Abstract
Neurophysiological investigations using transcranial magnetic stimulation (TMS) are a growing area of psychiatric research. Transcranial magnetic stimulation is implemented by passing a time-varying current through a coil held to the surface of the head. The resultant magnetic field passes easily through the skull and induces a small electric current in the cortex. Stimulation over the motor cortex results in an observable muscle movement and motor evoked potential (MEP). Motor evoked potentials from the contralateral abductor pollicis brevis can be measured via electrodes placed on the muscle surface. Studies using TMS have shown an increase in amplitudes of motor evoked potential after non-fatiguing exercise, a phenomenon called post-exercise facilitation.
To date, neurophysiological studies have focused on the motor cortex due to the ease of quantifying the effect of TMS. They have been split between assessing the integrity of cortical connections (e.g. central motor conduction time), and the measurement of the motor excitability (e.g. motor threshold, MEP input-output curves). Post-exercise facilitation is a period of increased motor excitability that occurs after brief muscle activation and decays to baseline over 2 to 4 minutes. The mechanisms underlying it have been established in normal subjects to be within the motor cortex [1]. The predominant action of TMS is to activate corticospinal cells trans-synaptically. There is no exercise facilitation when corticospinal cells are stimulated at the proximal axon by transcranial electric stimulation. This suggests an intracortical localization of facilitatory mechanisms induced by exercise [1]. Ultimately facilitation will be determined by cellular mechanisms, (e.g. neurotransmitter release, modification of calcium concentrations at this location).
Impaired facilitation has been reported in depressed patients [2, 3] with a call for independent replication [3]. These studies support the hypothesis that cortical regions are less excitable in depression. The addition of a group of patients with schizophrenia is included in this study to determine the specificity of the previous findings in depressed patients.
Subjects
The study was approved by the Royal Hobart Hospital Research and Ethics Committees, and informed written consent was obtained from all subjects. All patients and controls were right handed. Exclusion criteria were a history of head injury, epilepsy, significant neurological and medical disorders and substance abuse.
Patients were consecutively recruited from inpatients of the Royal Hobart Hospital, and outpatients referred specifically for the study by private psychiatrists. Patients underwent a clinical interview to confirm they fulfilled DSM-IV criteria for major depressive episode (unipolar or bipolar) or schizophrenia. All patients were taking psychotropic medication. Of the depressed patients three were taking fluoxetine (20–40 mg), three were taking venlafaxine (150–225 mg) with the remainder taking nefazodone (600 mg), fluvoxamine (200 mg), sertraline (150 mg) and moclobamide (600 mg). Of the patients with schizophrenia, four were taking risperidone (2–4 mg), three were taking olanzapine (10–30 mg), three were taking clozapine (200–325 mg) and one was taking quetiapine (400 mg). Thirteen healthy volunteers (nine females and four males aged between 25 and 54) were recruited from the staff employed at the Royal Hobart Hospital. A screening interview confirmed the absence of any medical or psychiatric illness.
Methods
Patients and controls had their motor cortex excited with transcranial magnetic stimulation at rest and after exercising the abductor pollicis brevis (APB) muscle. The resulting motor evoked potentials were measured from electrodes placed over the contralateral APB.
The stimulation procedure consisted of the subjects being seated comfortably with their forearms being supported by pillows. They were instructed to keep both hands still and relaxed as possible. Electromyograph (EMG) electrodes were placed on the right thenar eminence and the earth was placed on the back of that hand. Electromyographs were recorded using a Nicolet Viking IV (Nicolet Biomedical, Madison, Wisconsin). Transcranial magnetic stimulation was applied by a Magstim 2000 (Magstim Company, Whitland, UK) and a 70-mm figure of eight coil to the left motor cortex. The coil was placed over the primary motor cortex and systematically stimulated to elucidate the largest consistent MEP response from the APB. This site was marked with a skin pencil for future reference. Over this point on the scalp the motor threshold (MT) was determined as the minimum stimulus intensity required to cause an MEP response of at least 50 microvolts from a minimum of five out of 10 consecutive stimuli. Transcranial magnetic stimulation was administered to individual subjects at 110% of MT.
After mapping the motor cortex and determining motor threshold 16 baseline recordings were produced using trains of four with a interstimulus interval of 3 seconds. Trains were separated by 30 seconds, giving 16 recordings over two minutes.
The subjects were then asked to perform a hand exercise followed by a train of TMS at the same stimulus parameters creating facilitated MEPs, which were recorded. The hand exercise consisted of opposing the thumb using a dynamometer. Initially, subjects were asked to oppose their thumb as hard as possible to determine the maximal voluntary contraction (MVC). At least 10 minutes later the subject was asked to exercise for 30 seconds at 20% of their MVC using the same dynamometer. The figure of 20% was chosen to balance MEP enhancement with possible subject fatigue and enable comparison with the work by Shajahan [3]. On relaxation, a train of four TMS was given 3 seconds apart. The whole procedure was repeated four times with around 30 seconds between trains, resulting in 16 post-exercise MEP recordings. In summary 16 baseline recordings and 16 post-exercise recordings were produced for each subject in one session. This protocol was adopted due to equipment parameters and time constraints during this session, but ensured that recordings were within the time frame of facilitation decay. In contrast Shajahan had 50 baseline and 25 post-exercise stimuli recordings [3].
Data analysis
Data was analysed using the statview 5.0 (1998, SAS Institute, Cary, North Carolina) computer package. The measure of post-exercise facilitation was calculated in two forms to enable comparison with other studies [2, 3]. These were the difference between mean exercise and baseline amplitudes, and the percentage increase of mean exercise amplitudes over mean baseline amplitudes. The MEPs were normally distributed and parametric inferential statistics were used. One-way three group ANOVAS were used to analyse differences in continuous dependent variables. Pair wise comparisons using t-tests were used to further elucidate differences within significant main effects and interactions. χ2 was used to examine differences in proportions of males and females in each group.
Results
All subjects tolerated the procedure well, without any adverse effects. Table 1 summarizes the demographic and MEP data. Significant differences in age were seen between all groups, with schizophrenia group being the youngest (F2,31 = 13.85, p = 0.0001). There was a significant differences in the proportions of males and females in each group (χ2 = 6.78, df = 2, p = 0.03).
Mean demographic and motor evoked potential(MEP) results
TMS responses
Figure 1 shows a significant difference in pre-exercise MEPs for the 3 groups (F2,31 = 5.14, p = 0.01). T-tests revealed that the control group had significantly lower baselines than both the depression (p = 0.009) and schizophrenia groups (p = 0.012). Post-exercise MEP recordings for the three groups were more widely spread (F2,31 = 1.54, p = 0.23). The schizophrenia group was highest but failed to be significantly greater than both the control and depression groups. Motor threshold for the three groups were similar. A between groups ANOVA indicates no significant difference between the groups (F2,30 = 0.35, p = 0.71).
Motor evoked potential facilitation preand post-exercise. □, control; ⋄, depression; and ○, schizophrenia.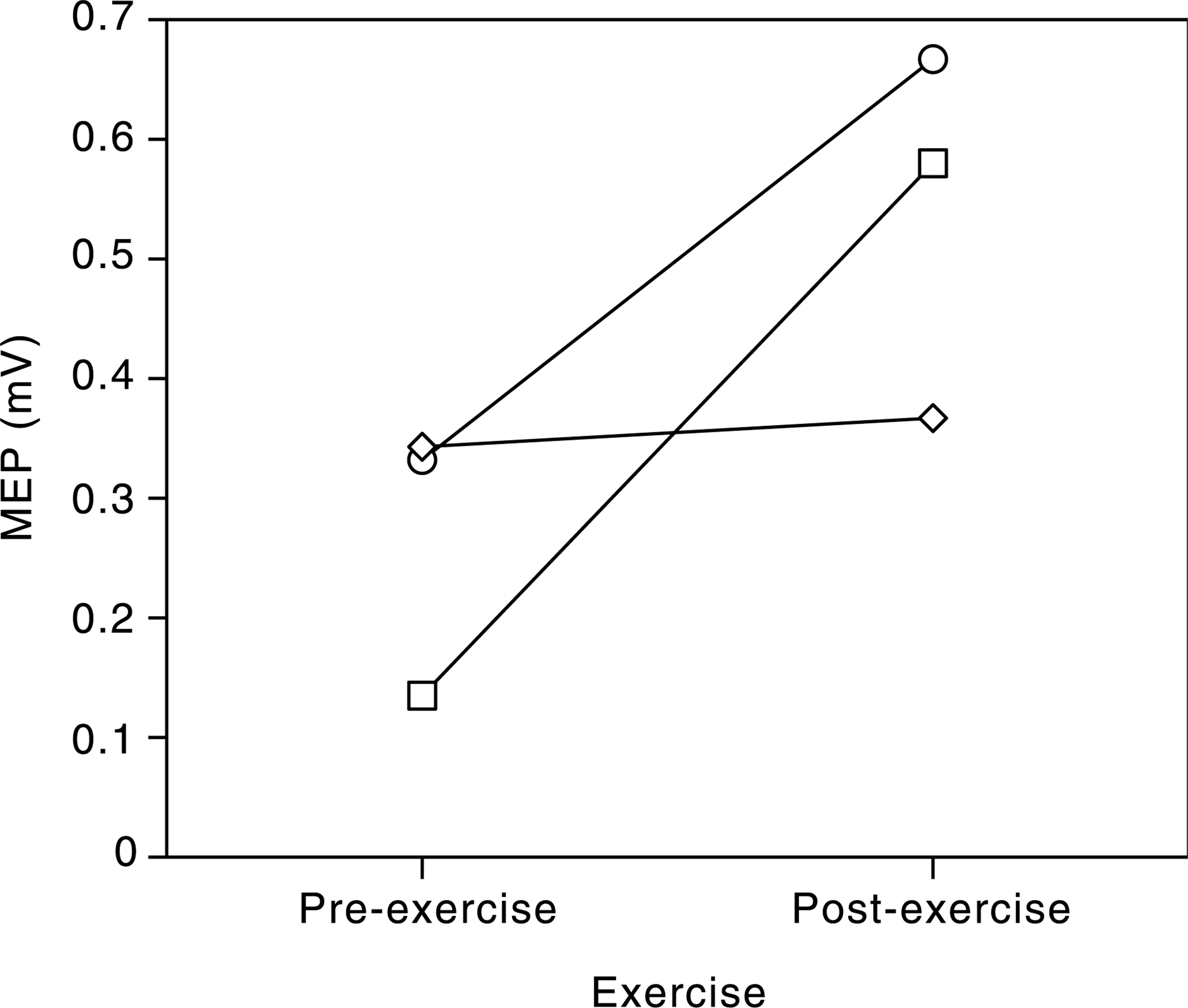
Measurement of facilitation
Table 1 shows facilitation as measured by post minus pre-exercise MEPs. There was a significant difference between the 3 groups (F2,31 = 5.04, p = 0.013). T-tests revealed that the depressed group was significantly lower than both the control (p = 0.004) and schizophrenia groups (p = 0.04). Table 1 also states facilitation as a percentage of baseline ratio. Again, there is significant difference between the groups (F2,31 = 12.02, p = 0.0001). T-tests revealed depression (p = 0.0001) and schizophrenia (p = 0.001) to be significantly lower than controls but not each other.
Discussion
This study confirms the work of others [2, 3] by demonstrating that post-exercise facilitation is reduced in depressed patients compared to controls. The main limitation is that the patient groups were medicated but the control group was drug free. However, Shajahan [4] repeated testing of post-exercise facilitation on recovered depressed patients taking antidepressant medication and found a return of facilitation similar to controls. This suggests a state variable that is largely independent of medication effects. Post-exercise facilitation has an intracortical mechanism. Facilitation reduction in depression may reflect a change in the balance of interneurone inhibitory and excitatory inputs at this level to corticospinal neurones [3]. Interestingly, post-exercise facilitation in the control group was relatively high compared to other studies [2, 3]. As facilitation is effected by exercise type (fatigue or non-fatiguing) and time course [1], subtle differences in protocols between studies may account for this.
Motivation is reduced in both schizophrenia and depression [5]. Apathy could possibly influence the results when performing any exercise procedure. However, the facilitation process occurs with minimal exercise and shows no significant difference over a range of different muscle activation [1]. This fact minimizes any effect that reduced motivation to perform the exercise paradigm will result in artificially lowering post-exercise facilitation.
Motor threshold reflects neuronal membrane excitability which is dependent on calcium channel conductivity [6]. The depressed group had the lowest mean motor threshold but was not significantly different to the other two groups. Abarbanel [7] studied electrophysiolgical responses to transcranial magnetic stimulation in depression and schizophrenia and also found motor threshold to be similar for medicated depressed patients and controls. In contrast, Made [8] found a non-medicated depressed group to be significantly lower than controls particularly of the left hemisphere. The contrast between the studies may reflect medication effects.
Facilitation results for the schizophrenia group were less emphatic. In this group MEPs increased after exercise but lay between the control and depressed participants. Other studies have provided post-exercise facilitation data in the form of percentage of base line and a quantitative figure of post-exercise MEPs minus pre-exercise MEPs. These alternate ways of expressing facilitation when applied to this data still gives a clear decreased result for depression. However, using the percentage baseline method, the schizophrenia group becomes significantly less than the control group but not statistically different from the depression group. The opposite occurs for the post minus pre-exercise method. We consider the percentage baseline method to be the most useful when evaluating facilitation, because there is considerable individual differences of motor threshold and baseline MEPs which are stable over time [9]. Considering a ratio tends to negate this factor.
There have been no other comparable studies looking at post-exercise facilitation in schizophrenia. Repeating the protocol with drug naive patients would be important to differentiate between the pathophysiology of the disease and a possible drug effect. Davey [10] looked at the effects of antipsychotic medication and found no difference in motor threshold between medicated and drug naive patients with schizophrenia but that it did effect latency and form of inhibitory periods to TMS. Boroojerdi [11] demonstrated delayed and prolonged transcallosal inhibition in schizophrenia, but also found no difference in motor threshold between medicated patients with schizophrenia and controls as we did. In contrast, Abarbanel [7] found significantly lower motor threshold in schizophrenia compared with depression and controls. Their schizophrenia group also exhibited increased amplitude of TMS elicited MEPs.
The other notable finding of our study was a significant difference between the control and patient groups for MEP baseline recordings. Patient groups were both considerably higher. Abarbanel felt muscle rigidity secondary to medication may influence results from patients with schizophrenia. Although our group were on atypical antipsychotics, extrapyramidal side-effects could account for baseline MEPs being higher than controls. However, this does not explain the depressive group being higher, which is in contrast to Shajahans group of depressives who had baseline recordings comparable to controls. The inconsistency between studies may have resulted from small sample size, minor differences in stimulus intensity (percentage of motor-threshold) and exercise intensity (percentage of MVC) used.
One of the limitations of the study was that the controls did not exactly reflect the age of the two groups. However, the mean age of controls lies between the mean age for schizophrenia and depression which largely negates age being a contributory factor to the differences in facilitation. The sex ratio between groups was also unmatched though this reflected the ward population of these illnesses. Other limiting factors include the small sample size to possibly enable depression to be distinguished further from schizophrenia. Providing more detail on the type and severity of the disorders may have aided further discrimination. Subjective fatigue was also not measured. However, excessive fatigue in subjects was not the experience of the researchers who guided the patients through the protocol.
Altered facilitation may reflect a general feature of these mental disorders both of which have common clinical abnormalities of motor function. Further investigation, paying greater attention to subtyping, age of onset and acuity of symptoms, may give a greater division between the groups. Despite this, post-exercise facilitation may be valuable as a potential tool to aid diagnosis, subtyping of illness, effect of treatment and recovery [4].
Probing the integrity of connections within the cerebral cortex is an expanding field in neuropsychiatry. It is particularly relevant as part of the pathophysiology of such disorders as schizophrenia and depression is presumed to be mediated through disordered cortical connectivity. Further research is needed to determine whether these studies are going to be relevant in clinical practice. Our study does not support post-exercise facilitation being specific for a particular clinical entity. In addition to understanding psychiatric disorders, potential therapies may ensue by identifying altered cortical responsiveness or excitability. There is evidence that TMS can alter excitability [12] and potentially stabilize this component of neuropsychiatric disorders.
Footnotes
Acknowledgements
We thank Gina Page for assistance in recording MEPs.
