Abstract
With approximately 12% of new mothers experiencing a major depressive episode, this is now one of the most common complications of childbirth [1]. Depression is associated with increased maternal and infant morbidity and mortality [2, 3]. This includes potential risks of pregnancy complications, impaired mother–infant interactions and poorer longer term child development outcomes [4–6]. There is also evidence that women who are already being treated with antidepressants in pregnancy have a significantly higher risk of relapse if they discontinue compared to those who maintain their antidepressant throughout pregnancy [7]. As a result there is an awareness of the importance of treating depression in pregnancy and in recent years there has been a significant increase in the prescription of antidepressants to pregnant women, in particular the selective serotonin re-uptake inhibitor (SSRI) class of antidepressants [8, 9].
This increasing prescription of antidepressants to women in pregnancy has resulted in a corresponding increase in the rate of in utero exposure of infants to antidepressants. Therefore the investigation of any potential pregnancy or neonatal complications that might arise from exposure is of growing importance. To date such investigations have found antidepressant exposure in pregnancy may be associated with an increased risk of prematurity, low birthweight and lower APGAR scores at birth [10]. There has been mixed data around a possible increase in the risk of both malformations [11–13] and pulmonary hypertension of the newborn [14, 15]. A range of neonatal symptoms has also been identified that are consistent with a discontinuation or withdrawal syndrome [16–21].
The term ‘discontinuation’ has been commonly used to describe neurobehavioral and gastrointestinal symptoms observed in neonates following in utero antidepressant and particularly SSRI exposure. It is unclear from the evidence available, however, if these observed neonatal discontinuation symptoms seen in the neonate are a result of withdrawal from the antidepressant or represent a serotonin toxicity state in the neonate. All antidepressants cross the placenta and therefore all antidepressants potentially carry some risk of a discontinuation syndrome in exposed neonates [22].
Symptoms of a discontinuation syndrome in neonates exposed to SSRIs were first reported by Chambers et al. in 1996 and have since been replicated in a number of studies and have been found with other classes of antidepressants [23, 24]. The symptoms include difficulties with feeding and sleep [19, 25], tremor [19, 20, 26], irritability [17, 25], increased tone [19, 25, 26], jitteriness [24], hypoglycaemia [17, 18, 24], gastrointestinal symptoms [19], jaundice [17] and respiratory distress [17–19, 24]. There have also been findings of an associated increased risk of prematurity, low birthweight and convulsions [18].
The variation in symptoms identified in different studies may in part be explained by the inconsistency across studies as to which specific neonatal symptoms were assessed. For instance studies that have utilized population-based databases have tended to examine jaundice and admissions to special care nursery, whereas smaller cohort studies have examined symptoms such as jitteriness and gastrointestinal symptoms.
Prescribed and illicit drugs that have also been associated with neonatal withdrawal syndrome have specific observed neonatal symptom patterns that vary between different drugs. For instance the opiate withdrawal symptom pattern differs significantly from the cocaine withdrawal symptom pattern in neonates [27]. Antidepressants have also been associated with a unique symptom pattern observed in neonates including an increased rate of respiratory distress, hypoglycaemia, lethargy, hypothermia, increased rates of sneezing, yawning and jitteriness [28]. To date, however, there is no validated scale or assessment tool to examine neonates at risk of a neonatal serotonin discontinuation syndrome [23]. The purpose of the present study was, therefore, drawing on the findings of previous studies, to compile a comprehensive set of neonatal symptoms that have been previously identified as associated with exposure to antidepressants in pregnancy.
The rate of occurrence of a neonatal discontinuation syndrome associated with antidepressant exposure has varied between studies [19, 24], but it has been suggested that approximately 22% and 30% of exposed neonates will develop a discontinuation syndrome [8, 17, 19, 24]. Some studies have attributed the observed variation in the frequency and severity of discontinuation symptoms to individual antidepressants’ placental passage and half-life [28], other studies suggested that it is related to the range in dose of medication that the infant was exposed to during pregnancy [19]. The symptoms usually have an onset somewhere between birth and day 5 and symptoms are thought to resolve by 1–2 weeks following birth [17, 29]. The role of breast-feeding as either a protective or risk factor is rarely examined.
The present study used a prospective and controlled design to examine symptoms of discontinuation in neonates who had been exposed to antidepressant medication in pregnancy. Based on previous findings it was hypothesized that within the first week mothers who had been prescribed antidepressant medication in pregnancy, and specifically in the third trimester of their pregnancy, would report observing known signs of discontinuation symptoms to a greater degree than women not prescribed antidepressants.
Methods
Participants
The Victorian psychotropic registry recruited 27 women who were treated with an SSRI, serotonin and noradrenaline re-uptake inhibitor (SNRI) or noradrenergic and specific serotonergic antidepressant (NaSSA) for depression during their pregnancy between June 2004 and July 2005. A total of 25 of these women remained on medication in the third trimester of their pregnancies; the period considered relevant to exposure related to discontinuation symptoms. Subjects were recruited at Mercy Hospital for Women, a tertiary obstetric hospital in Melbourne, Australia. A matched control group of 27 pregnant women not taking antidepressants and not depressed at the time of their recruitment were recruited prospectively via antenatal appointments at the Mercy Hospital for Women. The study has been approved by the Mercy Hospital for Women Human Research Ethics Committee. A written informed consent statement was obtained from each participant.
Of the 25 women included in the study group who continued antidepressants into their third trimester 14 participants were prescribed sertraline, two participants were each taking venlafaxine, fluoxetine, citalopram and one participant was each taking fluvoxamine, mianserin, mirtazepine, paroxetine and escitalopram oxalate, respectively. Two women were excluded from the final analysis due to incomplete data on discontinuation symptoms in their neonates.
Women were excluded from the study on the grounds of substance dependence and those unable to provide informed consent to participate. Inclusion criteria included English proficiency.
Measures
Maternal characteristics
A purpose-designed questionnaire was used to assess demographics, reproductive and medical history, prescribed medications, mental health service usage, maternal stress and coping. We also collected information on alcohol consumption, tobacco and recreational drug use. Within 1 week of delivery the mother was questioned about the course of her pregnancy, verification of the duration, dose and type of antidepressant treatment during pregnancy, maternal illnesses, and antenatal and/or postnatal complications. Information on delivery methods and birth outcomes was collected.
Depression
The Beck Depression Inventory, second edition (BDI-II) has been used to assess depression in all participants at each assessment point [30]. The BDI-II is a 21-item self-report instrument intended to assess the existence and severity of symptoms of depression as listed in the DSM-IV [31]. For the BDI-II a participant is asked to consider each statement as it relates to the way they have felt for the past 2 weeks. There is a 4 point scale for each item ranging from 0 to 3 and scores are added. A total score of 0–13 is considered minimal range, 14–19 is mild, 20–28 is moderate, and 29–63 is severe. The BDI-II has a high coefficient alpha (0.80). Its construct validity has been established and it is able to differentiate depressed from non-depressed patients. Test–retest reliability 1 week apart showed a correlation of 0.93. The BDI-II is worded specifically to assess the symptoms of depression listed in the DSM-IV and has strong content validity as a measure of depression. The original BDI has been used successfully as a treatment outcome measure for pharmacotherapy and has been shown to be a sensitive measure of change.
Neonatal discontinuation symptoms
Within 1 week of delivery a set of nine questions was used to assess serotonin withdrawal in the neonate. The questions asked were about symptoms observed in the infant including the nature of the infant's cry, length of sleep after feeding, tremor, yawning, sneezing, feeding problems, reflux, vomiting and diarrhoea. In addition information was collected on whether the infant was admitted to either the special care nursery or neonatal intensive care unit, and whether they had a diagnosis of respiratory distress, hypoglycaemia, jaundice or convulsions. Currently there is no validated scale to assess neonatal discontinuation symptoms for infants exposed to antidepressant medication [23]. Although there are scales validated for other drug withdrawal syndromes such as for opiates, these do not include all the symptoms that have been specifically found to be associated with antidepressant exposure in the neonate. Thus a purpose-designed questionnaire was used based on neonatal symptoms and complications found in previous studies of antidepressant exposure in pregnancy [17–19, 24, 25].
Medication
A component of the purpose-designed questionnaire included a section on medication prescription both antidepressants and other prescribed medication. Details were elicited concerning the time and duration of exposure to antidepressants and the dose and type of the antidepressant drug and any changes in pregnancy and early in the postpartum period. Because the majority of participants were taking sertraline (56%), other medications were converted to a sertraline equivalent dose using an equivalence chart for antidepressants published by Bezchlibnyk-Butler and Jeffries [32] for use in all data analysis.
Data analysis
Comparisons of discontinuation symptoms between the study and control groups were performed by calculating frequencies and percentages for the respective symptoms and then calculating an odds ratio for each comparison. The relationship between discontinuation symptoms that were coded as a binary was analysed using a tetrachronic correlation. Within the medicated group possible dose effects of serotonin exposure were tested comparing mean dosage for those displaying symptoms with those who did not display symptoms.
Results
Sociodemographic and delivery characteristics
Subject characteristics
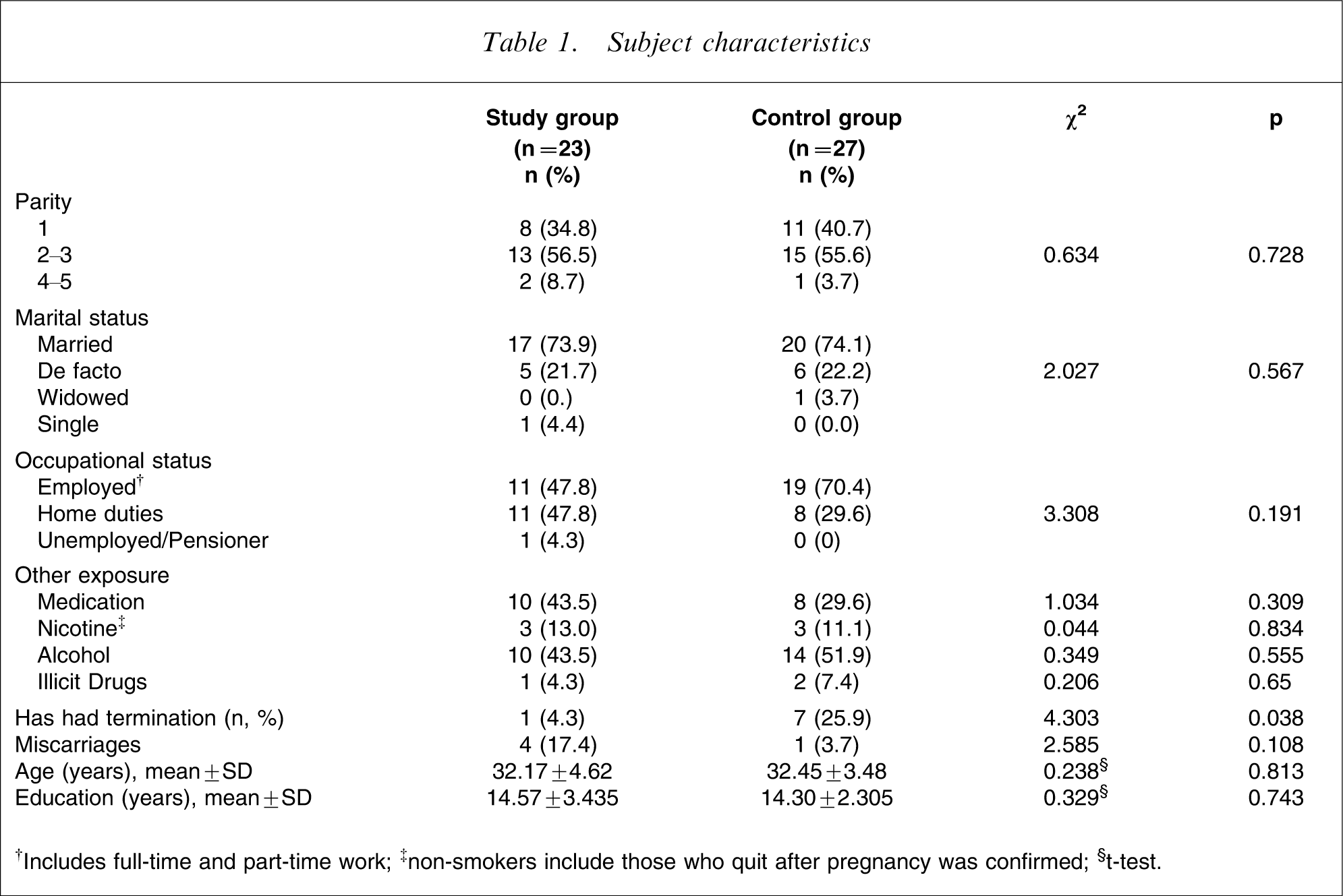
†Includes full-time and part-time work; ‡non-smokers include those who quit after pregnancy was confirmed; §t-test.
Method and complications of delivery vs group
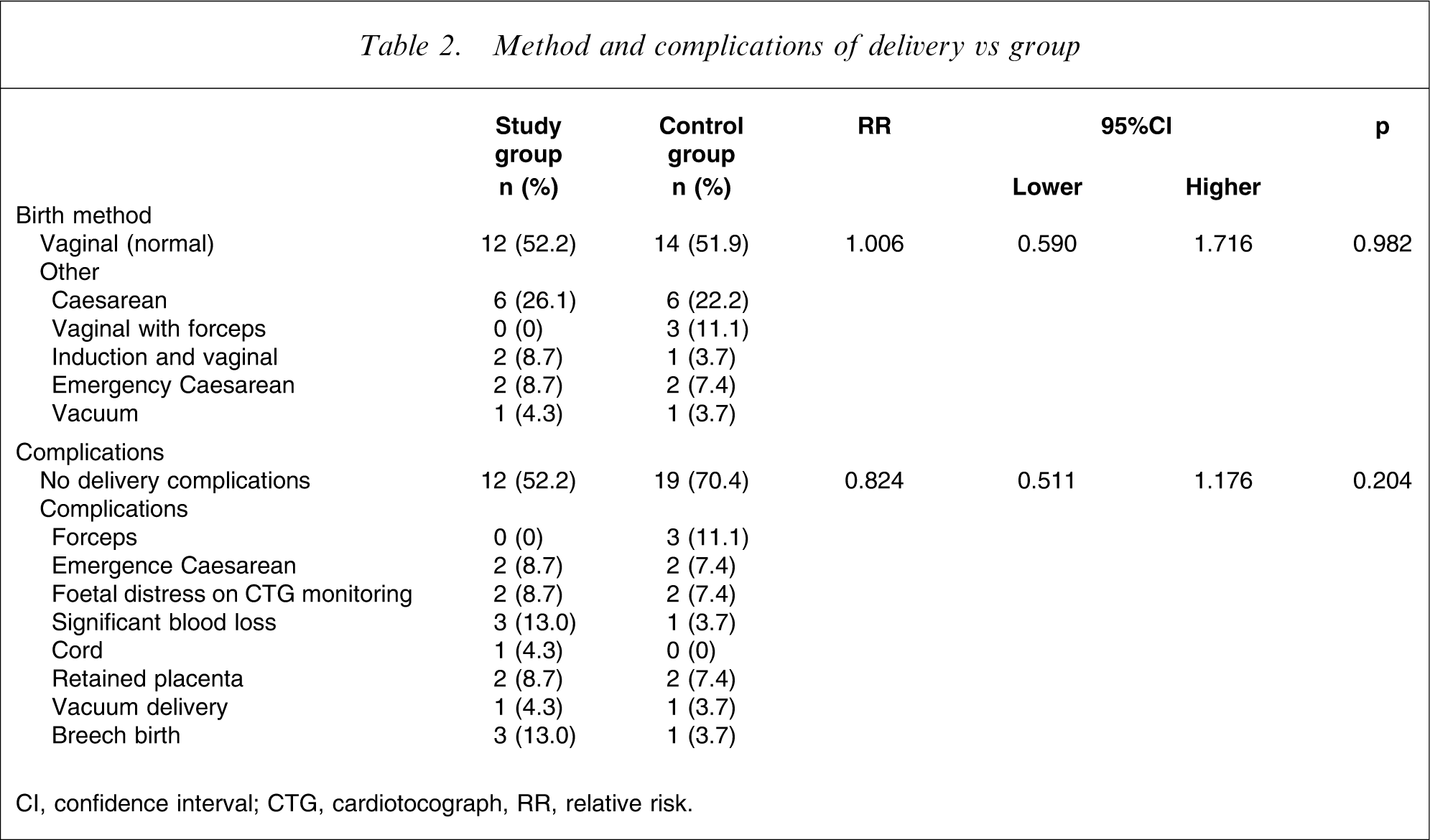
CI, confidence interval; CTG, cardiotocograph, RR, relative risk.
The mean gestational age at delivery in the study group was 38.5 weeks (SD = 2.39), and that in the control group was 39.9 weeks (SD = 1.14). All participants breast-fed their infants.
Discontinuation symptoms
Discontinuation symptoms vs group
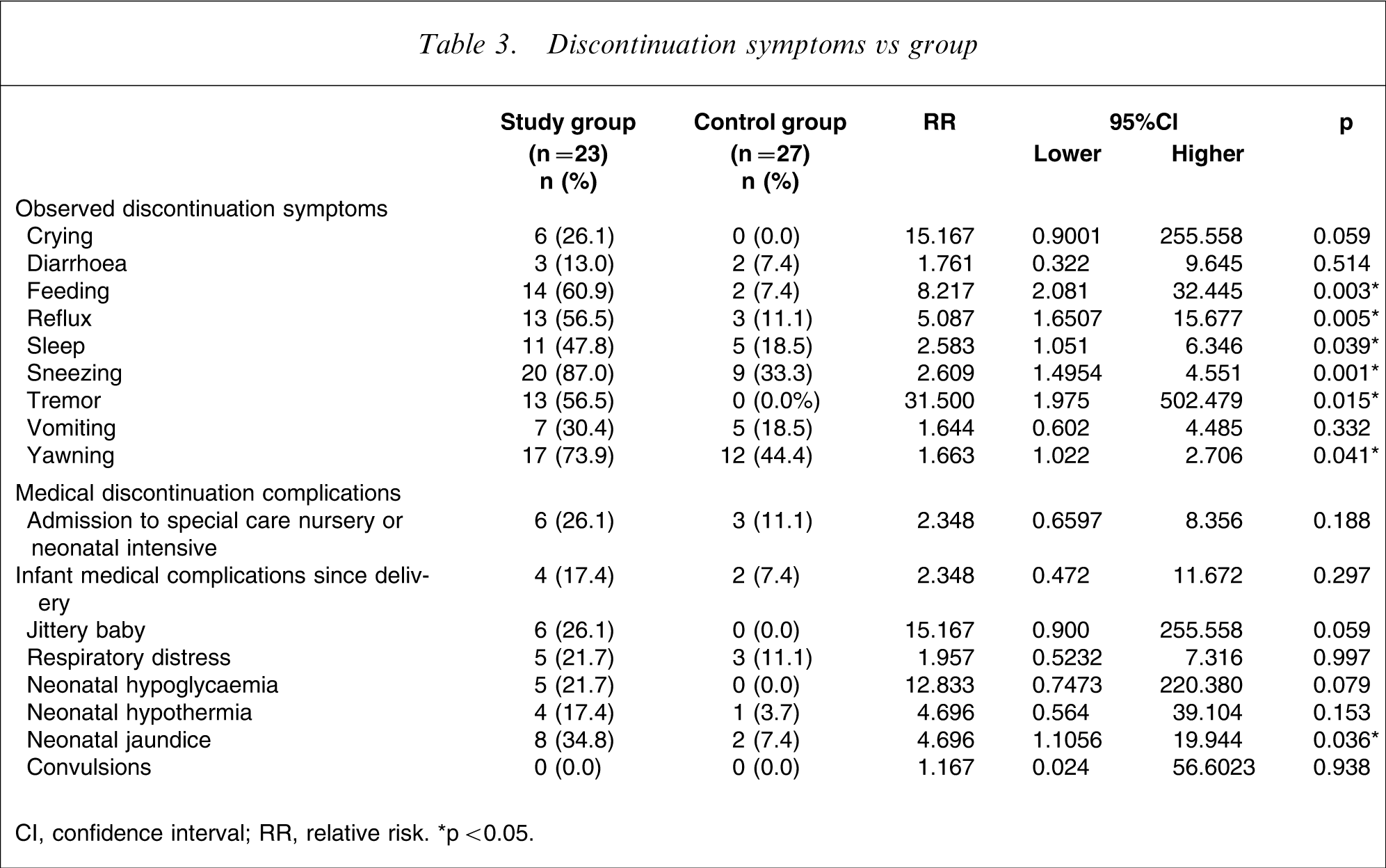
CI, confidence interval; RR, relative risk. ∗p < 0.05
Tetrachoric correlation between discontinuation symptoms (n = 23)
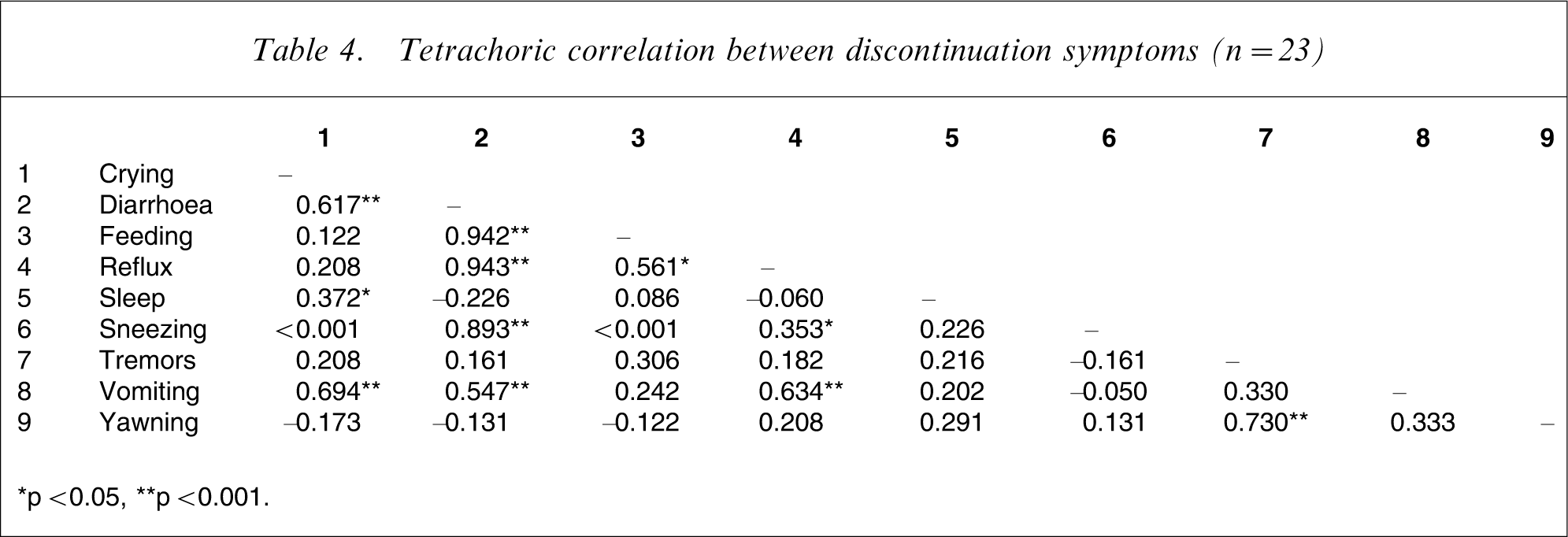
∗p < 0.05, ∗∗p < 0.001.
Dosage effects on discontinuation symptoms
Sertraline equivalent dose (mgs) vs discontinuation symptoms in the study group (n = 23)
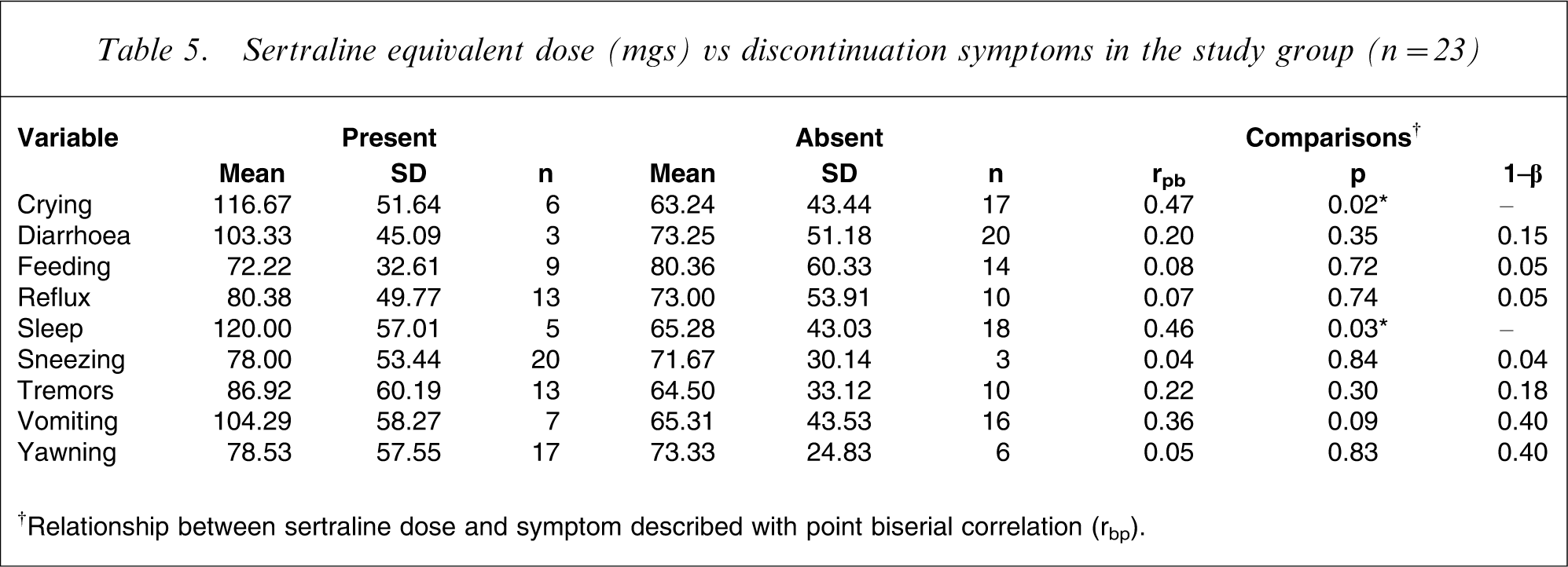
†Relationship between sertraline dose and symptom described with point biserial correlation (rbp).
To analyse this relationship further, in the medicated group only, we compared the mean dose of medication (and standard deviation)of those neonates who displayed each discontinuation symptom with those neonates who did not display that symptom. The results are presented in Table 5. Again, those neonates who displayed symptoms were on average exposed to slightly higher doses of sertraline equivalent dosages. It is also to be noted, however, that Table 5 suggests a high variance within these groups, suggesting a wide variety of outcomes. This may imply that other factors besides medication dosage are impacting on the likelihood of a neonate reacting to the withdrawal of serotonin exposure in the days after birth.
Discussion
Discontinuation symptoms have been associated with the sudden withdrawal of antidepressants in adults [33]. There is increasing evidence, however, that neonates exposed to antidepressants in late pregnancy are also at risk of discontinuation symptoms. This study has found a significant association between late pregnancy exposure and neonatal discontinuation symptoms. These symptoms include poor sleeping, poor feeding, crying, reflux, sneezing and tremor. The present study confirmed other previous studies that have found similar associated symptoms [16–19, 21, 24].
Some previous studies have shown an increased risk for discontinuation symptoms with specific antidepressants, such as paroxetine, but the present study did not find any difference between specific antidepressant medications and the risk of neonatal symptoms [17]. It must be noted, however, that the number of women on medications other than sertraline was low. The present study did find some tentative evidence of a dose effect for antidepressants and the risk of neonatal discontinuation symptoms. A dose effect was also found by Levinson-Castiel et al. for paroxetine but in that study no other antidepressants were examined for a dose effect [19]. These findings bring into question the theoretical view that individual antidepressant half-life and placental passage are the important factors in determining the risk of a neonate developing discontinuation symptoms following exposure to antidepressants in late pregnancy, and instead would suggest that dose may be the more important factor in determining risk. It must be noted, however, that the small numbers in each group for individual antidepressants in the present study make conclusions difficult to draw.
Neonatal symptoms associated with antidepressant exposure in late pregnancy have ranged from central nervous system symptoms, gastrointestinal symptoms, and respiratory symptoms to complications such as jaundice, hypoglycaemia and hypothermia [16–19, 21, 24, 25, 28]. In the present study the predominant symptoms identified in neonates following late pregnancy antidepressant exposure formed two groups and as clusters were correlated. These groups were gastrointestinal symptom clusters and central nervous system symptom clusters, and they are similar to those found in two other studies in this respect, in that both found greater central nervous system and gastrointestinal symptom clusters than respiratory symptoms [19, 34]. This finding is important given the lack of clarity of diagnostic criteria for neonatal antidepressant discontinuation syndrome in the literature. Identification of clear symptom patterns and symptom clusters will aid the future development of criteria for the assessment and diagnosis of a neonatal serotonin discontinuation syndrome.
This study has also found a higher rate of admission of neonates to special care nursery. This may reflect the current difficulty in making a diagnosis of antidepressant discontinuation syndrome in the neonate. This has the potential to lead to misdiagnosis or over-investigation, which may result in significant associated morbidity for the infant. Many of the manifestations of the discontinuation syndrome such as tremor, irritability, and jitteriness may be easily confused with convulsions, and again this suggests the need to clarify diagnostic criteria for the neonatal serotonin discontinuation syndrome to improve identification and management. It is important to note, however, that there are some presentations of neonatal serotonin discontinuation syndrome that may require monitoring and supportive treatment in a special care nursery setting. Distinguishing whether these admissions were warranted was not possible within the present study but may be useful to examine in future studies.
All infants in the present study were breast-fed. There has been little discussion in previous studies of the role of breast-feeding in the development of discontinuation symptoms. Depending on whether discontinuation symptoms are understood to be a process of withdrawal or of toxicity, breast-feeding could either be a contributing or a protective factor. For instance it may be argued that breast-feeding is protective given that there is breast milk excretion of all antidepressants and thus there is not the sudden withdrawal of medication. Given, however, the much higher rate of placental passage and a relative low rate of breast milk excretion for most antidepressants, this is questionable [22]. In addition studies in adults have shown that tapering dose as well as ceasing can result in adults experiencing discontinuation symptoms [33]. The present study showed that despite neonates being breast-fed they were still at significant risk of discontinuation symptoms.
The present study also confirmed the increased risk of neonatal jaundice for those neonates exposed to antidepressant medication in late pregnancy. Previous studies have varied in their findings: some have found a significant association [17] and some have not [18]. Given that this is a common neonatal complication often associated with breast-feeding, its association with antidepressant exposure in late pregnancy has been questioned. Using a case–control method, in the present study all neonates in both the control and medication groups were breast-fed and those exposed to medication in late pregnancy had a sevenfold higher risk of developing jaundice. Therefore in the present study breast-feeding was not the explanation for the increased rate of jaundice observed. Antidepressant medications are metabolized by the liver and neonatal hepatic maturation does not fully occur until 3 months of age [35]. The association between increased neonatal jaundice in neonates exposed to antidepressants in late pregnancy may well be explained through antidepressant medication toxicity. Indeed, maternal medications are a known risk factor for neonatal jaundice [35, 36]. Further investigation, however, would be required to confirm this hypothesis.
These findings, in terms of neonatal symptoms, complications and the need for assessment and monitoring over a number of days suggest that a short length of stay for many mothers and their newborns may be ill advised when there is a history of exposure to antidepressant medication in late pregnancy. Therefore, to ensure adequate management a length of stay measured in days rather than hours would seem good clinical care for both mother and baby.
Footnotes
Acknowledgements
The women and infants who participated in this study. Helena Sandahl, Gillian Opie. This study was supported by Neuroscience Research Grant, Pat and Toni Kinsman Scholarship, Pfizer, Lundbeck and Wyeth.
