Abstract
Major depression is a common, serious disorder [1, 2], and drug-resistant depression is a difficult, frequently encountered clinical problem [3]. Transcranial magnetic stimulation (TMS) [4] has antidepressant effects in drugresistant depression when applied at greater than 1 Hz to the left prefrontal cortex [5–13]. It does not appear to impair cognition [14].
Current depression treatment protocols rely on daily TMS (5 days per week), for 2–4 weeks. Longer courses yield better results [5–7,15], but it is unclear whether this is due to the greater accumulated dose, or to the longer duration of treatment. Antidepressant medication and electroconvulsive therapy take at least 2 weeks to produce beneficial effects. It is assumed that while active treatment is necessary to stimulate biological processes, time is also necessary to allow these processes to proceed to a critical point [16].
Daily TMS is time-consuming. It is burdensome for patients as they are required to attend daily, and it limits the number of patients who can be assisted. This study attempted to clarify the importance of time in TMS treatment. Two groups were given different numbers of treatments (5 and 10) over the same time period (12 days).
Method
Subjects
The study was approved by the Royal Hobart Hospital Research and Ethics Committee, and patients provided written informed consent. Patients were recruited from the inpatient unit of the Royal Hobart Hospital and from outpatients referred by local private psychiatrists. None of the treating/responsible psychiatrists were directly involved in the study.
Inclusion criteria were: (i) major depressive episode (DSM-IV); (ii) between 20 and 65 years; (iii) failure to respond to trials of at least two different antidepressants, at maximum manufacturer recommended doses, for at least 4 weeks; (iv) a score of at least 18 on the 17-item Hamilton Depression Rating Scale (HDRS) [17]; and (v) no medication change for a minimum of 2 weeks before commencement of the study. Exclusion criteria were: (i) concurrent neurological disorder (including epilepsy); (ii) other concurrent serious medical illness; (iii) history of significant head injury; (iv) recent alcohol or other drug misuse; and (v) intracranial metal object. Current antidepressant medication usewas not an exclusion criterion.
Details were collected of age, gender, duration of current episode, age at onset of first episode, diagnosis, history of electroconvulsive therapy (ECT) and concurrent medication. The diagnosis of major depression was made by the referring psychiatrist and confirmed by one investigator (Y T-S) at the intake interview.
Design
Patients were allocated to one of the two treatment groups in random order (decided by the flip of a coin) and treated over 2 weeks. One group received daily TMS (dTMS; days 1-5 and 8-12; a total of 10 treatments). The other group received spaced TMS (sTMS; days 1, 3, 5, 8 and 12; a total of five treatments). Medication was not altered during the study.
TMS treatment
Transcranial magnetic stimulation was administered using a Magstim Super Rapid device (Magstim Company, Wales, UK) and a handheld Magstim figureeight coil (9 cm). The motor threshold was determined using single pulses [18]. Treatment was applied 5 cm anterior to the point at which the motor threshold was determined, which is considered to be the region of the left prefrontal cortex. The coil was placed on the hair/scalp at the junction of the two coils with the handle at 45 degrees to the anterior-posterior line. Treatment consisted of 30 2-second trains of 20 Hz stimulation at 100% of motor threshold, in accordance with established safety guidelines [19, 20]. The inter-train interval was 28 seconds.
Measurements
Objective assessment
The HDRS was scored by blind raters before commencement (day 1), and on day 5 and 12 of treatment. Response was defined as a 50% reduction of the HDRS score; remission was defined as achieving an HDRS score of 8 or less.
Subjective assessment
A visual analogue scale (VAS) [21] using the anchor points (0 = ‘the worst I've ever felt’ and 10 = ‘the best I've ever felt’ [22]) was applied three times per week.
Data analysis
Comparison of the entry variables (gender, duration of current depressive episode, age of onset, current medication and previous ECT) was conducted using anova and chi-squared tests. The influence of time on mood was examined using a doubly multivariate anova with the independent variables of time (days 1, 5 and 12) and treatment group (dTMS, sTMS) with the dependent variables comprising the HDRS and VAS scores. Follow-up univariate analyses for individual depression ratings were conducted where appropriate. An alpha level of 0.05 was used throughout the analyses.
Results
Demographic and historical comparisons
At entry, there were no significant differences between the groups with respect to age, gender, duration of current episode, diagnosis, the number who had previously received ECT, or concurrent medication (Table 1).
Summary of demographic variables for patients in daily and spaced TMS treatment groups (standard deviations in parentheses)
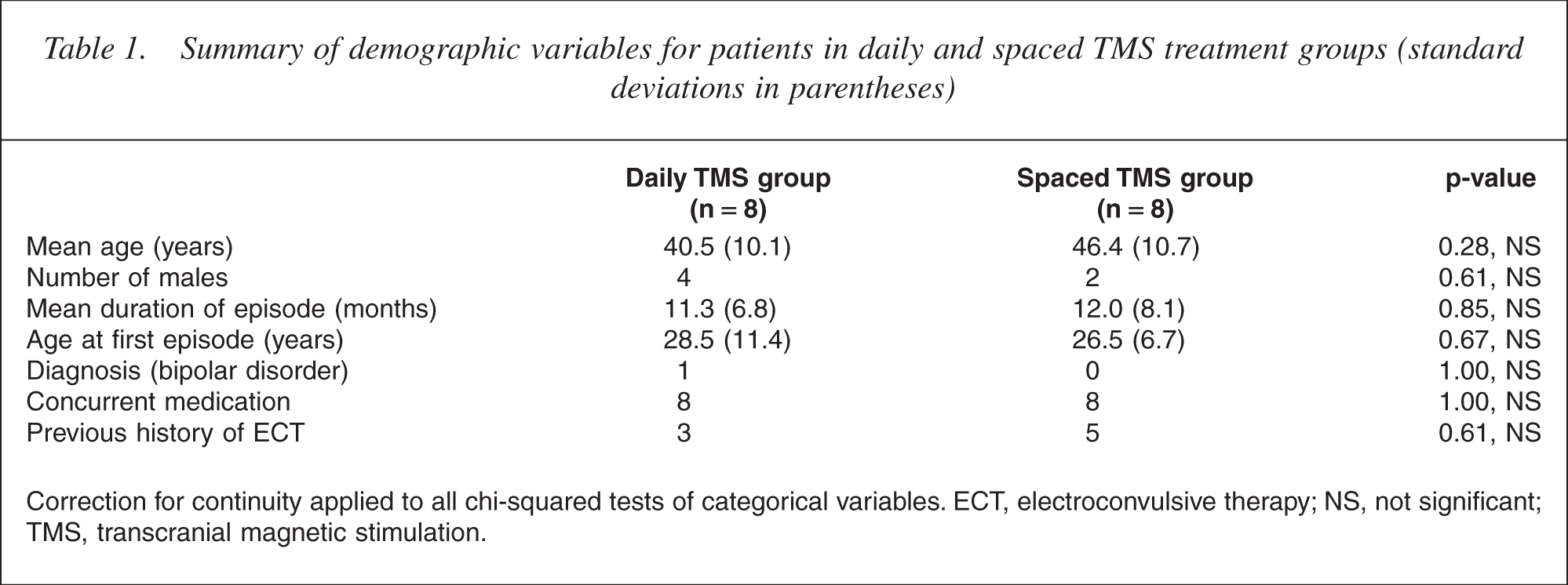
Correction for continuity applied to all chi-squared tests of categorical variables. ECT, electroconvulsive therapy; NS, not significant; TMS, transcranial magnetic stimulation.
Treatment comparisons over time
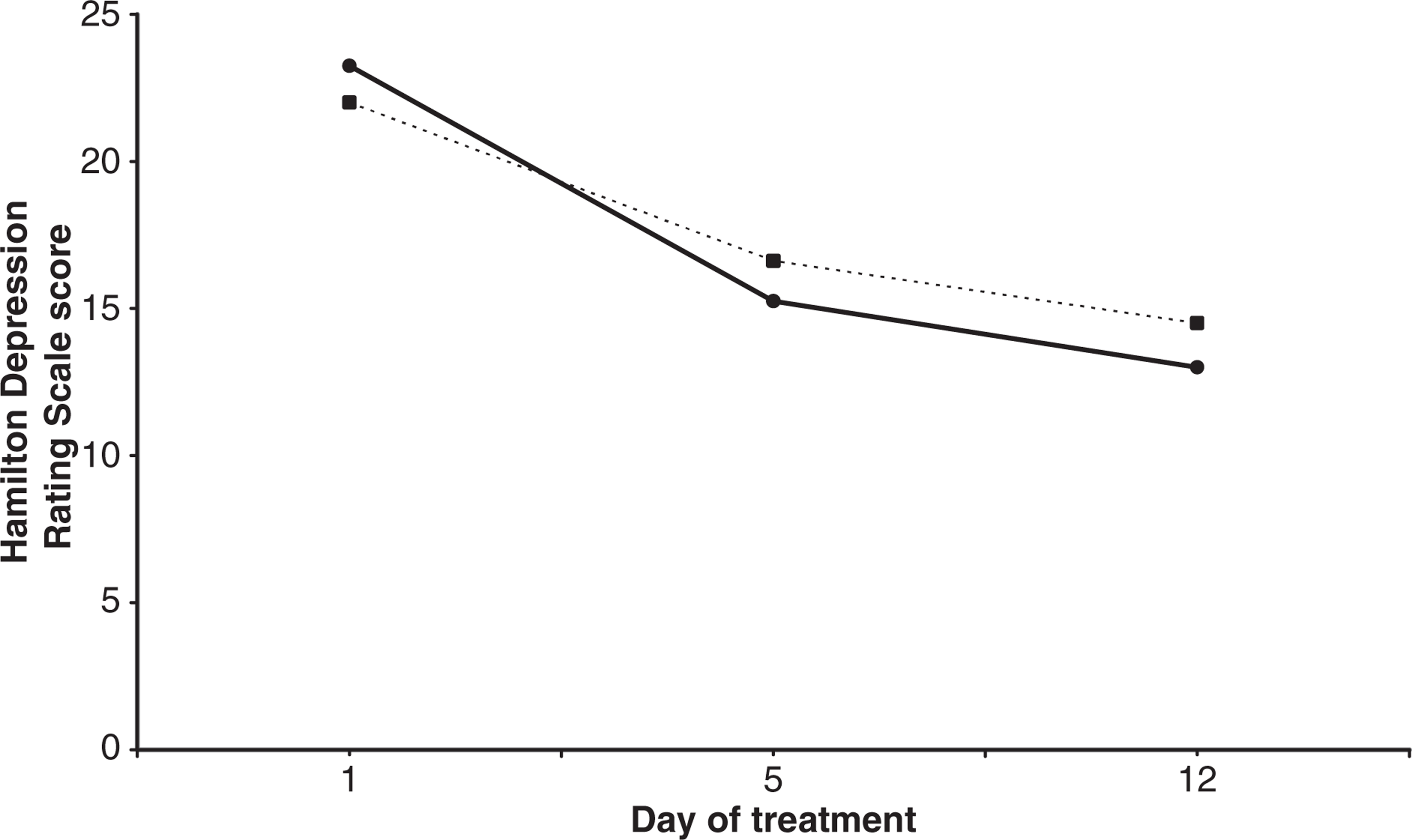
Multivariate tests showed a significant main effect for time (Pillai trace = 0.76, F(4,56) = 8.56, p < 0.001, power = 0.99) reflecting the improvement in both measures of depression during treatment (Table 2, Figs 1,2). There was no significant difference between the groups overall (Pillai trace = 0.25, F(2,5) = 2.12, p = 0.16, power = 0.36). Additionally, there was no significant interaction between time and treatment group (Pillai trace = 0.18, F(4,56) = 1.35, p = 0.26, power = 0.39) indicating that the significant improvement in depression over time was similar for both treatment groups. Univariate analyses of this design for individual depression rating scores reveal similar findings overall, and there were no significant differences between the treatment groups at days 1, 5 or 12 on either measure of depression (Table 2).
Mean visual analogue scale ratings of subjective mood over 12 days of treatment for patients with major depressive episode (MDE) who were treated with daily or spaced transcranial magnetic stimulation (TMS) treatment.(–––) Daily TMS; (······) Spaced TMS; (–––) Linear (daily TMS); (······) Linear (spaced TMS). Mean Hamilton Depression Rating Scale scores over 12 days of treatment for patients with major depressive episode (MDE) who were treated with daily or spaced transcranial magnetic stimulation (TMS) treatment. (–––) Daily TMS; (······) Spaced TMS.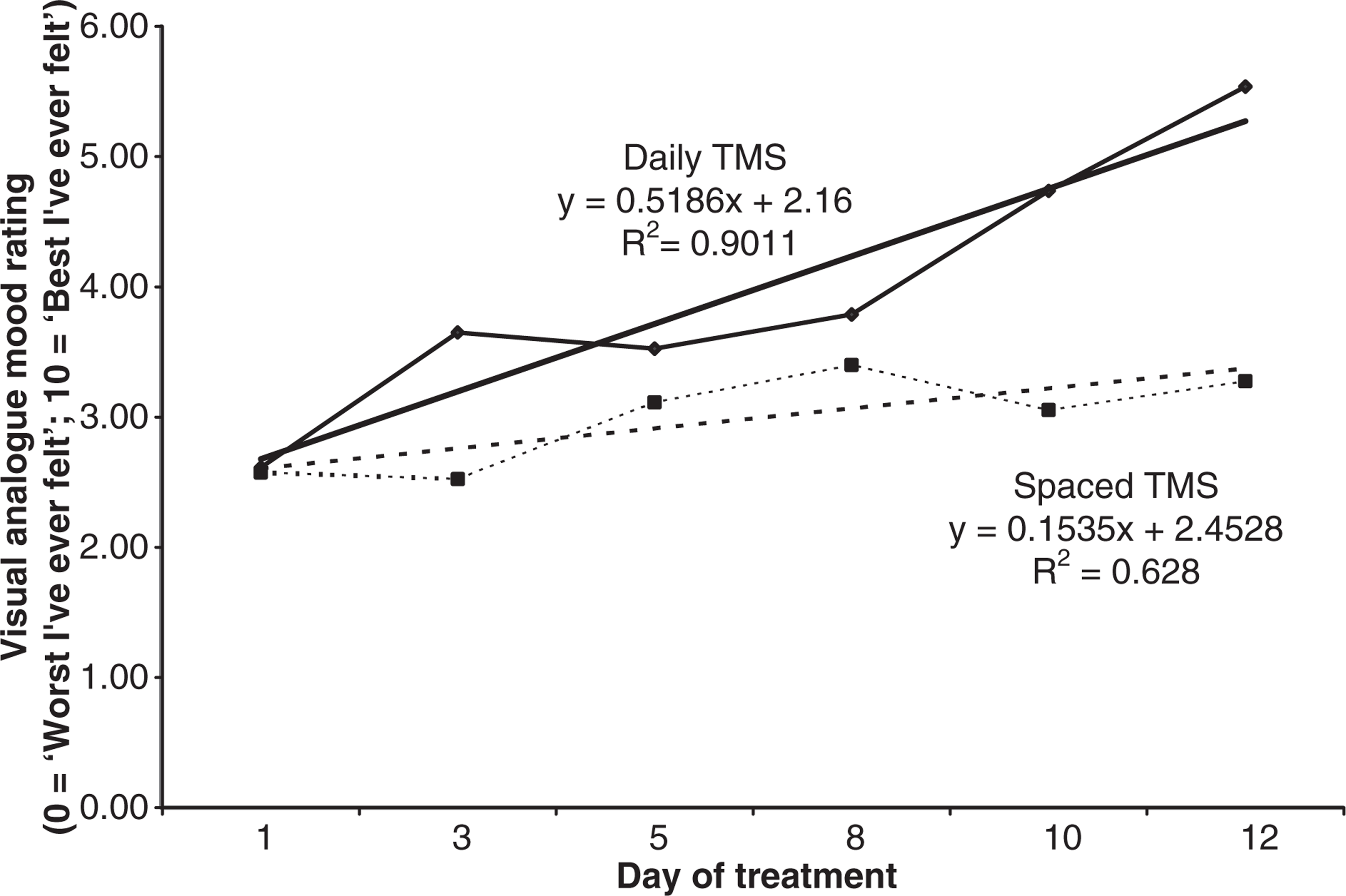
Summary of treatment variables for patients in daily and spaced TMS treatment groups (standard deviations in parentheses)
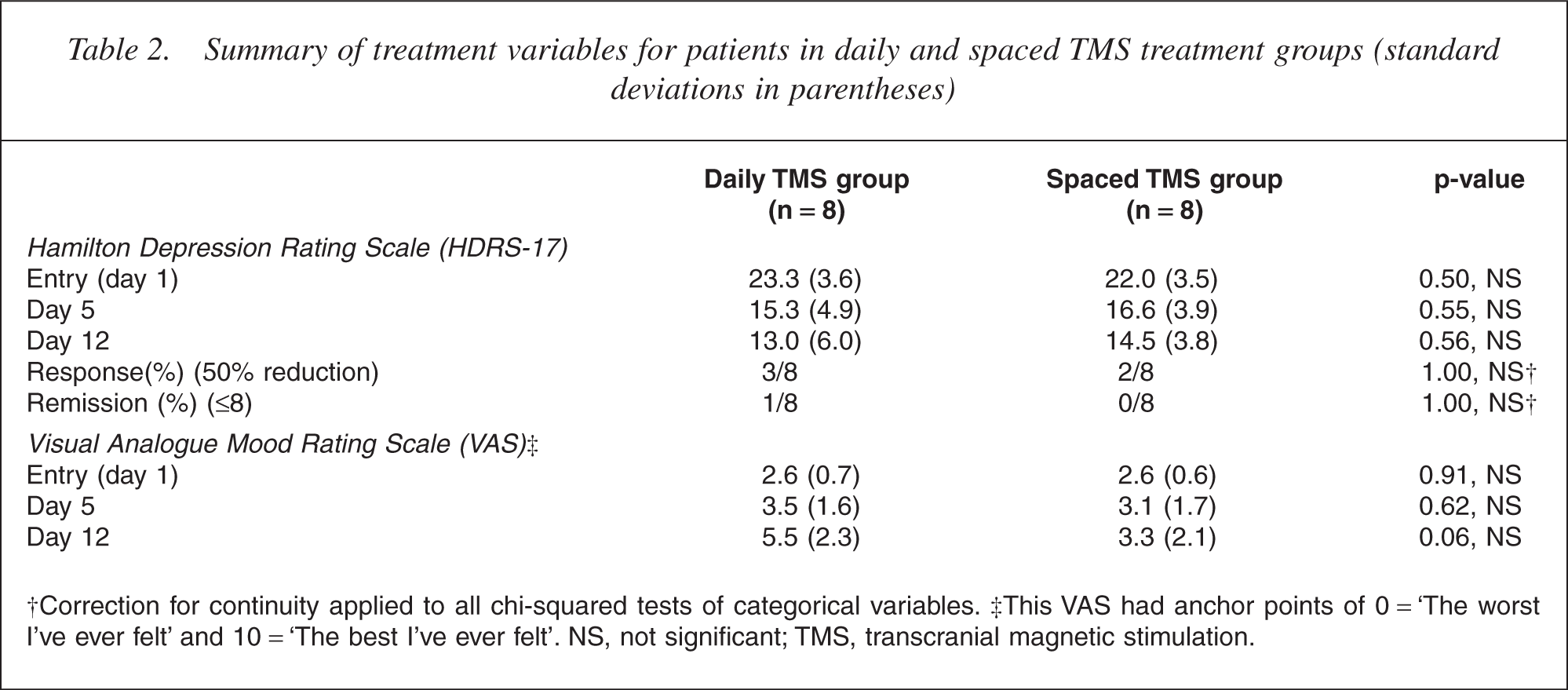
†Correction for continuity applied to all chi-squared tests of categorical variables. ‡This VAS had anchor points of 0 = ‘The worst I've ever felt’ and 10 = ‘The best I've ever felt’. NS, not significant; TMS, transcranial magnetic stimulation.
While there were significant improvements for both groups in depression over the course of the study, Bonferroni-adjusted pairwise comparisons indicated that, for both treatment groups, there was a significant improvement in objective ratings of depression (HDRS) between days 1 and 5, but not between days 5 and 12; however, for subjective (VAS) ratings of mood, there was no significant differences between ratings made between days 1 and 5, and between days 5 and 12, for either treatment group.
There were no significant differences between the treatment groups at day 12 in terms of the proportion of patients achieving response, achieved by three patients in the dTMS group and two in the sTMS group. Similarly, there were no differences in the proportion of patients achieving remission at day 12, which was achieved by one of the dTMS patients but none of the sTMS patients (Table 2).
TMS dose-response relationships
A significant linear trend showing continual improvement in VAS ratings of mood was shown for the dTMS group over the first 12 days of treatment (Fig. 1, F(1,42) = 15.78, p < 0.001). No higher-order trends were found to contribute significantly to this relationship (deviation F(4,42) = 0.54, p = 0.71). This suggests that dTMS treatment obeyed a dose-response relationship over this period of treatment.
However, while a linear trend provided a reasonable fit for the general improvement in VAS ratings of mood for the sTMS group over the first 12 days of treatment (Fig. 1), this linear trend was found not to produce a significantly better fit than a null model (F(1,42) = 1.33, p = 0.26). Inclusion of higher-order trends did not significantly improve this fit (deviation F(4,42) = 0.187, p = 0.94).
Discussion
We set out to examine whether time is an important factor in the TMS treatment of major depression [7]. As far as we are aware, this is the first reported comparison of sTMS and dTMS. Spaced TMS has rarely been described. A recent report [23] of low-frequency TMS applied to the right frontal area twice weekly for 4 weeks described significant improvement in depression scores. However, there was no comparison with either daily active or placebo treatment. There are two other reports of spaced treatments in the literature [24, 25], but these are attempts at maintenance rather than acute treatment.
We found no statistically significant differences in improvement for patients treated with 5 versus 10 treatments over a 2-week period. This suggests that the accumulated dose is not solely responsible for the efficacy, as in that case the dTMS group should have improved to a greater degree at day 12.
Medication deserves mention. In this study, all patients were receiving antidepressant medication, some were also receiving mood stabilizers or benzodiazepines. As patients had been unwell for an average of 11-12 months and were treatment-resistant, it would have been extremely difficult to collect a sufficient number of patients who were not taking psychotropic medication. We attempted to exclude medication effects by including only patients who had been on a stable dose of medication for at least 2 weeks before entry and making no changes during the study. We therefore believe that there was no effect of medication on outcome.
Limitations of this study include the small numbers, which were partially the result of strict inclusion and exclusion criteria and our relatively small patient pool. It is possible that the lack of difference between the groups represents Type II statistical errors and we considered increasing the number of subjects in this study. However, the observed differences between groups were small (about two points on the HDRS at most comparisons) and, given our circumstances, unattainably large samples would be required to achieve adequate statistical power to reliably detect such differences. A sample of 64 patients per group would be required to identify a twopoint difference on the HDRS (calculated for power of 0.8 and an alpha of 0.05). Such a small between-groupdifference is possibly of little clinical utility. This study may stimulate a group with greater resources to conduct a similar study with greater patient numbers.
An alternative would be to alter the design to increase the between-group difference. Instead of recruiting additional subjects for an extension of the present study, we have decided to follow this pilot with a study of improved design. To investigate the effect of spaced treatment further, we are considering comparing dTMS (10 treatments over 12 days) and sTMS (10 treatments over 26 days). In the current study, the sTMS group received only five treatments. Studying the effect of 10 treatments would more closely approximate current clinical practice as well as potentially increasing the effect size.
Overall, this study supports the efficacy of TMS in the treatment of treatment-resistant depression [5–13]. Five of 16 patients achieved response after having been unresponsive to a range of treatments for an average of 11–12 months treatment. This is a quite small response rate, but treatment was only studied over 2 weeks. Our clinical practice is to continue treatment as long as improvement is occurring [8, 26], and this is in accordance with the longer courses of treatment described by others [5–7].
A placebo group was not included. Previous studies have already established TMS to be superior to placebo [5–7,9, 15]. Rather than to add to the existing evidence of treatment efficacy, this study was designed to investigate the influence of time on treatment outcome.
Within the limitations of the study, we found time to be an important factor of treatment outcome. This is consistent with a range of medical treatments, from antibiotics to antidepressants and ECT. Accordingly, sTMS may be a clinical option. Spaced TMS would be less burdensome on both patients and staff, and allow greater numbers of patients to be treated.
