Abstract
Riociguat is approved for the treatment of pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension. Some patients have difficulty swallowing tablets; therefore, 2 randomized, nonblinded, crossover studies compared the relative bioavailability of riociguat oral suspensions and immediate-release (IR) tablet and of crushed-tablet preparations versus whole IR tablet. In study 1, 30 healthy subjects received 5 single riociguat doses: 0.3 and 2.4 mg (0.15 mg/mL suspensions), 0.15 mg (0.03 mg/mL), and 1.0 mg (whole IR tablet) under fasted conditions and 2.4 mg (0.15 mg/mL) after a high-fat, high-calorie American-style breakfast. In study 2, 25 healthy men received 4 single 2.5-mg doses: whole IR tablet and crushed IR tablet suspended in applesauce and water, respectively, under fasted conditions, and whole IR tablet after a continental breakfast. In study 1, dose-normalized pharmacokinetics of riociguat oral suspensions and 1.0-mg whole IR tablet were similar in fasted conditions; 90% confidence intervals for riociguat area under the curve (AUC) to dose and mean maximum concentration (Cmax) to dose were within bioequivalence criteria. After food, dose-normalized AUC and Cmax decreased by 15% and 38%, respectively. In study 2, riociguat exposure was similar for all preparations; AUC ratios for crushed-IR-tablet preparations to whole IR tablet were within bioequivalence criteria. The Cmax increased by 17% for crushed IR tablet in water versus whole IR tablet. Food intake decreased Cmax of the whole tablet by 16%, with unaltered AUC versus fasted conditions. Riociguat bioavailability was similar between the oral suspensions and the whole IR tablet; exposure was similar between whole IR tablet and crushed-IR-tablet preparations. Minor food effects were observed. Results suggest that riociguat formulations are interchangeable.
Keywords
Pulmonary arterial hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH) are progressive, life-threatening conditions characterized by increased pulmonary vascular resistance and vascular remodeling.1–3 Left untreated, they can lead to right heart failure and, eventually, death.
The stability of a low-pressure state in the healthy lung is dependent on a balance of vasodilatory agents, such as nitric oxide (NO) and prostacyclins, and vasoconstrictive agents, such as thromboxane A2 and endothelin. 4 In pulmonary hypertension, however, the balance of vasoactive agents is altered, with reduced bioavailability of NO and prostacyclins and increased production of endothelin, which results in chronic pulmonary vasoconstriction.3,5 NO mediates vasodilation via stimulation of the enzyme soluble guanylate cyclase (sGC) in endothelial cells, leading to increased production of cyclic guanosine monophosphate (cGMP). 6 In addition to inducing vasorelaxation, NO also inhibits vascular smooth muscle cell proliferation and migration.7,8 Thus, a target for treatment of PAH is stimulation of the NO—sGC—cGMP pathway.
Riociguat is a novel, oral sGC stimulator that stimulates the NO—sGC—cGMP pathway via a dual mechanism of action by sensitizing sGC to endogenous NO through stabilization of NO—sGC binding and directly stimulating sGC independently of NO. 9 In 2 pivotal phase 3 studies—PATENT-1 10 and CHEST-1 11 —riociguat (up to 2.5 mg 3 times daily) was well tolerated and showed significant and robust efficacy in improving exercise capacity and a range of functional and hemodynamic parameters in patients with PAH or CTEPH, respectively. The efficacy benefits of riociguat were sustained for up to 1 year in long-term extension studies (PATENT-2 and CHEST-2) with continued good tolerability.12,13 Accordingly, riociguat has been approved in the United States, Europe, and several other countries for the treatment of patients with PAH and inoperable or persistent/recurrent CTEPH.14,15
For some patients with PAH, for example, children and the elderly, swallowing tablets may be inappropriate or difficult; therefore, oral suspensions of riociguat and a crushed preparation of the riociguat immediate-release (IR) tablet suspended in applesauce were investigated for relative bioavailability. Here we report the findings from 2 studies that were conducted to evaluate the relative bioavailability of the 2 riociguat formulations under fed and fasted conditions compared with riociguat whole-IR-tablet dosing under fasted conditions. The safety and tolerability of the different riociguat formulations were also evaluated.
METHODS
Study designs
Participants
In study 1, healthy white male and female subjects aged 18–45 years with a body mass index of 18.0–29.9 were enrolled after providing written informed consent. Female subjects had to be without child-bearing potential or have had a negative pregnancy test at screening and agreed to use double contraception for the duration of the study. Inclusion criteria for study 2 were the same as for study 1, with the exception that female subjects were not eligible.
For both studies, subjects were excluded if they had clinically relevant findings in the initial electrocardiogram (ECG), physical, or laboratory examinations; systolic blood pressure <100 or >145 mmHg; diastolic blood pressure >95 mmHg (and <50 mmHg in study 2); or heart rate <45 (<50 for study 2) or >95 beats per minute. Other exclusion criteria were a history of coronary artery disease, bronchial asthma, or other airway disease; symptomatic postural hypertension or other medical condition that may affect drug metabolism or completion of the study; regular use or suspected abuse of medicines and alcohol; special diets; intake of grapefruit in the 2 weeks preceding study entry or regular daily intake of 1 L of xanthine-containing drinks; blood donation of 100 mL in the past 4 weeks or of 500 mL in the past 3 months; participation in another clinical trial in the preceding 3 months; and smoking in the past 3 months.
Outcome measures
The primary outcomes in both studies were the area under the plasma concentration versus time curve from time 0 to infinity (AUC) and maximum concentration in plasma (Cmax) of riociguat. Further pharmacokinetic parameters assessed included the ratio of AUC to riociguat dose (AUC/D); ratio of Cmax to riociguat dose (Cmax/D); time to reach riociguat Cmax (tmax); and riociguat half-life associated with the terminal slope (t1/2). Safety was assessed in terms of adverse events, vital signs, ECG, laboratory parameters, and physical examination.
Pharmacokinetic assessment
During each dose period, blood samples were taken from each subject before dosing and again at 0.25, 0.5, 0.75, 1, 1.5, 2, 3, 4, 6, 8, 12, 15, 24, 36, 48, and 72 hours after dosing. Samples were stored at or below −15°C until analysis. Bioanalytical assay of riociguat and its metabolite M1 in plasma was performed using a fully validated high-performance liquid chromatography-tandem mass spectrometry assay using [methoxycarbonyl-2H3]-riociguat and [2H3]M1 as internal standards. Quality control and calibration samples were analyzed concurrently with study samples. In study 1, the calibration range was 0.5–250 μg/L for both riociguat and M1. Quality control samples in the concentration range 1.5–200 μg/L were determined with an accuracy of 97.0%–98.3% for riociguat and 92.9%–99.4% for M1, with a precision of 4.3%–6.0% for riociguat and 4.6%–6.5% for M1. In study 2, the calibration range was 2.0–500 μg/L for riociguat and M1. Quality control samples in the concentration range 6–400 μg/L were determined with an accuracy of 92.0%–96.8% for riociguat and 93.2%–100% for M1, with a precision of 4.6%–5.3% for riociguat and 3.4%–4.6% for M1. Concentrations were calculated from the chromatographic raw data, and only values above the lower limit of quantitation were used to determine pharmacokinetic parameters. The bioanalyst was not blinded during analysis of study samples. Pharmacokinetic parameters were calculated using the model-independent (compartment-free) method and WinNonlin software (ver. 4.1; Certara, Princeton, NJ).
Taste assessment
In study 1, subjects receiving riociguat oral suspension (2.4, 0.3, or 0.15 mg) in the fasted state were required to complete a questionnaire to assess the taste immediately after intake of the riociguat suspension, in line with pediatric guidelines. Questions were designed to assess the following taste parameters: immediate impression, taste and mouth feeling, general aftertaste, and overall impression. All questions were answered using a 5-point scale. For statements such as “The oral suspension formulation feels sweet in the mouth,” possible answers on the 5-point scale were “Applies exactly,” “Applies somewhat,” “Neither nor,” “Applies rather less,” and “Does not apply at all.” For questions such as, “In general, to what extent did you like the oral suspension formulation?,” possible answers on the 5-point scale were “Very good,” “Good,” “Neither good nor bad,” “Bad,” and “Very bad.” A taste assessment was not conducted in study 2 as comparisons would be difficult owing to the applesauce masking the taste of the tablet.
Statistical analysis
The pharmacokinetic analysis set for each study comprised all subjects who completed that study with valid pharmacokinetic profiles taken at the scheduled times. The safety analysis set for each study comprised all subjects who received at least 1 dose of study drug in that study.
Unless otherwise indicated, all statistical analyses were prespecified. The statistical evaluation was conducted using SAS software, version 9.1 or higher (SAS Institute, Cary, NC). Logarithms of AUC/D and Cmax/D in study 1 and AUC and Cmax in study 2 were analyzed by analysis of variance with treatment, period, sequence, and subject (sequence) as factors. Point estimates (least squares means) and 90% confidence intervals (CIs) were calculated for ratios between dose groups. Although these studies were not prespecified to determine bioequivalence, the acceptance range of 80%–125% was applied in an exploratory manner for the assessment of the relative bioavailability and the potential food effect.
RESULTS
Participants
Study 1 enrolled 32 healthy subjects, of whom 30 (15 men and 15 women with mean age of 32.6 [range: 22–45] years) received the study drug and were valid for the safety analysis. A total of 29 subjects completed all 5 study periods with valid pharmacokinetic profiles and were included in the pharmacokinetic analysis population. In study 2, 26 healthy male subjects were randomized, and 25 (mean age 31.8 [range: 20–45] years) received the study drug and were valid for the safety analysis. A total of 24 subjects completed all 4 study periods with valid pharmacokinetic profiles and were included in the pharmacokinetic analysis set. Baseline characteristics for populations included in the pharmacokinetic analysis in studies 1 and 2 are shown in Table 1.
Baseline demographics of subjects included in studies 1 and 2
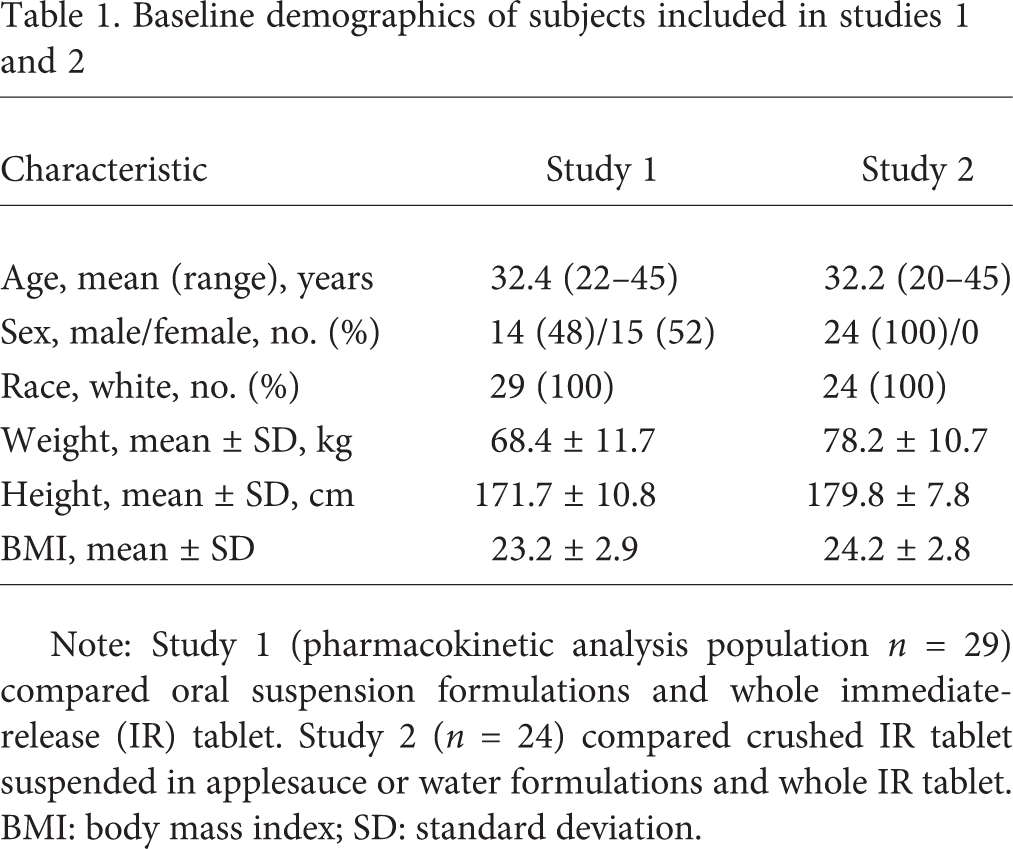
Note: Study 1 (pharmacokinetic analysis population n = 29) compared oral suspension formulations and whole immediate-release (IR) tablet. Study 2 (n = 24) compared crushed IR tablet suspended in applesauce or water formulations and whole IR tablet. BMI: body mass index; SD: standard deviation.
Pharmacokinetic analysis
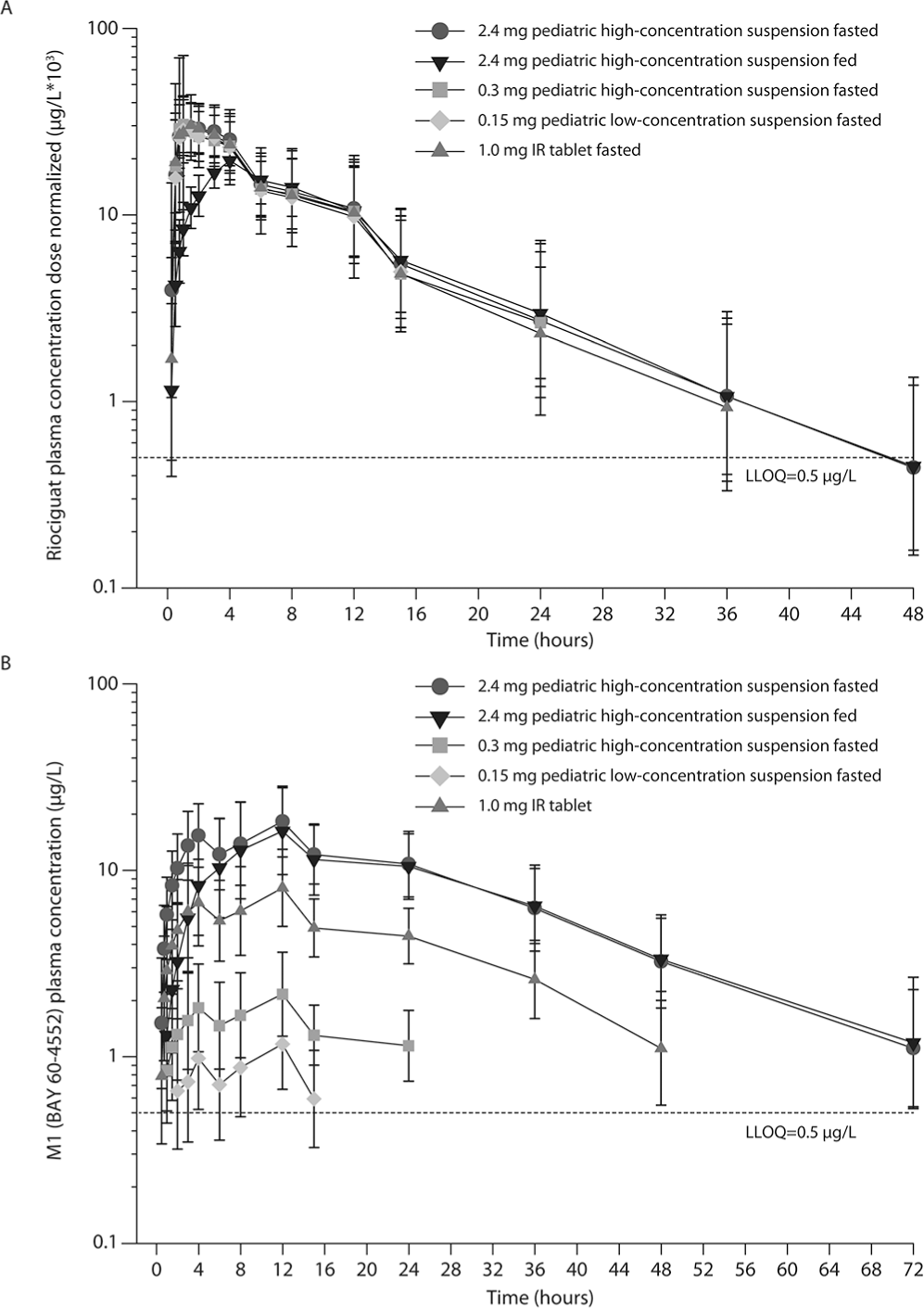
Dose-normalized plasma concentrations of riociguat (A) and its metabolite M1 (B) after a single oral dose of riociguat in study 1 (oral suspension formulations; n = 29). Data were calculated if at least two-thirds of individual values were above the lower limit of quantification (LLOQ) and are presented as geometric mean ± geometric standard deviation on a semilogarithmic scale. IR: immediate release.
Pharmacokinetic (PK) parameters of riociguat in plasma after a single oral dose in study 1 of oral suspension formulations and immediate-release (IR) whole tablet (n = 29)
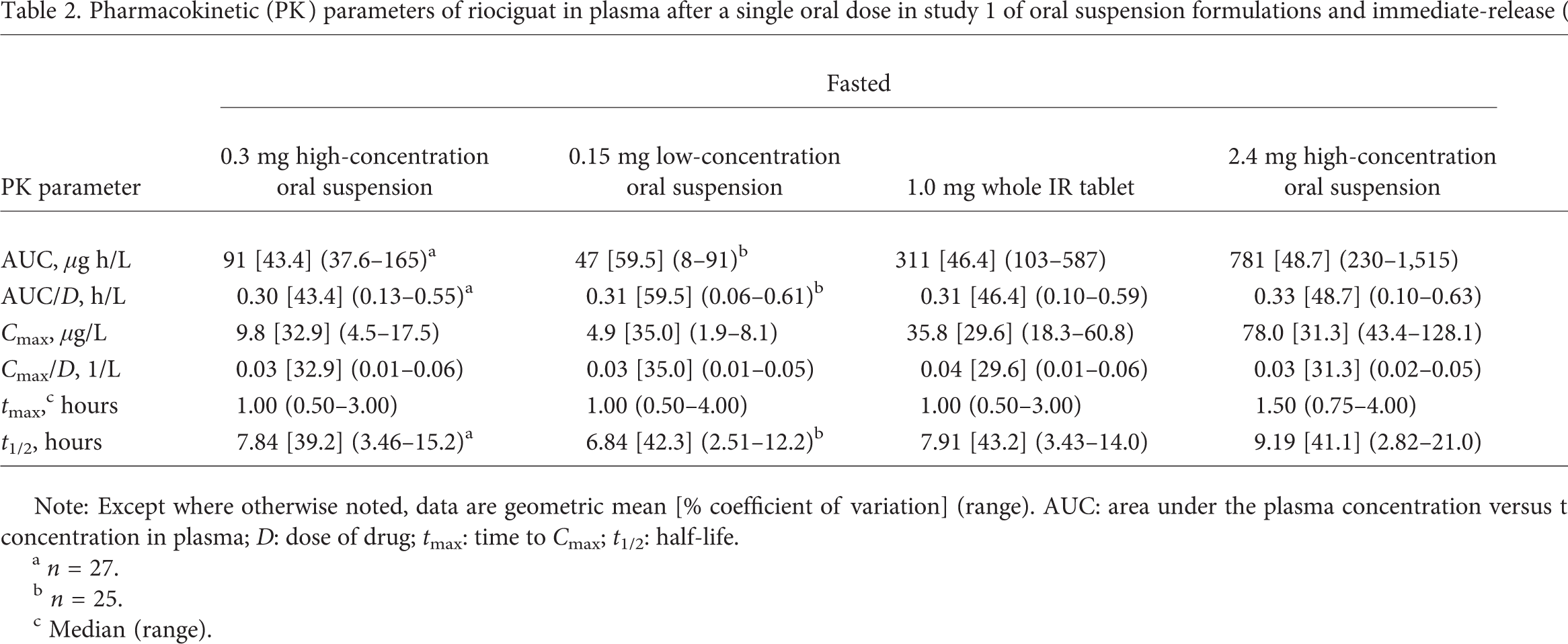
Note: Except where otherwise noted, data are geometric mean [% coefficient of variation] (range). AUC: area under the plasma concentration versus time curve; Cmax: maximum concentration in plasma; D: dose of drug; tmax: time to Cmax; t1/2: half-life.
n = 27.
n = 25.
Median (range).
Point estimates (least squares means) and 2-sided 90% confidence intervals (CIs) for selected pharmacokinetic parameters after administration of riociguat in study 1 of oral suspension formulations and whole immediate-release (IR) tablet (n = 29)

Note: AUC: area under the plasma concentration versus time curve; Cmax: maximum concentration in plasma; D: dose of drug; Est.: estimated.
A food effect on riociguat pharmacokinetics was observed for the 2.4-mg high-concentration suspension: when taken with a high-fat, high-calorie breakfast, absorption of riociguat was delayed, with a median tmax of 4.0 hours, compared with 1.5 hours in the fasted state, and the AUC/D and Cmax/D of riociguat were reduced by 15% and 38%, respectively (Table 2). The riociguat metabolite M1 showed a smaller food effect, with reductions of AUC/D by 8% and Cmax/D by 10%.
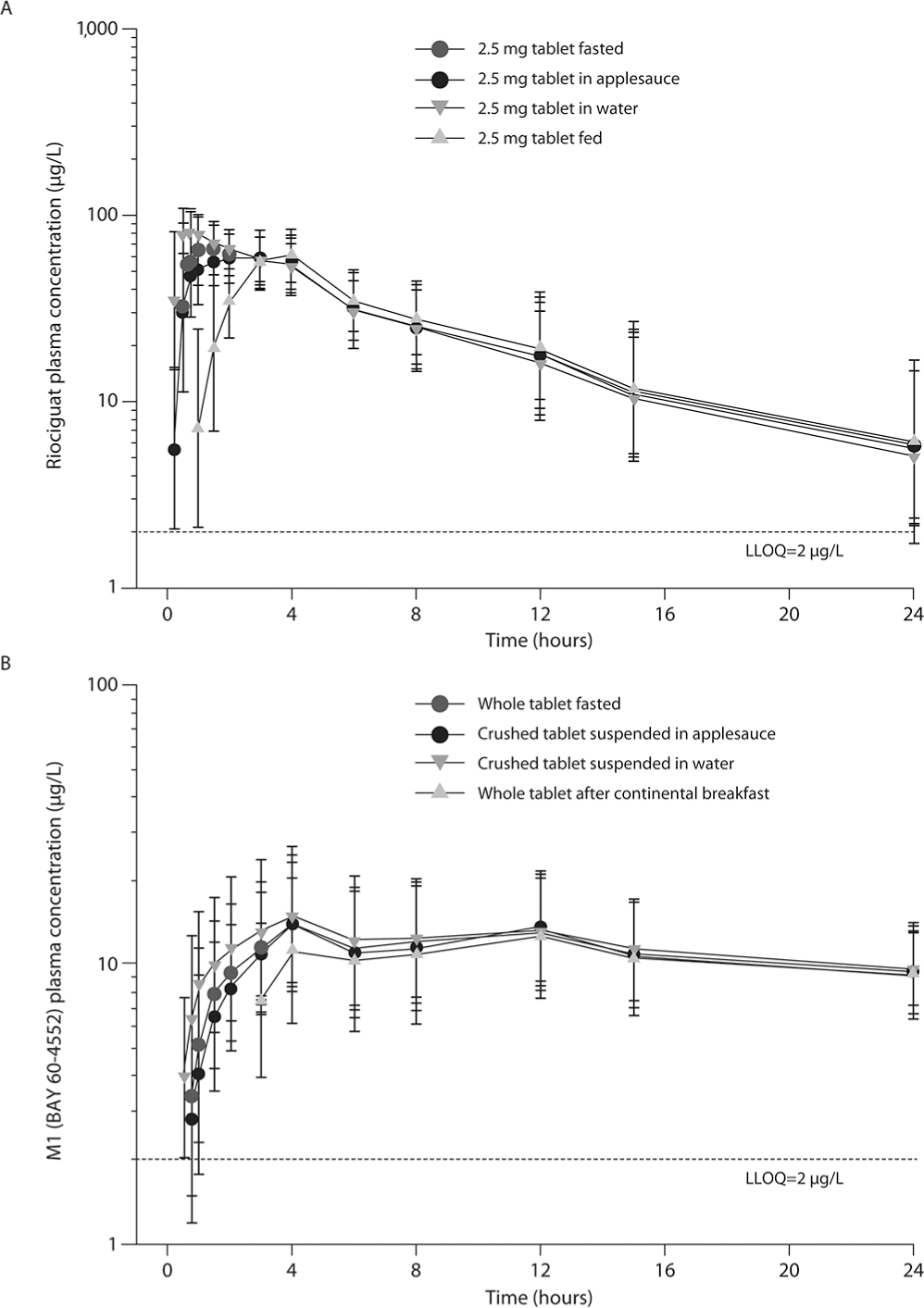
Plasma concentrations of riociguat (A) and its metabolite M1 (B) after a single oral dose of riociguat in study 2 (crushed immediate-release formulations; n = 24). Data were calculated if at least two-thirds of individual values were above the lower limit of quantification (LLOQ) and are presented as geometric mean ± geometric standard deviation on a semilogarithmic scale.
Pharmacokinetic (PK) parameters of riociguat in plasma after a single oral dose of 2.5 mg riociguat in study 2 of crushed immediate-release (IR) tablet suspended in applesauce or water formulations and whole IR tablet (n = 24)
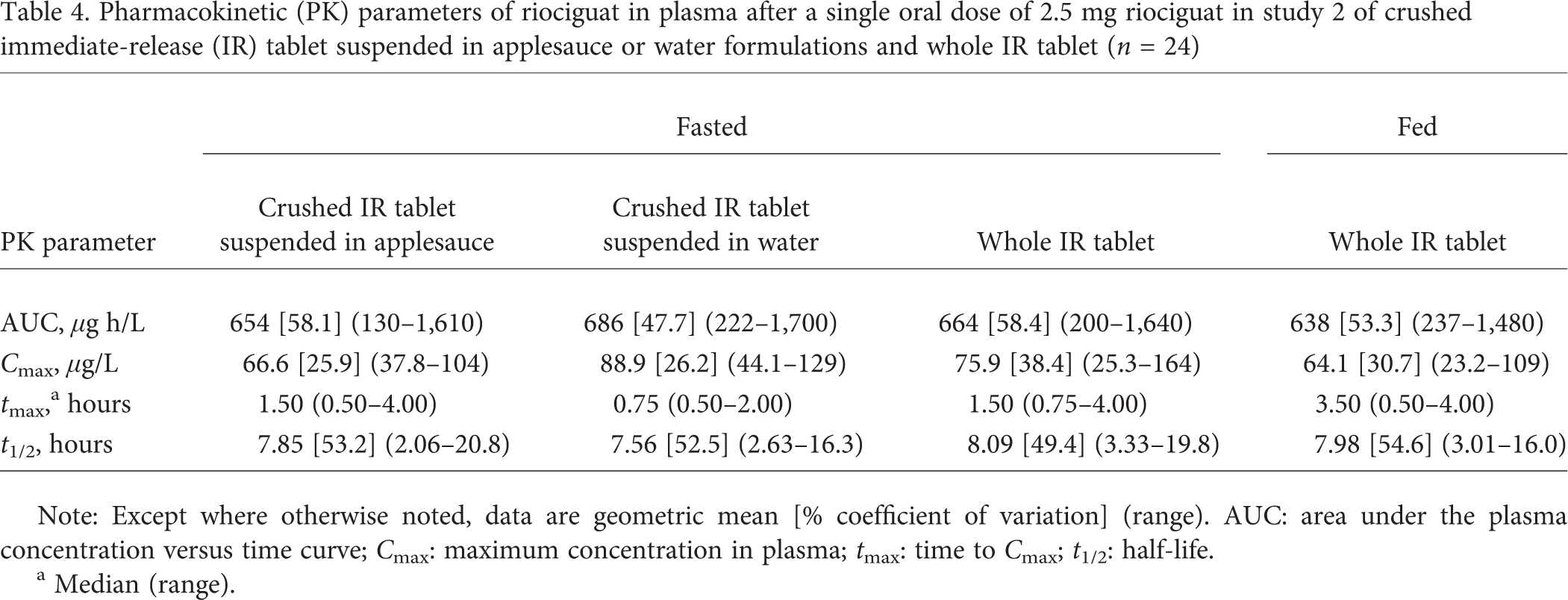
Note: Except where otherwise noted, data are geometric mean [% coefficient of variation] (range). AUC: area under the plasma concentration versus time curve; Cmax: maximum concentration in plasma; tmax: time to Cmax; t1/2: half-life.
Median (range).
The t1/2 for riociguat crushed IR tablet suspended in applesauce was 7.9 hours, compared with 8.1 hours for the whole IR tablet in the fasted state (Table 4). Although the main aim of the study was to establish relative bioavailability and not bioequivalence, the 90% CIs for the ratios of “2.5-mg crushed IR tablet suspended in apple-sauce/2.5-mg whole IR tablet fasted” for AUC and Cmax (Table 5) and M1 (data not shown) were entirely contained within the standard bioequivalence range of 80%–125%.
Point estimates (least squares means) and 2-sided 90% confidence intervals (CIs) for selected pharmacokinetic parameters after administration of riociguat in study 2 of crushed immediate-release (IR) tablet suspended in applesauce or water and whole IR tablet (n =24)
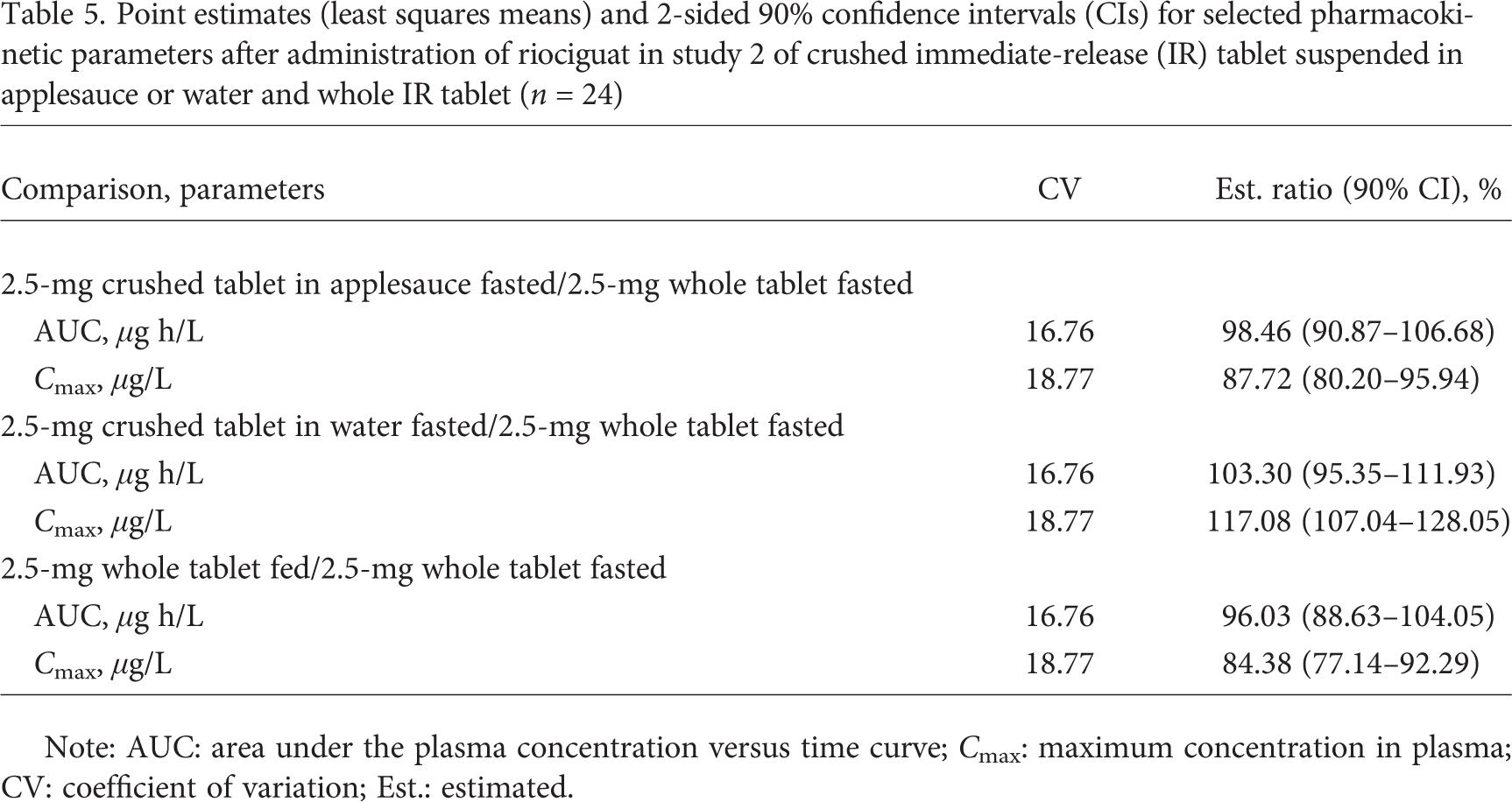
Note: AUC: area under the plasma concentration versus time curve; Cmax: maximum concentration in plasma; CV: coefficient of variation; Est.: estimated.
Compared with the riociguat 2.5-mg whole IR tablet in the fasted state, administration of riociguat as a crushed IR tablet suspended in water was associated with a 17% increase in Cmax (88.9 vs. 75.9 μg/mL) with similar AUC (664 vs. 686 μg h/L). Absorption of riociguat was also faster when the IR tablet was crushed and suspended in water compared with that for the whole IR tablet (median tmax of 0.75 vs. 1.5 hours, respectively; Table 4). The t1/2 for riociguat crushed IR tablet suspended in water was similar to that for the whole IR tablet in the fasted state (Table 4).
A food effect on riociguat pharmacokinetics was observed when taken after a continental breakfast: absorption of riociguat 2.5-mg whole IR tablet was delayed, with a median tmax of 3.5 hours compared with 1.5 hours in the fasted state (Table 4). In the fed state, the Cmax of riociguat was reduced by 16%, while overall AUC was unaltered compared with the fasted state (Table 4). Although the study was not designed to assess bioequivalence, the 90% CI of the AUC ratio “fed/fasted” was within the bioequivalence range of 80%–125%. Formation of the riociguat metabolite M1 was delayed after food intake by approximately 3 hours, which corresponded to the delayed absorption of riociguat in the fed state.
Taste assessment
When taken in the fasted state, 47%–57% of participants reported the aftertaste of the riociguat oral suspension as “Feels exactly or somewhat sweet in the mouth”; other forms of aftertaste such as “salty,” “bitter,” “sour,” and “astringent” were most frequently assessed as “Does not apply at all” (67%–80%, 70%–77%, 40%–63%, and 77%–90%, respectively). The overall taste of the riociguat oral suspension was well received, with 60%–77% of participants responding “Good” to the question, “In general, to what extent did you like the oral suspension formulation?”
Safety
The most commonly reported treatment-emergent adverse events reported in both studies are shown in Tables 6 and 7. The most common treatment-emergent adverse event in both studies was headache (study 1: 33%–47%; study 2: 21%–28%).
Most common treatment-emergent adverse events (AEs; more than 5% in any dose group) in study 1 of oral suspension formulations and whole immediate-release (IR) tablet of riociguat (n = 30)
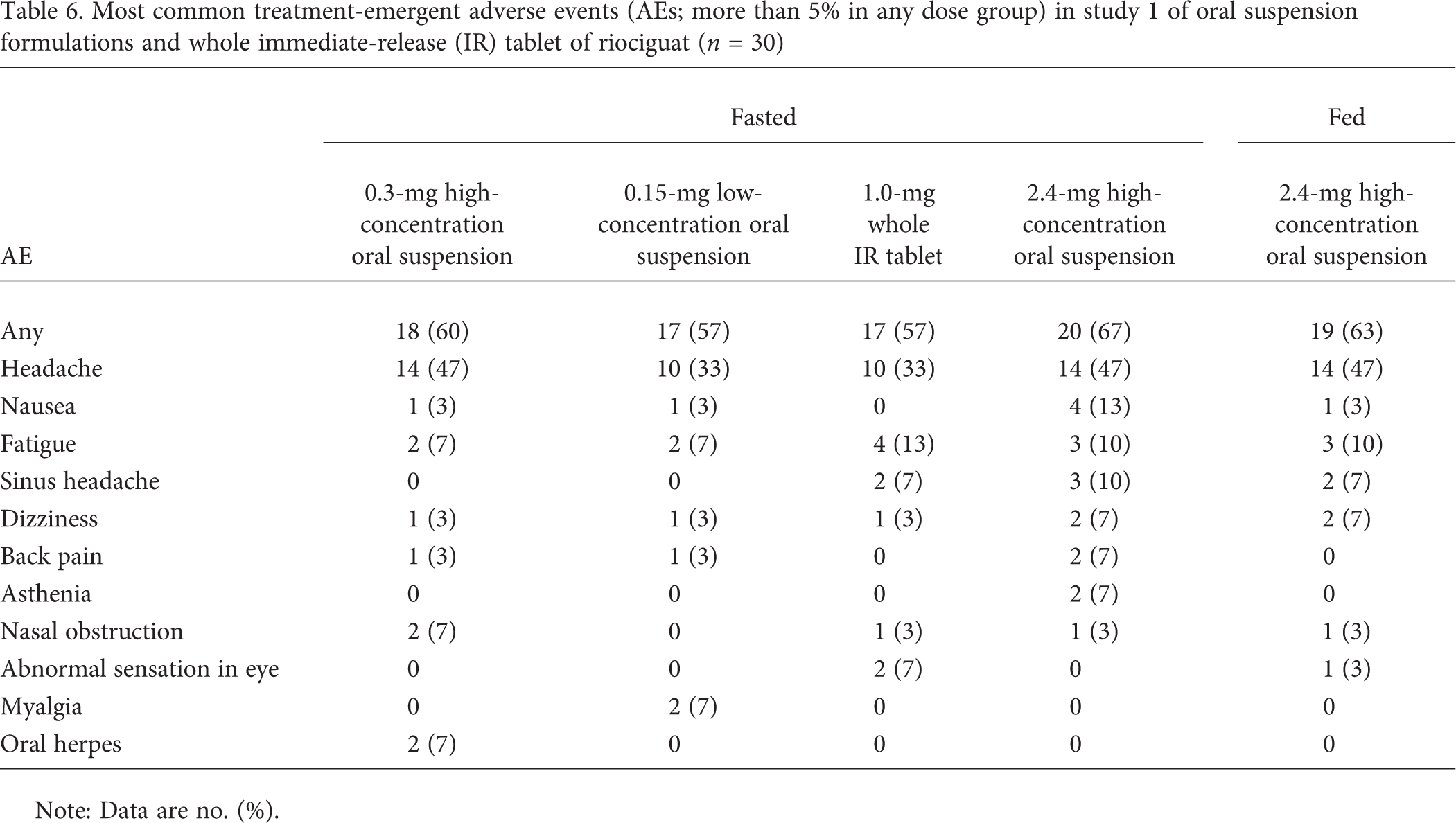
Note: Data are no. (%).
Most-common treatment-emergent adverse events (AEs; more than 5% in any dose group) in study 2 of crushed-tablet formulations and whole immediate-release (IR) tablet of 2.5 mg riociguat

Note: Data are no. (%).
The most frequently reported drug-related adverse events were generally associated with the mode of action of riociguat (i.e., vasodilation). These included headache (30%–47%), sinus headache (7%–10%), nausea (3%–13%), dizziness (3%–7%), and nasal obstruction/nasal congestion (3% for each) in study 1 and headache (13%–20%), nasal congestion (8%–21%), erythema (4%–16%), and dizziness and postural dizziness (0%–8% for each) in study 2. In both studies, there were no deaths or serious adverse events, and no adverse event led to premature study termination. In both studies, vital signs were in the expected range for healthy subjects, and ECG findings were inconspicuous. There was no signal for study drug-induced laboratory parameter changes in either study.
DISCUSSION
The clinical pharmacological studies reported here aimed to assess the relative bioavailability of different formulations of riociguat that may be of benefit for patients unable to swallow the standard whole (6-mm diameter) IR tablet formulation. In the fasted state, the bioavailability of riociguat was not altered for the low- or high-concentration oral suspension versus the 1.0-mg whole IR tablet or for the crushed 2.5-mg IR tablet suspended in applesauce or water versus the whole 2.5-mg IR tablet. Although the studies were not designed to show bioequivalence, an exploratory assessment suggested that standard bioequivalence criteria were met between the whole IR tablet and the oral suspensions and between the whole IR tablet and the crushed IR tablet suspended in applesauce. The crushed IR tablet suspended in water showed a comparable AUC with the whole IR tablet, although it was associated with faster absorption of riociguat and a 17% higher Cmax. Consistent with riociguat pharmacokinetics, no differences in relative bioavailability were observed between the different formulations for the pharmacologically active metabolite of riociguat, M1. Furthermore, although it was not a prespecified analysis, no apparent gender effect was observed on the pharmacokinetics for the riociguat 1.0-mg whole IR tablet, which was consistent with the findings of previous studies.16,17
The pharmacokinetic properties of riociguat in the current studies were generally similar to previous clinical pharmacology studies of riociguat administered to healthy volunteers in a fasted state (AUC and Cmax for riociguat 1.0-mg whole IR tablet of 171–244 μg h/L and 20.8–37.8 μg/L, respectively, in previous studies, compared with 311 μg h/L and 35.8 μg/L, respectively, in the current studies).18,19 Furthermore, the comparable bioavailability of the different formulations of riociguat in the current studies is consistent with previous studies that have shown no relevant differences in bioavailability between riociguat administered as a 2.5-mg whole 6-mm-diameter tablet or as a 2.5-mg solution 20 or riociguat administered as an oral 1.0-mg whole 6-mm tablet and as a 1.0-mg intravenous infusion. 18 The apparent interchangeability of riociguat administered as a whole IR tablet, oral suspension, or crushed IR tablet suspended in applesauce or water provides the potential to consider riociguat as a treatment option in eligible patient populations unable to swallow whole 6-mm-diameter tablets. Importantly, the taste of the oral suspension was well received by subjects in this study.
In the studies reported here, intake of food shortly before administration of riociguat delayed absorption of the riociguat 2.5-mg whole IR tablet compared with the same dose as an IR tablet in the fasted state; however, overall bioavailability was unaffected. This was consistent with the findings of another study on the effect of food on the pharmacokinetics of riociguat 2.5-mg tablets. 18 A minor food effect was also observed for the riociguat high-concentration oral suspension: intake of a high-fat, high-calorie breakfast delayed absorption of the riociguat high-concentration oral suspension compared with the fasted state; in this case, both AUC and Cmax were reduced. Thus, riociguat tablets can be taken with or without food.14,15
Riociguat was well tolerated in these studies, with no deaths or serious adverse events, and no adverse event led to premature study termination. As expected, the most common adverse events (headache, nausea, dizziness, and nasal obstruction/congestion) were consistent with the vasodilatory action of riociguat. The safety profile was consistent with previous studies of riociguat in healthy volunteers18–20 and in patients with PAH or CTEPH 10 and provides further evidence for the safety and tolerability of riociguat treatment.
In conclusion, the bioavailability of riociguat was similar between the oral suspensions and the standard whole IR tablet and between the crushed IR tablets suspended in applesauce or water and the whole IR tablet. A minor food effect was observed for the high-concentration suspension, which was consistent with the food effect seen for the whole IR tablet. These pharmacokinetic results suggest the interchangeability of these riociguat formulations for use in special populations such as children or the elderly.
