Abstract
Over the last 10 years, a considerable number of trials have been conducted of repetitive transcranial magnetic stimulation (rTMS) for treatment of depression (e.g. 1– [5]). The majority of these have focused on rTMS applied to the left dorsolateral prefrontal cortex (DLPFC) and several meta-analyses have indicated that this has effects on excess of sham stimulation [6–9]. Reports have also suggested that low-frequency stimulation applied to the right DLPFC [1, 10] may be effective and that the response rate to rTMS is similar to electroconvulsive therapy (ECT) in patients with non-psychotic depression [11–14]. Little is known about the effect of rTMS on the course of depression, a disorder typified by relapse and remission. There has been only one report, including only four patients, of the use of rTMS in the treatment of subsequent episodes of depressive relapse [15]. These patients were successfully retreated on average 6.9 ± 4.8 months after the original treatment [15]. The aim of the current study was to investigate the therapeutic efficacy of repeated courses of rTMS in the treatment of depressive relapse in patients who had previously responded to treatment.
Method
Subjects
Nineteen patients were included who received treatment in the rTMS program of Southern Health, Dandenong Hospital (2000–02) and Alfred Psychiatry (2002–04) in Melbourne, Australia. All patients had previously received rTMS in one of the three randomized controlled trials. The first trial consisted of three treatment arms, low-frequency right-sided PFC stimulation (LFR), high-frequency left-sided PFC stimulation (HFL) or sham [1]. Most sham treated patients subsequently received active treatment. The second study (unpublished data) consisted of the same two active treatment arms but no sham. The third study [16] consisted of either sham or bilateral stimulation involving both the treatment conditions used in study one provided sequentially in each treatment session. A total of 134 patients participated in these two studies between October 2000 and August 2004. At the completion of the initial treatment trials, patients who were clinically judged to have experienced a benefit from rTMS were informed that they could return for further treatment if there was a subsequent significant recurrence of depressive symptoms. The patients who participated in this study had been successfully treated with TMS and were re-referred by their treating psychiatrists following relapse. No formal criteria were used to define relapse. All patients who were re-referred for repeated treatment were provided further rTMS. All patients had initially received active treatment (in either double-blind or open label cross over phases of the initial trials). All patients provided written informed consent before each treatment session. Comprehensive data are not available on relapse rates in patients not referred for further treatment.
Of the 19 patients, 16 had a diagnosis of major depressive episode and three patients had bipolar disorder, depressive episode (DSM-IV criteria as of the time of the first episode). None were psychotic. The mean age was 45.6 ± 10.8 years. There were 11 male and eight female patients. During active treatment in the initial clinical trials, these patients experienced a decrease in Beck Depression Inventory (BDI) scores from 30.1 ± 10.9 to 10.1 ± 7.5 (p < 0.001) and Montgomery Asberg Depression Rating Scale (MADRS) scores from 35.9 ± 7.6 to 15.4 ± 8.0 (p < 0.001). Thirteen patients met criteria for clinical response (<50% reduction in MADRS scores) and the other six partial response (between 25% and 50% reduction). At initial treatment, they had unsuccessfully received treatment with 4.8 ± 4.4 antidepressants. Six patients had previously received ECT. Two of these had received ECT for a previous episode and achieved a response but significant sideeffect. The other four had received ECT during the episode in which they had received rTMS. None of these patients had responded to ECT. The mean age of onset was 32.3 ± 11.5 years and the patients had 2.9 ± 3.3 previous depressive episodes. At the time of their initial trial treatment, 15 patients were receiving antidepressant medication and five a mood stabilizer. Four were medication free. All patients continued to receive ‘treatment as usual’ between the treatment sessions. This usually consisted of antidepressant medication with a number of patients also receiving mood stabilizers at various times during the treatment courses. The dose of antidepressant medication prescribed was not permitted to change during any of the periods of rTMS treatment or the 4 weeks prior. The primary outcome measure used to assess treatment response was the BDI [17]. This was administered before the commencement of each session and within 24 hours of the end of each treatment course.
rTMS
For all patients, rTMS was provided in a similar fashion in which it was provided to an individual during the clinical trial in which they responded (Table 1). Therefore, treatment was provided with either HFL rTMS, LFR rTMS or a combination of both. Resting motor thresholds were measured by standard electromyography means before each treatment course [18]. Treatment was always provided at the same site and frequency. For HFL rTMS, 30 5-second trains were provided at 10 Hz and 100% of the resting motor threshold (RMT). Lowfrequency right-sided TMS was provided in a single 9-minute train of 1 Hz stimulation at 110% of the RMT. Where bilateral rTMS was used, left-sided stimulation was 15 5-second 10 Hz trains at 100% of the RMT, and right-sided stimulation was one 7-minute 1 Hz train at 100% of the RMT (right-sided stimulation always immediately preceded left in each treatment session). To maintain consistency with the initial clinical trial protocols, treatment was provided for a maximum of the same number of treatment sessions provided in initial treatment (20 for right- and left-sided treatment and 30 for bilateral treatment). The mean number of days per course and treatment type is presented in Table 1.
Treatment details for each episode
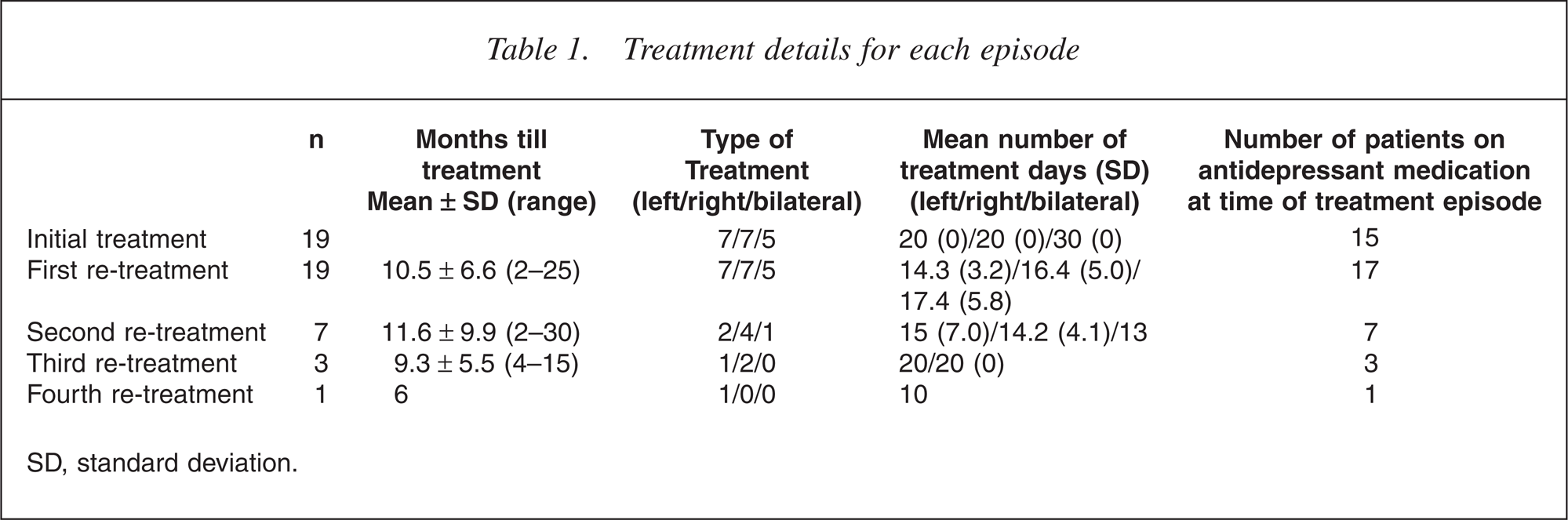
SD, standard deviation.
Stimulation was provided with a Magstim Super-Rapid magnetic stimulator (Magstim, Sheffield, UK) between 2000 and 2003 and with a Medtronic Magpro 30 (Medtronic, Minneapolis, MN, US) after this time. Stimulation was always provided using a stand-held 70 mm figure-of-8 coil. At all times, the coil was held tangential to the scalp with the handle pointing back and away from the midline at 45 degrees. The site of stimulation during the TMS treatment sessions was defined by a point 5 cm anterior to that required for maximum stimulation of the abductor pollicis brevis. Thresholds and the site for treatment were measured at the start of each treatment course and then kept constant.
Statistical analysis
The mean duration until each representation was calculated along with the change in depressive severity with treatment. Paired t-tests were calculated for the treatment response to each course of rTMS. The relationship between the degree of response (response, partial response) in the initial trial treatment and the response (response, partial response, non-response) was studies with correlation analysis. All procedures were two-tailed and significance was set at an alpha level of 0.05. All statistical analysis was conducted with spss 11.5 (SPSS Inc., Chicago, IL, US).
Results
Nineteen patients completed a total of 30 treatment courses: eight completed one course, seven completed two courses, three completed three courses and one completed four. Seven patients received HFL treatments, seven LFR and five bilateral. No major adverse events were experienced during any of the treatment episodes.
The duration between the end of participation in the initial clinical trial and presentation for the first treated relapse and between subsequent treatment courses is presented in Table 1. Mean pre- and postdepression scores for each episode are presented in Table 2. There was a mean improvement in BDI scores of 53.9 ± 26.0% for the first repeated treatment, 49.5 ± 33.2% for the second and 81.4 ± 3.8% for the third. For the first repeated course, 12 patients achieved a greater than 50% reduction in BDI score, five patients improved between 25% and 50% (two between 40% and 50%) and two patients did not respond. For the second repeated treatment, four (of seven) achieved a greater than 50% reduction (plus one of 48.6%). All of the patients having a third (n = 3) or forth treatment (n = 1) achieved a greater than 50% reduction.
BDI scores before and after each treatment episode
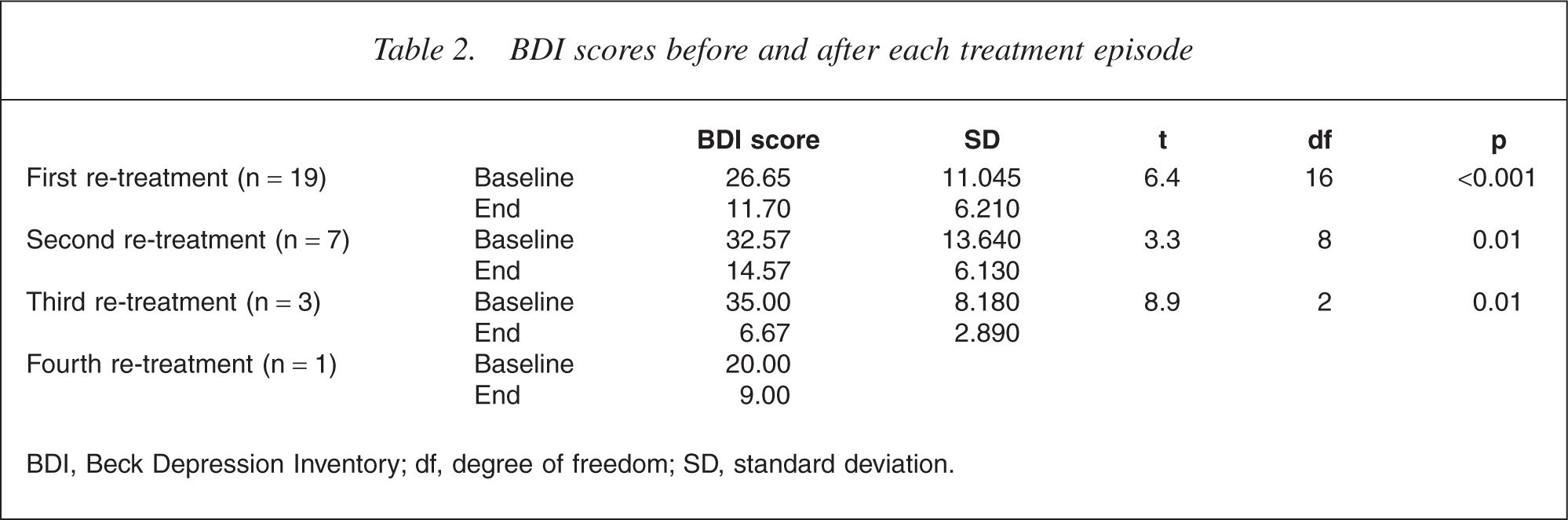
BDI, Beck Depression Inventory; df, degree of freedom; SD, standard deviation.
For the first relapse treatment, there was a significant reduction in BDI scores for patients having HFL rTMS (25.4 ± 12.7 to 13.2 ± 9.2, p < 0.005), LFR rTMS (30.0 ± 8.0 to 11.7 ± 5.5, p < 0.05) and bilateral rTMS at a trend level (29.7 ± 13.2 to 10.0 ± 4.7, p = 0.06). For HFL rTMS, three patients responded, three partially responded and one did not respond. For LFR rTMS, five patients responded, one partially responded and one did not respond. For bilateral rTMS, four patients responded and one partially responded.
A significant relationship was not present between those patients who achieved a full response in the baseline and first treatment episodes (Fisher's Exact Test, p = 0.62). Of the 13 responders in the baseline episode, nine responded, two had a partial response and two failed to respond at all. Of the six original partial responders, three had a full and three had a partial response. Of the four patients who made a full response to the third treatment course, three had made a full response originally and one had a partial response.
Discussion
The considerable majority of rTMS research studies to date have addressed issues about the efficacy of the technique in initial antidepressant treatment. To date, there has been little published research addressing issues relating to the ‘real world’ efficacy of the technique. The data provided in this report suggest that for the majority of patients, rTMS maintains its efficacy when repeatedly applied and it may prove to have treatment value in the treatment of repeated depressive relapses.
The major limitation of this study lies in the nature of the assessments conducted. The study was not blinded and we have only gathered comprehensive data on the patient rated BDI. This has established validity, however, and should realistically reflect the impact of the treatment of these patients and their lives in an unbiased fashion. There are considerable practical impediments to conducting this type of study in a true double-blind fashion. Most importantly, it is not practically possible to randomize patients who have received active treatment in one episode into groups that have a sham condition as the blinding would be markedly compromised. In addition, as all the patients were re-referred for treatment and a small group (four) had initially received treatment in the non-sham controlled trial, these factors may have created an expectation of response enhancing potential placebo effect. The other major limitation relates to the heterogeneity of antidepressant medication treatment that was not controlled between episodes and subjects. However, all patients were receiving good psychiatric care between episodes and relapsed despite the use of antidepressant medication. They were also multiply medicationresistant before inclusion in the original studies which limits the value that they were likely to have received from medication.
Although a large number of rTMS studies in depressed patients have been conducted, doubts about its efficacy and place in clinical treatment continue [19]. In addition to acute-phase clinical trials, it is important to assess the impact of a new treatment on the clinical course of depressive disorder. This will require long-term assessments of variables such as quality of life and functioning as well as depressive symptoms. It also requires consideration of the impact of the treatment on relapses of the illness. In this context, the data we have collected in this large series have several implications. Most importantly, given the relapsing nature of depression, it supports the notion that rTMS may be applied across time for the treatment of successive depressive episodes. Second, it suggests that this repeated efficacy applies to both leftand right-sided treatment and probably to bilateral treatment as well. To date, there have been no reports of the repeated efficacy of treatments other than HFL [15]. In addition to these reports, there are now a number of small case series of the use of rTMS as a maintenance treatment [20]. Despite the limitations of these studies, they provide accumulating evidence that there is therapeutic value in the provision of rTMS treatment in the management of depression post an acute episode. Further research is required to comprehensively assess the safety and cost-effectiveness of these treatment approaches.
Footnotes
Acknowledgements
This study was supported by a grant from National Health and Medical Research Council (NHMRC) (143651) and by Constance and Stephen Lieber through a NARSAD Lieber Young Investigator award (PF). Thanks to the patients whose participation was essential in the successful completion of the study.
