Abstract
In patients with idiopathic pulmonary arterial hypertension (iPAH), iron deficiency is common and has been associated with reduced exercise capacity and worse survival. Previous studies have shown beneficial effects of intravenous iron administration. In this study, we investigated the use of intravenous iron therapy in iron-deficient iPAH patients in terms of safety and effects on exercise capacity, and we studied whether altered exercise capacity resulted from changes in right ventricular (RV) function and skeletal muscle oxygen handling. Fifteen patients with iPAH and iron deficiency were included. Patients underwent a 6-minute walk test, cardiopulmonary exercise tests, cardiac magnetic resonance imaging, and a quadriceps muscle biopsy and completed a quality-of-life questionnaire before and 12 weeks after receiving a high dose of intravenous iron. The primary end point, 6-minute walk distance, was not significantly changed after 12 weeks (409 ± 110 m before vs. 428 ± 94 m after; P = 0.07). Secondary end points showed that intravenous iron administration was well tolerated and increased body iron stores in all patients. In addition, exercise endurance time (P < 0.001) and aerobic capacity (P < 0.001) increased significantly after iron therapy. This coincided with improved oxygen handling in quadriceps muscle cells, although cardiac function at rest and maximal
Iron deficiency is common in patients with idiopathic pulmonary arterial hypertension (iPAH) and is closely associated with poor survival and low exercise capacity.1–3 Compromised oxygen handling—that is, oxygen transport and consumption at a skeletal muscle level—has been suggested to cause the decreased exercise capacity in iron-deficient (ID) iPAH patients.2,4 Furthermore, iron deficiency might lead to deterioration of right ventricular (RV) function. 5 We have shown that oral iron supplements are ineffective in restoring body iron stores in iPAH.1,2 Recently, it was demonstrated that intravenous iron treatment in ID iPAH patients was associated with higher serum iron levels, improved 6-minute walk distance (6MWD), and quality of life.6,7 However, the exact mechanisms by which iron improves clinical performance in iPAH patients remain to be elucidated.
Therefore, the aim of the present study was twofold: (1) to investigate whether intravenous iron therapy in ID iPAH was safe and could indeed restore iron levels and improve exercise capacity in another iPAH cohort and (2) to explore whether altered exercise capacity can be explained in part by improved RV function and improved skeletal muscle oxygen handling.
METHODS
Patient inclusion
Patients who visited the Pulmonology Department of the Vrije Universiteit University Medical Center between January 2011 and January 2013 were approached to participate. Inclusion criteria were iPAH as defined by the European guidelines, 8 receipt of optimal PAH-specific treatment, being clinically stable for at least 3 months, and presence of iron deficiency (serum iron level of <10 μmol/L, transferrin saturation of <15% [women] or <20% [men], and serum ferritin level of <100 μg/L). Patients were excluded if they received iron therapy at admission, participated in another pulmonary hypertension study medication trial, or had a history of anemia, liver function impairment, or any other acute or chronic condition other than iPAH. The study was registered at ClinicalTrials.gov (NCT01288651) and was approved by the Institutional Review Board on Research Involving Human Subjects (Amsterdam, Netherlands). All patients provided written informed consent before inclusion.
Clinical study design
All end points were measured before (baseline) and 12 weeks after intravenous iron therapy. The primary end point was change in 6MWD after iron therapy. Secondary end points were change in blood iron parameters, change in maximal exercise parameters and endurance capacity determined by maximal and submaximal cardio-pulmonary exercise tests (CPETs), RV function determined by cardiac magnetic resonance imaging (MRI), pulmonary function determined by means of spirometry and diffusion capacity measurements, quality of life determined by the SF-36 questionnaire, and skeletal muscle oxygen handling at the cellular level determined by quadriceps muscle biopsy. A detailed description of all tests and methods is provided in the appendix (available online). After baseline measurements, an intravenous infusion of ferric carboxymaltose (Ferinject; Vifor Pharma, Glattbrugg, Switzerland) at 1,000 mg was given, in 20 mL of NaCl 0.9% over 2 hours. The study was not placebo controlled, since no permission for a placebo group was given by the medical ethics committee. At the end point of the study, 12 weeks after first admission, all measurements were repeated in the same order.
Statistical analysis
All data from patients before and after iron administration were analyzed using the paired Student t test or repeated-measures analysis of variance with Bonferroni post hoc tests to correct for multiple measurements. All analyses were performed with GraphPad Prism (ver. 5.00; GraphPad Software, San Diego, CA) and SPSS (ver. 20.0; SPSS, Chicago, IL). Log transformation was performed to obtain a normal distribution of the data when necessary. Data are presented as mean ± standard deviation, unless stated otherwise. Differences with a P value of <0.05 were considered statistically significant.
RESULTS
Tolerability and effects on blood iron levels
Eighteen iPAH patients were included in this study, of whom three did not finish the protocol. One patient presented with atrial flutter during second admission, 1 patient withdrew from the study before finishing the protocol, and 1 patient was excluded because erythropoietin was administered to that patient in another hospital during the follow-up period. Baseline characteristics of the 15 patients who were analyzed are shown in Table 1. All patients received the total amount (1,000 mg) of intravenous iron after the baseline measurements were obtained. Two patients had minor complaints of frontal headache during the infusion, which resolved shortly thereafter. No other adverse events were noted. Serum iron parameters were significantly increased 12 weeks after iron administration, without a change in N-terminal prohormone of brain natriuretic peptide levels. Serum hepcidin values were low at baseline and remained low until the end of the study (Table 2).
Baseline patient characteristics

Note: Except where otherwise noted, data are mean ± standard deviation. Hemodynamic parameters were obtained from right heart catheterization performed during the pulmonary hypertension diagnosis period. NYHA: New York Heart Association; ERA: endothelin receptor antagonist; PDE5: phosphodiesterase 5.
Laboratory parameters in idiopathic pulmonary arterial hypertension patients at baseline and end point
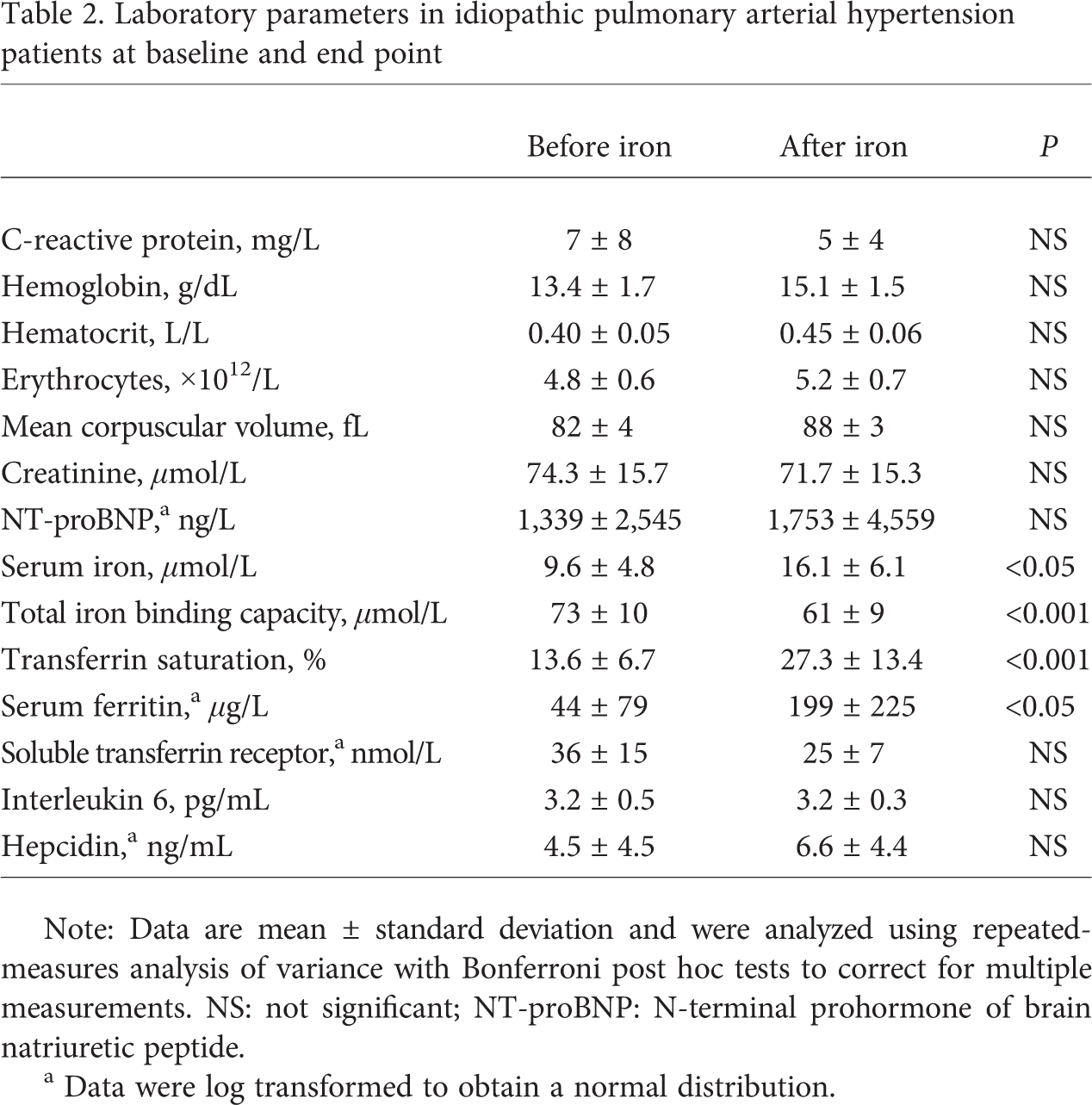
Note: Data are mean ± standard deviation and were analyzed using repeated-measures analysis of variance with Bonferroni post hoc tests to correct for multiple measurements. NS: not significant; NT-proBNP: N-terminal prohormone of brain natriuretic peptide.
Data were log transformed to obtain a normal distribution.
Iron improved submaximal exercise capacity
Intravenous iron therapy did not significantly change 6MWD after 12 weeks (409 ± 110 m before vs. 428 ± 94 m after; P = 0.07; Fig. 1A). Maximal workload (55 ± 23 W before vs. 59 ± 27 W after; not significant [NS]) and peak oxygen uptake (0.97 ± 0.22 L/min before vs. 0.97 ± 0.26 L/min after; NS), determined by maximal CPET, were also unchanged by iron therapy (Table S1; Tables S1–S4 are available online). However, the time to reach anaerobic threshold was significantly increased after iron therapy (175 ± 33 seconds before vs. 238 ± 43 seconds after; P < 0.001; Fig. 1B; Table S1). Exercise endurance capacity was markedly improved, as iPAH patients were able to exercise 51% longer after iron therapy compared with baseline (269 ± 89 seconds before vs. 405 ± 210 seconds after; P < 0.001; Fig. 1C; Table S2). Spirometry parameters were unchanged after iron therapy (Table S3).
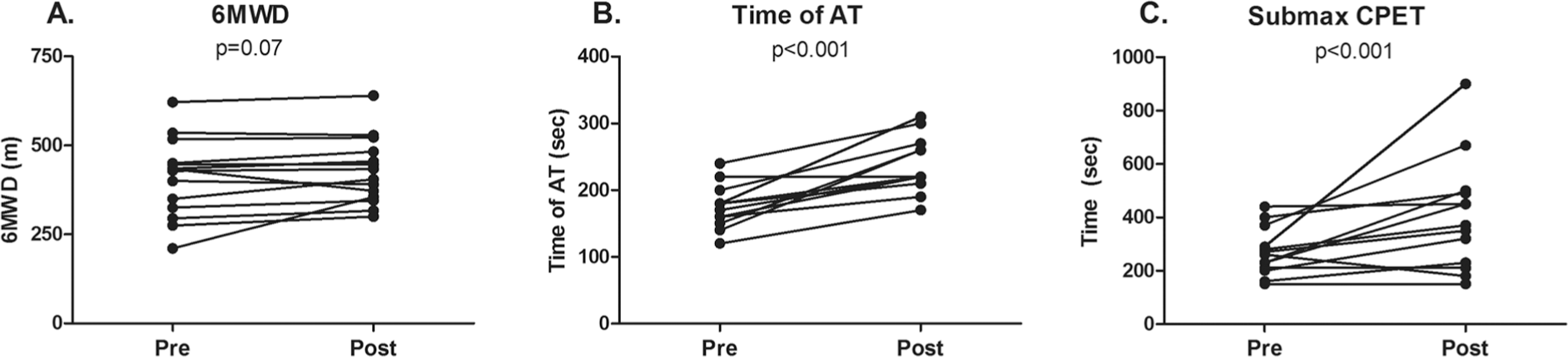
Exercise capacity in iron-deficient idiopathic pulmonary arterial hypertension patients before and after iron administration. Six-minute walk distance (6MWD) was unchanged after iron therapy (A). Time to anaerobic threshold (AT) during maximal cardiopulmonary exercise testing (CPET) was significantly postponed after iron therapy (B). At a submaximal level (75% of maximum achieved workload during CPET), patients were able to bike 51% longer after iron therapy (C).
Iron administration improved quality of life
Quality of life was improved after iron treatment, as indicated by a higher total score in the SF-36 questionnaire (47% ± 19% before vs. 56% ± 19% after; P < 0.05). Subdivision of the different components of the questionnaire showed that mental health was significantly better after iron treatment (49% ± 10% before vs. 60% ± 17% after; P < 0.01), whereas physical health was unaltered (39% ± 20% before vs. 46% ± 20% after; P = 0.09).
Improved exercise capacity could not be explained by improved RV function
Three patients could not undergo cardiac MRI because of obesity or claustrophobia; therefore, cardiac MRI was performed in a subset of 12 patients. Cardiac function at rest, represented by cardiac index (2.8 ± 0.9 L/min/m2 before vs. 2.5 ± 0.8 L/min/m2 after; NS) and ventricular ejection fraction (left ventricle [LV]: 62% ± 12% before vs. 59% ± 14% after; RV: 40% ± 21% before vs. 39% ± 21% after; both NS), was unchanged by iron therapy. In addition, LV and RV free wall mass index remained stable (LV: 59 ± 15 g/m2 before vs. 62 ± 17 g/m2 after; RV: 51 ± 29 g/m2 before vs. 56 ± 31 g/m2 after; both NS; Table S4).
Skeletal muscle oxygen transport was enhanced after intravenous iron therapy
Twelve patients consented to a quadriceps muscle biopsy. In 2 patients, the biopsy procedure did not result in sufficiently high-quality material. Biopsy analysis was therefore performed in a subset of 10 patients. Myoglobin concentration (0.34 ± 0.17 mM before vs. 0.44 ± 0.11 mM after; P < 0.05) and mitochondrial oxidative capacity (0.06 ± 0.01 nmol/mm 3 /s before vs. 0.09 ± 0.02 nmol/mm 3 /s after; P < 0.05) were both significantly increased in low oxidative cells after iron therapy, as was total myoglobin protein content (0.5 ± 0.4 AU before vs. 1.1 ± 0.6 AU after; P < 0.05; Figs. 2, 3). Furthermore, the number of capillaries per myocyte in the quadriceps muscle (1.0 ± 0.4 cap/myocyte before vs. 1.2 ± 0.2 cap/myocyte after; P = 0.37) was similar after iron therapy (Figs. 2, 3).
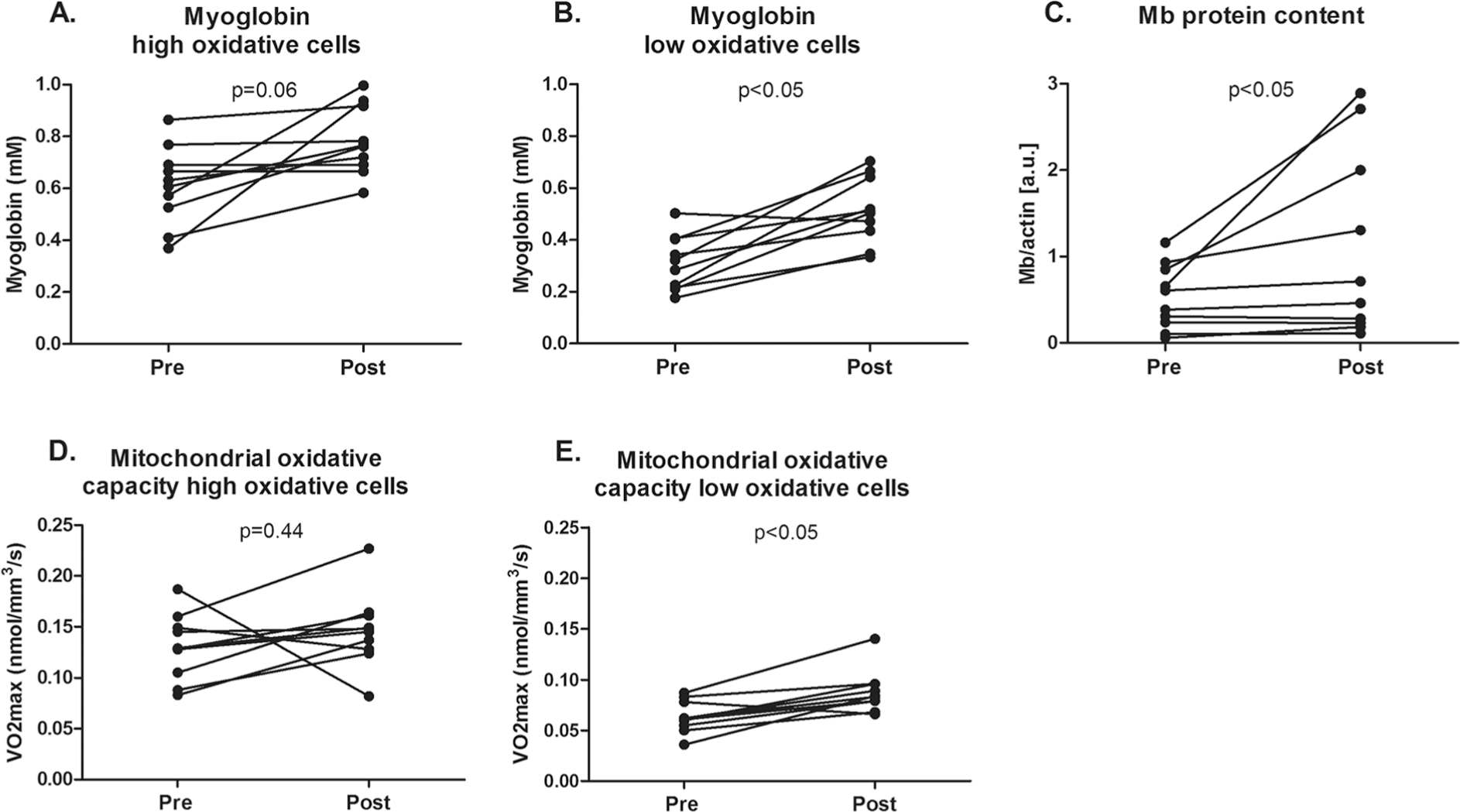
Oxygen handling in quadriceps muscle fibers after iron therapy in iron-deficient idiopathic pulmonary arterial hypertension patients. Myoglobin concentration was unaltered in high oxidative cells after iron therapy (A); however, a significant increase was observed in low oxidative cells (B) and total myoglobin protein content (C). Mitochondrial oxidative capacity, expressed as maximal oxygen consumption (
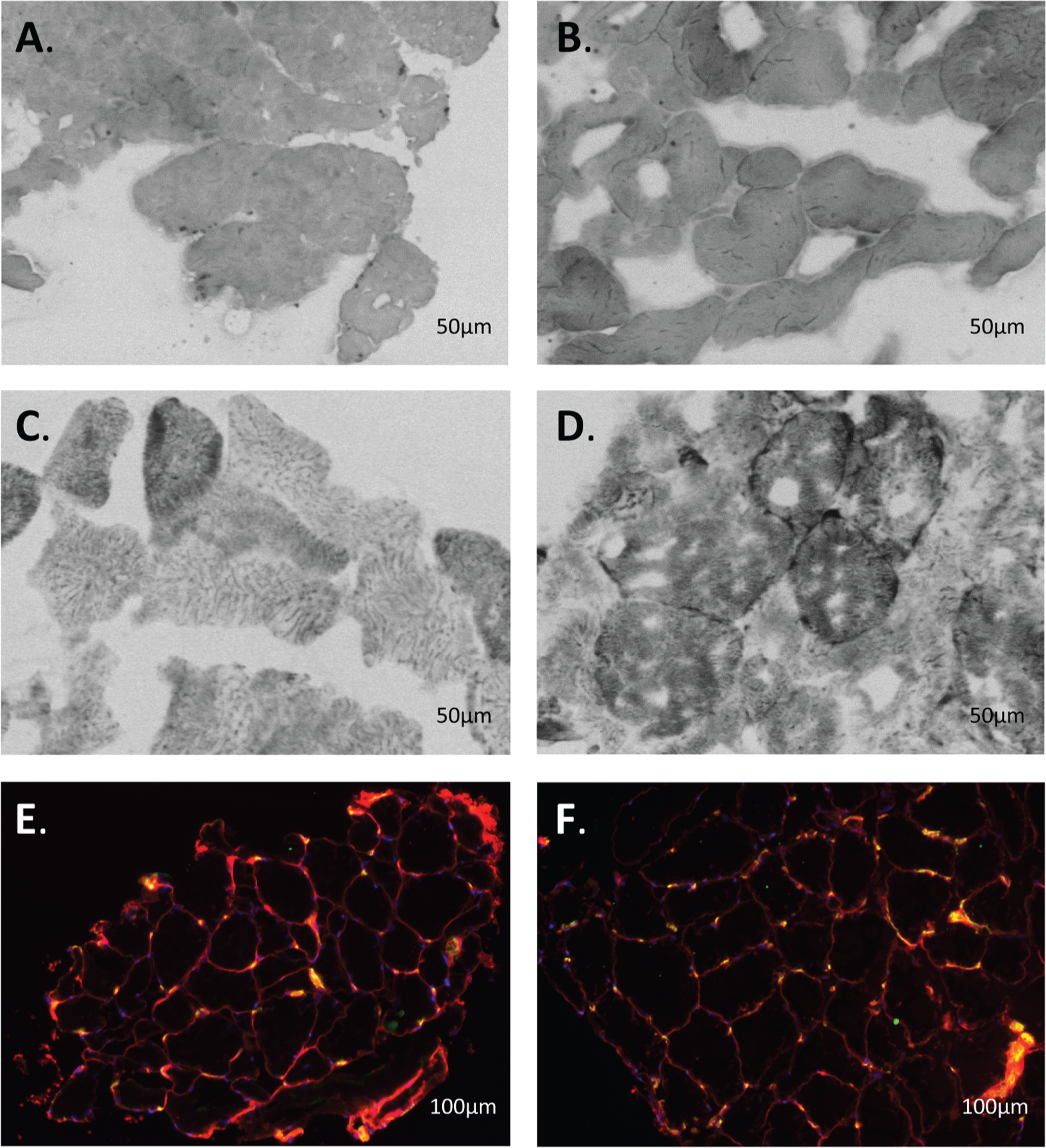
Quadriceps biopsy samples from idiopathic pulmonary arterial hypertension (iPAH) patients. Shown are typical examples of quadriceps biopsy samples from iPAH patients before (A, C, E) and after (B, D, F) intravenous iron therapy. A, B, Myoglobin staining. C, D, Succinate dehydrogenase activity staining. Type I and II cells are distinguished by color, as type I cells have more myoglobin and succinate dehydrogenase activity (darker cells) than type II cells. E, F, CD31 staining. Every yellow dot represents a capillary.
DISCUSSION
In the present study, we investigated the effects of iron therapy in ID iPAH patients. We showed that intravenous iron therapy in ID iPAH patients increased exercise endurance capacity. This could not be explained by better RV function; however, skeletal muscle biopsies of these patients revealed improvements in oxygen handling capacity after iron treatment.
Intravenous iron safely increased body iron stores and quality of life
Patients received a single fixed high dose of intravenous iron. An alternative approach would have been to supply multiple smaller doses using the Ganzoni formula to calculate the total amount of iron needed to restore body iron.5,9–11 One reason for using the present approach was that ferric carboxymaltose (Ferinject) is safe to administer in high iron dosages (up to 1,000 mg at one time) and allowed a single visit for every patient. 12 Second, use of the Ganzoni formula may not be reliable in iPAH patients with hypoxemia-driven erythropoiesis and consequent hemoglobin (Hb) levels within the normal range despite iron deficiency.11,13 Third, previous studies with iron therapy showed that the mean dose of iron needed was around 1,000 mg or even higher. 10 One of the concerns was that patients might increase their Hb levels above reference values, which could be detrimental since this increases blood viscosity. However, although we observed a slight increase in Hb after iron treatment, it was not above the upper limit of normal. In addition, in contrast to oral iron treatment, which does not produce a response in 44% of ID iPAH patients, ferric carboxymaltose led to higher iron stores in all patients, as represented by increased ferritin levels. 2
Furthermore, intravenous iron therapy was accompanied by improved quality of life, as shown by a mean increase of 9 points on a 100-point scale. Better quality of life after iron therapy has been previously described in ID patients with left heart failure as well as those with pulmonary hypertension.5,6 Whether the observed improvement is a result of better exercise capacity or an independent result of iron therapy, however, is difficult to determine.
Iron and exercise capacity
The achieved distance in the 6-minute walk test (6MWT) was unchanged by iron therapy. In addition, maximal CPET results were also unaltered. However, in the submaximal CPET, a significant improvement in exercise endurance time and improved aerobic capacity was demonstrated. It seems contradictory that the 6MWT, which is mostly regarded to be a submaximal exercise test, was unchanged by iron therapy. This might be explained in part by the very different type of exercise (i.e., biking vs. walking). In addition, the 6MWT has been associated with higher aerobic capacity and less metabolic stress than the CPET, which might indicate a different effect of iron therapy in both exercise tests. 14 Also, any potential beneficial effect on the 6MWT is difficult to extrapolate to resting pulmonary hemodynamics. 15 To observe changes in hemodynamics after iron therapy in future studies, right heart catheterization should be performed.
Iron deficiency by itself (without pulmonary hypertension) has been associated with diminished exercise capacity and fatigue in otherwise healthy subjects,16–18 although improvements in exercise capacity were observed only when anemia was also present.4,18–21 In the present study, increased Hb or higher maximal
Intravenous iron did not alter cardiac function but enhanced skeletal muscle oxygen handling
Iron therapy did not improve RV function in iPAH patients. However, it is difficult to relate cardiac function at rest with exercise parameters. In addition, restoring iron levels at the cellular level might have beneficial effects in the long term by improving cellular oxygen handling in the RV, similar to the alterations in skeletal muscles after intravenous iron treatment.
Alterations in skeletal muscle oxygen handling may have been responsible for the improved exercise endurance capacity with iron treatment. A relationship between skeletal muscle ID and reduced exercise capacity has been suggested previously in ID rats and humans (without PH).17,19,22,23 We showed an increase in myoglobin and mitochondrial oxidative capacity in low oxidative cells after iron therapy, suggesting a higher capacity for intracellular oxygen transport.
Iron and the lungs
Besides negatively influencing oxygen transport, it has been suggested that ID also has direct effects on the lungs. Under hypoxic conditions and at high altitude, ID resulted in an increase in pulmonary arterial pressure in otherwise healthy subjects.24,25 This has been mainly attributed to stabilization and transcription of hypoxia-inducible factors linked to contraction, proliferation, and migration of pulmonary artery smooth muscle cells.25–27 Another proposed concept is that (ID-induced) anemia may limit the conversion of nitrite to nitric oxide, thereby inhibiting the antiproliferative and vasodilatation effects of nitric oxide on the pulmonary vasculature.28–30 Because the present study did not include right heart catheterization, we can only hypothesize about the alterations in the pulmonary circulation in ID iPAH patients. However, Howard et al. 31 are currently performing a similar clinical study in the United Kingdom in which right heart catheterization is performed, and this study may provide more mechanistic insights into pulmonary vascular changes.
Limitations
The two main limitations of the present study were that we did not include a placebo group and right heart catheterization was not performed. However, a larger placebo-controlled trial that includes this measurement is currently pending in the United Kingdom. 31
Conclusions
Intravenous iron therapy in ID iPAH patients increased iron stores and was well tolerated without significant adverse events. 6MWD was unaltered by iron therapy, but endurance capacity improved significantly after iron therapy. This could not be explained by altered RV function at rest; however, increased skeletal muscle oxygen handling at the cellular level might be a cause.
Footnotes
ACKNOWLEDGMENTS
We thank Frank Oosterveer, Iris van der Mark, Martha Wagenaar, Colette ter Heerdt, Pia Trip, Sylvia Bogaards, John Wharton, Patrick Jak, Jerica Sinkeldam, Rita Visser, Tim Marcus, and Lara Konijnenberg for their help in patient inclusion and clinical patient care.
