Abstract
Approximately 15% of women experience an episode of perinatal depression (PND), antenatally and/or postnatally [1]. Because there are increasing concerns about possible adverse effects of antidepressant medication use in pregnancy and in breast-feeding mothers [2] it is important to investigate possible alternative treatments. One candidate antidepressant strategy is the use of fish oil, in light of its omega-3 constituent.
We have reviewed epidemiological, biological marker and treatment studies suggesting that deficits in dietary-based omega-3 may contribute to mood disorders (including perinatal depression) and that fish oil supplementation (as monotherapy or as augmentation) may provide a therapeutic strategy [3], [4].
Many theories have been proposed to suggest how brain omega-3 polyunsaturated fatty acids (n-3 PUFAs) affect receptors and neurotransmitters implicated in depression. One view is that changes in fatty acid concentrations in cells affect membrane fluidity [5]. Another theory holds that the n-3 PUFAs (eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA)) have effects on inflammatory and immune systems via eicosanoids and cytokines [6] and which have been linked to depression via effects on the hypothalamic–pituitary–adrenal (HPA) axis [7]. Brain-derived neurotrophic factor, which has antidepressant effects, is also affected by n-3 PUFAs [8].
The possible relevance of n-3 PUFAs in PND is supported by several epidemiological studies. Hibbeln reported a strong negative correlation between fish consumption and major depression in a cross-national study [9] and an inverse relationship between the prevalence of post-partum depression and seafood consumption as well as levels of DHA in mother's milk [10]. DHA is a long chain polyunsaturated fatty acid that is depleted perinatally due to fetal and neonatal accretion [11].
Using fatty acid analysis, n-3 PUFA plasma and red blood cell levels have been shown to be decreased in depressed compared to non-depressed subjects [12–14]. Perinatal studies are, however, inconclusive at this stage. Two prospective case-controlled studies of depressed perinatal women have identified fatty acid blood levels as distinctly lower than in non-depressed women (n = 48 and 114, respectively) [15], [16]. A negative study (n = 52) reported that fish consumption during pregnancy (measured using a food frequency questionnaire) was not predictive of postnatal depression and that the postnatal omega-3 fatty acid blood level status was not associated with postnatal depression [17]. The Avon Longitudinal Study of Pregnancy and Childhood recruited 14 541 women. Omega-3 deficient dietary intake during pregnancy was associated with a near doubling of risk of depression antenatally and postnatally [18]. But another fairly large trial of 865 Japanese women who completed diet history questionnaires during pregnancy and an Edinburgh Postnatal Depression Scale (EPDS) postnatally, showed no dose-response associations between omega-3 and postnatal depression [19].
Studies have established that maternal long chain omega-3 fatty acids are depleted during pregnancy, particularly during the third trimester and the postnatal period, with post-mortem studies confirming that this depletion is due to fetal accretion of the omega-3 [20].
Thus, the pregnant woman is seemingly at high risk for omega-3 depletion during the perinatal period. Because depression rates are high at that time, a causal sequence is suggested, as is a therapeutic approach for evaluation.
Treatment studies in the perinatal period are very limited so far. A randomized controlled trial of 89 breast-feeding women assessed the use of 200 mg day−1 DHA supplementation compared to placebo as a preventative treatment for postnatal depression. The result was not significant but the sample size was likely to be too small to detect a difference for such a preventative strategy [21]. In a secondary prevention, open-label study, in which euthymic pregnant women were recruited with a past history of postnatal depression, and supplemented from the mid-third trimester with 3 g day−1 fish oil (1.4 EPA:DHA), four out of seven women developed depression at 12 weeks’ post-partum. Although the authors reported that this was not a promising result, the sample size was still too small to draw conclusions [22]. A randomized non-placebo controlled dose-ranging trial for post-partum depression (0.5 g day−1, n = 6; 1.4 g day−1, n = 3; 2.8 g day−1, n = 7) with an EPA: DHA ratio of 1.5:1, did demonstrate significant improvement in all groups [23].
Because omega-3 fatty acids may be more acceptable to women in the perinatal period compared to current antidepressants, it is important to assess whether omega-3 has superior efficacy over placebo as a monotherapy treatment for PND.
Methods
Depressed women were recruited for a 6 week double-blind randomized placebo-controlled trial.
Subjects were recruited over a 15 month period via the perinatal depression clinic and midwives’ antenatal clinic at Royal Hospital for Women, Sydney As well as distributing trial descriptions and invitations to local general practitioners and relevant nursing staff, recruitment was also assisted by newspaper, television and radio advertisements and interviews.
The study was carried out in accordance with the latest version of the Declaration of Helsinki. All subjects received a study information sheet and completed a consent form. The study was approved by the South Eastern Sydney Area Health Service and the University of New South Wales Human Research Ethics Committee.
We required subjects to meet criteria for a current episode of major depression or dysthymia, according to DSM-IV criteria, and confirmed this by both CIDI structured interview criteria [24] and clinical assessment by a psychiatrist.
Subjects were required to be >21 years of age and, in regard to perinatal status, from the third trimester of pregnancy to 6 months postnatal. Exclusion criteria included bipolar disorder, psychosis, drug and alcohol abuse, obsessive–compulsive disorder, eating disorder or personality disorder, an unstable medical condition, diabetes, receipt of anticoagulants or having a fish allergy. We also excluded those already receiving an antidepressant or any psychological therapy, as well as those taking fish oil supplements or eating more than three oily fish portions per week. We did not exclude those with comorbid anxiety disorders.
Subjects were initially screened via the telephone. Basic demographics, clinical information and approximate fish intake were checked to assess eligibility for the trial and the EPDS was conducted. If subjects appeared initially suitable, then the trial psychiatrist conducted a baseline assessment.
At baseline assessment the clinical interview and CIDI structured interview were performed. Subjects were also required to score ≥ 13 on the EPDS [25] and either >14 on the 17- item Hamilton Depression Rating Scale (HDRS) [26] or >25 on the Montgomery Asberg Depression Rating Scale [27]. Demographic information was collected, and medical, psychiatric and smoking histories recorded. An assessment of each subject's diet was made, including fish intake, vitamin supplements and omega-3 enriched foods. This dietary assessment was not used for formal fatty acid intake assessment but simply to assist in assessing whether a subject met study criteria in terms of excluding those with high omega-3 intake. High omega-3 intake was deemed to be consumption of >3 oily fish meals per week, and/or fish oil capsules and/or consumption of omega-3 supplemented foods. During the study, subjects were requested not to receive any independent omega-3 supplementation and not to change their usual fish intake. Dietary assessment was then completed at the end of the study to ensure compliance with this study requirement. At baseline (and at the end of the trial) subjects were required to have a fasting blood test for plasma fatty acid analysis.
If eligible for the study, subjects were randomized to receive either 6 g day−1 fish oil or placebo in divided doses, dispensed by the hospital pharmacy, every 2 weeks, to ensure blinding of the investigators and raters. A randomization sheet was generated by a statistician who was not directly involved in the study. This involved a computer-based random number generation method. Subjects were interviewed by the first author, who remained blind to treatment assignment, and assessed weekly by her. The blind was not broken until the study had been completed.
The fish oil soft gelatin capsules and matching placebos were dispensed in identical plastic containers. The fish oil capsules contained 27.3% DHA, 6.9% EPA (total omega-3 fatty acids = 35.6%) and 3.3% omega-6 fatty acids. The remainder of the capsule content consisted of monounsaturated fats and a small amount of saturated fat. Vitamin E (80 mg) was added to prevent oxidation of the oil. Sunola oil was used to constitute the placebo, which consisted mainly of monounsaturated fatty acids (85%) and a small amount of saturated fat (7%) and polyunsaturated fats (8%). Peppermint oil was added to all capsules to disguise any fish taste and may also have minimized the gastrointestinal side-effects [28].
Outcome measures consisted of change from baseline to week 6 in the EPDS, HDRS and MADRS scores. Adverse effects were recorded in terms of time of onset, intensity and outcome. Study compliance with medication was assessed on weekly patient report and changes in fatty acid analysis from the beginning to the end of the trial. Subjects were asked if they could taste fish or peppermint in the capsules as a means of assessing the adequacy of the study blind.
Statistical analysis included conducting Student's t-test and χ2 analyses to assess differences in demographic and clinical variables. t-tests were also conducted to measure significant changes in mean scores during the trial. Repeat measures analysis of variance was used to examine depression score results in terms of differences over the trial period between the fish oil and the placebo groups. The impact of possible covariates on the results was checked using analysis of covariance. t-tests were used to measure significant changes in fatty acid analysis over the trial period and χ2 analyses were conducted to examine for differential rates of adverse effects in each group.
Results
A total of 98 women were screened initially (Figure 1). Of these, 49 women did not meet study criteria, mainly due to scoring too low on the depression rating scales or already receiving antidepressant treatments. Another 23 women refused to participate after showing initial interest, most commonly expressing concern that they may receive the placebo treatment.
CONSORT diagram: subject flow through the trial.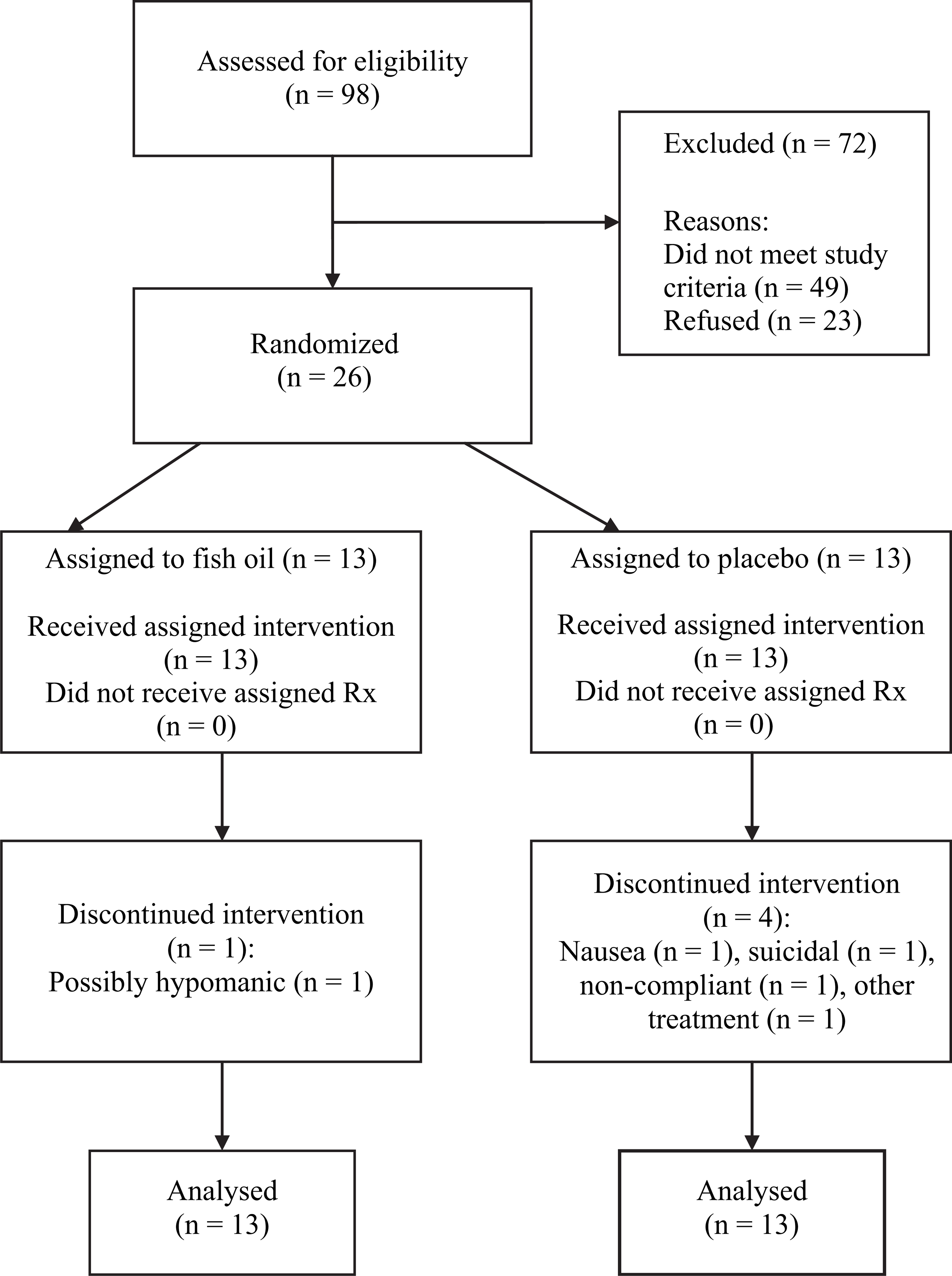
Twenty-six women were recruited into the study, 12 in the antenatal period and 14 in the postnatal period, with equal numbers (n = 13) randomly assigned to the treatment and placebo groups. Most subjects were medically well, although two subjects in each group had had thyroid disease (albeit all adequately treated and now euthyroid). Alcohol intake was either nil or very minimal in all subjects.
There were two early dropouts. One (who had been assigned to placebo) withdrew from the trial after 1 week, electing to receive counselling instead. The other (on fish oil) became hypomanic after 3 days of treatment and was withdrawn from the trial. Three other subjects (all on placebo) dropped out between weeks 3 and 5 of the study: one due to nausea; another became suicidal and was referred to an independent psychiatrist; while the third subject was non-compliant with follow-up review sessions. All 26 women were included in the analyses using an intention-to-treat statistical strategy, and with their depression scores extrapolated using the last-observation-carried-forward method.
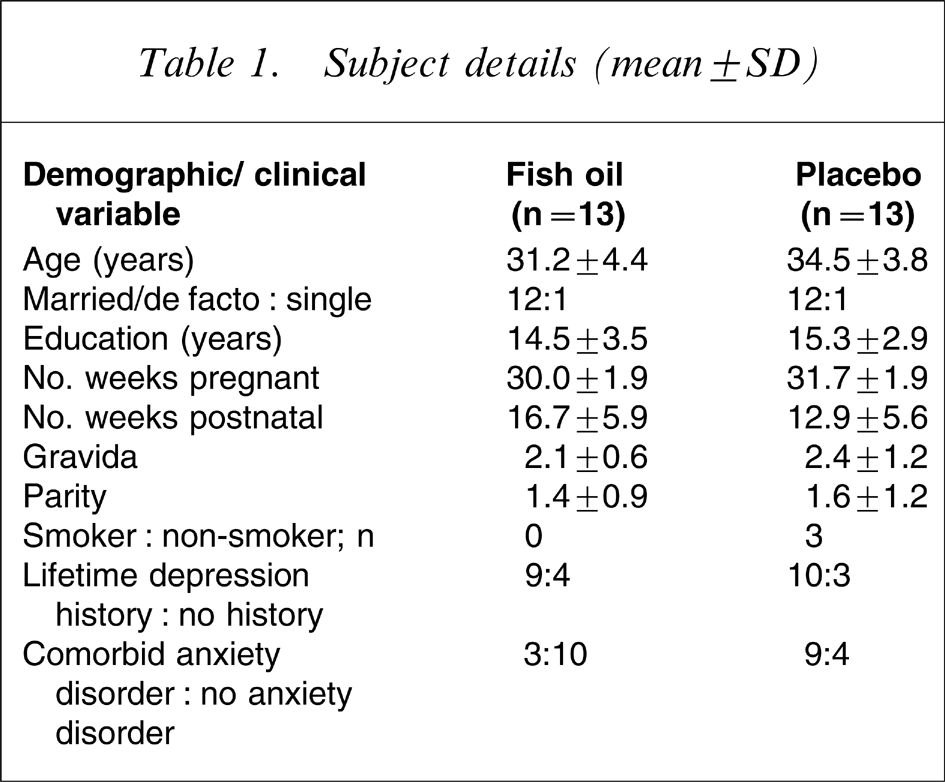
Demographic and clinical variables are detailed in Table 1. Using independent two-tailed Student's t-tests and χ2 analyses, there were no statistically significant differences (p > 0.05) between baseline characteristics of the treatment and placebo groups, apart from the placebo group being more likely to have a comorbid anxiety disorder.
Subject details (mean±SD)
There were no statistically significant differences between baseline depression scores across those receiving fish oil or placebo (i.e. EPDS 17.3 vs 16.5; HDRS 19.7 vs 19.0; MADRS 30.2 vs 29.3, respectively). Table 2 reports weekly scores on the depression variables from baseline through to the end of the 6 week trial. Mean change scores from baseline to the 6 week assessment quantified that improvement was significant (p < 0.001) across each of the depression measures and for both those receiving fish oil or placebo (EPDS 8.8 vs 7.5; HDRS 11.8 vs 9.3; MADRS 16.8 vs 14.1, respectively).
Mean (±SD) depression scores during trial
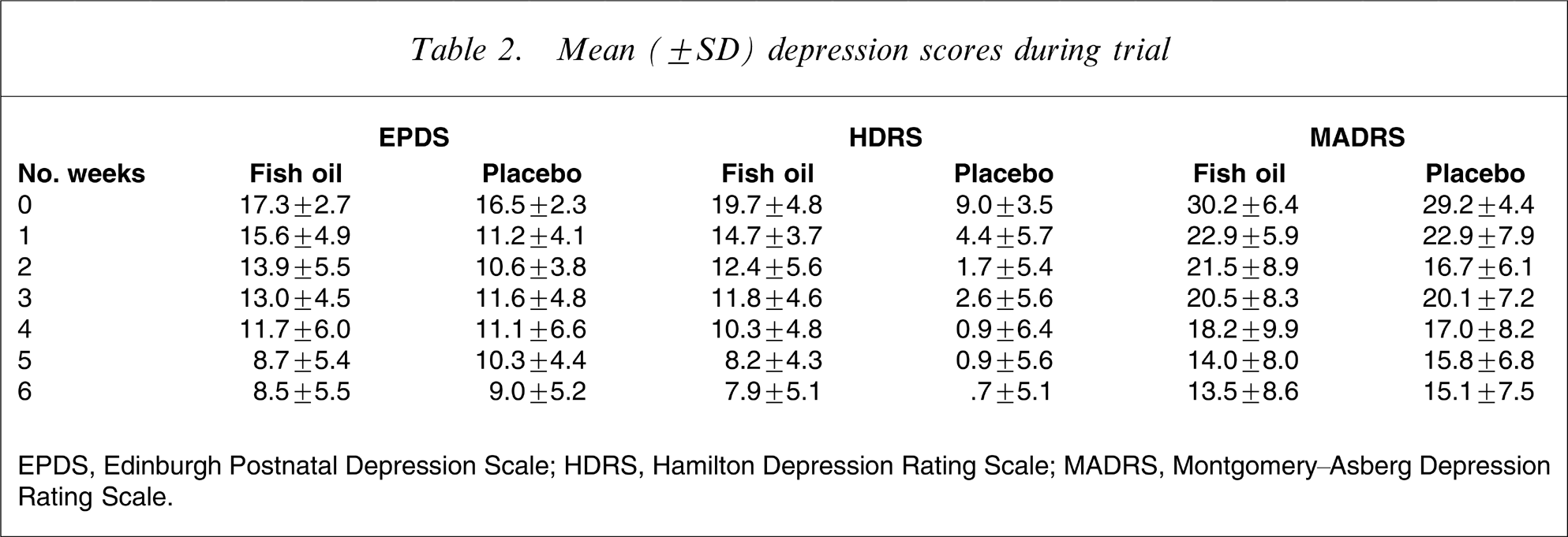
EPDS, Edinburgh Postnatal Depression Scale; HDRS, Hamilton Depression Rating Scale; MADRS, Montgomery–Asberg Depression Rating Scale.
Repeated measures analyses of variance (ANOVA) showed no statistically significant difference between depression scores (across the three measures) in the fish oil group compared to the placebo group using intention-to-treat analyses (EPDS, F = 0.79; HDRS, F = 0.01; MADRS, F = 0.17), with Figure 2 illustrating the EPDS results. In view of group difference in relation to comorbid anxiety disorder diagnosis (p < 0.05) and possibly age (p = 0.1), these two variables were both entered as covariates, using analysis of covariance, with no effect on the results. When antenatal and postnatal groups were separated, no statistically significant differences were identified.
Changes in mean Edinburgh Postnatal Depression Scale (EPDS) score.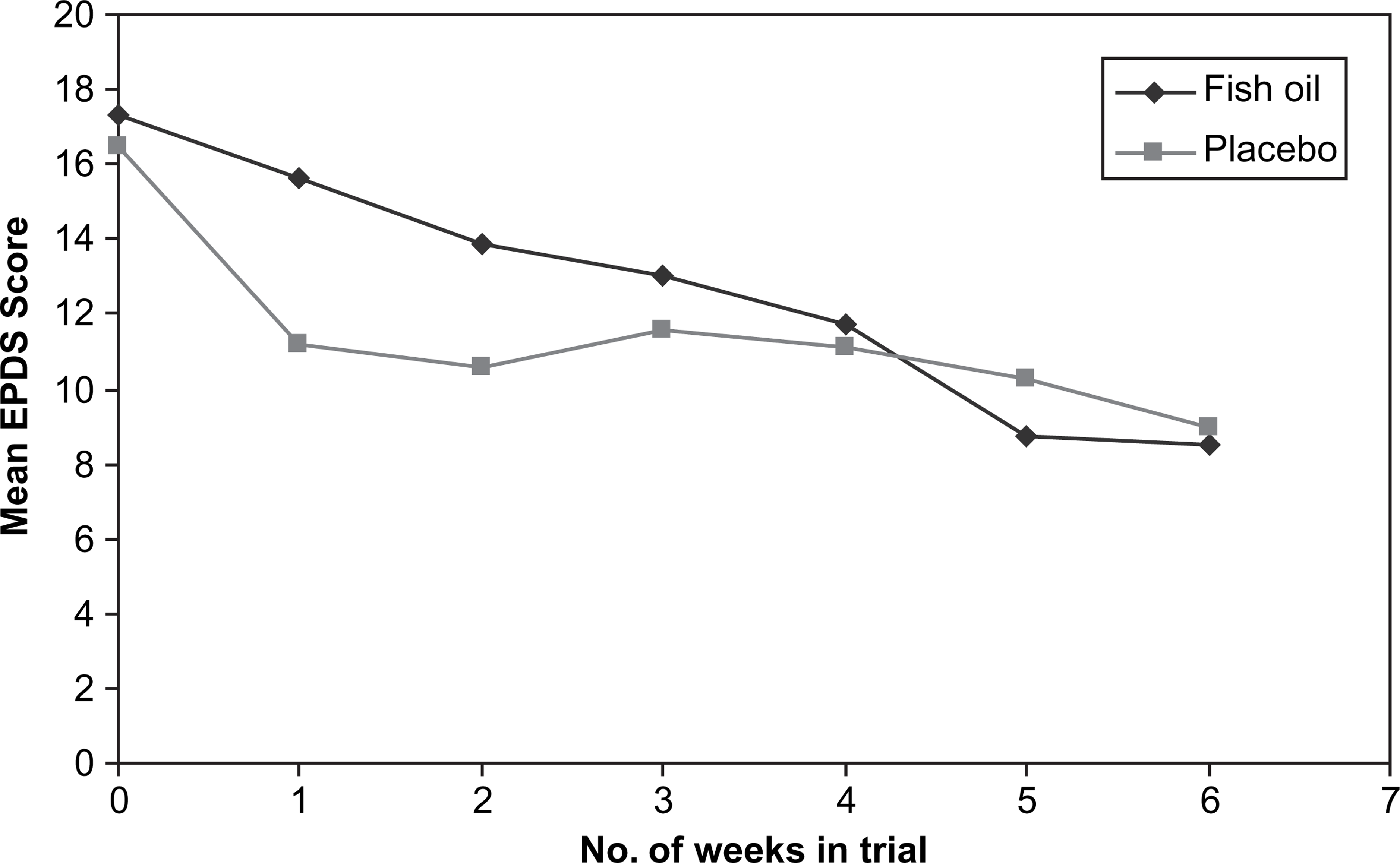
Subjects’ reports of compliance indicated that forgetting to take capsules occurred only occasionally across the trial, and equally across both treatment groups. Analyses of plasma samples at baseline and at the end of the trial demonstrated rising plasma levels for each subject receiving active treatment and supported compliance. Specifically, plasma total omega-3 mean (±SD) was 83.7±26.5 µg mL−1 at baseline, and 139.7±56.1 µg mL−1 at trial end. A paired t-test demonstrated that this was a significant increase (t = − 4.0, df = 10, p = 0.002). As expected, there was no significant change in the placebo group fatty acid levels. All subjects also reported adhering to the study criteria in relation to not taking additional omega-3 supplements and not increasing their individual fish intake during the trial. The fatty acid plasma levels reflected this. Blinding appeared adequate. A fishy aftertaste was reported by only one subject, but six reported a peppermint taste (four in the treatment group and two in the placebo group).
The χ2 analyses were undertaken to examine for differential rates of adverse effects in each intervention group. No significant differences were found between the two groups. Four subjects reported mild reflux at some stage during the trial (two in the treatment group and two in the placebo group). Three subjects reported mildly increased stool frequency, two being in the treatment group and one receiving placebo. Two subjects (one in the treatment group, one receiving placebo) reported nausea.
Discussion
This paper reports the first randomized double-blind placebo-controlled trial of fish oil as a treatment for depression in the perinatal period. The other omega-3 clinical trials for the treatment of PND reported in the literature have had small sample sizes and either not been placebo controlled or have been of a preventative nature rather than treatment studies of existing PND.
Overall, this was a very difficult trial to complete due to slow recruitment. Feedback indicated that many potential subjects commenced self-medicating in response to the extensive recruitment drive, having concluded that the study was an endorsement of its benefits and that it was wiser to commence the readily available product themselves than chance allocation to a placebo trial arm. There were also ethical issues related to the need to treat significantly depressed women with evidence-based treatments in order to ensure optimal management. It is likely that many of the women recruited into the study had milder depression (as confirmed by mean baseline HDRS scores of 19.7 and EPDS scores of 17.3 in the fish oil groups).
Hence the sample size in the present study was small. But other important clinical trials published in the literature that assess omega-3 as a treatment for depression in general have reported positive results with small sample sizes [29–31]. When considering these trials, it could be argued that the present sample sizes were appropriate.
Once recruited, subjects tolerated the treatment reasonably well and there were few side-effects reported by those in the active treatment group. They were also largely compliant. The blind did not appear to have been affected by a fishy aftertaste, which was well masked by the addition of peppermint oil. The dropout rate of 20% was similar, if not superior, to rates expected in antidepressant trials.
The randomization process resulted in baseline differences in comorbid anxiety disorders between the two groups. But when this variable was tested as a covariate there was no evidence that it related to the change in mood scores.
Overall the study yielded a non-significant difference in depression scores between the fish oil and the placebo groups, suggesting that fish oil supplementation is ineffective for PND.
There has been some recent work indicating that fish oil may be superior as a treatment for melancholic depression (as against the non-melancholic disorders) [7], and because it is likely that few of the present subjects had a melancholic disorder, a negative result could be so explained.
Assuming that the present result is a true negative one, consideration of omega-3 nuances is also in order. One issue contributing to any such result includes the controversial issue as to whether DHA or EPA is the most important therapeutic ingredient. In view of the results of randomized controlled trials for the treatment of major depression in the general population, it has been suggested that the EPA content of the fish oil should be higher than the DHA content [32], despite DHA being the most abundant in the brain. The two negative clinical trials in the general depression literature that have been published after the present trial commenced, have used a DHA-rich fish oil similar to that used in the present trial. It is, however, clear in the non-depression perinatal literature, that DHA is the n-3 PUFA most significantly depleted during the perinatal period due to fetal and neonatal accretion [11], [20]. The present test product contained significantly more DHA than EPA. This difference in types of omega-3 content may have affected the results but we speculate that, in perinatal depression, DHA and EPA are both likely to be important. Specifically, we theorize that replenishment of the depleted DHA may restore low EPA levels, because there is less pressure on the n-3 PUFA metabolic pathway to metabolize EPA into DHA if there is an external source of DHA. However, the replenishment of DHA may need to occur either before or during pregnancy. This is clearly an area where further research is needed.
Another issue relates to whether omega-3 works best via augmentation mechanisms and therefore should be added to antidepressants as opposed to being trialled as monotherapy. The statistically significant treatment trials in non-perinatal depressed groups have principally been augmentation trials [3].
Another interpretation of these results is also worthy of discussion. It is possible that omega-3 was beneficial (presumably only partially or for a subset) and that any such efficacy was masked by the well-recognized problem of large placebo responses in antidepressant trials (reports of up to 50% in some trials) [33].
Trial publicity and the intensive weekly follow up mandated in the present trial would have created a therapeutic environment activating placebo benefits in both trial arms. Spontaneous remission is also common in those with non-melancholic disorders [34] and would have contributed to improvement patterns in both arms, perhaps reflecting people with depression commonly seeking help when at their worst. Of all antidepressants licensed by the FDA over a 12 year period, analyses have shown that drug–placebo differences were minimal, with formal separation occurring only in 50% of such trials [35]. Presuming that those formalized antidepressant drugs are effective, then such data indicate that even when large sample sizes are recruited, a truly effective drug may appear ineffective if a sizeable percentage of trial subjects (in both the active and placebo group) experience a placebo response or have a spontaneous remission. The early responses in both arms of this study are suggestive of a placebo effect being prominent.
A significant improvement in depression scores was recently reported in a randomized dose-ranging trial of omega-3 fatty acids for postnatal depression [23]. In light of the improvement trajectories demonstrated across the present active and placebo groups – and the problems of depression trials in general, the absence of a placebo group in the Freeman et al. study[23] disallows any firm conclusion. Those authors noted the ethical problems associated with recruiting a placebo group in the perinatal period, highlighting the difficulties in conducting gold standard trials in certain settings.
It is particularly important that the status of omega-3 as monotherapy is clarified as a treatment for PND because it is during the perinatal period that patients are more likely to resist treatment with antidepressants due to potential fetal effects. In view of the small numbers in the present trial, as well as the likely problems with spontaneous remission, the authors suggest that this trial should not be viewed as definitive and there is a need for a larger, multicentre trial, using lead-in times [36] to minimize the placebo effects.
Footnotes
Acknowledgements
We acknowledge research grants from the NSW Institute of Psychiatry, a Neuroscience Research Grant from Pfizer Australia, an NHMRC Program Grant (222708) and an Infrastructure Grant from the NSW Department of Health. We are grateful to Numega Ingredients and Clover Corporation for supplying fish oil and placebo capsules for the study only. Sincere thanks to Kerry-Ann Egliston and Dusan Hadzi-Pavlovic for statistical assistance and to the Royal Hospital for Women's Pharmacy.
