Abstract
Pulmonary hypertension has a marked detrimental effect on quality of life and life expectancy. In a mouse model of antigen-induced pulmonary arterial remodeling, we have recently shown that coexposure to urban ambient particulate matter (PM) significantly increased the thickening of the pulmonary arteries and also resulted in significantly increased right ventricular systolic pressures. Here we interrogate the mechanism and show that combined neutralization of interleukin 13 (IL-13) and IL-17A significantly ameliorated the increase in right ventricular systolic pressure, the circumferential muscularization of pulmonary arteries, and the molecular change in the right ventricle. Surprisingly, our data revealed a protective role of IL-17A for the antigen- and PM-induced severe thickening of pulmonary arteries. This protection was due to the inhibition of the effects of IL-13, which drove this response, and the expression of metalloelastase and resistin-like molecule α. However, the latter was redundant for the arterial thickening response. Anti-IL-13 exacerbated airway neutrophilia, which was due to a resulting excess effect of IL-17A, confirming concurrent cross inhibition of IL-13- and IL-17A-dependent responses in the lungs of animals exposed to antigen and PM. Our experiments also identified IL-13/IL-17A-independent molecular reprogramming in the lungs induced by exposure to antigen and PM, which indicates a risk for arterial remodeling and protection from arterial constriction. Our study points to IL-13- and IL-17A-coinduced inflammation as a new template for biomarkers and therapeutic targeting for the management of immune response–induced pulmonary hypertension.
INTRODUCTION
Pulmonary hypertension has a major effect on quality of life and life expectancy1–3 with impairment of two organ systems, the lungs and the right heart. Remodeling and constriction of the pulmonary arteries are causes of the increase in pressure in the pulmonary circulation. 1 Inflammation with an increase in the activity of pathogenic T cells 4 and macrophages 5 is a hallmark of different types of pulmonary hypertension, 6 particularly when associated with autoimmunity (systemic sclerosis, systemic lupus erythematosus), 7 chronic obstructive pulmonary disease, 8 or helminth parasite infections (schistosomiasis),9,10 and also of idiopathic pulmonary hypertension. 11 The right heart develops structural changes (hypertrophy, dilation, fibrosis) as well as metabolic and molecular reprogramming.12–14
Although often unrecognized, a variety of environmental and occupational exposures are associated with the development of pulmonary hypertension. Prime examples are silicosis and cigarette smoke–induced pulmonary hypertension associated with chronic obstructive pulmonary disease.15,16 Our group has recently shown that exposure of immunized mice to a weak antigen that induces type 2 T helper (Th2) responses results in severe thickening of approximately one-quarter of the pulmonary arteries. 17 Further studies showed that when the immunized mice were coexposed to the weak antigen and particulate matter (PM) collected from urban air, the abundance of severely thickened arteries in the lungs was significantly increased, and pulmonary hypertension, measured as an increase in right ventricular pressures, was induced. 18
In this study, we sought to determine the molecular mechanism of pulmonary hypertension induced by coexposure to antigen and PM. We focused our studies on interleukin 13 (IL-13) and IL-17A and the mediators controlled by these cytokines as well as on known risk markers of pulmonary arterial remodeling and constriction (Fig. 1). Clinical data point out the significance of IL-13 and IL-17A for pulmonary hypertension associated with chronic inflammation. In particular, studies of pulmonary hypertension associated with systemic sclerosis showed correlation of markers for Th17 and Th2 responses, including IL-17A and IL-13, with disease severity.19–21 Additionally, two Th17-associated markers, neutrophils 22 and IL-6, 23 have long been implicated in idiopathic pulmonary hypertension. Th17 responses eliciting IL-17A have been identified as pathogenic mechanisms in many autoimmune diseases24–27 and in silica particle exposure–induced lung inflammation in mice. 28 IL-13 is an important Th2 effector molecule, particularly in asthma.29–32 IL-13 and IL-17A are thought to be independent and cross-inhibitory inducers of inflammation and associated tissue destruction/remodeling.24,25 Surprisingly, our studies show cooperative effects despite concurrent subtle cross inhibition, pointing to a therapeutic strategy of simultaneously targeting both IL-13 and IL-17A for immune response–induced pulmonary hypertension.
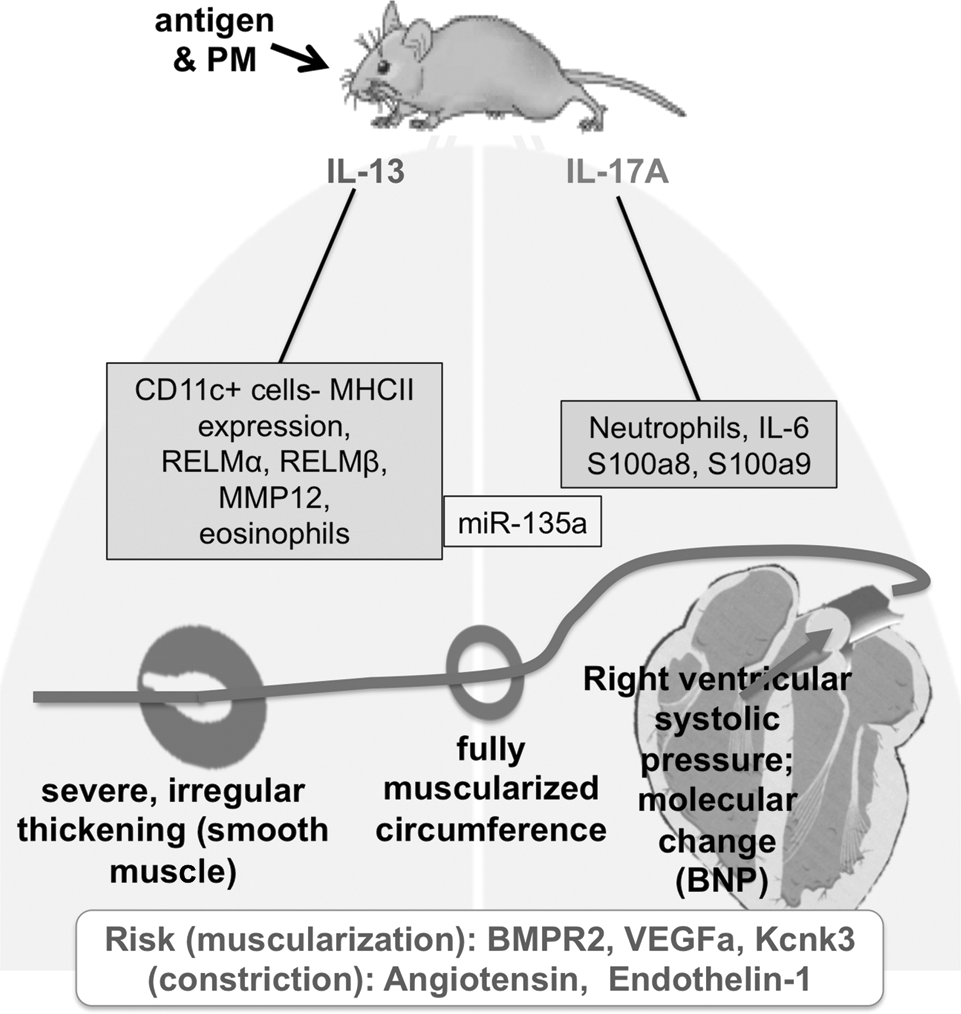
Illustration depicting the major pathways being examined in mice exposed to antigen and particulate matter (PM) with respect to responses in the pulmonary artery (severe thickening, circumference muscularization) and the right heart (right ventricular systolic pressure, molecular expression changes). We tested the hypothesis that the interleukin 13 (IL-13)- and the IL-17A-dependent molecular pathways would be independent mechanisms that induce the pulmonary hypertension phenotype. CD11c+ cells: bronchoalveolar lavage macrophages or dendritic cells that express CD11c; MHCII: major histocompatibility complex class II; RELM: resistin-like molecule; MMP: matrix metallopeptidase; miR: microRNA; S100a: S100 calcium binding protein A; BNP: brain natriuretic peptide; BMPR: bone morphogenetic protein receptor; VEGF: vascular endothelial growth factor; Kcnk3: potassium channel, subfamily K, member 3. A color version of this figure is available online.
METHODS
Ethics statement
All animal experiments were performed according to guidelines outlined by the US Department of Agriculture and the American Association of Laboratory Animal Care under the supervision and specific approval of the Institutional Animal Care and Use Committees (IACUCs) at New York University Medical Center, New York, New York (IACUC no. 111107-03; most recent approval date, November 18, 2013).
Mice
C57BL/6J (wild-type) mice were purchased from the Jackson Laboratory. Stock Retnla?/? BALB/c background mice were provided by Dr. Marc E. Rothenberg (Cincinnati Children's Hospital Medical Center).33,34 In the Grunig laboratory, the animals were backcrossed to the C57BL/6 strain for six generations, and heterozygous Retnla+/? mice were interbred to generate wild-type and knockout littermate mice.
Mostly female mice (90% of the animals were female, 10% male) were used for the study, being 5–7 weeks of age at the start of the experiment. This is an important aspect of the animal model because women are overproportionally affected by pulmonary hypertension. For each experiment, littermate mice were randomized into cages holding up to four mice each. The mice were housed under specific pathogen–free conditions at the Department of Environmental Medicine, New York University (NYU) Medical Center, Tuxedo, New York.
Urban PM18
Urban PM was captured from a roof of a low-rise building in New York City. 35 A large batch of PM2.5 from New York ambient air has been prepared and is available to all interested investigators on request. This is size fractionated, and only the PM2.5 fraction was used. This PM2.5 was provided by Dr. Terry Gordon's laboratory at the NYU Medical Center, resuspended, and ultrasonicated before use. 35 This fraction was chosen because the size of <2.5 μm in diameter allows the particles to travel via the airways into the alveoli. The dose chosen was at half the concentration of the dose reported to induce significant airway inflammation using PM2.5 sampled in New York City, 35 Beijing, 36 and Baltimore.37–39 PM2.5 was given intranasally at a dose of 25 μg/50 μL in phosphate-buffered saline (PBS) combined with ovalbumin (OVA; see below). When given without OVA, PM2.5 at the 25-μg/50-μL dose did not elicit significant airway inflammation or vascular remodeling. 18
Antigen immunization and exposure to antigen and PM
Animals were primed and challenged with antigen as described elsewhere17,18 and schematically presented in Figure 2A. In brief, mice were injected intraperitoneally with OVA (grade 5; Sigma, St. Louis, MO; 50 μg/dose) adsorbed to alum (Imject Alum; Thermo Fisher Scientific, Rockford, IL; 2 mg/dose) to prime the immune response at a 2-week interval. Two weeks later, the mice were intranasally challenged with either PBS or combined OVA (100 μg/dose) and PM2.5 (25 μg/dose) two times each week, for a total of six doses given over a 3-week period. This exposure regimen has been shown to induce severe arterial remodeling17,18 (just like antigen exposure by nebulization 17 ), and when antigen exposure was combined with PM, significant increases in right ventricular systolic pressures (RVSPs) were measured. 18 To verify that the antigen and PM delivery elicited the expected inflammation and that the PM dose used for the study did not induce microscopic signs of inflammation when given alone, the lungs were scored for airway and interstitial (alveolar) inflammation (Fig. 2B, 2C) as described elsewhere.17,40–42 Airway (peribronchial, perivascular) inflammation was scored on 20 or more consecutive view fields, as follows: 1, normal with very few inflammatory cells; 2, scattered inflammatory cells up to two rings in depth; and 3, cuffs of inflammatory cells measuring three rings or more in depth. Interstitial (alveolar) inflammation was scored on 20 or more consecutive view fields, as follows: 1, normal; 2, increased numbers of cells within the alveoli; and 3, consistent increase in the numbers of cells within the alveoli, appearance of multinucleated giant cells, and thickening of the alveolar septa. All measurements were performed without prior knowledge of the group designation of the lungs.
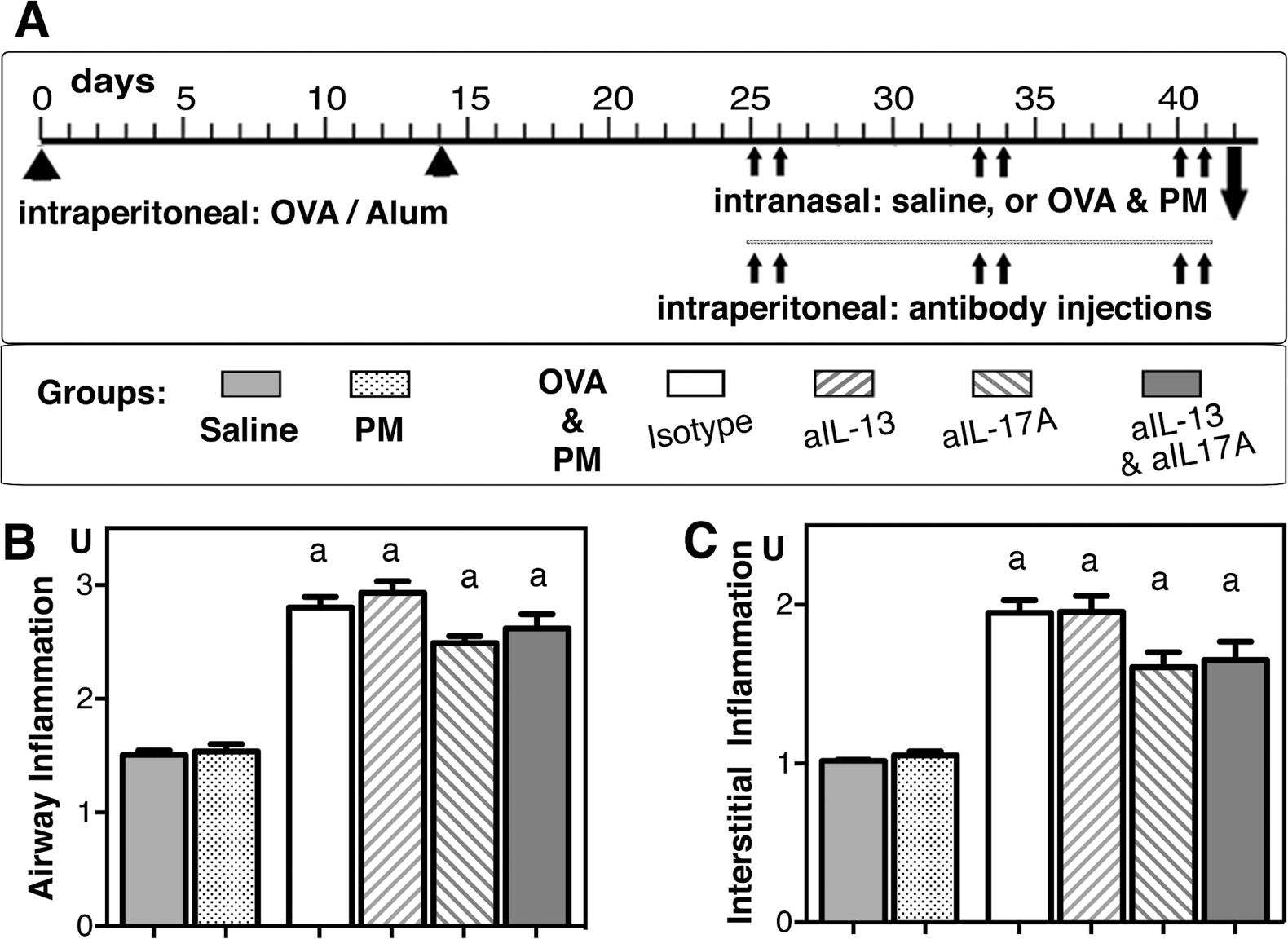
Experimental schedule and inflammation (airway/interstitial) induced by exposure to antigen and particulate matter (PM). A, Timeline showing the timing of the priming injections with ovalbumin (OVA)/alum, the dosing of intranasal challenges with saline or antigen and PM (OVA&PM), and the antibody injections administered intraperitoneally. B, C, Bar graphs showing scores for airway (peribronchial/perivascular) inflammation (B) and interstitial (alveolar) inflammation (C). Groups of mice were exposed to saline, PM, or OVA&PM and given isotype control or neutralizing antibodies. A score of 1 was given for normal morphology, 2 for mild infiltrates, and 3 for severe infiltrates of inflammatory cells. Data were pooled from 2–3 experiments, n = 5–20 per group. P < 0.0001 for group comparisons with saline (a). IL: interleukin. A color version of this figure is available online.
Antibody injections
Wild-type mice were injected intraperitoneally with 150 μg of anti-mouse-IL-13 (Ebioscience, San Diego, CA; clone eBio1316H), anti-mouse-IL-17A (BioXCell, West Lebanon, NH; clone MM17F3 43 ), or isotype control (rat immunoglobulin G [IgG] 1, mouse IgG1; Biolegend, San Diego, CA, or BioXCell, respectively) monoclonal antibodies prior to each of the intranasal challenges. The antibody preparations were of cell culture/experimental animal usage grade (<2 EU lipopolysaccharide/mg). The dose of monoclonal antibody used (6.8 mg/kg body weight) is within the dose range used clinically in human patients (2–3 mg/kg body weight).
Functional evaluation and tissue collection
RVSP was measured after inserting a catheter via the jugular vein in anaesthetized, spontaneously breathing mice.40,44 Right ventricular hypertrophy was determined as right ventricular weight relative to the weight of the left ventricle and septum. 44
Bronchoalveolar lavage (BAL) and tissue harvest were performed after right heart catheterization and euthanasia of animals.17,40,44 BAL was performed by gently washing with three 1-mL aliquots of Hank's balanced salt solution. Following BAL, lungs and ventricles were recovered. The right lung lobe was snap-frozen in liquid nitrogen, and the remainder of the lungs were inflated with buffered formaldehyde and placed in formaldehyde for histology. In our laboratory, BAL does not wash out all inflammatory cells from the lungs and is performed gently to minimize the potential for mechanical damage of the tissue. The above protocol allows us to reduce the number of animals used for each study. All measurements were performed without prior knowledge of the group designation of mice.
Cirumferential muscularization of lung blood vessels was measured following immunohistochemical staining of lung sections with anti–von Willebrand factor and anti–smooth muscle actin, as described elsewhere. 40 Blood vessels <100 μm in diameter were analyzed for the circumference length of smooth muscle actin staining relative to the length of the vessel circumference. For each lung, at least 20 vessels (mean, 128) were analyzed. Dual immunohistochemistry was performed with a rabbit anti–von Willebrand factor antibody (Dako, Glostrup, Danemark) recognizing endothelial cells and goat anti–smooth muscle actin antibody (Everest Biotech, Upper Heyford, United Kingdom) to detect muscularization, as described elsewhere.40,45,46 Slides were scanned using an Aperio XX slide scanner, and the images were analyzed using VisiomorphDP software (Visiopharm, Hoersholm, Denmark). From each slide at least 20 vessels (mean, 128) were identified and analyzed for muscularization by calculating the length of smooth muscle actin staining relative to the length of the vessel circumference for vessels <100 μm in diameter.
The vessels were categorized into three groups—nonmuscularized (<0.3), partially muscularized (0.3–0.8), and fully muscularized (>0.8)—and for each lung the percentage of vessels per group was calculated (sum of nonmuscularized + partially muscularized + fully muscularized was 100). For each lung, the ratio of nonmuscularized to fully muscularized blood vessels was also calculated, using the formula (% nonmuscularized)/(% fully muscularized).
Severe arterial thickening was determined on lung sections stained with hematoxylin and eosin. The severe arterial thickening change induced by prolonged exposure to antigen has been described in detail by our group. 17 The number of arteries (blood vessels adjacent to airways) at 200× magnification that showed severely thickened walls characterized by disorganized layers of cells (cells in the blood vessel wall assume a pattern that differs from that of the lumen) 17 were counted relative to all arteries scored per lung. All arteries from at least 20 consecutive view fields comprised the sum of arteries with normal (thin) walls, mildly thickened walls, or the above-mentioned severely thickened walls. For each lung, severe arterial thickening was calculated using the following formula: 100/number of all arteries examined × number of severely remodeled arteries. The muscularized nature of the severe thickening lesions 17 was confirmed by immunohistochem-istry with anti–smooth muscle actin staining. Digital photomicrographs were obtained from images captured by scanning the lung slides using the Leica SCN400F whole-slide scanning system at the Histopathology Core of the NYU Medical Center and capturing images via SlidePath's Digital Image Hub (Leica Biosystems, Buffalo Grove, IL).
Messenger RNA (mRNA) and microRNA (miRNA) expression
Total RNA from lung or right heart tissue was isolated using the RNeasy Mini Kit (Qiagen, Valencia, CA). Reverse transcription was performed using the High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems/Life Technologies, Grand Island, NY) for mRNA and the Universal RT cDNA Synthesis Kit (Exiqon, Woburn, MA) for miRNA, with locked nucleic acid–modified primers (Exiqon). Quantitative polymerase chain reaction (qPCR) was performed in duplicate with 20 ng of complementary DNA (cDNA) for mRNA or 0.1 ng of cDNA for miRNA, using the 7900HT Fast Real-Time PCR System (Applied Biosystems).
qPCR for the detection of mRNA expression was performed with SYBR Green (Invitrogen, Grand Island, NY). For the specific detection of the homologous genes Retnla and Retnlb, we used the TaqMan Gene Expression Assay (Applied Biosystems) based on a FAM-labeled probe and the corresponding TaqMan Gene Expression Assay for β-actin. The sequences for primers and probes are indicated in Table 1. The following conditions were used: 95°C for 10 minutes, followed by 45 cycles of 95°C for 15 seconds and 60°C for 1 minute, followed by a hold at 4°C. The miR-135a expression level was determined by using miR-135a primer (Exiqon) and SYBR Green Master Mix (Exiqon) with these conditions: 95°C for 10 min, followed by 45 cycles of 95°C for 10 seconds and 60°C for 1 minute, followed by a hold at 4°C.
Sequences of primers and probes

Note: BMPR: bone morphogenetic protein receptor; F: forward; R: reverse; VEGF: vascular endothelial growth factor; IL: interleukin; IFN: interferon; MMP: matrix metallopeptidase; BNP: brain natriuretic peptide; RELM: resistin-like molecule.
Raw data were analyzed with SDS Relative Quantification Software (ver. 2.3; Applied Biosystems) to determine cycle threshold (Ct). Mean Ct values were standardized by calculating ΔCt using the housekeeping gene β-actin (mRNA) or 5S (miRNA) and calculating 1.98ΔC × 10,000.
Immune responses
BAL cells were analyzed by flow cytometry as described elsewhere,17,41,47 using a MACS Quant instrument (Miltenyi Biotec, Auburn, CA) and FloJo software (TreeStar, Ashland, OR). Cell counts were performed using the feature provided by the MACS Quant flow cytometer. All cell analysis was performed without prior knowledge of the group designation of the samples. BAL samples were analyzed as described elsewhere40,41 for the presence of eosinophils (CD11bhigh, CCR3high, GR1low, CD11clow-intermediate, MHCIIlow-intermediate), neutrophils (CD11bhigh, GR1high, CD11clow-intermediate, MHCIIlow-intermediate), lymphocytes (cells expressing CD4 or CD8 and falling into the lymphocyte gate by forward- and side-scatter profile), and CD11c+ cells that coexpress CD205. The mean fluorescence intensity of CD11c+ cells stained with anti-MHCII antibody was determined as a measure of the capacity of these airway dendritic cells/airway macrophages to present antigen.41,47 The cell populations were analyzed using monoclonal antibodies tagged with Pacific Blue, AlexaFluor, fluorescein isothiocyanate, phycoerythrin, peridinin-chlorophyll, allophycocyanin, or cyanine tandem dyes that were purchased from BD Biosciences (San Jose, CA), Ebioscience (San Diego, CA), or Biolegend (San Diego, CA).
Statistical analysis
Statistical analysis was performed with Aable (ver. 3; Gigawiz, Tulsa, OK) or Prism (ver. 6; Graphpad, San Diego, CA) software. Two-group comparisons were conducted with the unpaired two-tailed Mann-Whitney U test or the unpaired two-tailed t test with Welch's correction for unequal variances. Data sets were analyzed for correlation using the Spearman rank correlation test. Contingency tables were analyzed with the χ 2 test for multigroup comparisons and the two-tailed Fisher exact test for two-group comparisons. Differences with P < 0.05 were considered significant.
RESULTS AND DISCUSSION
Exposure to antigen and PM induces increased RVSP and circumferential pulmonary arterial muscularization via IL-13 and IL-17A
To understand the mechanism by which exposure to a weak antigen (OVA) and ambient PM2.5 induced this phenotype, 18 groups of mice were given neutralizing anti-IL-13 and/or anti-IL-17A antibodies or control antibodies. RVSPs are correlated with pulmonary arterial pressures. We analyzed the medians and data spread by box plot (Fig. 3A) as well as the distribution of data within each group of mice by showing the number of animals with RVSP values below or above a 26-mmHg threshold (Fig. 3B). Both types of analysis showed that the increase in RVSP induced by exposure to OVA and PM was significantly attenuated by coneutralization of IL-13 and IL-17A. Blocking either IL-13 or IL-17A (Fig. 3A, 3B) had no significant effects. Furthermore, the circumferential muscularization of blood vessels in the lungs (Fig. 3C) was significantly attenuated by coneutralization of IL-13 and IL-17A, as indicated by the restoration of the predominance of nonmuscularized vessels over fully muscularized vessels (Fig. 3D).
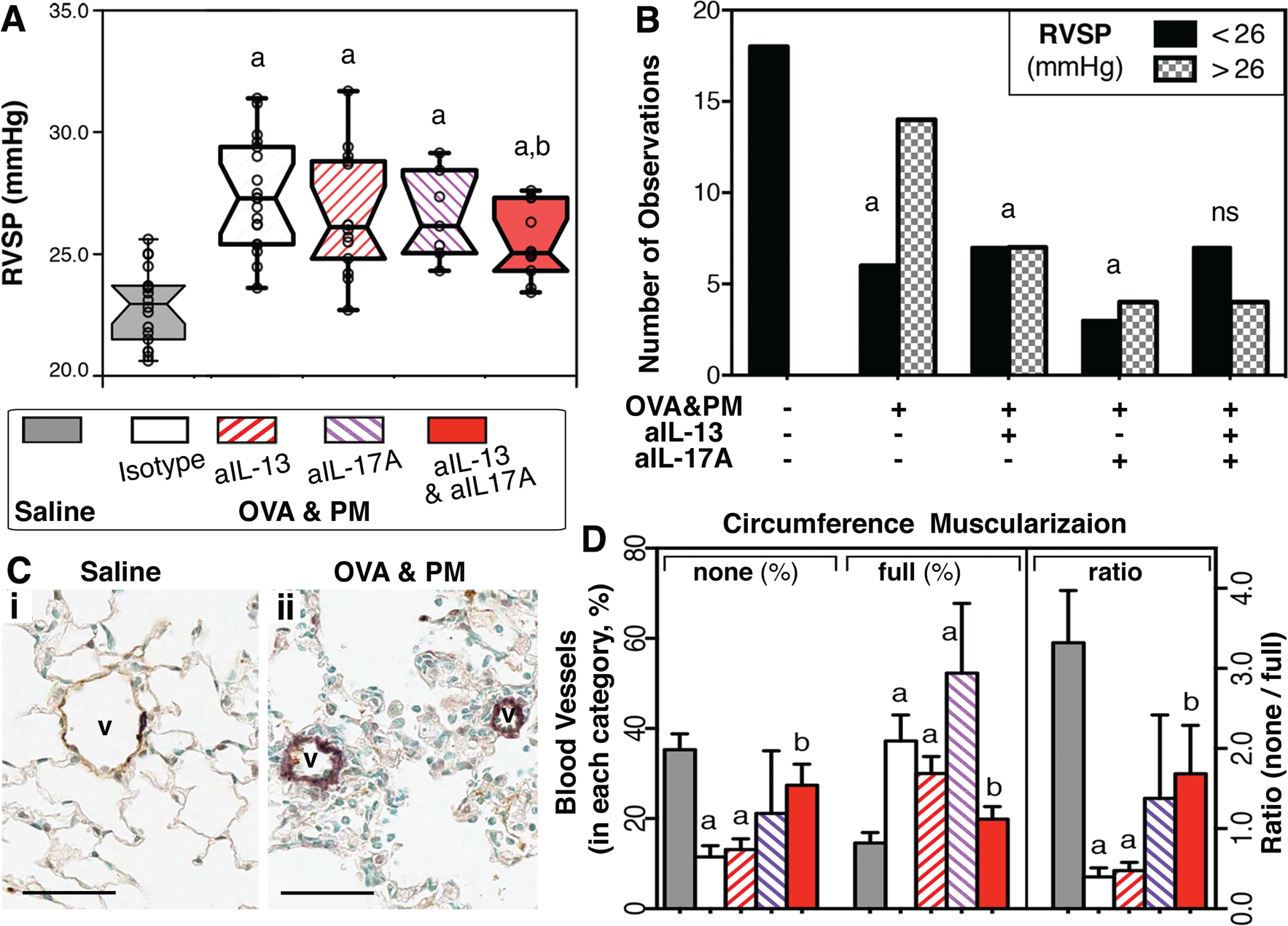
Coblockade of interleukin 13 (IL-13) and IL-17A inhibits the increase in right ventricular systolic pressure (RVSP) and the circumferential muscularization of lung blood vessels in response to exposure to antigen and particulate matter (PM). RVSP data are shown as a box plot with circles indicating individual data points (A) or as a bar graph showing the number of observations of RVSP less than or greater than 26 mmHg (B); n = 7–18 per group. C, Photomicrographs of representative sections of lungs stained with anti–smooth muscle actin (brown; scale bars: 50 μm; v: blood vessel). D, Bar graphs showing mean and SEM values (n = 5–12 per group) for circumferential muscularization as a percentage of blood vessels that were in the category none (<30% of the circumference positive for smooth muscle actin staining) or full (>80% of the circumference positive) as well as for the ratio of none to fully muscularized arteries. Mean and SEM values for the category of partially muscularized arteries were as follows: 50.08 ± 2.47 (saline), 51.2 ± 4.65 (ovalbumin [OVA] and PM [OVA&PM], isotype), 56.8 ± 3.91 (OVA&PM, anti-IL-13), 26.6 ± 7.64 (OVA&PM, anti-IL-17A), and 52.7 ± 3.62 (OVA&PM, anti-IL-13 and anti-IL-17A). Data from groups of mice exposed to saline or antigen and PM and given neutralizing or isotype control antibodies were pooled from 2–3 experiments. P < 0.05 for group comparisons with saline (a) or OVA&PM–control antibody (b). ns: not significant for comparison with the saline group.
Exposure to antigen and PM induces molecular change in the right ventricle that is dependent on IL-13 and IL-17A
The data in Figure 3 show that coexposure to antigen and PM induced modest but significant increases in RVSP. This was similar to that reported for other mouse models of pulmonary hypertension induced by hypoxia, 48 a strong antigen, 49 or a weak antigen combined with the presence of a transgene with a hypomorphic mutation in bone morphogenetic protein receptor type 2 (BMPR2). 40 To understand the biological significance, we examined right ventricles for signs of molecular change because right ventricular remodeling and ultimately failure are the cause of the impairment of physical activity and mortality associated with pulmonary hypertension in human patients.12,13 We examined the expression of brain natriuretic peptide (BNP, Nppb) because it is recognized as a marker of right ventricular strain in humans. 3 Compared with the saline-exposed group, the mice exposed to antigen and PM had significantly increased expression of Nppb in the right ventricle, and this was inhibited by coneutralization of IL-13 and IL-17A (Fig. 4A). Right ventricular Nppb expression was significantly associated with right ventricular weight, a measure of right heart hypertrophy (P = 0.003), and RVSP (P = 0.029; Fig. 4B, 4C). The tightest correlation was between right ventricular Nppb and the numbers of BAL neutrophils (P = 0.0002; Fig. 4D), while there was no correlation with BAL eosinophils (Fig. 4E). Right ventricular weights were also significantly correlated with RVSP (P = 0.016; Fig. 4F). These correlations suggest that the right ventricular molecular change in Nppb expression was associated, as expected, with the increased pressures in the pulmonary circulation and independently with inflammation in the lungs. The latter association has also been suggested by studies in human patients. 12 Right ventricular weights (weight of right ventricle/weight of the left ventricle + septum) were not significantly increased by exposure to OVA and PM (mean ± SD, 0.204 ± 0.019 for saline vs. 0.222 ± 0.028 for OVA and PM). This is likely due to the ability of the mice to adapt to the pulmonary arterial remodeling and the increase in right ventricular pressure or to the insensitivity of this measure for mild changes.
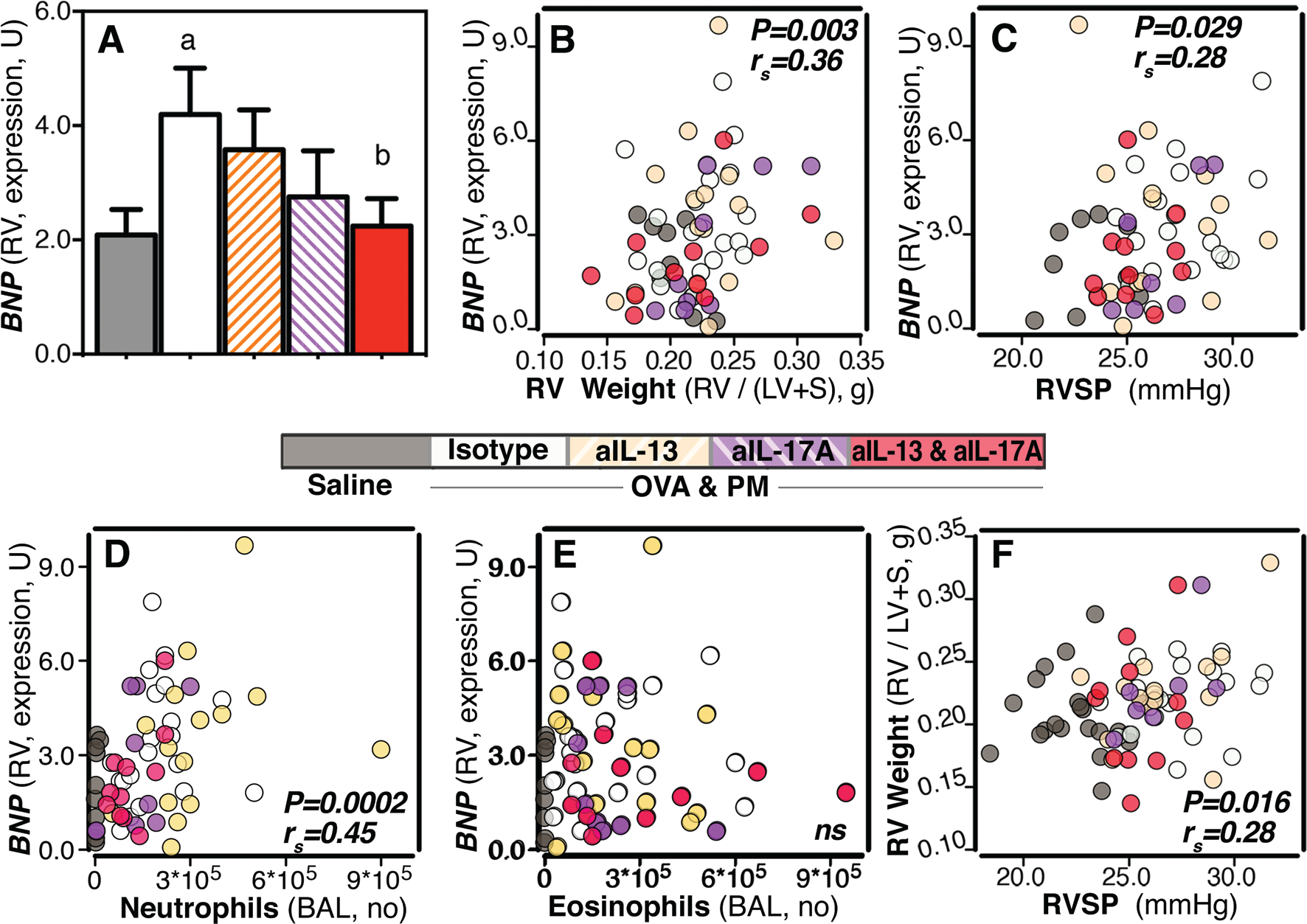
Molecular change is induced in the right ventricle (RV) by exposure to antigen and particulate matter (PM) via interleukin 13 (IL-13) and IL-17A and is correlated with airway neutrophils. A, Bar graph of expression of the brain natriuretic peptide (BNP) gene Nppb in the RV in mice exposed to saline or to antigen (ovalbumin [OVA]) and PM (OVA&PM) and given neutralizing or isotype control antibodies. Data are means and SEM. P < 0.05 for group comparisons with saline (a) or OVA&PM–control antibody (b). B–E, Nppb expression in the RV plotted against RV weight (RV/left ventricle plus septum [LV+S]; B), right ventricular systolic pressure (RVSP; C), numbers of neutrophils in bronchoalveolar lavage (BAL) fluid (D), and numbers of eosinophils in BAL fluid (E). F, RV weight (RV/LV+S) plotted against RVSP. P and rs (tie corrected) values for Spearman's rank correlation test are shown (ns: not significant). Data were pooled from 2–3 experiments, n = 5–20 per group. Nppb expression is given relative to that of β-actin (set at 10,000 U).
Neutralization of IL-13 and IL-17A reveals their concurrent activity in the lungs of mice exposed to antigen and PM
The cooperative activity of IL-13 and IL-17A for right ventricular pressure, circumferential pulmonary vascular muscularization, and right ventricular strain was surprising because of the cross inhibition of Th17 and Th2 development and the reported metastability and adaptability of Th17 cells.24,50 Therefore, we wanted to know whether IL-13- and IL-17A-dependent changes would occur at the same time (concurrently). BAL cells and lung RNA were probed for direct downstream effectors in the Th2 (Fig. 5A–5E) and Th17 (Fig. 5F–5I) pathways, respectively. The levels of MHCII expression by CD11c+ cells in BAL fluid, as a marker of activation of macrophages and dendritic cells, 47 and expression of Retnla, Retnlb, and matrix metallopeptidase 12 (Mmp12) were decreased by IL-13 neutralization (Fig. 5A–5D). The numbers of BAL eosinophils remained unaffected (Fig. 5E). The numbers of BAL neutrophils and expression of the myeloid cell effector molecules 51 S100 calcium binding protein A8 (S100a8), S100a9, and Il6 were decreased by IL-17A neutralization (Fig. 5F–5I).
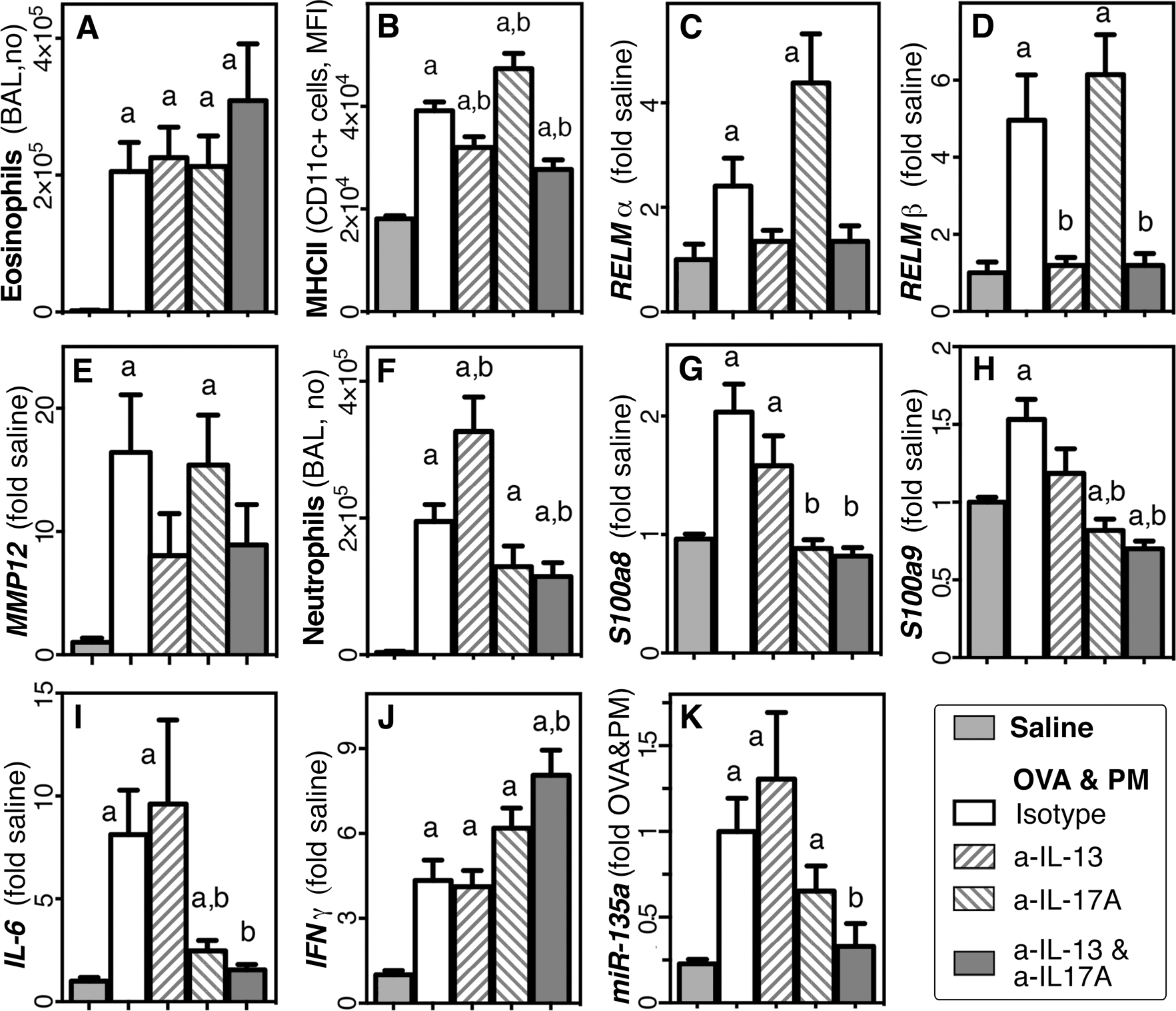
Concurrent cellular and molecular activity of interleukin 13 (IL-13) and IL-17A in the lungs of mice exposed to antigen and particulate matter (PM). Bar graphs show mean and SEM values for numbers of eosinophils in bronchoalveolar lavage (BAL) fluid (A); major histocompatibility complex class II (MHCII) expression by CD11c+ cells from BAL fluid (mean fluorescent intensity [MFI]; B); RNA expression in the lungs for the resistin-like molecule α (RELMα) gene (Retnla; C), the RELMβ gene (Retnlb; D), and the matrix metallopeptidase 12 (Mmp12) gene (E); numbers of neutrophils in BAL fluid (F); and RNA expression in the lungs for S100a8 (G), S100a9 (H), the IL-6 gene (Il6; I), the interferon γ gene (Ifng; J), and miR-135a (K). Groups of mice were exposed to saline or to antigen (ovalbumin [OVA]) and PM (OVA&PM) and given neutralizing or isotype control antibodies (n = 5–20 per group, pooled from 2–3 experiments). P < 0.05 for group comparisons with saline (a) or OVA&PM–control antibody (b). RNA expression levels were calculated relative to the means of the saline or OVA&PM group, as indicated. A color version of this figure is available online.
Th2 and IL-17A responses to Aspergillus conidia challenges 52 or interferon γ (IFN-γ)- and IL-17A-producing T cells from atherosclerotic coronary arteries in humans 53 and inflamed mouse tissue 54 have been reported to coexist consecutively or concurrently. To distinguish between consecutive and concurrent activity of IL-13 and IL-17A, we looked for cross inhibition of effector responses. Our data showed significant increases in (a) MHCII expression by BAL CD11c+ cells in mice given anti-IL-17A (Fig. 5B) and (b) numbers of BAL neutrophils in mice given anti-IL-13 (Fig. 5F). In both cases, coneutralization of IL-13 and IL-17A showed that the exacerbated response elicited by single-cytokine blockade was due to enhanced IL-17A or IL-13 effects, respectively (Fig. 5B, 5F). Coneutralization of IL-13 and IL-17A significantly enhanced the expression of Ifng (Fig. 5J), suggesting the independent activation of the Th1 pathway in mice exposed to antigen and PM, and significantly inhibited the expression of miR-135a (Fig. 5K). miR-135a is upregulated in the allergen-challenged lung, 55 targets several components of cytokine signaling, and may participate in the control of the IL-13/IL-17A-dependent response to coexposure to antigen and PM.
Severe pulmonary arterial thickening induced by antigen and PM has an independent mechanism
Severe thickening and irregular pattern formation by smooth muscle cells is induced in pulmonary arteries in mice exposed to only antigen via IL-13, 17 and this response occurs in IL-17A-deficient mice. 52 IL-13 has also been shown to be a critical mediator in experimental Schistosoma infection–induced pulmonary arterial remodeling. 56 As shown schematically in Figure 1, this type of remodeling is phenotypically different from circumferential muscularization. Circumferential muscularization is typically induced by hypoxia, 48 and in our studies of mice exposed to antigen and PM it was inhibited by IL-13 and IL-17A coneutralization (Fig. 3). The simultaneously occurring arterial wall thickening was quantified by determining for each lung the percentage of pulmonary arteries that were severely thickened and also showed irregular sheets of smooth muscle cells (Fig. 6A).
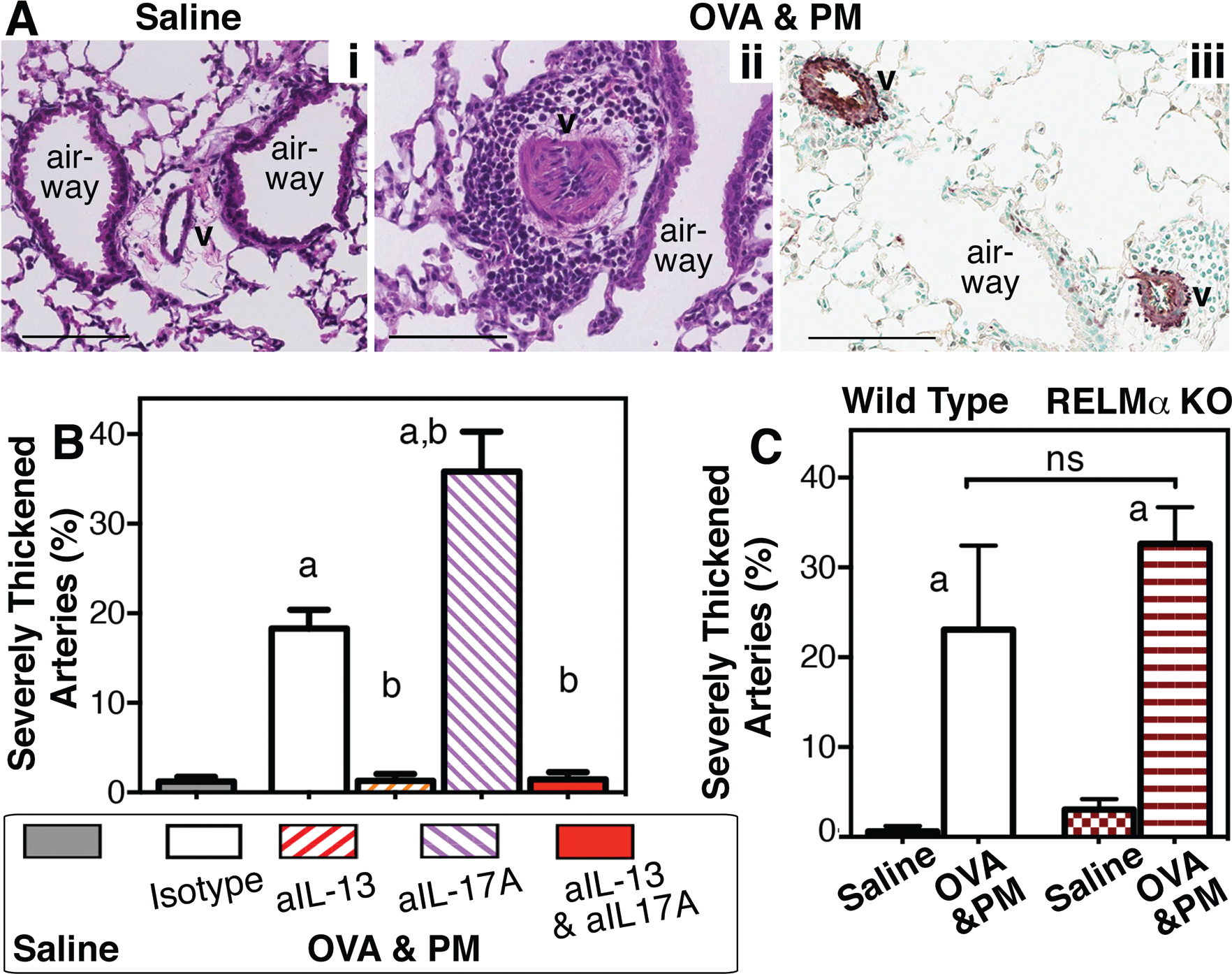
Interleukin 17A (IL-17A) protects against widespread severe thickening of the pulmonary arteries in response to exposure to antigen and particulate matter (PM) by inhibiting IL-13-controlled effects with a redundant role of resistin-like molecule α (RELMα). A, Representative lung sections from a saline-exposed mouse (i) and a mouse exposed to antigen (ovalbumin [OVA]) and PM (ii, iii), stained with hematoxylin and eosin (i, ii) or with anti–smooth muscle actin (brown, iii). Vessels (v), airways, and scale bars (100 μm) are indicated. Bar graphs show mean and SEM values for percentages of severely thickened arteries per lung in groups of wild-type mice exposed to saline or OVA&PM and given neutralizing or isotype control antibodies (n = 10–27 per group, pooled from 2–3 experiments; B) or in groups of wild-type and Retnla knockout (KO) mice exposed to saline or OVA&PM (n = 7–10 per group, pooled from 2 experiments; C). P < 0.05 for group comparisons with saline (a) or OVA&PM–control antibody (b). ns: not significant.
To our surprise, injection with neutralizing anti-IL-17A antibody significantly exacerbated the severe remodeling response (Fig. 6B). This was reminiscent of findings in a mouse model of atherosclerosis. 43 In our mice, the IL-17A-mediated protection was due to the inhibition of IL-13-dependent effects because coinjection of neutralizing anti-IL-17A and anti-IL-13 antibodies resulted in profound inhibition of the severe arterial thickening response, just like anti-IL-13 antibody alone (Fig. 6B). Resistin-like molecule α (RELMα) was not necessary for the severe remodeling response that was fully present in knockout mice (Fig. 6C). This was unexpected because Retnla levels in the lungs of antigen- and PM-exposed mice were controlled by IL-13 (Fig. 5C). Furthermore, mouse RELMα and its human homologue, RELMβ, are known as hypoxia-induced mitogenic factors and are smooth muscle cell mitogens.48,57–60 As an explanation for our unexpected data, the role of RELMα in arterial thickening and remodeling could vary depending on the type of challenge (e.g., hypoxia vs. antigen).48,61
Comparing the data shown in Figures 5 and 6 suggests that airway eosinophils, neutrophils, myeloid cell activation markers S100a8 and S100a9, and IL-6 are not critical promoters of the severe arterial remodeling response and that IFN-γ is not a critical inhibitor. With respect to the role of IL-6 in severe pulmonary arterial thickening, it is of great interest that in experimental schistosomiasis the IL-6 pathway has recently been shown to have a protective function. 62 In contrast, the IL-13-dependent upregulation of MMP12 metalloelastase expression (Fig. 5E) could represent a critical step because the fragmentation of the internal elastin barrier of the vasculature allows smooth muscle cells to invade. 1 The ability of IL-13 to directly induce proliferation of smooth muscle cells is controversial;20,63 therefore, the effects could be indirect by inactivating a suppressor of the response to a mitogen that may be present ubiquitously or that may be upregulated due to exposure to antigen and PM.
Comparing Figures 3 and 6, our study suggests that circumferential blood vessel muscularization and severe pulmonary arterial thickening have different control mechanisms in mice exposed to antigen and PM (IL-13 and IL-17A induced vs. IL-17A protected via inhibition of IL-13 effects, respectively). This is surprising because one may assume that arterial thickening is a more severe form of circumferential muscularization. Our study refutes this notion in the lungs of mice exposed to antigen and PM. Instead, circumferential muscularization that organizes smooth muscle cells in a chain-like configuration versus the severe muscularization resulting in irregular sheets of smooth muscle cells could recapitulate distinct ancestral responses. For example, choanoflagellates, the closest living eukaryotic relative of animals, use distinct molecular mechanisms to form chain-shaped or rosette-shaped multicellular colonies.64,65
Exposure to antigen and PM induces a molecular risk profile in the lungs for pulmonary arterial remodeling and a protection profile for pulmonary arterial constriction
Our data left two questions open. The first question was, is the role of IL-13 in controlling severe pulmonary arterial thickening supported by molecular reprogramming in the lungs of mice exposed to antigen and PM? To address this, we looked for expression in the lungs of key molecules known to predispose for the development of pulmonary arterial remodeling by controlling smooth muscle and endothelial cells in humans and animals when under-expressed.66–70 These key molecules are BMPR2, vascular endothelial growth factor a (VEGFa), and the potassium channel Kcnk3/TASK-1.40,71–74 Here we show significantly decreased expression of all three molecules in the lungs of animals exposed to antigen and PM (Fig. 7A–7C), constituting a molecular risk profile for pulmonary arterial remodeling.
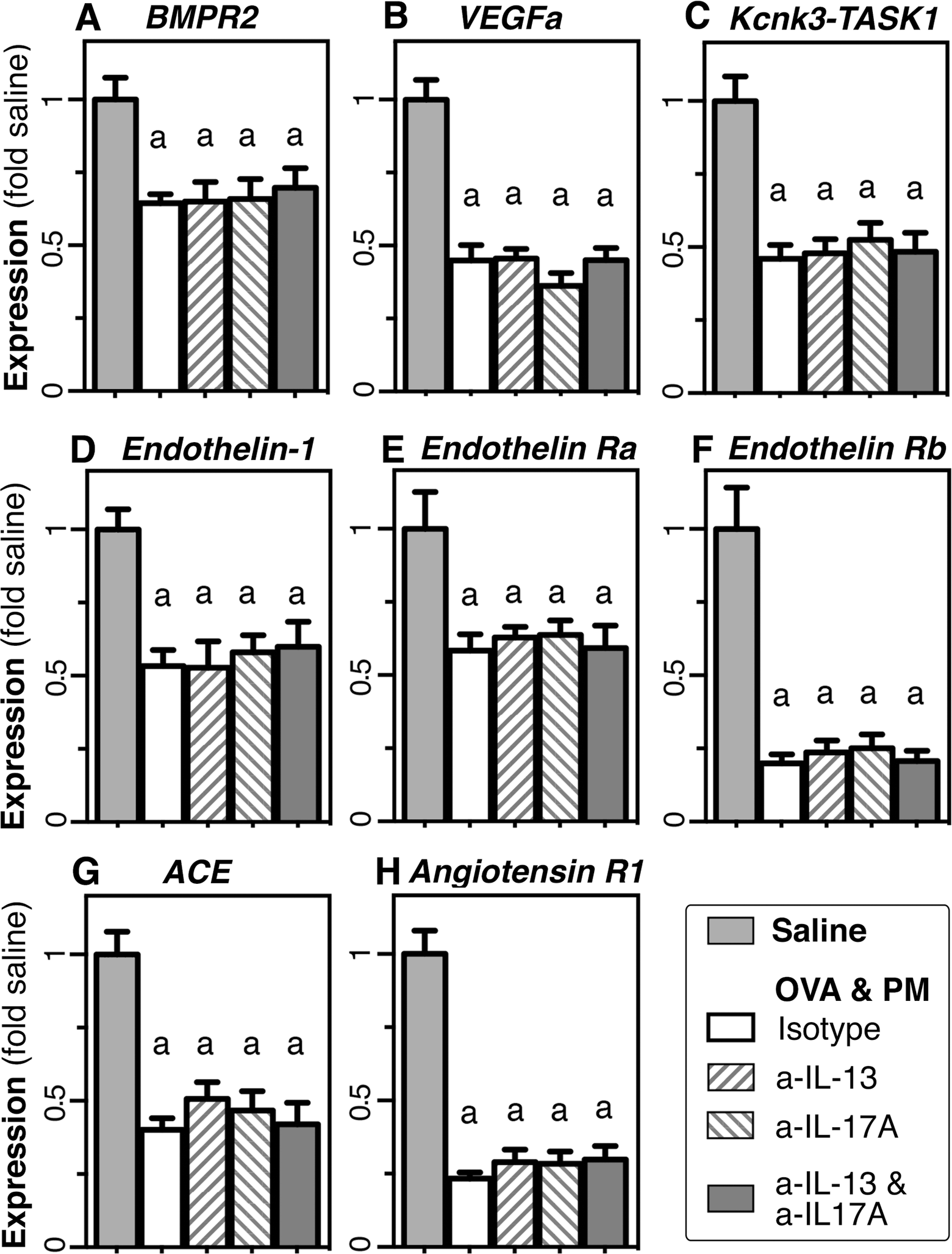
Molecular at-risk profile for arterial remodeling and protection profile for vasoconstriction in the lungs of mice exposed to antigen (ovalbumin [OVA]) and particulate matter (PM). Bar graphs show mean and SEM values for bone morphogenetic protein receptor type 2 (BMPR2; A), vascular endothelial growth factor a (VEGFa; B), the potassium channel Kcnk3/TASK-1 (C), endothelin 1 (D), endothelin receptor (R) a (E), endothelin Rb (F), angiotensin-converting enzyme (ACE; G), and angiotensin R1 (H) RNA expression in the lungs (calculated relative to the means for the saline group). Groups of mice were exposed to saline or to antigen and PM (OVA&PM) and given neutralizing or isotype control antibodies (n = 5–12 per group, pooled from 2–3 experiments). P < 0.05 for group comparisons with saline (a) or OVA&PM–control antibody (b). A color version of this figure is available online.
The second question was, why do mice exposed to antigen and PM not develop a higher degree of pulmonary hypertension despite the widespread circumferential muscularization and severe thickening of pulmonary arteries? To address this, we determined the expression of endothelin 1, angiotensin, and their receptors/enzymes needed for activation. Endothelin 1 and angiotensin are major arterial constrictors and play significant roles in pulmonary hypertension.2,75,76 Our data showed a significant downregulation of these molecules (Fig. 7D–7H), constituting a molecular protection profile for pulmonary arterial constriction.
Our data showed clearly that the concerted molecular profiles of risk for arterial wall thickening and protection from arterial constriction elicited in the lungs of animals exposed to antigen and PM are not controlled by IL-13 and/or IL-17A (Fig. 7). The transcriptional control mechanism is currently under intense inquiry because this molecular profile may enable the right ventricle to adapt to the changes in the pulmonary vasculature.
Relevance and limitations
Because of the inherent limitations of animal experimentation, our study will require follow-up work to understand its relevance for the human condition. Our study has clinically translatable implications by identifying a therapeutic target of two distinct cytokine-controlled pathways, IL-13 and IL-17A, for antigen- and PM-induced increases in RVSP, circumferential muscularization, and right ventricle molecular changes and a single gene target (IL-13) for the severe thickening of pulmonary arteries. This is exciting because of the emerging clinical success of neutralizing IL-13/IL-13 signaling30–32 and IL-17A/IL-17 signaling26,27 in diseases associated with chronic inflammation (asthma and psoriasis, respectively). Careful studies in human patients have shown increased IL-13-dependent, IL-17A-dependent, or IFN-dependent markers in pulmonary hypertension.19–23 Further translation of the findings from our study is dependent on the identification of biomarkers for an immune response that elaborates coexisting IL-13- and IL-17A-driven inflammatory pathways in pulmonary hypertension patients. This would be a necessary step toward evaluating reagents that neutralize IL-13 or IL-17A as potential therapeutic options for the clinical management of pulmonary hypertension. In addition, this would help to design studies to further understand how fine-dust exposure from air pollution could affect exacerbations of pulmonary hypertension.
Footnotes
ACKNOWLEDGMENTS
We thank Carol Hoffman and Elena Thompson for expert technical help. Airborne particulate matter (PM2.5) collected from New York City is available to investigators on request.
