Abstract
Clozapine is a dibenzodiazepine derivative and a truly atypical antipsychotic [1]. Clozapine may, however, unfavourably alter the lipid profile in some patients [2, 3]. Clozapine appears to particularly increase triglyceride levels [3], which is associated with an increased risk of coronary artery disease [4]. Finding effective and safe lipid-lowering treatments for patients taking clozapine is of value.
Fish oils contain high concentrations of omega-3 fatty acids in the forms of eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) [5]. Omega-3 fatty acids have been shown in studies to reduce both triglyceride levels [6–8] and incidence of coronary artery disease [9–11]. The American Heart Association recommends the use of fish oils in the treatment of people with hypertriglyceridaemia [12]. Fish oil supplementation appears to be very safe, with studies providing no indication of onerous harmful effects associated with increased omega-3 intake [13]. When utilized in patients with schizophrenia, a recent review by the Cochrane Group concluded that fish oil supplementation is possibly associated with mild diarrhea, but produces no other side-effects or interactions [14]. Omega-3 fatty acids have been trialled in patients taking clozapine without the emergence of interactions or significant side-effects [15]. Therefore, omega-3 fatty acids are likely to be the safest of all currently available lipid-altering treatments, in patients taking clozapine. To date, there appear to be no studies utilizing omega-3 fatty acids for their lipid-lowering effect in patients taking clozapine. If fish oils were found to be efficacious with no associated side-effects or interactions, then this treatment could have a positive impact on current clinical practice. Because lipid abnormalities may occur not just with clozapine, but with other atypical antipsychotics and the phenothiazines [16], there is a potential for utilizing fish oils across a broader spectrum of patients.
The aim of this study was to explore the efficacy and tolerability of omega-3 fatty acid supplementation in a group of patients taking clozapine, with a particular focus on triglyceride levels. We hypothesized that omega-3 supplementation would result in significant reductions in triglyceride levels, with minimal side-effects or interactions.
Method
Participants
Participants were recruited through the outpatient clozapine clinic of the Townsville Mental Health Service (located in North Queensland, Australia). Patients who had received clozapine continuously for at least 6 months and were not currently taking omega-3 fatty acid supplements were individually approached and invited to volunteer for the study.
Patients were excluded if they had any severe or unstable physical illnesses, including cardiac, renal, pancreatic or hepatic disease. Patients with ongoing alcohol or substance dependence or abuse were excluded. Patients were excluded if their psychiatric illness was sufficiently severe or unstable that their treating psychiatrist was of the opinion that participating in the trial could be harmful to the patient.
Once patients consented to enter the study, they received a physical examination. Screening blood investigations, including full blood count, liver function tests, clozapine serum levels and fasting lipid levels were carried out. Patients with any changes in blood parameters that were deemed to require immediate treatment or further investigation were excluded.
In all, from 115 patients registered in local area as receiving clozapine, 65 patients were deemed ineligible to enter the study. Primary reasons for exclusion of patients included: active substance use (21), insufficient time taking clozapine (14), physical illness (12), unstable psychiatric illness (8), and current use of omega-3 fatty acids (3). Seven participants could not be contacted and were therefore excluded. Of the 50 eligible patients approached, 31 patients consented to enter the study. Three patients were subsequently excluded from the study because of inability to comply with the fasting requirements.
Supplements
Supplements were given in the form of gelatin capsules (encapsulation by Laboratories Pharma-A Care, Pty Ltd, NSW, Australia) and contained 1000 mg of salmon oil. Each capsule contained approximately 180 mg of EPA and 120 mg of DHA.
Laboratory analysis
The primary outcome measures were fasting triglyceride and cholesterol levels. Fasting total plasma cholesterol, high-density lipoprotein (HDL) cholesterol and triglyceride concentrations were quantified at the local hospital using standard automated enzymatic lipid assays. Low-density lipoprotein (LDL) cholesterol was calculated using the Freidwald equation [17]. Very low-density lipoprotein cholesterol (VLDL) was calculated using the formula as follows: VLDL = TC (total cholesterol) − (HDL + LDL).
The Freidwald equation reduces in accuracy as triglyceride levels rise [18]. When triglyceride levels are above 4.5 mmol L−1, the local laboratory does not calculate VLDL and LDL levels, citing unreliability of results. This resulted in missing data of some VLDL and LDL levels.
Serum lipid estimations, particularly triglyceride levels, are unreliable in the non-fasting state [19]. Participants were contacted the night before their lipid assays, and reminded to fast. If participants had not fasted, the process was repeated on subsequent days, until a fasting sample was obtained.
Procedure
The study was a 4 week, open-label design. All participants were given supplements of omega-3 fatty acids in the form of salmon oil. Participants were requested not to make any significant changes to their diet or lifestyle during the study period.
The timeframe of 28 days was chosen in keeping with a number of studies of similar duration that have demonstrated lipid-lowering effects of fish oil within such a time period [6, 8]. The lipid-altering effect of omega-3 fatty acids can be seen within 1–2 weeks of commencement [20].
The study was approved by the Townsville Hospital ethics committee and performed in accordance with Good Clinical Research Practice Guidelines.
Participants received 3 g of EPA + DHA (10 capsules) in two daily doses. A dose of 3 g was chosen to reflect an intermediate dose in relation to the recommendations of the American Heart Association of 2–4 g of EPA and DHA in people with high triglyceride levels [12].
Subjects provided venous samples after fasting overnight at days 0 (commencement) and 28 (completion). The initial samples were analysed for the following parameters: total cholesterol, LDL cholesterol, HDL cholesterol, triglyceride levels, clozapine levels and full blood count.
Subjects were interviewed at the commencement and after completion of the trial period. A psychiatric assessment, mental state examination, medical review and a physical examination were conducted at each visit. Diagnosis was recorded and based on the most recent identified diagnostic entry in the medical records. Participants were asked to complete a feedback questionnaire at the completion of the study period. The questionnaire addressed compliance, attitudes to the fish oils, sideeffects as well as alcohol and cigarette use over the 28 days of the trial.
Statistical analysis
Lipid values and lipid changes were reviewed to confirm that results were normally distributed and suitable for parametric analysis. Two tailed, paired sample t-tests were utilized to compare differences in lipid measures on days 0 (commencement) and 28 (completion). Unless otherwise stated, significance levels were at p < 0.05. Unpaired t-tests were utilized to assess the effects of gender, smoking status and initial triglyceride levels on subsequent lipid changes. Descriptive statistics were also utilized.
Missing data
Of the 31 persons who entered the study, only the data for 28 are presented. For the other three participants, it was not possible to obtain accurate initial lipid assays because of non-compliance with fasting requirements. These participants were excluded from the analysis.
Two HDL values are missing. Owing to laboratory problems, the tests were not done and could not be repeated. Subsequently, LDL and VLDL estimates were not possible for these participants.
Results
Participant differences
The participant characteristics are outlined in Table 1. The group consisted of patients with a diagnosis of schizophrenia and schizoaffective disorder. No standardized diagnostic interviews were used. Diagnosis was based on the last charted diagnostic entry that could be identified. There was a high incidence of obesity in the group, based on measurements of body mass index. Women, on average, had a higher body mass index than men (36.7 kg m−2 for women; 30.7 kg m−2 for men). Seventeen (60%) of the participants were current smokers. High triglyceride and cholesterol levels were noted at baseline.
Baseline characteristics of participants
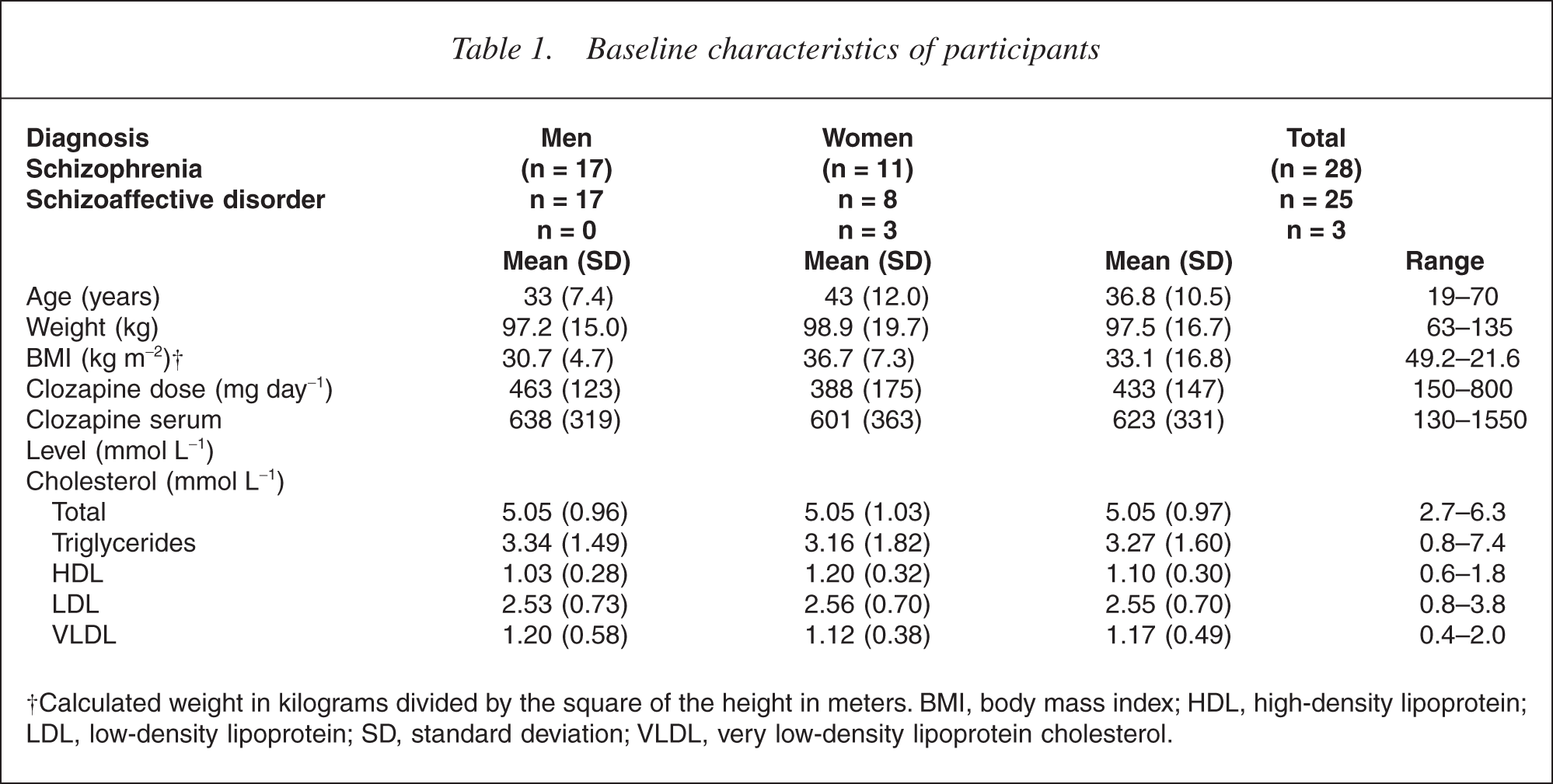
†Calculated weight in kilograms divided by the square of the height in meters. BMI, body mass index; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SD, standard deviation; VLDL, very low-density lipoprotein cholesterol.
Triglyceride changes
The study participants demonstrated a mean decrease in triglyceride levels of 0.72 mmol L−1 (22%) (Table 2). Most of the participants (79%) experienced a reduction in serum triglyceride levels. The Australian National Heart Foundation recommends that target triglyceride levels should be below 2.0 mmol L−1 [21]. At the completion of the trial, 11 participants (39%) registered triglyceride levels at or below 2.0 mmol L−1, compared with seven participants (25%) at the commencement.
Mean lipid changes over the study period †
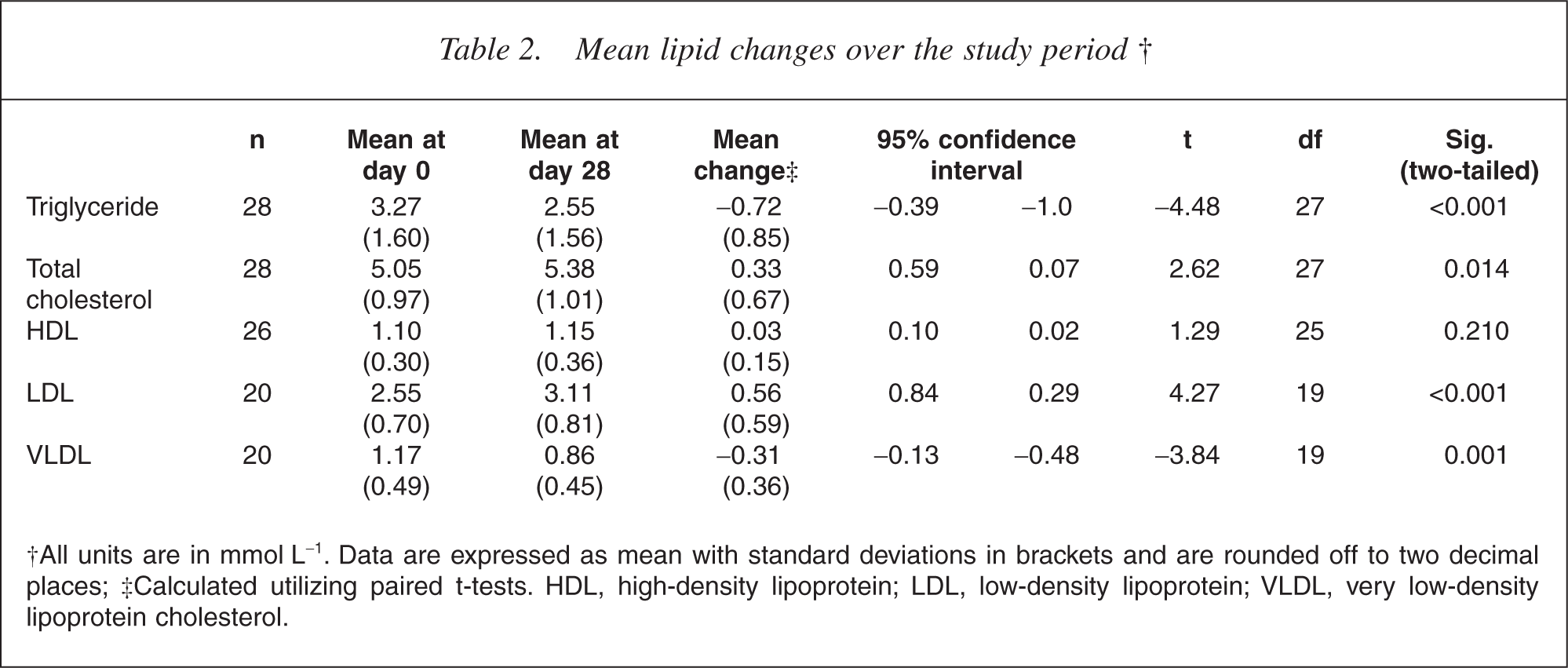
†All units are in mmol L−1. Data are expressed as mean with standard deviations in brackets and are rounded off to two decimal places; ‡Calculated utilizing paired t-tests. HDL, high-density lipoprotein; LDL, low-density lipoprotein; VLDL, very low-density lipoprotein cholesterol.
We attempted to analyse the data in order to identify meaningful subgroups of participants who may have responded preferentially to the intervention. Mean triglyceride changes were compared according to gender, smoking status and initial triglyceride level utilizing independent sample t-tests (Tables 3–5).
Triglyceride (TG) changes according to gender

CI, confidence interval; df, degrees of freedom; SE, standard error.
Triglyceride (TG) changes according to smoking status

CI, confidence interval; df, degrees of freedom; SE, standard error.
Triglyceride (TG) changes according to initial TG level

CI, confidence interval; df, degrees of freedom; SE, standard error.
Men had a slightly greater reduction in triglyceride levels than women (0.87 mmol L−1 vs 0.50 mmol L−1), but this was not statistically significant. Smoking status did not alter triglyceride reductions in participants.
Participants whose initial triglyceride levels were above or equal to 3.0 mmol L−1 demonstrated greater mean reductions in triglyceride levels than those whose levels were below 3.0 mmol L−1 (0.37 mmol L−1 vs 0.99 mmol L−1). This difference was statistically significant though not especially surprising. When represented as a percentage change from the participants' initial triglyceride levels, however, there was no difference between the two groups (20.0% vs 22.8%).
Changes in other lipid parameters
Mean changes in total cholesterol, HDL, LDL and VLDL are outlined in Table 2. Participants experienced a 0.33 mmol L−1 (6.6%) increase in total cholesterol and a 0.04 mmol L−1 (3.4%) increase in HDL levels.
There was a 0.57 mmol L−1 (22%) increase in LDL and a 0.31 mmol L−1 (26%) reduction in VLDL in the standard analysis (Table 4). In both cases, these results were statistically significant. However, seven participants had triglyceride levels above 4.5 mmol L−1, so LDL and VLDL values were not calculated. This represents significant missing data.
Tolerability and side-effects
Participants were given a simple feedback questionnaire (available from the author on request), which addressed compliance, side-effects, acceptability and desire to continue taking the fish.
Self-reported compliance was high. Most participants (64%) reported that they had taken the fish oils ‘all the time’. Only three participants reported ceasing their fish oils completely before the completion of the study. A significant proportion of participants (36%) felt the fish oils had been beneficial or very beneficial.
No serious side-effects were described by any of the participants. The most common side-effects were fishy burps or fishy breath, which where reported by 43% of the participants. Other reported side-effects included diarrhea (two participants), flatulence (one participant) and nausea (one participant). Over 60% of participants reported at least one side-effect. The most common complaint about the trial medication was the large dose and relatively large capsules that needed to be swallowed.
No medical problems were noted in any of the participants. No changes in psychiatric symptomatology were observed during the study period. No weight gain was observed during the study period.
Discussion
At the commencement of our study, the participants demonstrated significantly elevated lipid levels according to current Australian National Heart Foundation guidelines (Table 6). Mean triglyceride levels, at 3.3 mmol L−1 (<2 mmol L−1, optimal levels recommended by the Australian National Heart Foundation), were especially elevated. While mean total cholesterol was elevated at 5.1 mmol L−1 (<4 mmol L−1), mean LDL cholesterol at 2.55 mmol L−1 (<2.50 mmol L−1) was only marginally above optimal levels. Mean HDL cholesterol, at 1.10 mmol L−1 (<1 mmol L−1), was within the recommended range.
Optimal lipid levels†
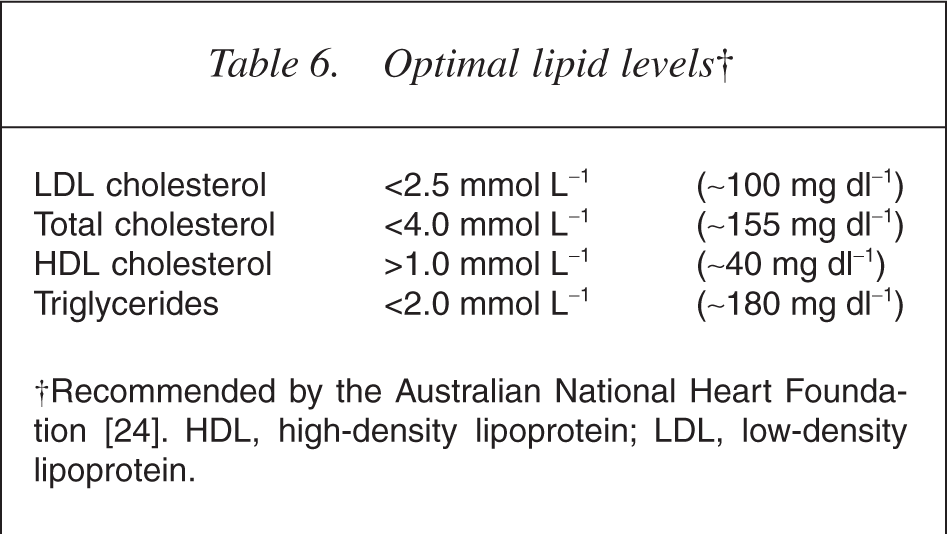
†Recommended by the Australian National Heart Foundation [24]. HDL, high-density lipoprotein; LDL, low-density lipoprotein.
Over the study period, participants demonstrated statistically significant reductions in triglyceride levels. Literature indicates that elevated triglyceride levels are an important independent risk factor for coronary heart disease [17, 22, 23]. A recent meta-analysis estimated that a 0.1 mmol L−1 increase in triglyceride levels resulted in an increase of unadjusted relative risk of cardiovascular disease of 3.2% in men and 7.2% in women [17]. Extrapolating these data to our study population would indicate approximate reductions in unadjusted cardiovascular risk of 27.5% for men and 36% for women. This study indicates that there could have been significant health benefits in our experimental group, if the triglyceride changes were sustained over a long-term period.
The lipid changes observed in our study were of a slightly greater magnitude than those of a recent review by the Cochrane Group [7]. Their meta-analysis estimated a reduction of 0.56 mmol L−1 (95% CI = −0.71–0.40 mmol L−1) in triglycerides and an increase of 0.21 mmol L−1 (95% CI = 0.02–0.41 mmol L−1) in LDL cholesterol for patients with diabetes who were taking omega-3 fatty acid supplements. This review confirmed a greater triglyceride-lowering effect in hypertriglyceridaemic subjects and in studies with higher fish oil dosages (more than 2 g). High initial triglyceride levels and the high dose of fish oils utilized may therefore explain why our group had greater reductions in triglyceride levels and greater increases in LDL cholesterol than those estimated by the Cochrane Group. Conceivably, a direct interaction between fish oils and clozapine could have been responsible for the observed changes.
Low-density lipid cholesterol is traditionally regarded as the most atherogenic of the lipid estimates in current routine use [24]. The rise in total cholesterol and LDL cholesterol in the participants in this study is therefore of concern. The elevation in cholesterol from omega-3 fatty acids supplementation, however, may not be a harmful one. The increase in cholesterol appears to be primarily due to elevations in larger, cardioprotective apolipoprotein subfractions rather than atherogenic subfractions [25]. This evidence is in keeping with the many trials that demonstrate an antiatherogenic effect of fish oil supplementation [10, 11]. Increased levels of HDL and reduced levels of VLDL cholesterol, both of which are cardioprotective, may also help offset the any detrimental rise in cholesterol [25].
The clinical validity of various currently utilized lipid parameters is complex and is currently the focus of much research and debate. The understanding of lipids is advancing beyond cholesterol and triglyceride measurements. Some authors have focused on apolipoprotein (apo) levels. In at least four recent studies, apolipoproteins levels have been shown to be a more robust indicator of cardiac risk that any other lipid markers [24]. Jacotot reported that omega-3 fatty acids, despite in some cases increasing LDL concentrations, favourably altered the ratio of apoA-1 to apoA-2 [25]. Some research suggests that the reductions in apoC-III may explain the cardio-protective effects of omega-3 fatty acids [23]. It would have been of interest to assay markers for cardiovascular risk, such as apolipoprotein and arachidonic acid levels in our study group. Unfortunately, this was prohibitively expensive, but might be possible in future trials.
Omega-3 fatty acid dosages utilized in this study reflected the recommendations of the American Heart Association for the treatment of hypertriglyceridaemia [12]. Different trials, however, have utilized different dosages. The GISSI-Prevention trial, for instance, was able to demonstrate reductions in cardiac mortality utilizing only 850–882 mg of omega-3 fatty acid daily [13]. In order to achieve a triglyceride-lowering effect, however, doses of at least 3 g daily are generally required [14]. Optimal dosing for patients taking clozapine remains to be established. The large number of tablets that needed to be taken by the participants represents a concern, especially in regards to compliance over the longer term. Higher potency formulations of omega-3 fatty acids are available, but their cost is currently higher than standard marine oil formulations.
The fish oils consumed represented a significant intake of calories; however, no increase in weight was observed in our study. While the timeframe of 28 days was relatively brief, the results are in keeping with large meta-analyses, which have not detected weight increase with fish oil supplementation [7, 13].
The study has major limitations in both its size and design. An obvious limitation of this study is its openlabel design. Therefore, the underlying premise in this study is that lipid levels would not have changed spontaneously in this group without the intervention. Confounding factors, such as changes in lifestyle and diet, as a result of increased awareness, might have been responsible for observed lipid changes. Although asked participants not to make changes to their diet and lifestyle during the study, clearly utilizing a control group would have been of great value in this study. In a number of previous studies, as reviewed by the Cochrane Group, lipid variables appear to have been reasonably stable in the placebo arm of the trial [7], suggesting that the observed changes were likely due to our intervention. Of relevance is that fasting state of the participants was only assessed through self-report, and this may not have been reliable.
Following fish oil supplementation, participants demonstrated reduced triglyceride and VLDL levels and raised total cholesterol, HDL and LDL levels. This specific pattern of change corresponds to observations in other trials and to the expected changes from fish oil supplementation. This would support the likelihood that omega-3 fatty acids were most responsible for the observed changes.
The small size and short duration of the study limit its power to detect lipid change as well as side-effects and adverse reactions. It is not possible to establish long-term efficacy, safety and tolerability because of this. The selection process was not random, and limited extrapolation of the results to general clinical populations. The study was only intended as a pilot study, in the absence of other research in the area. While initial results are promising, further controlled studies are needed.
Lipid biology has been implicated in the pathogenesis of schizophrenia [26]. Clinical trials remain inconclusive, but indicate that omega-3 fatty acids may be of value for the treatment of schizophrenic patients [14]. A review by Peet et al. suggests that essential fatty acid supplementation may be particularly efficacious in treating symptomatology in patients taking clozapine [27]. These factors may be added to the therapeutic appeal of utilizing omega-3 fatty acids in psychiatric populations.
Conclusion
In summary, this study suggests that the lipid-altering profile of fish oils is similar in people taking clozapine as it is in other populations. Overall, fish oils were well tolerated, with no serious side-effects noted. Omega-3 fatty acids are recommended for the treatment of raised triglyceride levels. This trial indicates that these supplements are likely to be safe and efficacious for patients taking clozapine. The high rates of lipid abnormalities and other cardiovascular risk in the study sample suggests that we should be vigilant in monitoring and treating these conditions.
Footnotes
Acknowledgements
This study was partially funded by a $2000 grant by Novartis, the makers of ClozarilTM (clozapine). In no other way was the company involved in this trial. Additional funding was provided by Townsville Mental Health Services. Thanks to Satish Karunakaran, John Allan and June Ling.
