Abstract
Two very different claims have been made about the effects that the selective serotonin reuptake inhibitor (SSRI) antidepressants have on suicide risk in depressed patients. First, some authors have argued that increased SSRI use has reduced population suicide rates, supporting their case with ecological evidence that suicide rates have fallen as SSRI prescribing has increased (e.g. [1]). Second, critics of SSRI prescribing have claimed that SSRIs have increased suicide risk among a minority of patients [2]. Healy et al., for example, have reported that two of 20 healthy volunteers given reboxetine and sertraline for 2 weeks developed suicidal ideation [3].
A large number of randomized controlled trials (RCTs) have established the effectiveness of SSRIs in treating depression in adults [4,5], but they have provided limited data on their impact on suicide risk for the following reasons. First, the sample sizes sufficient to establish efficacy in reducing depressive symptoms (typically fewer than 100) are too small to detect changes in suicide risk [6–8]. Second, the chances of detecting changes in suicide risk are reduced even further by the fact that RCTs often exclude patients who are adjudged to be at high risk of suicide [9]. Third, few studies have been designed to investigate the iatrogenic effects of SSRIs (or other antidepressants) on suicidal thoughts and behaviour [9].
We review the evidence relating to these two causal claims about SSRIs and suicide from three relevant study types: (i) meta-analyses of RCTs of the safety and effi- cacy of SSRIs; (ii) observational studies of the relationships between suicidal behaviour and SSRI and other antidepressant prescribing; and (iii) ecological studies of the relationships between SSRI prescribing and suicide mortality. Each of these methods has its strengths and limitations that are discussed below and summarized in Table 1. We looked for a convergence of results from these three types of study while taking account of their complementary advantages and disadvantages.
Strengths and weaknesses of available evidence on SSRIs and suicide
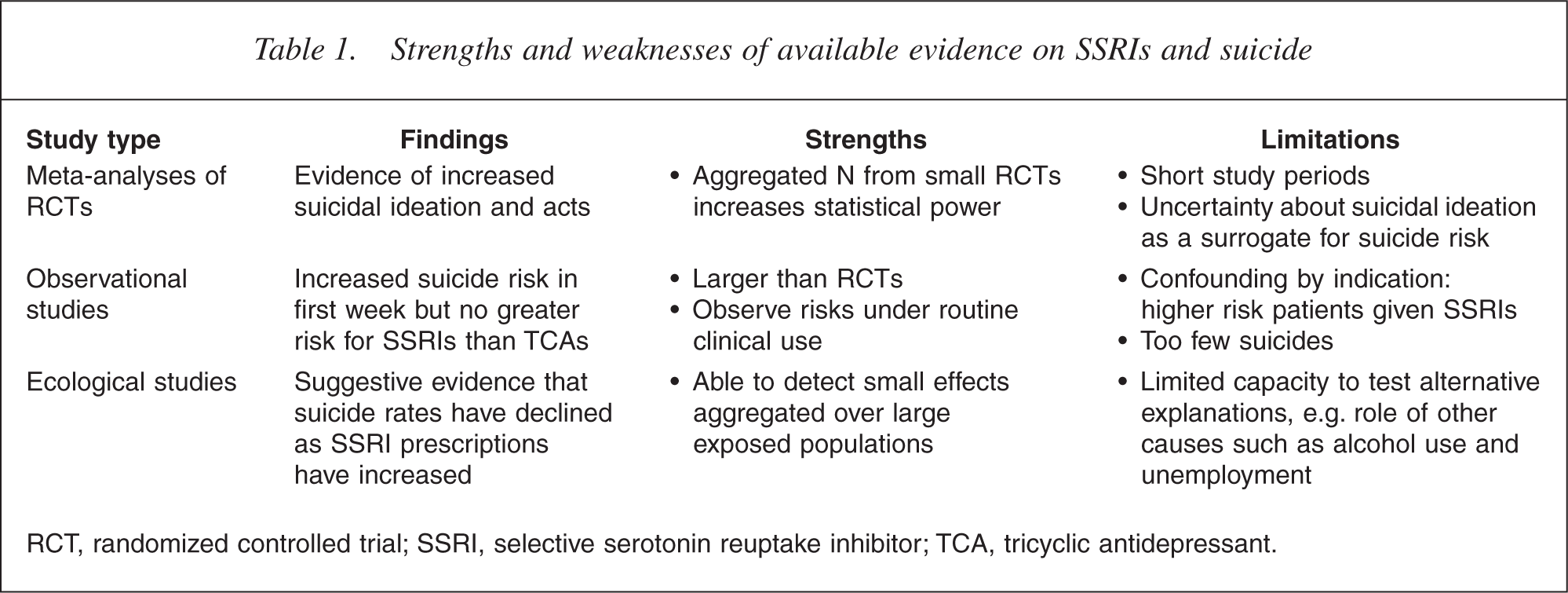
RCT, randomized controlled trial; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant.
Method
A series of computer searches was conducted on PubMed of published research on SSRI antidepressants and suicide. We also handsearched the reference lists of papers identified in this initial search strategy and also assessed papers that were cited in reviews and commentaries on the issue. The review was limited to studies published in English.
Approach used
We extracted different measures of relationship between SSRIs and suicidal behaviour and suicide mortality in each of the three types of study identified above. We used the pooled estimate of the relative risk (RR) of suicidal ideation from SSRIs versus placebos reported in metaanalyses of clinical trials of SSRI antidepressants. There were too few suicides in these trials to provide useful estimates of mortality risk. We extracted odds ratios (OR) from case-control and observational studies of associations between antidepressant use and suicide attempts. A variety of different measures of association were reported in ecological studies of relationships between SSRI prescribing and population suicide rates that precluded a simple summary measure.
A formal meta-analysis was not attempted because of: (i) the varying types of study design used; (ii) the overlapping sets of studies included in the different meta-analyses; (iii) the different comparisons of risks between specific antidepressant drugs that were reported in different observational studies; and (iv) different measures of statistical effect reported in ecological studies. Instead, we provide a systematic description of the findings of each type of study as well as a qualitative, narrative assessment of how the evidence from different study types coheres.
Results
Meta-analyses of randomized controlled trials
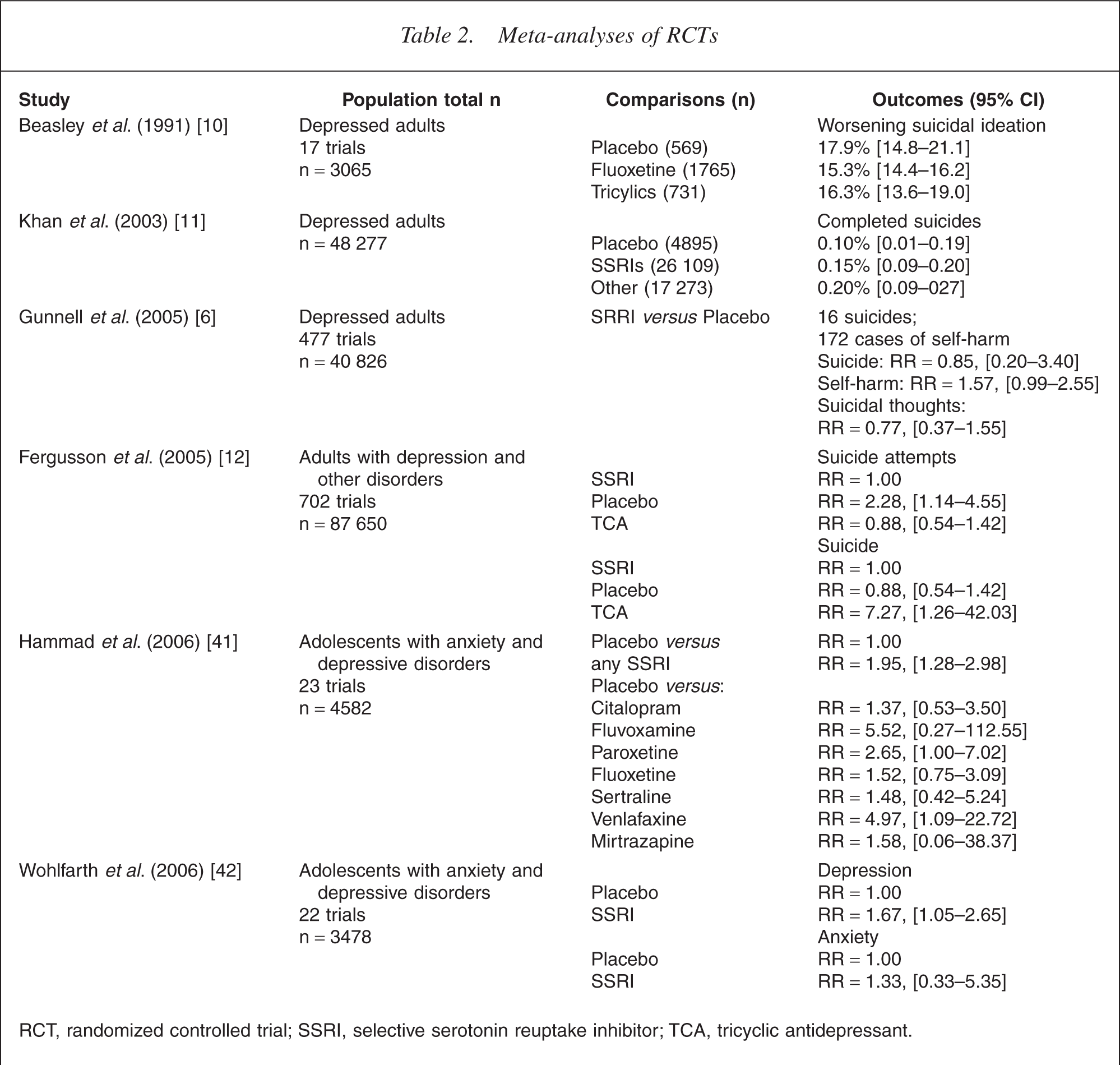
A small number of meta-analyses found no evidence of increased suicide risk from SSRI antidepressants [10,11]. An analysis of data from 17 RCTs of fluoxetine conducted in the late 1980s found no evidence that SSRIs increased suicidal thoughts in depressed people more than placebo or other antidepressants (see Table 2) [10]. Analyses of the aggregate data from trials of antidepressants submitted to the FDA between 1985 and 2000 failed to find any difference in suicide rates between SSRIs (0.15%, 95% confidence interval [CI] =0.10– 0.20), other antidepressants (0.20%, 95% CI =0.09–0.27) and placebo (0.10%, 95% CI =0.01–0.19]) [11]. These analyses, however, have included relatively small aggregate numbers of patients and they have not performed formal statistical meta-analyses [6].
Meta-analyses of RCTs
RCT, randomized controlled trial; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant.
More recent formal meta-analyses of larger sets of trials (including unpublished ones) have found an increased risk of self-harm in patients given SSRIs or placebos. Gunnell et al. reported a meta-analysis of 342 RCTs that compared SSRIs with placebo that had been submitted by pharmaceutical companies to the Medicines and Healthcare Products Regulatory Agency in the UK [6]. They found suggestive evidence that SSRIs increased self-harm (RR =1.57, 95% CI =0.99–2.55), but the evidence on suicide risk was inconclusive because there were only 16 suicides among 40 826 patients in these studies (RR =0.85, 95% CI =0.20–3.40) [6]. They estimated that two million people would need to be randomized to either SSRI or placebo to provide an 80% chance of detecting a clinically significant increase in suicide mortality.
Fergusson et al. reported a systematic review and meta-analysis of 702 RCTs that included 87 650 patients [12]. They found a higher rate of suicide attempts among patients receiving SSRIs compared with placebo (RR =2.28, 95% CI =1.14–4.55) but no difference between SSRIs and tricyclic antidepressants (TCAs) (RR =0.88, 95% CI =0.54–1.42). There was no difference in suicide rate between SSRI and placebo but the small number of events produced wide CIs around this estimated risk (RR =0.95, 95% CI =0.24–3.78). Paradoxically, despite no difference in suicide attempts between SSRIs and TCAs, and no difference in suicide rate between SSRIs and placebo, Fergusson et al. found a higher risk of suicide in patients receiving SSRIs compared with TCAs (RR =7.27, 95% CI =1.26–42.03) [12].
Meta-analyses partially overcome the problem of the limited statistical power in RCTs by aggregating large numbers of studies but they still have a number of limitations. First, they still lack sufficient statistical power to detect clinically significant effects on suicide. As Gunnell et al. noted, two million individuals need to be randomized to SSRI or placebo to detect an increase in completed suicide [6] and the total sample sizes in the largest meta-analysis are still below 100 000. Second, meta-analyses are also subject to the limitations of the individual RCTs whose results they summarize, namely, their short duration and exclusion of patients at high risk of suicide (e.g. [6]). Third, without access to the individual data, meta-analyses cannot adjust for differences between the patients in different trials. This may lead to an underestimation of the variance between trials and subsequent masking of effects [6]. Fourth, analyses of published studies may overestimate SSRI efficacy because studies with null results are less likely to be published.
Observational studies of suicide and SSRIs
Observational studies typically use record linkage in administrative and clinical data bases to assess the strength of associations between SSRI and other antidepressant use and suicide attempts or episodes of self-harm. This type of evidence is evidentially inferior to that from RCTs because patients are not randomly assigned to receive an antidepressant drug or not, thereby raising doubts about the equivalence in suicide risk before exposure to the antidepressant. The potential confounding by indication can only be imperfectly addressed by statistical methods such as covariate adjustment and propensity score analysis. The strength of observational studies is that they often involve large numbers of patients who are prescribed these drugs under conditions of routine clinical practice, precisely the situation in which we wish to make inferences about suicide risk (Table 3).
Observational studies of SSRI use and suicide
ADP, antidepressant; HMO, health maintenance organization; RCT, randomized controlled trial; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant.
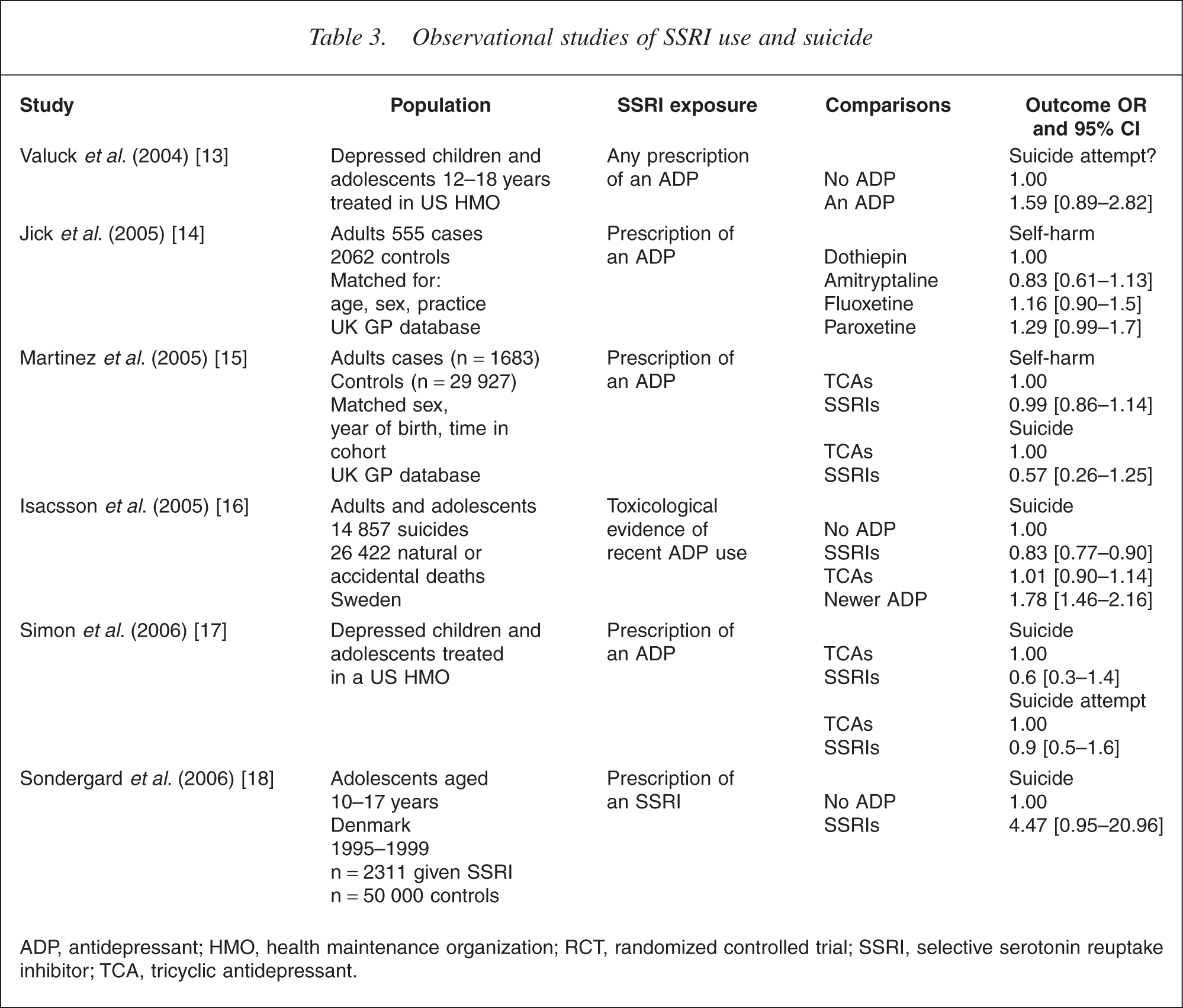
Valuck et al. compared suicide risk among adolescents aged 12– 18 years who received a diagnosis of major depressive disorder and either did or did not receive a prescription for an antidepressant [13]. Participants were treated in a large health maintenance organization in the US over a 6 year period and the outcome measured was suicide attempts in the 6 months following diagnosis. Two-thirds (68%) of these prescriptions were for SSRIs. A crude analysis showed an increased risk of a suicide attempt among adolescents who were prescribed any antidepressant (OR =1.93, 95% CI =1.56–2.39) or an SSRI (OR =1.95, 95% CI =1.53–2.49). A propensity analysis that adjusted for potential confounding by indication showed that the relationship was still elevated but no longer statistically significant (OR =1.59, 95% CI =0.89–2.82). A separate analysis found a reduction in suicide attempts among those were treated with an antidepressant for greater than 180 days (OR =0.34, 95% CI =0.21–0.55) compared with those treated for less than 180 days.
Jick et al. compared the risk of suicide and suicide attempts in patients who received the four most common antidepressants prescribed in UK general practice between 1993 and 1999, namely, amitryptyline, a TCA, and the SSRIs fluoxetine, dotheipin and paroxetine [14]. They selected cases and controls from the UK General Practice database that included 159 810 patients who had been prescribed one of these drugs during this period. They compared 555 cases who attempted suicide within 90 days of being prescribed one of these drugs and 2062 controls who had attempted suicide but had not been prescribed one of these drugs (matching cases and controls for sex, age and general practice).
Jick et al. found that patients with a previous history of suicide attempts were more likely to receive an SSRI than the most commonly prescribed tricyclic [14]. There was also a higher risk of a suicide attempt in the first 9 days after a prescription (OR =4.07, 95% CI =2.89–5.74), but there were no marked differences in risk between the four drugs. The relative risks (compared with dothiepin) were 0.83 (95% CI =0.61–1.13) for amitrytyline, 1.16 (95% CI =0.80–1.50) for fluoxetine, and 1.29 (95% CI =0.99, 1.70) for paroxetine. There were too few suicides (n =17) in the cohort to detect any differences in suicide risk between the different drugs.
Martinez et al. reported an observational study of suicide risk from SSRIs, tricylic antidepressants and other antidepressants in the UK GP database [15]. They selected cases (of persons who had an episode of self-harm) and up to 20 controls (who had not) for each case from 146 095 patients who received an antidepressant prescription between 1995 and 2001. They found that patients with a history of self-harm were more likely to receive an SSRI but there was no difference in the risk of self-harm between patients receiving an SSRI and those receiving a TCA (OR =0.99, 95% CI =0.86–1.14). There was possibly a higher risk among patients in the 10–19 year age group, with an OR of 1.50 (95% CI =1.01–2.50). Among the 69 suicides in this cohort the risk of suicide did not differ between those prescribed an SSRI and those prescribed a TCA (OR: 0.57, 95% CI =0.26–1.25).
Isacsson et al. compared toxicological evidence of use of SSRIs, TCAs and other antidepressants in 14 857 suicides and 26 422 controls who died from natural causes or accidents in Sweden between 1992 and 2000 [16]. Antidepressants were detected in 20% of suicides (compared with 6% of the controls), but the SSRIs were less likely to be detected in suicides than the newer selective norepinephrine reuptake inhibitors antidepressants (OR =0.83, 95% CI =0.77–0.90) and just as likely to be detected as TCAs (OR =1.01, 95% CI =0.90–1.14). Selective serotonin reuptake inhibitor were also less likely to be found among suicides in persons younger than 19 years (OR =0.14, 95% CI =0.05–0.43). Treatment refractory depressed patients were more likely to be prescribed the newest antidepressant at the time of their treatment. This was fluvoxamine in the early 1990s and moclobemide, mianserin, reboxetine and venlafaxine in the late 1990s.
Simon et al. used computerized medical records in a health maintenance organization in Washington State and Idaho to assess the association between antidepressant use and suicide and suicide attempts in 82 285 episodes of depression involving 65 103 patients who were prescribed an antidepressant between January 1992 and June 2003 [17]. They examined the risk of suicide attempts in the 6 months after the index prescription. There were 31 suicides and 76 suicide attempts that led to hospitalization during the 10.5 years of observation. They found the highest risk of suicide attempts in the month after a prescription (OR =2.5, 95% CI =1.6–3.8) but no increase in suicide deaths in the first month of treatment (OR =1.2, 95% CI =0.5–2.9). Nor was there any increased risk attributable to the specific antidepressants that had been included in the FDA warning about increased suicide risk. There were higher rates of suicide attempts among adolescents than adults but no evidence (within limitations of the small numbers involved) of an increased risk in the first month of treatment.
Sondergard et al. reported a small pharmacepidemiological study of antidepressant use among adolescents age 10–17 years in Denmark between 1995 and 1999 [18]. During this period, prescribing of SSRIs in this age group increased substantially. A linked study of antidepressant prescriptions and suicide found an increased risk of suicide in adolescents prescribed an SSRI but the number of suicides (n =42) was very small, and there was marked confounding by indication in that adolescents with a history of past psychiatric hospitalization for depression were most likely to be prescribed antidepressants (and a variety of other psychotropic medications). The study had limited capacity to adjust for the effects of this confounding. The fact that there was no increase in suicide rate among Danish adolescents during the period that SSRI use increased suggests that this relationship probably reflected increased pharmacological treatment of severely depressed adolescents in Denmark over the study period.
All of the observational studies have found ‘confounding by indication’ [19,20], that is, patients who were at increased risk of suicide were more likely to be prescribed SSRIs than TCAs. Despite many of these studies having only limited ability to adjust for such confounding (e.g. via the use of a limited number of variables in covariate adjustment and propensity analyses), none of the studies found a strong association between the SSRIs and self-harmful acts. The effect of antidepressants on suicide risk was certain because the number of suicides involved in these studies has been too small to permit one to infer the absence of an increased suicide risk.
Ecological studies of suicide and SSRI use
When individual level data are not available on SSRI use and suicide ecological analyses [21], researchers have explored associations between (i) population indicators of SSRI use (e.g. total SSRI sales in kilogram or daily dose equivalents); (ii) population suicide mortality; and (iii) controlling where possible for changes in other factors that might explain the associations (or lack thereof) [22].
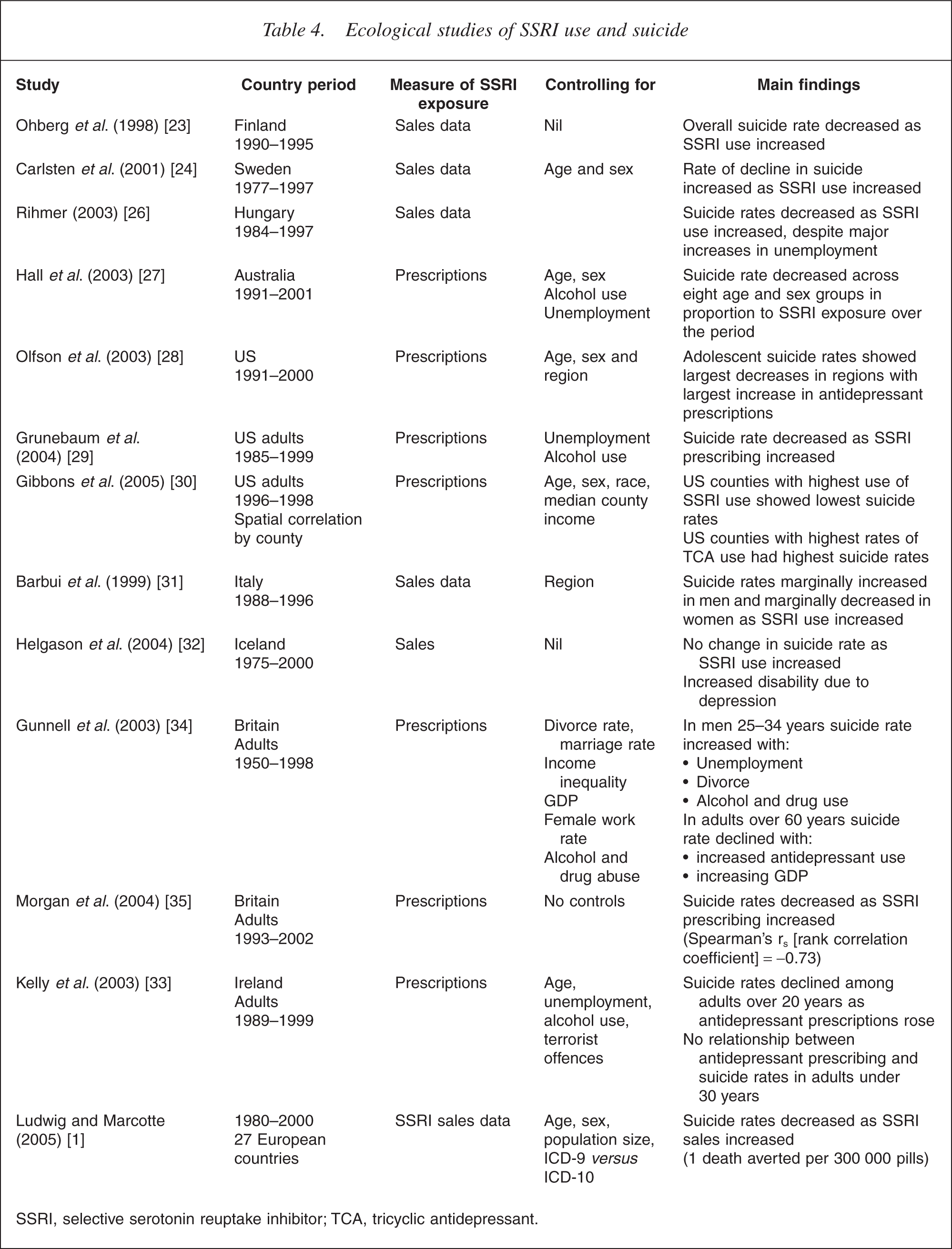
A number of studies have found inverse associations between suicide mortality and SSRI use, that is, suicide mortality has declined as SSRI use has increased (Table 4). Ohberg et al. found that suicide mortality declined between 1990 and 1995 in Finland as SSRI use increased [23], but they did not have any quantitative data on rates of SSRI use and they were unable to assess alternative explanations of the trend. Carlsten found that suicide rates in Sweden decreased by 31% in men and 34% in women between 1977–1979 and 1995–1997 while SSRI use increased markedly between 1993 and 1996 [24]. The decline in suicide rates could not be wholly attributed to SSRI use because it began before their introduction. Fazel et al. have more recently reported an ecological study of trends in suicide in Sweden between two 5 year periods, 1989–1994 and 1995–2000 [25]. Over this period, rates of SSRI prescribing increased tenfold in Swedish adults while suicide rates declined in all age groups and both sexes.
Ecological studies of SSRI use and suicide
SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant.
Rihmer et al. found that suicide deaths declined in Hungary after antidepressant prescribing increased in the early 1990s, despite steep increases in unemployment and per capita alcohol consumption, factors that would be expected to predict an increased suicide rate [26]. Hall et al. found a dose–response relationship between SSRI use and suicide rates in Australian men and women between 1991 and 2000: in men and women the higher the exposure to antidepressants in each age group, the larger the decline in suicide rate between 1991 and 2000 [27]. These trends were not explained by changes in per capita alcohol consumption or unemployment rate.
Three studies in the US have found associations between increased SSRI use and declining suicide rate. Olfson et al. examined the relationship between regional trends in antidepressant use and suicide mortality among adolescents between 1990 and 2000 in the US [28]. They found that a 1% increase in antidepressant use in a region was associated with a decrease of 0.23 suicides per 100 000 per year. Grunebaum et al. analysed the relationship between adult suicide rates between 1985 and 1999 and SSRI sales data, controlling for the effects of unemployment and per capita alcohol use [29]. They found that suicide rates declined as SSRI use increased. Gibbons et al. studied the relationship between US county suicide rates and SSRI and TCA sales over the period 1996–1998, controlling for county, age, sex, race and median income [30]. Antidepressant sales were uncorrelated with suicide rates overall, but different types of antidepressants had different types of relationships to suicide rates: higher sales of TCAs were correlated with higher county suicide rates while higher sales of non- TCAs (which included the SSRIs and all other newer antidepressants) were associated with lower county suicide rates.
Other ecological studies have found more mixed results. Barbui et al. found that while SSRI use increased 53% in Italy between 1988 and 1996, suicide rates increased in men and only marginally declined in women [31]. In Iceland a time series analysis by Helgason et al. found that sales of SSRIs increased by 16% per year after 1989 but rates of suicide did not change while psychiatric treatment rates and rates of disability from depressive disorders increased [32]. Sondergard et al. did not find any change in suicide rates among Danish adolescents as SSRI use increased between 1995 and 1999 [18].
In Northern Ireland, Kelly et al. did not find an association between antidepressant use and suicide in adults under 30 years but suicide rates declined as antidepressant use increased among those over the age of 30 years [33]. Similar trends emerged from a time-series analysis of suicide between 1950 and 1998 in the UK [34]. This study found that antidepressant use was associated with declines in suicide among adults over the age of 60 years but not among younger men (aged 25– 34 years) who had much smaller increases in antidepressant use than older men and women. Morgan et al. reported an inverse correlation between antidepressant prescribing in England between 1993 and 2001 but did not examine the relationship by age or gender [35].
Ludwig and Marcotte [1] have reported the largest ecological study of SSRI use and suicide to date. They analysed the relationship between SSRI sales data and suicide rates in 27 countries between 1980 and 2000 while controlling for different ways of coding cause of death (e.g. ICD-9 vs. ICD-10) and for differences between populations in age and sex, employment and divorce rates. They also assessed the robustness of their findings to the method of statistical analysis and to the measures of suicide and SSRI exposure that were used. They generally found an inverse relationship between per capita SSRI sales and population suicide mortality, indicating that the higher the sales of SSRIs in a country, the lower the suicide rate. An increase of one pill per capita was associated with a 2.5% reduction in suicide rate.
These ecological studies have a number of limitations. First, antidepressant sales data do not necessarily describe patterns of drug use and sales data typically do not partition SSRI use by age and sex (e.g. [1]). Second, most studies have not been able to control for the effects of risk factors for suicide, such as alcohol and other substance use [24,28,31]. The most likely effect of these limitations would be to reduce the magnitude of associations observed between SSRI use and suicide. It is therefore impressive that increased SSRI use has so often been correlated with declining suicide rates. The analysis of data from 27 countries over 20 years has supported the findings of the majority of the analyses in individual countries in finding that suicide deaths declined as SSRI use increased.
High risk populations: adolescents
Concern about the risk of SSRI use was originally prompted by cases of suicide in adults but attention has more recently focused on the risk of self-harm and suicide among depressed adolescents treated with SSRIs [36–38]. Because there have been many fewer placebo controlled RCTs of SSRIs in adolescents the evidence of the efficacy of SSRIs other than fluoxetine is much less impressive in adolescents than in adults [39,40]. Recent meta-analyses of clinical trials have found evidence of increased suicide ideation or suicide attempts like that in trials in adults. An FDA-commissioned study that reviewed 24 placebo controlled trials with over 4582 children and adolescents found a higher incidence of suicidal ideation in those receiving SSRIs compared with placebo (RR =1.66, 95% CI =1.02–2.68) [41]. A meta-analysis of 22 paediatric trials submitted to the European registration authorities found a similar increase in the risk of events related to suicidality (RR =1.33, 95% CI =0.33–5.35) [42].
Adolescents have not been well represented in the larger observational and ecological studies so the effect of SSRI use on suicidal risk is less certain in adolescents than in adults [37,38]. Nonetheless, Jick et al. [14] found only equivocal evidence of an increased risk of suicide in adolescents while Isacsson et al. [16] found evidence of a protective effect of SSRIs in young adults. The results of three recent observational studies in adolescent populations have been a bit more equivocal: Simon et al. and Valuck et al. did not find a large increase in the rate of suicide attempts among adolescents who had been prescribed an SSRI, after account was taken of confounding by indication [13,17]; Sondergaard et al. found an increased risk of suicide among adolescents prescribed an SSRI which was no longer significant after adjustment for confounding by indication but the upper limit of the CI did not exclude an increased rate of suicide [18].
Summary
In the absence of infallible ways of assessing the impact of SSRI use on suicide risk we have to look for a convergence of evidence from different types of studies using multiple methods of independent imperfection. A reasonably coherent picture is emerging from such research about the effects of SSRI use on suicide risk.
Concerns that SSRIs may increase suicide risk in the short term were raised by case studies and volunteer studies and have been supported by the biological plausibility of the hypothesis that SSRIs have a short-term activating effect [2] that may increase suicidal impulses [8,43] or may improve patients' energy before they improve mood, thereby making it easier for patients to act on suicidal thoughts.
In principle, the best evidence comes from RCTs of the effects of SSRIs on depressed patients but these trials have involved too few patients, observed for too short a time, and not well assessed on suicidal ideation [37]. The most recent and largest meta-analyses of placebocontrolled RCTs of SSRIs have found suggestive evidence that SSRIs increase suicidal ideation and attempts early in treatment. Randomized controlled trials are unlikely to assist in deciding whether SSRIs increase suicide mortality because an extremely large number of patients (of the order of several million) would need to be randomized to detect effects on a rare outcome like suicide [6,7]. It is uncertain whether suicidal ideation as assessed in clinical trials predicts suicide mortality well enough to serve as a useful surrogate marker for suicide mortality risk [7].
The evidence that SSRIs increase suicidal ideation in the meta-analyses has not generally been supported by observational studies of self-harm and suicide or ecological studies of suicide trends. Several observational studies have found an increased risk of self-harmful acts within 9 days to a month of an antidepressant drug being prescribed but the increase in risk has been similar for the older TCAs and SSRIs. If SSRIs increase suicide risk in some patients, then the number of additional suicide deaths is very small because no increase in suicide rates has been detected in the case–control studies or ecological studies to date. The ecological studies also suggest that the risks of SSRIs are outweighed by their benefits in reducing suicide since suicide rates have declined as SSRI use has increased in developed countries.
The observational studies provide some support for the clinical wisdom that suicide risk increases as psychomotor retardation begins to lift, an effect that may occur before mood improves, thereby enabling depressed patients to act on suicidal thoughts [44]. A plausible hypothesis is that any increase in suicide risk arising from initial improvement in depression would be more noticeable during induction onto SSRI antidepressants because (i) SSRIs are the most commonly prescribed antidepressant drugs; (ii) they are more likely to be prescribed to patients at risk of suicide; and (iii) their lower side-effect profile means that patients are more likely to take them for long enough to experience a therapeutic effect than was the case with older antidepressants.
Our understanding of the evidence on the relationship between SSRIs and suicide may be improved by epidemiological modelling of their effects on suicide rates using synthetic cohorts to model the likely effects of SSRIs on the natural history of depressive illness and suicide risk. There may also be value in pharmacogenetic studies of polymorphisms that are associated with an increased or reduced risk of developing suicidal ideation when taking SSRIs, or older antidepressant drugs. The discovery of any interactions between polymorphisms and suicide risk would strengthen the case for a causal relationship in a high-risk subset of depressed patients. If such relationships existed, the polymorphisms would also potentially provide a way of identifying the vulnerable minority of depressed patients before antidepressant treatment was initiated.
Implications
On the available evidence, any increase risk of suicide that SSRIs may produce in a subset of depressed patients appears to be offset by the public health benefits of the increased diagnosis and treatment of depression in primary practice. Given the residual uncertainty about whether SSRIs increase suicide risk in a small subset of patients, it would be prudent for those who prescribe SSRIs (and the newer selective norepinephrine reuptake inhibitors) antidepressants to carefully monitor suicidal ideation in the first few weeks after prescribing them to depressed patients with a history of suicidal ideation. Closer monitoring of suicidal ideation is also advisable in depressed adolescents [45] because of the possibly greater risk of suicide, and the uncertainty about the costbenefit ratio of SSRIs (other than fluoxetine) in this population.
Footnotes
Acknowledgements
This work was undertaken as part of a review funded by the National Prescribing Service (NPS), Sydney, Australia, on methods of assessing the population health impact of prescribed medicines. NPS is an independent Australian organization for the Quality Use of Medicines that is funded by the Australian government and does not receive funding from the pharmaceutical industry. The views expressed are solely those of the authors.
