody for recognizing phosphorylated Kinase I was made in-house, [γ-33P]-ATP was purchased from PerkinElmer (Product No.: NEG302H001MC), and streptavidin-coupled polystyrene imaging beads were purchased from GE Healthcare (Cat. No.: RPNQ0261). All other general chemicals were purchased from Sigma-Aldrich (St. Louis, MO) and were of analytical grade unless otherwise specified.
All measurements were performed on a Wallac ViewLux™ µHTS microplate imager manufactured by PerkinElmer except the FP readout of Kinase I, which was measured on an Analyst GT multimode reader (Molecular Devices, Sunnyvale, CA). Compounds were stamped using Hummingbird™ dispenser (Genomic Solutions, Ann Arbor, MI). Assay liquid was dispensed into 384- or 1,536-well plate (Greiner Bio-one, Monroe, NC) with either Multidrop Combi™ (Thermo Scientific, Waltham, MA) or CyBi®-Well (CyBio, Jena, Germany) dispenser.
ADP-Glo Assay for Kinase I
Compound profiling for Kinase I using ADP-Glo was comprised of 3 main steps: the primary enzyme reaction, depletion of remaining ATP, and conversion of ADP product into ATP for luminescence detection.
The primary enzyme reaction was carried out as follows: 50 nL of inhibitors in DMSO (or DMSO controls) were stamped into 384 white, low-volume plates (Greiner, Catalog # 784075). The 2.5 µL of assay buffer was dispensed, as negative control, into column 18 of the plate using Multidrop Combi™, followed by the addition of 2.5 µL of 2× enzyme (final concentration 10 nM) mix to the remaining columns of the plate. The plate was then spun at 500 rpm for 30 s and incubated at room temperature for 30 min. After that, 2.5 µL of 2× ATP (final concentration 10 µM) was added to entire plate and the plate was then sealed and spun at 500 rpm for 30 s and incubated at room temperature for 6 h.
At the end of incubation, the reaction was stopped by adding 5 µL of ADP-Glo detection Reagent I and the plates were then spun at 500 rpm for 30 s and incubated at room temperature for 1 h to allow complete ATP depletion. And then 10 µL of ADP-Glo detection Reagent II was added and plate was covered (ADP-Glo detection Reagent II is light-sensitive) and spun at 500 rpm for 30 s. The plate was then incubated at room temperature for 1 h. The luminescence was read on ViewLux™ using the following condition: clear filter, measurement time 5 s, speed slow, gain low, and binning 4 times.
For determination of the ATP K m values of Kinase I using ADP-Glo, 30 nM Kinase I enzyme was used for a 2 h reaction at varying ATP concentrations. At each ATP concentration, a control containing no Kinase I was included to calculate the assay background. At the end of the reaction, the remaining ATP was depleted by adding Reagent I for 1 h and the ADP product was converted into ATP for signal readout by adding Reagent II for another hour. The rates of Kinase I reaction were calculated as the difference between the enzyme reactions and the corresponding no enzyme controls.
Fluorescence Polarization (FP) Assay for Kinase I
This assay measures the displacement of the FP ligand from the active site of Kinase I. The protocol is summarized as below. First, 2 solutions were made, solution 1 was 5 nM FP ligand in 20 mM Tris, pH 7.5, 0.5 mM DTT, 15 mM MgCl2, and 0.08% (w/v) CHAPS, which was used as control; and solution 2 was a mixture of 5 nM FP ligand and 20 nM Kinase I in the same buffer. Then, 5 µL of solution 1 (the FP ligand alone) was added into column 18 of a 384-well low-volume Greiner black plate prestamped with 50 nL of inhibitors in DMSO and 5 µL of solution 2 (Kinase I and the FP ligand) to columns 1–17 and 19–24. Plate was then spun at 500 rpm for 30 s and incubated at room temperature for 60 min. The FP signal was then measured on Analyst GT using the following settings: lamp continuous, excitation filter 485–25 nm, emission filter 530–25 nm, dichroic mirror 505 nm, target SD 1 mP, attenuator out, Z-height middle of well, integration time 100,000 µs, and raw data RFU.
Scintillation Proximity Assay (SPA) for Kinase I
This SPA assay was configured to measure the autophosphorylation of Kinase I. In brief, 5 µL of Kinase I (final concentration 10 nM) was added into a 384-well Greiner flat bottom, polypropylene white plate with prestamped compounds; the plate was then spun for 10 s at 1,000 rpm and incubated at room temperature for 30 min. The 5 µL [γ-33P]-ATP (final concentration 10 nM) was then added and Kinase I autophosphorylation was carried out at room temperature for 7 h. The reaction was then quenched by adding 20 µL of 30 mM EDTA, followed by an addition of 60 µL of streptavidin-coupled polystyrene imaging beads pretreated (preincubated for 90 min) with biotinylated anti-phospho-antibody. The plate was then sealed and sat for overnight at room temperature before being read on ViewLux™ (60 s SPA with dual exposures (613 nm filter), cosmic ray detection, speed slow, gain high, and binning 4 times).
ADP-Glo Assay for Kinase II
The ADP-Glo assay for Kinase II was run in a similar fashion as that for Kinase I. In brief, 2.5 µL of 2× enzyme solution (final enzyme concentration was 1 nM in 100 mM Hepes, pH 7.2, 100 mM KCl, 10 mM MgCl2, 0.05% (w/v) CHAPS, 0.05 mg/mL BSA, 10 µM DTT) was dispensed into assay plate with inhibitors prestamped, followed by addition of 2.5 µL of 2× substrate solution that contains both ATP (to make a final concentration of 1 mM) and Kinase II substrate (final concentration of 0.2 mM). The reaction was then incubated at room temperature for 30 min. After that 5 µL ADP-Glo™ Reagent I (prepared by adding 10 µL of Enzyme I into 1 mL Buffer I) was added into the assay wells to stop the reaction and deplete unconsumed ATP for 60 min at room temperature; then, 10 µL of Reagent II (prepared by adding 10 µL of Enzyme II into 1 mL Buffer II) was added into the assay wells, followed by 60 min incubation in dark to develop luminescence. The luminescence was then read on ViewLux™ using the following settings: clear filter, measurement time 5 s, speed slow, gain low, and binning 4 times.
Data Analysis
For dose–response analysis, normalized data were fit by ActivityBase™ (IDBS) using the equation y = a + (b−a)/(1+(10ˆx/10ˆc)ˆd), where a is the minimum % activity, b is the maximum % activity, c is the pIC50, and d is the Hill slope. Assay comparison was analyzed using Spotfire DecisionSite (TIBCO, Sommerville, MA).
The ATP K m of Kinase I autophosphorylation was calculated using Michaelis–Menten equation v = V max[S]/(K m + [S]), where v is the enzyme reaction rate, V max is the calculated maximal reaction rate at saturation concentration of substrate, [S] is the substrate concentration, and K m is the substrate concentration that produces a reaction rate that is half of the maximal reaction rate. The reaction rate v was taken as the difference of that between Kinase I reaction and the corresponding no enzyme control as stated in the Materials and Methods section.
RESULTS
Assay Development
One of the purported advantages of ADP-Glo assay format is the broad range of ATP concentrations that are tolerated. To evaluate this, an ATP–ADP standard curve was performed at different concentrations ranging from low micromolar up to 1 mM. As shown in Figure 2, the assay signal is linear to percentage conversion of ATP to ADP at 1 mM (Fig. 2A) and 10 µM (Fig. 2B), respectively. At 1 mM ATP, about 5% conversion is required to achieve 3:1 signal-to-background ratio, which typically yields a robust assay. However, at 10 µM ATP, 2% conversion is sufficient to generate a S/B ratio of 3:1. Comparing these 2 conditions, the assay is more sensitive at lower ATP concentration. The reason is that commercial ATP is typically contaminated by ∼1% ADP. At high ATP concentration, the effect of the contaminated ADP is more pronounced than at lower ATP concentration, which resulted in a relatively smaller assay window. Using 10 µM ATP as an example in a 10 µL assay, the assay was able to reliably detect 0.2 pmoles ATP conversion to ADP.
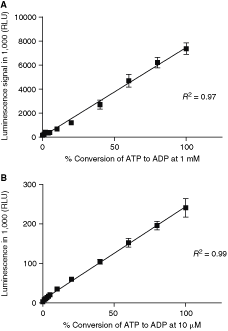
The standard curves of ADP-Glo™ signal vs. ATP to ADP conversion at fixed total ATP and ADP concentration of 1 mM (
DMSO Effect
Compound libraries are typically made in dimethyl sulfoxide (DMSO). We, therefore, performed DMSO tolerance experiment for Kinases I and II and found that both assays can tolerate up to 5% DMSO (data not shown), which is higher than the DMSO content of typical HTS conditions and therefore the presence of DMSO in compound libraries is not a concern of ADP-Glo assay.
Z′ Factor Determination
The Z′ factor is a commonly used parameter in determining the robustness of an assay.14 It is an indication of how well the positive control can be reliably separated from the negative control and its value ranges from 0 to 1, the higher the number, the greater the separation between the positive and negative control. In drug discovery, single dose is often performed during primary screening and therefore, it is critical to be able to distinguish “hits” from “noise,” which usually requires a Z′ > 0.4. The Z′ of ADP-Glo™ assay was assessed for Kinases I and II and in both assays, the Z′ > 0.4 (Fig. 3), which demonstrates that ADP-Glo assay is suitable for screening compounds at single concentration.
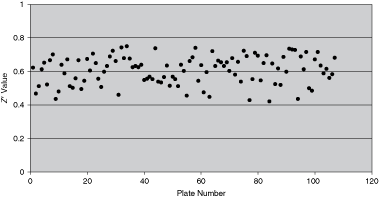
Z′ of ADP-Glo™ assay for Kinase II. The Z′ was calculated between the means and standard deviations of both the positive (uninhibited Kinase II reaction) and the negative (100% inhibited Kinase II reaction) controls using this equation from Zhang et al.14 Z = 1 − 3 × (™p + ™n)/(|µp − µn|), where ™p and ™n are the standard deviations of positive and negative controls, and µp and µn are the means of positive and negative controls.
Assay Signal Stability
In a HTS campaign, hundreds or thousands of plates are screened and the process is handled by automation. Usually, there is a delayed or waiting period between the reactions; after the reaction is completed the plate is read, which requires the assay signal to be stable over that period of time. This is especially true given the batch mode operation, where reactions were completed in assay plates and then the plates are stacked onto a reader. The signal stability of ADP-Glo assay was evaluated and it was stable for at least 2 h for both Kinases I and II assays, which allows enough flexibility to process hundreds of plates during the HTS process.
Steady-State Enzyme Kinetic Characterization
The success of a HTS campaign is determined in part by a sensitive and robust assay. Therefore, preliminary work leading to a desired screening assay condition is critical. For example, to run a balanced condition for a kinase assay, an ATP concentration equivalent to the K m of the kinase is usually employed. This would allow equal chance of identifying compounds with different modes of inhibition, such as competitive, noncompetitive,15 and uncompetitive.16 In order to generate this data, an assay that can easily characterize steady-state enzymatic kinetics is preferred. To fulfill this criterion, we performed ATP K m experiments using ADP-Glo assay for Kinases I and II. Figure 4 shows the Michaelis–Menten plot of ATP plot of Kinase I. The tolerance of high ATP concentration in the mM range and direct measurement of ADP make this assay ideal to perform kinetic parameters of kinases. The substrate K m of ATP of Kinase I was similar to that determined in a SPA format (data not shown).
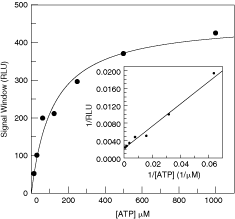
K m, ATP determination of ATP for Kinase I. Data were collected at varying ATP concentrations and fitted using Michaelis–Menten equation. The insert is a double-reciprocal plot of the same set of data. Abbreviation: RLU, relative light unit.
Compound Inhibition
A fit-for-purpose screening assay not only has to be robust, measured by a standard parameter Z′ factor,14 but also has to be sensitive.17 To this end, we performed dose–response curves of a few known inhibitors of Kinase I. As shown in Figure 5, ADP-Glo assay was able to determine the potency of 3 compounds whose potency are in 3-log unit different. This result shows that ADP-Glo™ assay can be used to confirm primary hits from HTS, but also can be used to profile compounds with various potencies.
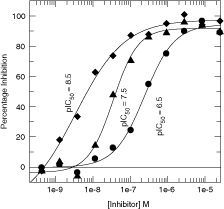
Dose–response curves of Kinase I inhibitors with different potency. The data were collected and analyzed as described in the Materials and Methods section.
Assay Variability and pIC50 Comparison
To meet the criterion of a fit-for-purpose profiling assay, especially for determining SAR, the assay is required to generate reproducible data. To evaluate this feature of ADP-Glo assay, the pIC50 values of a number of Kinase I inhibitors that were obtained from 2 independent experiments and a good agreement was observed (data not shown).
In addition, the pIC50 values of 13 program compounds were compared in SPA and ADP-Glo assays both of which measure inhibition of enzyme activity. It is clear from Table 1 that pIC50 of each compound is in good agreement (within 0.5 log unit difference between these 2 methods). The result of this comparison indicates that ADP-Glo can reach the sensitivity of radioactivity-based assay without using radioactive materials, which is a main advantage, especially for enzyme of low activity, such as Kinase I.
Another assay we developed for the Kinase I program is an FP assay using fluoro-tagged ATP pocket binder. Upon binding to the target, a high mP signal is generated. However, if a compound binds to the ATP-binding pocket and replaces the fluoro-ligand, a low mP will occur. In order to compare ADP-Glo and FP assays, we selected a number of program compounds to be tested in these 2 assays and the results are shown in Figure 6. Below pIC50 values of 7.5, there is a good correlation between the 2 assays, but above this value only the ADP-Glo assay can distinguish compound SAR. This is typical when one assay (in this case the FP assay) hits the tight binding limit, and demonstrates that the ADP-Glo assay can offer advantages in sensitivity for compound profiling.
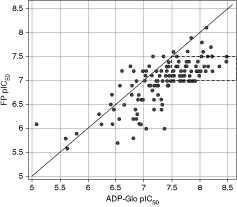
Comparison of Kinase I inhibitors’ pIC50s obtained from ADP-Glo™ assay (x-axis) and fluorescence polarization (FP) assay (y-axis). R 2 is 0.6. It is indicated in the boxed area that ADP-Glo™ demonstrated higher resolution for more potent Kinase I inhibitors under the conditions specified in Materials and Methods section.
Superior Assay Sensitivity of Kinase II
Kinase II is a nonprotein kinase. In order to demonstrate the generality of the ADP-Glo assay, an enzyme titration was done and the results are shown in Table 2. Excellent S/B ratio could be obtained down to low pM concentrations of Kinase II, demonstrating, again, the excellent sensitivity afforded with this format.
DISCUSSION
Many kinase assay technologies are commercially available and they were reviewed recently.5 , 18 Among them 2 were recently developed to detect the production of ADP. One is ADP Quest™ developed at DiscoverX, Fremont CA, which couples the production of ADP to the product of pyruvate via pyruvate kinase. Pyruvate is then oxidized by pyruvate oxidase to yield hydrogen peroxide as a product, which is ultimately linked to the oxidation of Amplex red to resorufin by peroxidase.12 The other one is Transcreener™ (Bellbrook Labs, Madison, WI), which utilizes an anti-ADP antibody and fluorescently labeled ADP analog. The assay can be configured as either a TR-FRET or FP.19 The displacement of fluorescently labeled ADP analog from the antibody by the ADP generated via target enzyme reaction leads to signal decrease.
However, the Transcreener assay cannot tolerate high ATP concentration, as the ADP antibody also has significant affinity for ATP.19 In addition, the assay signal is nonlinear with response to ADP generated in the reaction. Therefore, it is not suitable for kinetic studies as ATP–ADP standard curve is required to quantify the amount of ADP is produced. As for ADP Quest™, it is sensitive to the reducing agent used in the reaction. In addition, it is reported that high ATP concentration cannot be tolerated as increasing ATP concentration can negatively affect the assay performance.20 In contrast, ADP-Glo is a signal increase assay and the assay signal is linear to ADP production. This has been shown for Kinase I kinetic studies in Figure 4. The excess amount of coupled enzymes in Reagents I and II makes this assay less likely causing false positives in HTS.
ADP-Glo assay can tolerate a dynamic range of ATP. As unused ATP in the reaction is depleted prior to conversion of ADP to ATP by coupled enzymes in Reagent II (Fig. 1), the background is extremely low. On one hand, this allows robust detection of small amount of ADP generated in the reaction. For example, in the ATP–ADP standard curve shown in Figure 2, as little as 0.2 pmoles of ADP can generate an S/B ratio of 3:1. On the other hand, high ATP concentration is well-tolerated. As shown in our studies of Kinase II, we employed 500 µM ATP in the reaction and our result shows that as little as 10 pM of enzyme can produce a signal-to-background ratio of 2.5. However, at high concentration of ATP, trace amount of ADP contamination in ATP solution will ultimately requires >3% turnover of ATP at any concentration in the ADP-Glo assay. We find that in the 1 mM ATP standard curve experiment, 5% turnover is needed to generate a signal-to-background ratio of 3:1. Compared with other universal kinase assays regardless of ATP depletion or ADP detection where >10% turnover is required to generate >3-fold of assay signal to background, ADP-Glo still requires lowest ATP turnover while meeting the criteria of a robust assay.14
Tolerance of broad range of ATP concentration provides many advantages. First of all, it allows easy determination of ATP K m as varied ATP concentration does not interfere with ADP production. Second, it can accommodate assay development for a range of kinase targets whose K m, ATP values differ dramatically. This feature potentially allows ADP-Glo assay to be used as single platform to perform kinase profiling (Jasbinder S. Sanghera, personal communication). In addition, tolerance of high ATP concentration allows ATP competitiveness studies, a key experiment for MOA studies.
The superior sensitivity of ADP-Glo assay with broad range of potencies was demonstrated in Kinase I study. Figure 5 showed 3 Kinase I inhibitors, whose potency differ by 2-log unit with a pIC50 of 8.5 of the most potent compound. As shown in Figure 6, ADP-Glo offers a better resolvable range of high potency compounds when compared with FP assay side-by-side, which is a true advantage when it comes to use SAR to progress compound. Also showed in Figure 6, compounds appear to have a bias toward ADP-Glo compared with the FP assay and this was mainly caused by different conditions of these 2 assays, especially the concentration of enzyme, ligand, and substrate used to optimize the individual assays. In addition, superior sensitivity will reduce the amount of enzyme used in the assay so it can reduce cost per well. With the ever-increasing size of screening libraries, miniaturization and cost reduction per well become a big factor in choosing an assay for HTS.
Compared with ratiometric readout such as FP or TR-FRET, which is insensitive to assay volume, the robustness of ADP-Glo assay relies on good liquid handling. This may explain the lack of consistency in the Z′ factor shown in the Kinase II pilot screen (Fig. 3). Although the majority of the screening plates passed the Z′ criteria (0.4) with average Z′ score of 0.6, some plates failed. One factor to be considered here is that the ADP-Glo reagents we used are pre-production and we had to mix enzymes provided separately to make ready-to-use Reagents I and II prior to addition. The marketed product has eliminated this step and will potentially help homogeneity and therefore improve assay performance.
In summary, the ADP-Glo assay is an antibody-free, homogeneous and nonradioactive assay that is applicable to all kinases. The direct and accurate measurement of ADP from a kinase reaction makes it a fit-for-purpose assay for HTS, profiling assay as well as mode of action (MOA) studies. The detection of ADP rather than peptide substrate allows it suitable not only for protein kinases, but also for any ADP production enzymes with limited adjustment of assay protocol. In fact, ADP-Glo assay has been used to measure ATPase activity of Hsp90 as well as Na–K ATPase activity (Said Goueli, personal communication). This assay will not replace other kinase platforms, but provide a new alternative to complement exiting assays available on the market.
Footnotes
ACKNOWLEDGMENTS
We thank our BRAD colleagues Huizhen Zhao and Hongwei Qi for the purification of Kinase I and John D. Martin and Stephanie Chen for the purification of Kinase II. We thank SMtech Management group of GSK for providing compound plates and MDR-IT of GSK for data management. We would also like to thank Drs. Bob Ames, Gordon McIntyre, and Thomas Meek for their support in evaluating ADP-Glo technology.
AUTHOR DISCLOSURE STATEMENT
H.L., R.D.T., L.A.L., B.S., P.C., A.J.J., and G.Z. are employees of GlaxoSmithKline.
