Abstract
The neuropsychological and immunological domains are closely interrelated, and psychoneuroimmunological homeostasis is important to maintain a healthy life [1]. Immunological dysregulations play a role in the pathogenesis of major psychiatric disorders such as schizophrenia [2, 3] and bipolar disorders [4].
Autoimmunity is an immunological dysregulation, in which the host generates antibodies to act against selfantigen. Psychiatric symptoms are commonly seen in primary autoimmune disorders, such as systemic lupus erythromatosus [5]. Some studies have also showed that patients with primary psychiatric disorder have a high prevalence of some autoantibodies [6–8]. Patients with schizophrenia have higher rates of anti-heat shock protein (anti-HSP) antibodies [8–11]. Previous autoimmunity studies of patients with bipolar disorder were focused on the antithyroid antibody, which is increased in patients with bipolar disorder, and its increase is independent of lithium treatment [6, 7]. A recent study of autoimmunity profile showed that patients with bipolar I disorder had increased levels of HSP40 [12]. An extensive survey also showed that patients with bipolar disorder had increased antibodies against thyroperoxidase, H/K adenosine triphosphatase and glutamic acid decarboxylase-65 [13]. Furthermore, antithyroglubulin antibody titres have been found to be associated with the symptoms of borderline personality disorder [14]. Taking these findings together, we thought that increased autoantibody level might be associated with psychiatric illness. Thus, the aim of this study was to test the hypotheses that serum autoantibody levels might be changed during different clinical phases (mania, remission and depression) in patients with bipolar disorder.
Method
Subjects
We recruited patients with bipolar mania from inpatients of the Department of Psychiatry, Taipei City Hospital, Taipei, Taiwan. This study was approved by the institutional review board of Taipei City Hospital. The study patients were clinically diagnosed by Drs S.-Y. Tsai and K.-P. Chen using a validated semistructured schedule, the Psychiatrist Diagnosis Assessment, which was described elsewhere [15]. They met the DSM-IV diagnostic criteria for bipolar disorder I, manic phase. The severity of patients' manic symptoms was rated with the Young Mania Rating Scale (YMRS) [16]. The decisions for patients' final diagnoses were reached at consensus meetings after consideration of all available information, including family members' confirmation and review of medical records.
Eligible for the study were physically healthy bipolar patients who were younger than 45 years of age and had YMRS scores greater than 29. Excluded were patients with current mixed episode or comorbid substance-use disorders. When blood samples were obtained from acute mania patients, 57 of them were free of any psychotropic drugs for at least 2 weeks (designated as unmedicated subgroup), and 37 patients had been treated with psychotropic agents (designated as medicated acute subgroup). During the index hospitalization, all study patients were treated with various combinations of lithium, carbamazepine, valproate and antipsychotics. The follow-up blood samples were collected when patients were in consequent remission with YMRS scores lower than 12 and were free from any sign of depression. We also recruited healthy control subjects from our hospital staff and Taipei Medical University students. After signed informed consent, all the subjects received physical examinations and laboratory tests, including complete blood counts with differentials, serum enzyme and metabolite screenings, urine analysis and thyroid function tests. Excluded from the study were subjects who had shown symptoms of chronic or acute infection, allergies, history of autoimmune diseases or other conditions known to affect the immune system for at least 2 weeks before the study. They were also free from using other concomitant drugs known to alter immune or endocrine functions.
Following an overnight fasting from 24.00 hours the preceding night, the study patients and the normal controls gave blood samples between 08.30 hours and 09.30 hours to control for variations in circadian rhythmicity. Heparinized bloods were drawn by venous puncture, and the sera were collected and stored at −80°C until assay.
Western blot analysis of antibodies to U373 MG cell lysate and anti-HSP antibody detection
We first used human glioblastoma cell line U373 MG cell lysate to screen the autoantibodies for the study subjects. Equal amounts of U373 MG cell lysate (50 μg mL−1) were loaded to the 10% SDS-PAGE and hybridized with patients' sera (1:200 dilution). In addition, we used pure HSP (HSP60, 70 and 90, from StressGen, Ann Arbor, Michigan, USA) for 10% SDS-PAGE. Briefiy, we mixed recombinant HSP60, 70 and 90 (100 ng well−1) with 4X SDS loading buffer, boiled it for 95°C for 5 minutes and then separated it on 10% SDS-PAGE. Gels were transferred to the NC paper with 100 mA at 4°C for 12–14 hours. Blots were blocked with 5% non-fat dry milk at 4°C for 8 hours, and then cut into strips and blotted with 1:200 dilutions of sera from our patients and controls at 4°C for overnight. After washing, a secondary antibody with a 1:4000 dilution of the mixture of antihuman κ and antihuman λ antibodies (Cappel, Irvine, CA, US) was incubated at room temperature for 2 hours. After washing, we exposed the blots with enhanced chemiluminescence (Amersham Pharmacia Biotech, Buckinghamshire, UK).
ELISA analysis of anti-HSP60, 70 and 90 in patients with bipolar mania
The next step was to measure the levels of these antibodies. We used enzyme-linked immunosorbent assay (ELISA) with biotin-advidin amplify system for quantification. Pure HSP60, 70 and 90 antigens (StressGen) were coated on the 96-well plates with a concentration of 1 μg mL−1 at 4°C overnight. Then, we blocked the plates by 1% bovine serum albumen in phosphate buffer saline at room temperature for 2 hours and then washed three times for use. We added patients' sera with proper dilutions to the plates in duplicate and incubated them at 37°C for 2 hours. The antibody was then detected by the secondary antibody, which was the mixture of horseradish peroxidase-conjugated antihuman κ and antihuman λ antibodies (Cappel) incubated at room temperature for 2 hours. After washing, we added substrate tetramethyl benzidine and then the stop solution (2 N HCl). For standard curve, we measured the protein concentrations of anti-HSP 60, 70 and 90 antibody (StressGen), and added twofold dilutions of anti-HSPs to the plates. After washing, we also added secondary antibody with HRPconjugated antirabbit IgG antibody (KPL, England, UK). We measured the absorbance with ELISA plate reader (Microplate Reader Emax, Molecular Devices, Sunnyvale, CA, US) at 450 nm, and calculated the concentrations of the anti-HSP antibodies according to the standard curve. The serum samples from normal controls were also assessed side by side. The ELISA data were represented as mean of the duplicated well ±SD.
Statistical analyses
We used the one-way analysis of variance (anova) to analyse the differences of autoantibody levels between bipolar patients and normal controls. We also used paired t-test to compare antibody-level changes in patients in acute mania with those in remission. The differences were considered significant if p-value was equal or less than 0.05.
Results
We enrolled a total of 94 acute bipolar mania patients (M/F = 45/ 49), with 37 of them medicated and 57 unmedicated at the time of blood sampling. The samples also consisted of 44 bipolar patients in remission and another 48 normal controls (male/female = 27/21). The mean age in years (±SD) of the bipolar mania patients was significantly greater than that of the normal controls (32.1 ± 9.0 vs 25.3 ± 4.5, p < 0.001, independent t-test). The mean duration of illness was 7.3 ± 5.6 years. The mean YMRS score of acute mania was 33.3 ± 7.6 points (range 26–49). The mean YMRS score was 4.0 ± 2.1 points (range 0–12) during the remission state.
Glial cells, which can provide trophic support to neurones, neuronal metabolism and the formation of synapses and neurotransmission, are one of the possible pathogenesis factors in major psychiatric illnesses [17, 18]. In this study, we used human glioblastoma cell line U373 MG cell lysate screened for the autoantibodies for the study subjects, and we found that patients had more antibodies against proteins with molecular weights of 60, 70 and 90. Patients with abundant HSPs or with schizophrenia were found to have increased levels of anti-HSP antibodies [9–11,19]. Those findings led us to hypothesize that those autoantibodies might be targeted at HSPs. To confirm this hypothesis, we used pure HSP (HSP60, 70 and 90) as antigen for SDS-PAGE and hybridized with patients' sera. We found that for anti-HSP60, 19.2% of acute mania patients and 15.8% of normal controls had this autoantibody. AntiHSP70 was weakly detected but could be detected in all tested subjects. Anti-HSP90 was detected in 53.1% of patients with acute mania, 26.2% of those in remission state and 47.4% of normal controls (Fig. 1).
Anti-HSP antoantibody detection by Western blot to pure HSP proteins. Lanes 1, 7 and 8 were blotted with rabbit polyclonal anti–HSP90, anti-HSP70 and anti-HSP60 antibodies, respectively. Lanes 2–6 were blotted with subjects' sera at a dilution of 1:200 sera. Lanes 2–4 were from mania patients and Lanes 5 and 6 were from normal subjects. Anti–HSP90 autoantibody was present in Lane 6. Anti–HSP70 autoantibody was present in all tested subjects but weak in Lanes 5 and 6. Anti–HSP60 was also present in Lanes 3, 6 and 7. HSP, heat shock protein.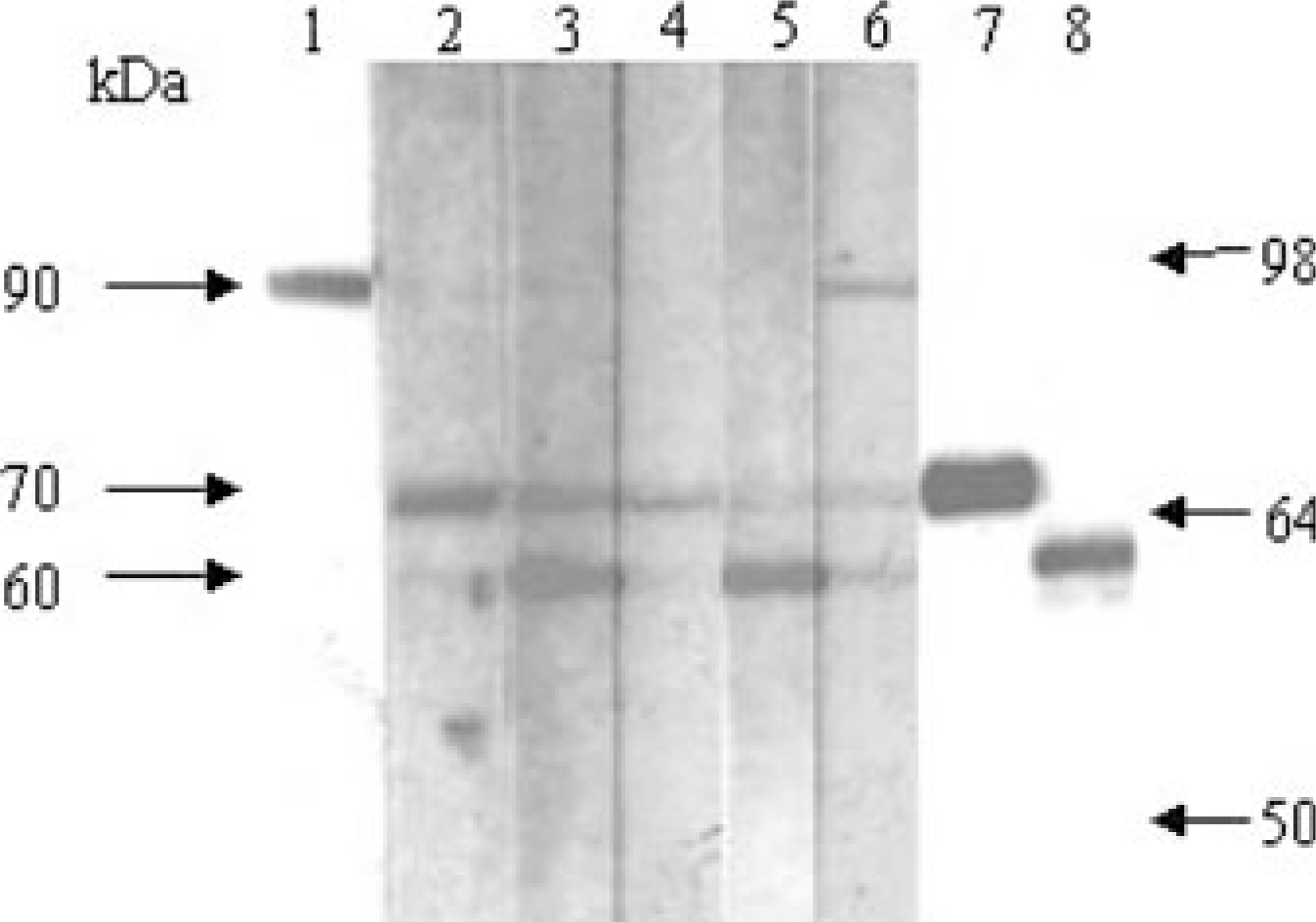
Table 1 lists anti-HSP60, 70 and 90 autoantibody levels in ng mL−1 (mean ± SD) in patients with bipolar mania and in normal controls. There was a non-significant trend of higher anti-HSP90 autoantibody levels in acute unmedicated patients compared with those in medicated manic patients (33.16 ± 21.48 ng mL−1 vs 21.9 ± 14.2 ug mL−1, F = 3.691, df = 1,65, p = 0.059, one-way anova).
Anti-HSP60, 70 and 90 autoantibody levels in ng mL−1(mean ± SD) in patients with bipolar mania and in normal controls
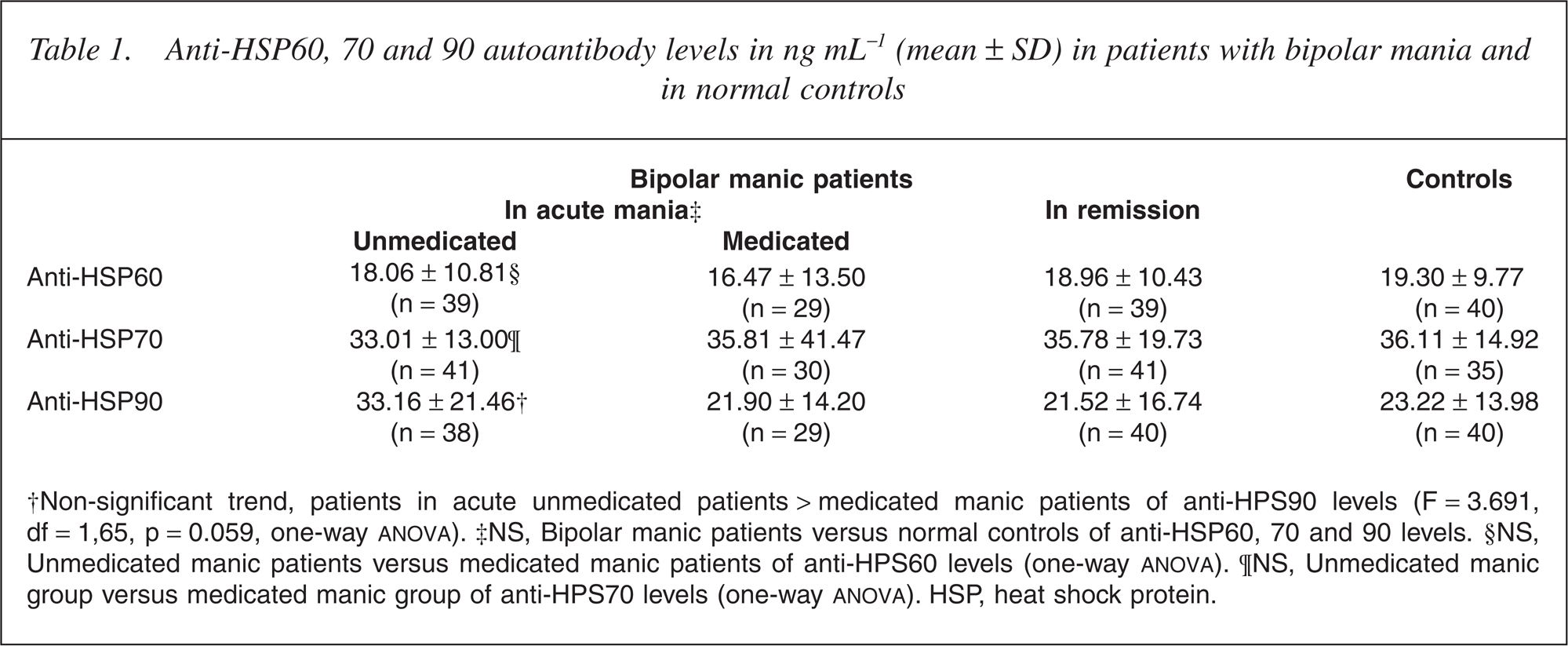
†Non-significant trend, patients in acute unmedicated patients < medicated manic patients of anti-HPS90 levels (F = 3.691, df = 1,65, p = 0.059, one-way ANOVA). ‡NS, Bipolar manic patients versus normal controls of anti-HSP60, 70 and 90 levels. §NS, Unmedicated manic patients versus medicated manic patients of anti-HPS60 levels (one-way ANOVA). ¶NS, Unmedicated manic group versus medicated manic group of anti-HPS70 levels (one-way ANOVA). HSP, heat shock protein.
We examined the changes in anti-HSP autoantibody levels between patients in acute mania and patients in remission (Table 2). The total serum anti-HSP90 autoantibody levels were significantly increased in patients in acute mania compared with those in remission (26.35 ± 21.17 ng mL−1 vs 21.52 ± 16.74 ng mL −1 , p = 0.002, paired ttest). As shown in Table 2, the differences were also noted in both medicated and unmedicated subgroups (p < 0.05, paired t-test).
Changes in anti-HSP autoantibody levels (ng mL−1) ± SD between patients in acute mania and patients in remission
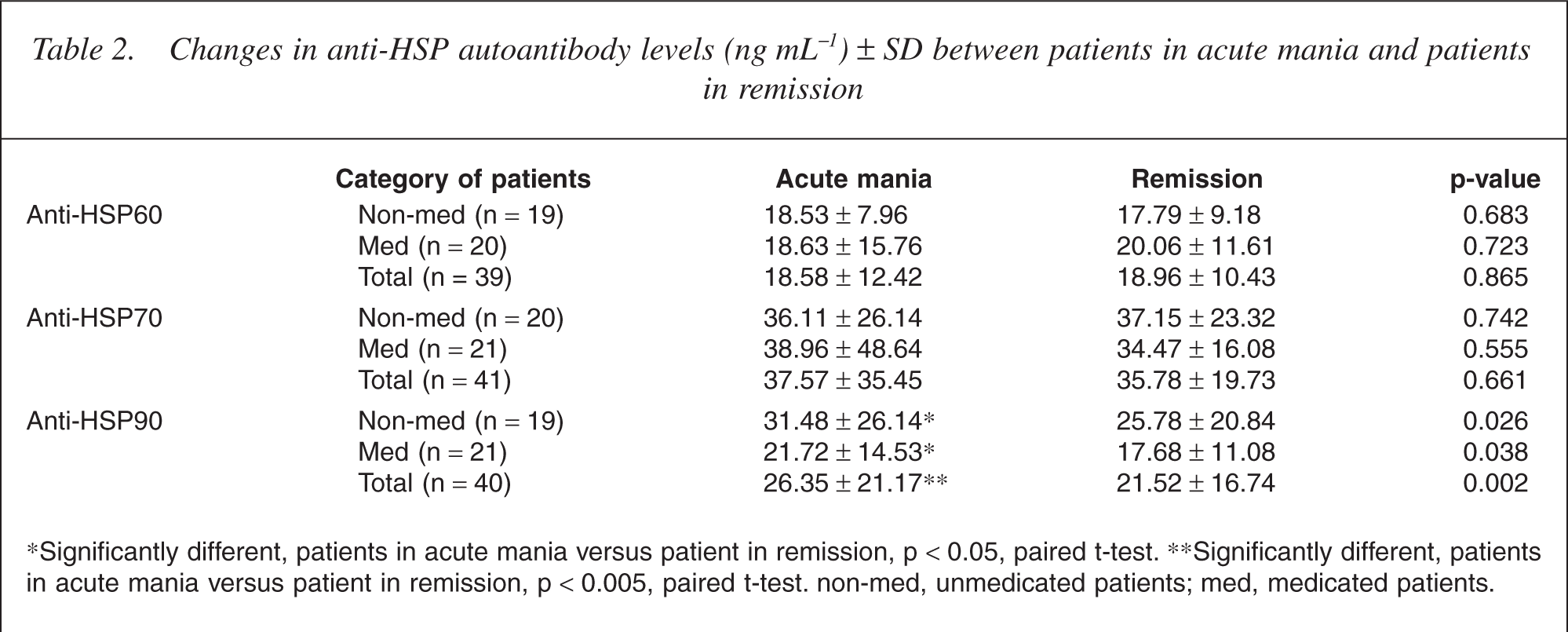
∗Significantly different, patients in acute mania versus patient in remission, p < 0.05, paired t-test. ∗∗Significantly different, patients in acute mania versus patient in remission, p < 0.005, paired t-test. non-med, unmedicated patients; med, medicated patients.
Discussion
As shown in Table 2, the major finding of our study showed that anti-HSP90 antibody levels in patients with bipolar mania were significantly higher in acute mania than in remission (p = 0.002). There was a significant trend of higher anti-HSP90 levels in the unmedicated subgroup than in the medicated subgroup in acute manic state (p = 0.059) (Table 1).
In this study, patients in the unmedicated group were those who did not receive psychotropic agents for at last 2 weeks before undergoing blood sampling. The findings of the study suggest that anti-HSP90 level might be highest in patients in unmedicated acute manic state, that this level decreases gradually after the patient received psychotropic agents, and that the level was lowest in patients in the remission state. Those findings were also found in the data from the Western analysis of pure HSP. Anti-HSP90 was detected in 53.1% of patients in acute mania but only in 26.2% of patients in remission upon a 1:200 dilution of sera.
In our study, the decrease in the anti-HSP90 level in our patients in manic state (Table 2) may have resulted from the effect of drug and/or clinical improvement. In 2001, Kim et al. reported that a subset of schizophrenia patients had a high level of anti-HSP90 and/or antiHSP70, that their clinical severity was positively correlated with anti-HSP70 level, and that the levels were decreased after clinical improvement, but that the antiHSP90 level was not associated with clinical severity. Therefore, the results in Kim et al.'s study in 2001 suggest that treatment with antipsychotic drugs does not lower the anti-HSP90 level.
The relationship between treatment with antipsychotics and the level of anti-HSP antibodies is complicated. Anti-HSP60 levels were reported to be increased in medicated schizophrenia patients [10]. We did not control the psychotropic agents in this study. Our patients received various combinations of medications, including lithium, carbamazepine, valproic acid and antipsychotics. Those psychotropic agents may have influenced the anti-HSP level. Although there were potential drug effects, the significant decrease in the anti-HSP90 level when the patients remitted from manic episode suggested that elevated anti-HSP90 might be a state marker for bipolar mania patients in acute manic state, although the potential drug effect directly on the anti-HSP level cannot be ruled out.
The biological significance of anti-HSP90 antibodies in the central nervous system is not well known. The HSP90 has been shown to have neuroprotective effects [20–22]. Then the antibody against HSP90 would have the potential to compromise normal neuronal functions. The anti-HSP90 antibody was shown to be toxic to oligodendrocytes, impairing the re-myelination process in patients with multiple sclerosis [23]. Anti-HSP90 is also found in patients with various autoimmune disorders such as rheumatoid arthritis, ankylosing spondylitis, systemic lupus erythematosus and inflammatory bowel diseases [24, 25]. The findings in this study have added another evidence that immune mechanism may play a role in the pathogenesis of bipolar disorder [4].
The major weakness of this study was that we did not control the medications in the treatment course. The drug effect on anti-HSP90 level could be shown by other studies in which patients' medications were well controlled. The second weakness of this naturalistic treatment study was that our enrolled subjects had a high dropout rate. We enrolled 94 patients in acute mania; however, only 44 of them in remission were seen in follow-up. Thus, a larger sample size is needed to confirm our findings. The third weakness of this study was that our control subjects were much younger than our study patients. Because age is an important factor in autoimmune disturbances [26], we suggest that a study with age-matched control subjects is needed in the future.
In conclusion, the findings of this preliminary study showed that a high anti-HSP90 autoantibody level might be a state marker for patients in acute mania, and its level was reduced after they remitted from mania. Although the findings need to be duplicated in a study with a larger sample size and long-term follow-up, we contend that autoimmune mechanism involving HSP90 may play a role in causing manic episodes in patients with bipolar disorder.
Footnotes
Acknowledgements
We thank Dr Shang-Ying Tsai, in the Department of Psychiatry, Taipei Medical University and Hospital, Taipei, Taiwan, for patients' sample and data collection. This study was supported by Taipei Medical University, Wan Fang Hospital (Grant 93TMU-WFH-15), National Science Council of Taiwan (Grant NSC 90–2314-B-038–019) and Taipei Medical University (Grant TMC 89-Y05-A101; principal investigator, Dr Sy-Jye Leu).
